Abstract
Ferroptosis is a regulatory cell death characterized by intracellular iron accumulation and lipid peroxidation that leads to oxidative stress. Many signaling pathways such as iron metabolism, lipid metabolism, and amino acid metabolism precisely regulate the process of ferroptosis. Ferroptosis is involved in a variety of lung diseases, such as acute lung injury, chronic obstructive pulmonary disease (COPD) and pulmonary fibrosis. Increasing studies suggest that ferroptosis is involved in the development of asthma. Ferroptosis plays an important role in asthma. Iron metabolism disorders, lipid peroxidation, amino acid metabolism disorders lead to the occurrence of ferroptosis in airway epithelial cells, and then aggravate clinical symptoms in asthmatic patients. Moreover, several regulators of ferroptosis are involved in the pathogenesis of asthma, such as Nrf2, heme oxygenase-1, mevalonate pathway, and ferroptosis inhibitor protein 1. Importantly, ferroptosis inhibitors improve asthma. Thus, the pathogenesis of ferroptosis and its contribution to the pathogenesis of asthma help us better understand the occurrence and development of asthma, and provide new directions in asthma treatment. This article aimed to review the role and mechanism of ferroptosis in asthma, describing the relationship between ferroptosis and asthma based on signaling pathways and related regulatory factors. At the same time, we summarized current observations of ferroptosis in eosinophils, airway epithelial cells, and airway smooth muscle cells in asthmatic patients.
Introduction
Regulatory cell death (RCD) is an important mode of cell death, which has an essential role during a variety of physiological processes, including organogenesis and maintenance of homeostasis. 1 Currently, growing numbers of novel forms of RCD are identified, such as apoptosis, autophagy, pyroptosis, and ferroptosis, etc. 2 Apoptosis is widely known as RCD eliciting no inflammatory responses, relying on the activation of caspase protease to induce rapid cell death under the action of serine protease.3,4 Autophagic cell death or autophagy, a lysosomal mediated RCD, is a molecular pathway in which intracellular components are conveyed to the lysosomal compartment for degradation. 5 Pyroptosis is a primitive form of RCD mediated by inflammasome activation. 6 Occurring with the rupture of plasma membrane and the release of a large number of pro-inflammatory factors, pyroptosis results in osmotic swelling and rupture of cells, and ultimately cell death. 7 Ferroptosis is an iron-dependent death pattern caused by accumulation of intracellular lipid peroxides,8,9 in which repair of oxidative poly unsaturated fatty acids (PUFAs) is impaired and lipid peroxides are increased, eventually leading to cell death. 10 In addition, ferroptosis is obviously different from that of other RCD mode in the morphology, biochemical and immunological characteristics. 11 The characteristics of ferroptosis are as follows, (1) The iron-dependent accumulation of lipid peroxides and production of mitochondrial reactive oxygen species (ROS) lead to lipid peroxidation of PUFAs. Fe2+ promotes ROS production via Fenton reaction, that causes lipid peroxidation. Fenton reaction generates a large number of free radicals, such as the hydroxyl⁃ (OH−), hydroxyl radical (●HO), and lipid hydroperoxides (LOOH), which lead to strong oxidative stress.12,13 Lipid hydroperoxides increasing result from the dysmetabolism of intracellular lipid oxides is the essence of ferroptosis. 14 And the many different aldehydes like malondialdehyde (MDA), propanal, hexanal, and 4-hydroxynonenal (4-HNE) can be formed as secondary products during lipid peroxidation. 15 (2) Immunological features of ferroptosis are intracellular components released into damage-associated molecular patterns, such as ATP, high mobility histone B1 (HMGB1) and pro-inflammatory cytokines (e.g. IL-33 and interferon γ (IFNγ)), which promote inflammatory response.16–18
Asthma is a complex chronic and heterogeneous lung disease, whose common clinical symptoms include dyspnea, chest tightness, wheezing, and cough.19,20 A large number of studies suggest that inflammatory response plays a key role in the process of asthma.21–23 In an inflammatory environment, many innate and adaptive immune system cells interact with airway epithelial cells to cause airway structural changes, such as mucous metaplastic, airway hyperresponsiveness (AHR), airway remodeling, and airway inflammation, etc.24–26 Asthma mainly involves type 1 (T1) and type2 (T2) inflammation. T1 inflammation is primarily associated with neutrophilic, and T2 inflammation is primarily associated with eosinophils. 27 Most asthmatic patients present with T2 inflammation, and generally go hand in hand with eosinophilic inflammation, characterized by increased interleukin (IL)-4, IL-5, and IL-13.28,29
At present, the treatment of asthma mainly focuses on targeting T2 inflammation, inhibiting the aggregation of T2 inflammatory cells in the lung or the release of T2 inflammatory mediators, which has therapeutic effect on asthma. Inhaled corticosteroids and long-acting β2-agonist (LABA) are known as the most common pharmacological options for management of asthma, but some asthmatic patients respond poorly to them. 30 Increasingly, studies focus on therapeutic target for asthma. 31 The molecular pathways that can be targeted include T2 cytokines, IgE, thymic stromal lymphopoietin (TSLP), and IL-33 etc.32–34 The latest research shows that ferroptosis may be an emerging target in inflammatory diseases. 10 Authors identified that ferroptosis inhibitor alleviates bronchial epithelial cell injury in asthma.35,36 It is shown that T2 inflammation is closely related to ferroptosis. In ferroptosis, airway epithelial cells release large amounts of proinflammatory cytokines, leading to inflammation. Inhibiting ferroptosis has an important inhibitory effect on T2 inflammation.37,38 Accordingly, ferroptosis is involved in asthma and is an important target for treatment of asthma. 39
Currently, an increasing number of studies suggest that ferroptosis is involved in asthma. 14 The effect of ferroptosis on asthma is mainly achieved by three special cells. In asthma, due to the autophagy of ferritin, a large amount of free iron is produced. Iron catalyzes the formation of highly destructive hydroxyl radicals and ROS through Fenton reaction, resulting in ferroptosis of airway epithelial cells. ROS promotes T2 inflammation by destroying cell membranes and promoting the release of many T2 cytokines by airway epithelial cells. 40 Excessive infiltration of eosinophils is the main manifestation of airway inflammation in asthma. Eosinophils produce arachidonic acid-derived lipid mediators that contribute to the development of ferroptosis in asthma through the lipid peroxidation signaling pathway, leading to airway inflammation, AHR, and mucus overproduction. 41 Studies suggest that the up-regulation of the extracellular matrix protein periostin (POSTN) in smooth muscle cells increases the sensitivity of the cells to ferroptosis. And a ferroptosis-related gene solute carrier family 7 member 11 (SLC7A11) is identified in airway smooth muscle cells (ASMs). Hypertrophy of ASMs is an important cause of airway remodeling and AHR in asthma, but its association with ferroptosis in asthma has been unclear. This review aimed to summarize the ferroptosis in asthma. In addition, modulating ferroptosis signaling pathways may be a new target to design new drugs to treat asthma.
Ferroptosis in asthma
Asthma is closely related with infiltration of inflammatory cells and AHR. Recent studies suggest that elevated oxidative stress has been considered as a driving force behind the inflammation and AHR.30,42 And biomarkers of lipid peroxides are elevated in asthma, which suggested that lipid peroxidation is related to asthma pathophysiology and severity. 43 Simultaneously, large lipid peroxides accumulation is a hallmark of ferroptosis, indicating that ferroptosis may be involved in asthma. 44 A recent study shows that a high iron level in lung cells is associated with asthma exacerbations. In asthma, high levels of iron cause airway inflammation and hyperoxidative state, leading to ferroptosis.45,46 In addition, exposure to allergens is an important cause of elicitation of symptoms of asthma.47,48 Allergens’ exposure increases the production of oxidants such as ROS and reactive nitrogen species, which contributes to the imbalance between oxidants and antioxidants, leading to oxidative stress.49,50 And oxidative stress further activates pro-inflammatory genes such as nuclear factor κB (NF-κB) and p53, leading to release of a large number of proinflammatory cytokines. 51 It is reported that apoptosis of bronchial epithelial cells in asthmatic patients, and ROS in peripheral blood eosinophils of asthmatic patients is significantly higher than that of healthy people. ROS plays an important role in asthma by activating inflammatory signals.52–55 In addition, severe allergic and eosinophilic asthma are both driven by T2 inflammation. Both are the cardinal cell type associated with T2 asthma. 56 In recent years, accumulating studies suggest that ferroptosis regulates oxidative stress and inflammatory responses of asthma. Inhibition of ferroptosis contributes to reduce airway inflammation.57,58 There is a study revealing that lipid peroxidation and ROS levels were elevated in asthma, indicating that ferroptosis was activated during the development of asthma. 59 In addition, IL-6 promotes ferroptosis in bronchial epithelial cells and accelerates the progression of allergic asthma by inducing ROS-dependent lipid peroxidation and destroying iron homeostasis. 58 Accordingly, ferroptosis in involved in asthma.
Ferroptosis signaling pathways in asthma
An increasing number of studies suggest that a variety of signaling pathways related to ferroptosis play an important role in asthma, such as iron metabolism, lipid metabolism, and ROS production, amino acid metabolism and other potential signaling pathways.60,61
Iron metabolism
As the essential trace element, iron mainly exists in the form of bivalent and trivalent iron ions in the body and participates in cell transport and metabolism. 10 Iron metabolism is a dynamic process. 62 Normally, Fe2+ in vivo is oxidized to ferric ion (Fe3+) with the help of ceruloplasmin, and is transported intracellularly in a complex formed with transferrin (TF) for a series of biochemical reactions. 63 In cell, Fe3+ is reduced to Fe2+ under the action of ferric reductase (prostate six transmembrane protein 3). Furthermore, Fe2+ is stored in the ferritin in the cytoplasm or pumped out of cells by the iron transporter on the membrane, participating in iron recycling and maintaining iron homeostasis. 64 Iron metabolism is important for the ferroptosis now that it is closely related with the accumulation of lipid peroxides. 14 Iron metabolism plays an important role in ferroptosis by promoting the release of ROS and causing tissue damage through its effects on lipids, proteins, and even DNA. 65
On the one hand, iron catalyzes the formation of highly destructive hydroxyl radicals and ROS through Fenton reaction. 62 Redox-active iron is a catalyst that generates hydroxyl free radicals through Fenton reaction and directly catalyzes lipid peroxides, triggering a strong oxidative stress, generating a large number of ROS and then inducing ferroptosis.66,67 On the other hand, as a cofactor, iron enhances the activity of various metabolic enzymes (such as lipoxygenase (LOX) proline dehydrogenase 1 (PDH1)) in the process of lipoxygenase-mediated peroxidation and promotes the generation of lipid ROS. 68 Fe2+ promotes the decomposition of phospholipid hydroperoxide to alkoxyl phospholipid radical, which by attacking PUFAs promotes lipid peroxidation. 69 Studies suggest that TF, ferritins, ferroportin (FPN) exert crucial functions in the maintenance of systemic iron homeostasis.70,71 Nuclear receptor co-activator 4 (NCOA4) is a unique transport receptor for ferritinophagy, which delivers ferritin to lysosomes for degradation to release free iron. 72 Increased levels of TF and transferrin receptor 1 (TFR1) promote ferroptosis in cells. 73 New research suggests that the heat shock protein β-1 (HSPB1) reduces intracellular iron content by inhibiting the expression of TFR1, thereby inhibiting ferroptosis. 63
Studies show that asthma is associated with increased cellular iron and ferritin levels in airway epithelial cells, possibly due to increased ROS production.38,74 Metal chelators closely bind redox-active metals like iron and copper, preventing redox-active metals from participating in Fenton reactions, thus reducing oxidative damage and even the production of pro-inflammatory cytokines.75–77 Studies suggest that metal chelators relieve clinical symptoms of asthma, confirming that changes in iron levels and iron-related gene expression in the airway are related to lung function of patients with asthma.78,79 In addition, a high iron level in lung cells is associated with asthma exacerbations. 43 It is reported that elevated iron levels induce an increase of pro-inflammatory cytokines in human ASMs and fibroblasts in vitro, and contribute to the appearance of key asthma characteristics, such as AHR, T2 inflammation. 80 Moreover, pseudomonas aeruginosa leads to the accumulation of phospholipid peroxide in human bronchial epithelial cells via enhancing expression of 15-lipoxygenases (15-LOXs), which in turn triggers ferroptosis of the epithelial cells.81,82 Accordingly, iron accumulation in the lungs contributes to the asthma (Figure 1).

A flow-through of iron metabolism in a healthy versus inflamed/asthmatic cell.
Lipid metabolism and ROS
Intracellular lipid peroxidation is divided into two types, enzymatic lipid peroxidation and non-enzymatic lipid peroxidation. 83 Enzymatic lipid peroxidation refers to the generation of various lipid hydroperoxides under the catalysis of lipoxygenases (LOXs). 84 Non-enzymatic lipid peroxidation, also known as lipid autoxidation, is a chain reaction mediated by free radicals. 14 In non-enzymatic autooxidations, Fe2+ reacts with hydrogen peroxide (H2O2), generating Fe3+ and a hydroxyl radical. 85 The hydroxyl radical initiates the chain oxidation of polyunsaturated phospholipids, thus leading to impairment of membrane function. 86 At the molecular level, lipid peroxides are further broken down, which consume nucleic acids and proteins, leading to cell ferroptosis. 87 Lipid peroxidation is an important process leading to ferroptosis.88,89 In ferroptosis, lipid peroxidation leads to oxidative degradation of the two important biological membrane components, PUFAs and phosphatidylethanolamines (PE). PUFAs are the components of lipid bilayer of cytomembranes and are also the molecular basis of the fluidity and deformability of the cytomembrane. 90 However, PUFAs have a high affinity with hydroxyl radicals and the hydrogen atoms between its double bonds are easily oxidized by radicals to generate lipid peroxides.91–93 By changing the molecular configuration of PUFAs, lipid peroxidation destroys the fluidity and stability of cell membrane structure, increasing the permeability of cell membrane, subsequently. These changes make the cells vulnerable to rupture and death. 14 However, the affinity between PE and free radicals is low. Before the occurrence of lipid peroxidation, PE needs to bind to two fatty acyl-arachidacyl-adrenoyl groups (AA) and adrenoyl (AdA) to form oxidation site PE-AA/AdA. This process requires the participation of acyl-CoA synthetase long-chain family member 4 (ACSL4) and lysophosphatidylcholine acyltransferase 3 (LPCAT3). 94 Subsequently, PE-AA/AdA is easily oxidized by Arachidonate 15-Lipoxygenase (ALOX15) to form PE-AA/AdA-OOH with cytotoxicity, thus promoting ferroptosis. 83 It is showed that arachidonic acid-derived lipid mediators are mainly produced by eosinophils and mast cells while excessive infiltration of eosinophils and mast cells is the main manifestation of asthmatic airway inflammation. These lipid mediators are further responsible for activating pro-inflammatory molecules as well as broncho-constriction of airways through lipid peroxidation signaling pathway of ferroptosis. 95
Although ROS is essential for the immune defense system against inflammatory stimuli, excess ROS may lead to lipid peroxidation. 96 Lipid ROS is formed by enzymatic or non-enzymatic oxidation of PUFAs on lipid membranes. 94 In addition, allergic asthma is often accompanied by increased ROS production and decreased antioxidant system capacity. 97 In asthma, oxidative stress is generated due to the imbalance between excessive ROS generation and reduced antioxidant defense mechanism. 98 It is reported that increased ROS promotes tissue damage in asthmatic airways. 99 In asthma, excessive ROS induces ferroptosis, leading to direct oxidative damage and cell abscission of bronchial epithelial cells in asthma. ROS increases lipid peroxidation and the permeability of the alveolar epithelial cells by destroying the cell membrane and promoting the release of proinflammatory cytokines from epithelial cells and alveolar macrophages, which increases the AHR.100,101 In addition, ROS activates epithelial cells and releases cytokines such as IL-25, IL-33, and TSLP. These cytokines promote the production of T2 cytokines from Th2 cells and ILC2s through the activation of dendritic cells (DCs), promoting T2 inflammation. 37 Furthermore, ROS activates transcription factors such as NF-κB and AP-1, which promote the release of IL-6, IL-8, and TNF-α, thus activating the T2 inflammation and resulting in impaired airway epithelium and capillary endothelial barrier function. 102 Accordingly, ferroptosis is involved in the occurrence and development of asthma through lipid metabolism and ROS signaling pathway (Figure 2).
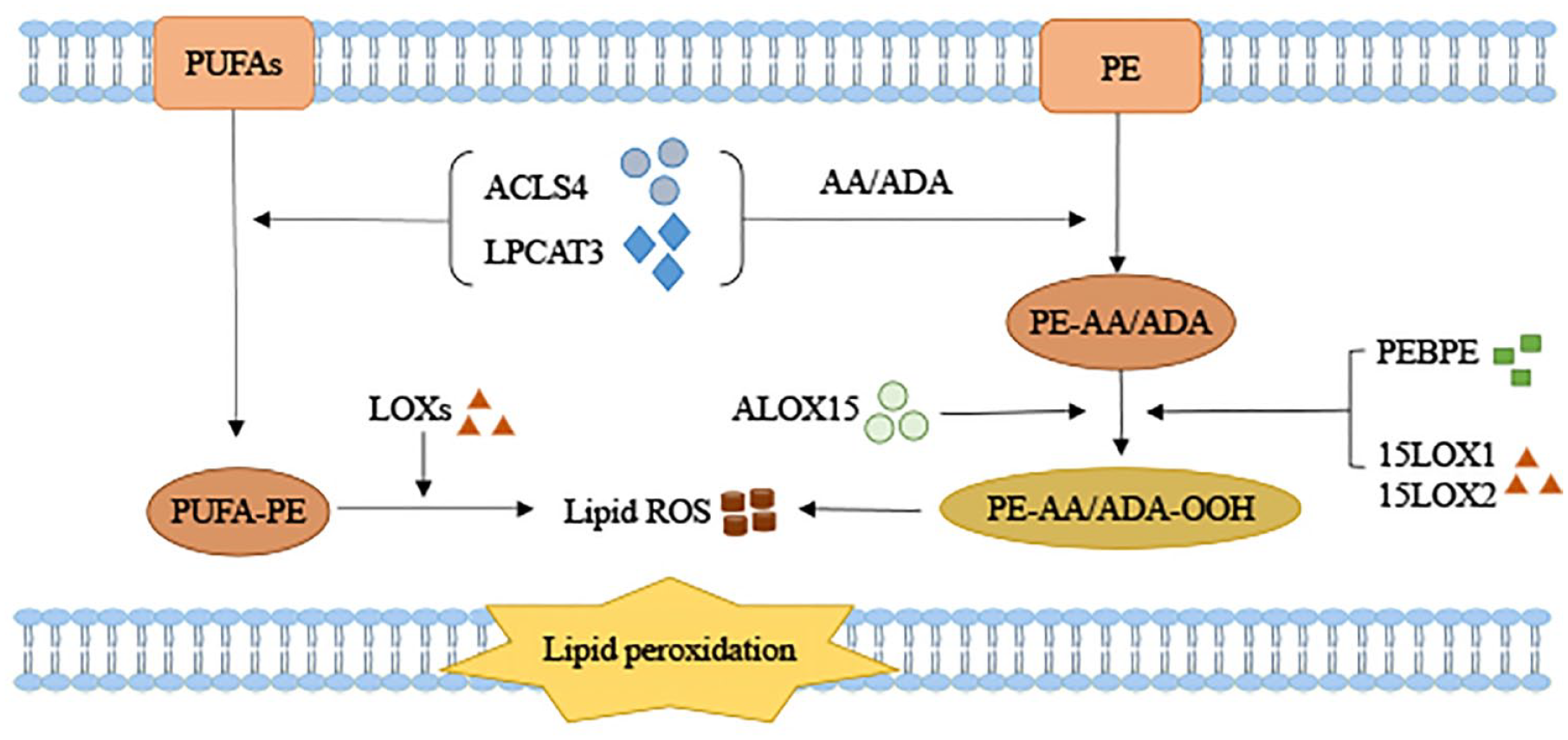
The reactive pathways between lipid peroxides, PUFAs, and PE degradation.
Amino acid metabolism
Glutathione (GSH) is the most abundant antioxidant in the airway epithelial lining fluid and is involved in various important functions relevant to asthma. 103 It is suggested that the airway GSH/glutathione oxidized (GSSG) redox balance is unbalanced in asthma. 104 Ferroptosis caused by abnormal metabolism of amino acids is mainly related to abnormal metabolism of GSH. 14 Reduced GSH is an essential antioxidant in human body. 8 Cysteine is a key substrate for GSH to protect cells from oxidative damage. Cysteine can be further converted to reduced GSH by ATP-dependent cytoplasmic enzymes glutamate-cysteine ligase and glutathione synthase. 105 Glutathione peroxidase 4 (GPX4) is a phospholipid hydroperoxide glutathione peroxidase, which increases the activity of peroxidase, and then reduces the toxicity of lipid peroxides, thus maintaining the stability of membrane lipid bilayer.106,107 With the help of GPX4, GSH reduces H2O2 to H2O, scavenge free radicals, and maintain the equilibrium state of intracellular free radical content. Moreover, as a cofactor of GPX4, GSH participates in the reducing of lipid peroxides LOOH and converts the LOOH into non-toxic lipid alcohols (L-OH) to prevent ferroptosis.10,108 In this process, GSH works as a reducing agent and GPX4 works as a key enzyme to mediate the reducing of lipid peroxides, which negatively regulates ferroptosis. 109 Accordingly, GPX4 plays an important role in ferroptosis.
Cysteine, one of the major components of the synthesis of GSH, enters cells in two main ways. One of the ways depends on system Xc- and the other depends on the sulfur-transfer pathway.14,105 System Xc- is a cysteine/glutamate reversal transporter consisting of two subunits, which is consisted of SLC7A11 and the solute carrier family 3 member 2 (SLC3A2). System Xc- mediates the exchange of cysteine and glutamate across the membrane in a 1:1 ratio. 110 Cystine is reduced to cysteine in the body, which facilitates the production of GSH and then uses GSH to consume excess hydroxyl peroxides. When systemic Xc- function is damaged, the exchange between glutamate and cystine is impaired and the cystine content in cells is reduced, leading to insufficient cysteine synthesis in cells. This subsequently affects the synthesis of GSH, and then leads to the decrease of GSH peroxidase activity. These changes result in the decrease of antioxidant capacity of cells and the accumulation of lipid activity, ultimately leading to oxidative damage and ferroptosis.111,112 The sulfur-transfer pathway is important for the synthesis of cysteine through transferring a sulfur atom from methionine to serine, and upregulating genes related to the sulfur-transfer pathway can inhibit intracellular iron migration, leading to iron metabolism disorders. 14 Therefore, impairing these two pathways may promote the ferroptosis. Studies suggest a variety of drugs could induce ferroptosis by targeting the system Xc-, GSH, and GPX4, such as erastin and its analogs, 113 sulfanilamide pyridine, glutamate, and sorafenib, etc. 13 Also, study suggests that plasma cysteine levels were significantly decreased in the asthma compared with healthy controls. In summary, loss of extracellular cysteine, damage of system Xc-, reduction of intracellular glutathione and inactivation of GPX4 promote ferroptosis through amino acid metabolism. 9 It is found that phospholipid peroxide generated by 15 lipoxygenase 1 (15LO1) inhibits SLC7A11, thereby reducing intracellular GSH, promoting ferroptosis and T2 inflammatory protein expression. 114 Therefore, amino acid metabolism plays an important role in ferroptosis in asthma (Figure 3).

Amino acid metabolism for the synthesis of GSH in healthy and inflamed/asthmatic cells.
Asthma and regulators of ferroptosis
Glucocorticoids and bronchodilators are current treatments for asthma. These treatments reduce symptoms, but do not cure asthma. 91 At present, there is no more effective treatment in clinical practice than these two. Accordingly, many studies focus on finding new therapeutic targets to break through the dilemma of asthma treatment. A growing number of studies suggest that regulators of ferroptosis may be new targets for asthma. 115 There are many common factors that regulate ferroptosis including nuclear factor erythroid 2-related factor 2 (Nrf2), heme oxygenase-1 (HO-1), mevalonate pathway, ferroptosis inhibitor protein 1 (FSP1), PE-binding protein-1 (PEBPE1), and vitamin E etc. Increasing number of studies confirm the role of these factors in the pathogenesis of asthma.115–117
Nuclear factor erythroid 2-related factor 2
The Nrf2 is an important endogenous antioxidant that regulates cellular defense system and antioxidant response, and is also the main ferroptosis signal molecule in the nucleus.54,118 Under the unstressed conditions, Nrf2 binds to Kelch-like ECH-associated protein 1 (Keap1) and remains inactivated in the proteasome through ubiquitination and degradation. 119 Nevertheless, oxidative stress impairs the ability of Keap1 to target Nrf2 for ubiquitination and degradation, and induces newly synthesized Nrf2 to translocate to the nucleus to bind with transcriptional activation of the antioxidant response element (ARE) gene. 120 Nrf2, which is associated with ARE, is beneficial in asthma, acting through anti-inflammatory mechanisms. 121 It is confirmed that Nrf2 inhibits the expression of T2 cytokines in airway epithelial cells and ASMs, thereby inhibits the oxidative effects and reduces the severity of asthma. 122 Nrf2 increases the antioxidant genes, including HO-1, GPX4, which serves an important role in maintaining redox balance in asthma. 103 It is reported that Nrf2-Keap1 signaling pathway promotes the activation of system Xc-, the expression of GPX4, and regulates the activity of SLC7A11,123,124 to reduce the production of ROS, which ultimately inhibits ferroptosis. 125 It is suggested that Nrf2 activator ameliorates airway inflammation and oxidative stress in asthmatic mice.126,127 Therefore, Nrf2 inhibits the progression of asthma by negatively regulating ferroptosis. 128 A new study suggests that 18β-GA, the main active component of licorice, has a protective effect on airway allergic inflammation by increasing the expression of Nrf2, which may provide a new treatment for asthma. 129
Heme oxygenase-1
HO-1 presents anti-cancer, anti-inflammatory, anti-apoptotic, anti-proliferative, and antioxidant effects. 130 It is suggested that HO-1 expression is elevated in the airway of asthma.97,131 In addition, HO-1 effectively reduces airway inflammation and mucus secretion in asthmatic patients. 49 However, HO-1 is overactivated under the condition of enhanced oxidants. Overactivation of HO-1 leads to large amounts of iron ions and ROS accumulating, which increases the lipid peroxidation, and then leads to the ferroptosis. 132 Therefore, HO-1 plays an important role in the ferroptosis. Clinically, the HO-1 inhibitor Zinc protoporphyrin IX (ZnPP) prevents Erastin-induced ferroptosis. 133 Meanwhile, the administration of formononetin (FMT) which is a bioactive isoflavone obtained from Chinese herb Radix Astragali attenuate airway inflammation and oxidative stress in asthma, and significantly improve lung function, by significantly elevating the expression of HO-1. 30
Mevalonate pathway
Two basic products of mevalonate pathway, namely isopentenyl pyrophosphate (IPP) and antioxidant coenzyme Q10 (CoQ10), are both involved in the regulation of ferroptosis. 134 IPP is a chemical substance produced by decarboxylation after the activation of mevalonate, which biosynthesizes lipophilic free radicals to capture the CoQ10. 14 Inhibition of CoQ10 activity reduces the antioxidant capacity of cells and increases lipid peroxides, thus inducing ferroptosis. 135 In addition, CoQ10 inhibits allergic inflammatory response, oxidative stress, and the production of T2 cytokine by increasing the expression of Nrf2. 136 Therefore, mevalonate pathway plays an important role in the progression of asthma by up-regulating Nrf2 and inhibiting ferroptosis.
Ferroptosis inhibitor protein 1
Ferroptosis inhibitor protein 1 (FSP1), also known as AIFM2 or AMID, is a key component of a non-mitochondrial CoQ antioxidant system and acts as an inhibitor of ferroptosis in vitro and vivo. 81 FSP1 plays a role as an oxidoreductase on the plasma membrane, which catalyzes the regeneration of CoQ10 using NAD(P)H. 137 As a lipophilic radical-trapping antioxidant, CoQ10 halts the accumulation of lipid peroxides by capturing antioxidant (RTA). 138 It is reported that CoQ-FSP1, a key downstream effector of KEAP1-NRF2 pathway, suppresses ferroptosis. 139 Thus, the FSP1-CoQ10-NAD(P)H pathway works in parallel with the typical glutathione-dependent GPX4 pathway, inhibiting lipid peroxidation, and then inhibits ferroptosis. 140 However, the mechanism of FSP1 involved in asthma is still unclear.
PE-binding protein-1
PEBP1 is a scaffold protein inhibitor of protein kinase cascade. It is reported that the number of PEBP1/15LO-1 in the airway epithelial cells have a strong correlation with increased FeNO in T2 asthma patients. 141 PEBP1/15LO1-driven AA-PE oxidation is important in the T2 airway inflammation associated with asthma.53,142 Under high IL-13 conditions, 15LO1 binds to PEBP1 to oxygenate PE on the membrane, thereby damaging the membrane and driving ferroptosis.53,140 It is confirmed that 15LO1 interacting with PEBP1 enhances critical inflammatory pathways of asthma. 143 Furthermore, ALOX15 is induced of T2 cytokines, which supports the release of chemokines to recruit activated T cells in lung epithelial cells, promoting inflammation in asthma. 144 PEBP1 interacting with ALOX15 enhances the level of inflammation in airway epithelial cells. 145 Accordingly, PEBP1, as a major regulator of ferroptosis, plays an important role in asthma.
Vitamin E
As a lipophilic antioxidant, Vitamin E inhibits NF-κB pathway and restores Nrf2, further reducing oxidative stress, inflammation, and cell death. 146 It is showed that vitamin E can relieve asthma. 147 And vitamin E could reduce or terminate the damaging chain reaction caused by ROS to alleviate ferroptosis, thus preventing the associated cell membrane damage, and assisting in the maintenance of membrane integrity.148,149 Other studies showed that vitamin E could act synergistically with GPX4 to prevent lipid peroxidation. 150 Vitamin E reduces PGE2 production by inhibiting enzymatic activity of cyclooxygenase 2 (COX-2) and exerts indirect regulatory effects on T cells.151,152 Therefore, vitamin E may inhibit ferroptosis by reducing lipid peroxidation, thereby reducing asthma-related symptoms.
Current observations of ferroptosis in specific cell types in asthma
Airway epithelial cells
More and more studies suggest that ferroptosis of airway epithelial cells plays an important role in asthma pathogenesis.38,143 On the one hand, the increase of iron content causes the Fenton reaction in the airway epithelial cells and induces the occurrence of ferroptosis. 153 On the other hand, ROS activates airway epithelial cells, releasing many pro-inflammatory factors (IL-6, IL-8, IL33, IL25, and TSLP), and chemokines (CCL5, CCL7, and CCL22).154,155 These cytokines promote the production of T2 cytokines from Th2 cells and ILC2s through the activation of DCs, promoting T2 inflammation. 37 Studies show that IL-6 promotes lipid peroxidation, disrupts iron homeostasis and induces ferroptosis in cultured human bronchial epithelial cells. 58 The 15-LO1/PEBP1 complex activates lipid peroxidation, resulting in ferroptosis of the airway epithelial cells in asthmatic patients.53,156 Ferroptosis of airway epithelial cells result in the destruction of the airway barrier, which induces airway remodeling and AHR, and stimulates the differentiation of airway epithelial cells into goblet cells, promoting the secretion of mucus.38,157
Airway smooth muscle cells
ASMs secrete a variety of inflammatory mediators such as growth factors, T1 and T2 cytokines, peptides (bradykinin), and chemokines, which are involved in the occurrence of asthma inflammation. 158 Hypertrophy of ASMs is an important cause of airway remodeling and AHR in asthma. 159 POSTN is considered to be a marker of high Th2 asthma. Studies suggest that up-regulation of POSTN in smooth muscle cells increase cell sensitivity to ferroptosis.156,160 And a ferroptosis related gene, SLC7A11, was identified in ASMs. 161 In conclusion, ferroptosis of ASMs may occur and play a role in asthma, but relevant studies are relatively lacking at present.
Eosinophils
Eosinophils play an important role in the pathophysiology of asthma. 56 Upon recognizing allergens, T2 cytokines (IL-4, IL-5, IL-9, and IL-13) lead to the accumulation of high numbers of eosinophils in the airway wall.154,162 Over-production of ROS resulting from infiltrating eosinophils lead to development of oxidative stress in asthma, promoting airway inflammation, AHR, and mucus overproduction. 163 Affected by pulmonary inflammatory factors, the half-life of eosinophils is significantly prolonged, and the cells are continuously activated, which leads to the aggravation of asthma inflammatory response.164,165 Studies have found that eosinophils have higher levels of iron ions than other inflammatory cells and are more sensitive to the stimulation of ferroptosis. 166 New research suggested that ferroptosis inducers relieve asthma symptoms by inducing cytoplasmic ROS-mediated iron-dependent ferroptosis in eosinophils. 163
Summary and prospect
Asthma is a chronic inflammatory disease with clinical manifestations about mucous metaplastic, including goblet cell hyperplasia and excess mucous production, AHR, airway remodeling, and inflammatory cell infiltration. Ferroptosis is involved in asthma. Iron metabolism disorders, lipid peroxide accumulation, amino acid metabolism disorders lead to the occurrence of ferroptosis, and then promote the occurrence and development of asthma. Also, other regulators, such as Nrf2, HO-1, mevalonate pathway, FSP1, and PEBP1, are crucial to ferroptosis in asthma. Accordingly, ferroptosis and its potential regulatory signaling pathways not only provide new insights into the pathogenesis of asthma, but also provide a traceable target for the disease. However, the specific mechanism of ferroptosis in asthma remains unclear. It is necessary to explore the mechanism of ferroptosis in asthma and strive to find effective ferroptosis inhibitors to direct the treatment of asthma.
