Abstract
Background:
Arterial lactate is a recognized biomarker associated with death in critically ill patients. The prognostic role of arterial lactate in acute respiratory failure due to the novel coronavirus disease 2019 (COVID-19) is unclear.
Objectives:
We aimed to investigate the prognostic role of arterial lactate levels at admission in patients with COVID-19-related acute respiratory failure.
Design and Methods:
Cohorts of consecutive patients admitted to nonintensive care units (ICU) at study centers for COVID-19-related respiratory failure were merged into a collaborative database. The prognostic role of lactate levels at admission was assessed for continuous values and values ⩾2.0 mmol/l, and lactate clearance at 24 h through delta-lactate (ΔLac). The study outcome was 30-day in-hospital death. Cox proportional regression model was used to assess independent predictors of the study outcome.
Results:
At admission, 14.6% of patients had lactate levels ⩾2 mmol/l. In-hospital death at 30 days occurred in 57 out of 206 patients; 22.3% and 56.7% with normal or ⩾ 2 mmol/l lactate at admission, respectively. The median lactate level was 1.0 [interquartile range (IQR) 0.8–1.3] mmol/l and 1.3 (IQR 1.0–2.1) mmol/l in survivors and nonsurvivors, respectively (p-value < 0.001). After adjusting for age, relevant comorbidities, acidemia, and the severity of respiratory failure, lactate ⩾2.0 mmol/l was associated with in-hospital death (HR 2.53, 95% CI 1.29–4.95, p-value 0.0066), while Δ Lac ⩾0 was not (HR 1.37, 95% CI 0.42–4.49). These results were confirmed in patients with a pO2/FiO2-ratio (P/F ratio) ⩽300 mmHg.
Conclusions:
In our study, increased arterial lactate at admission was independently associated with in-hospital death at 30 days in non-ICU patients with acute respiratory failure related to COVID-19.
Introduction
The novel coronavirus disease 2019 (COVID-19) had dramatic consequences on global health worldwide. 1 Clinical presentation ranges from asymptomatic (about 33%) to severe (14%) or critical disease (5%) in infected patients.2,3 Progression from mild to severe disease is mainly due to rapidly developing acute respiratory distress syndrome (ARDS). In patients with critical disease, the observed case fatality rate is as high as about 49%. 2
Several demographic features, laboratory abnormalities, and procedures have been associated with disease severity and clinical outcome.4–9 Mortality in ARDS has been mainly related to the severity of hypoxia.10,11 In this setting, the role of lactate levels as a severity marker is unclear. However, lactate is rapidly available at blood gas analysis and is a marker of severity in several clinical conditions in the emergency department (ED) and intensive care unit (ICU), like major trauma or different type of shock.12–22 Historically, lactate was considered a dead-end waste product of anaerobic metabolism triggered by inadequate oxygen delivery and tissue hypoxia 23 ; more recently, impaired oxygen extraction, peripheral shunting, stress, and increased adrenergic stimulation have been recognized as triggers for anaerobic metabolism and increased lactate levels.18,22,24
For community-acquired pneumonia, lactate is already known as a parameter associated with the severe course of the disease and mortality.25–27 However, COVID-19 pneumonia has different pathophysiology, probably due to the viral etiology, and the prognostic role of lactate in this setting remains uncertain.
An association between increased lactate levels and adverse outcomes has been suggested in studies of COVID-19 patients admitted to ICU.28–30 Nevertheless, the association between lactate levels and mortality in COVID-19 patients in the non-ICU setting has been explored in a few studies.31,32 Prompt identification of COVID-19 patients likely to deteriorate by a simple test such as lactate assessment would be crucial for tailoring patient management.
Our study aims at investigating the association between arterial lactate levels and death in patients with respiratory failure related to COVID-19 pneumonia admitted outside the ICU setting.
Methods
Study design, setting, and selection of participants
We considered for inclusion in the study consecutive adult patients admitted due to COVID-19 pneumonia at the Department of Internal and Cardiovascular Medicine – COVID-19 of the University of Perugia from February to May 2021 and at the Department of Medicine at the University of Pisa from March to April 2020. Patients were included in the study if the following criteria were fulfilled: (1) diagnosis of COVID-19 confirmed by real time-polymerase chain reaction testing and (2) pneumonia detected by imaging techniques (chest X-ray, computed tomography, or lung ultrasonography). Exclusion criteria were lack of serum lactate measurement at admission and pregnancy.
Outcomes
The study outcome was in-hospital death at 30 days.
Measurements
The following data were collected for each patient: age, sex, comorbidities (congestive heart failure, history of chronic obstructive pulmonary disease, cancer, systemic arterial hypertension, obesity, chronic kidney disease, chronic liver disease, and diabetes mellitus), use of supplemental oxygen therapy, and noninvasive respiratory strategies (noninvasive mechanical ventilation or continuous positive airway pressure).
Data from arterial blood gas (ABG) analysis, including pH, lactate, pO2, pCO2, and FiO2, were recorded. To account for the severity of respiratory failure at admission, the pO2/FiO2-ratio (P/F) was calculated.
According to previous literature, lactate was considered increased if ⩾2.0 mmol/l. 33
Analysis
Descriptive statistics were used to summarize demographic characteristics, ABG parameters, and respiratory indexes at admission. Categorical variables were reported as frequencies, while numerical variables were given as mean ± standard deviation if normally distributed or median and interquartile range (IQR) if non-normally distributed. Differences in continuous variables were tested by independent t-test or the Kruskal–Wallis test as appropriate, and differences in categorical characteristics by the χ2 test or Fisher exact test.
The association between lactate at admission as continuous values or as normal/increased according to a cutoff ⩾2.0 mmol/l and death were assessed 10 ; an analysis was also performed for lactate with a cutoff level ⩾4.0 mmol/l.16,33 To analyze the association between lactate clearance and death, delta-lactate (Δ Lac) at 24 h was calculated. Δ Lac was defined as the ratio of the difference between serum lactate at admission and on the following day, divided by the first lactate, all by 100. 12 According to this formula, positive values indicate a decrease in serum lactate, while negative values indicate an increase in serum lactate. The P/F was used to express the severity of respiratory failure, categorized into four stages: >300, 300–200, 200–100, and <100 mmHg. 34
Univariate and multivariate logistic regression was used to assess factors associated with increased lactate at admission. Cox proportional regression model was used to evaluate factors independently associated with in-hospital death at 30 days. Multivariable analyses were constructed from univariable variables having p <0.10 at entry. Lactate levels were added to multivariable models as variables of clinical interest. pH was added to multivariable models to assess the independency of the prognostic role of lactate from acidemia. Survival analysis for in-hospital death at 30 days was presented by Kaplan–Meier curves with the log-rank test and by Cox proportional hazard model. Subgroup analyses for patients with P/F ⩽300 mmHg, or with PaCO2 ⩽ 45 mmHg, or with pH ⩽ 7.45 at admission were performed.
We assessed the discriminatory ability for lactate levels at admission by calculating the area under the curve (AUC), with AUC = 0.50 for prediction no better than chance and AUC = 1.00 for perfect discriminatory ability. Sensitivity, specificity, and positive and negative predictive values were also calculated.
With an expected incidence of in-hospital death of 20% (consistent with contemporary literature) and 40% in the elevated lactate group, a two-sided test at a significance level of 0.05 and a power of 90%, we estimated that about 200 patients with COVID-19-related respiratory failure were required to assess the prognostic role of lactate levels after adjusting for age and the severity of respiratory failure. 35
All tests were two-sided, and a p-value of <0.05 was considered statistically significant.
All analyses were performed in R version 3.6.2 (R Foundation for Statistical Computing).
The study was conducted according to the amended Declaration of Helsinki. It was approved by the local ethics committee (Comitato Etico Regionale dell’Umbria, approval number CRU Unipg 20-05 SARS-CoV-2). Standard written informed consent at hospital admission has been obtained from all subjects.
Results
Characteristics of study subjects
During the study period, 383 patients with SARS-CoV-2-related respiratory failure were admitted to the study centers. After excluding 177 patients due to the lack of arterial lactate measurement at admission, 206 patients were included in the study (Figure 1). Table 1 shows the main patients’ features and ABG measures at admission according to vital status at 30 days. The median age was 71 (IQR 57–81) years. The most prevalent comorbidity was systemic arterial hypertension (54.4%).

Flowchart of patient selection.
Main characteristics or measures of study patients at admission according to in-hospital death at 30 days.

IQR, interquartile range; P/F, P/F ratio; RI, respiratory index; ROX, respiratory rate-oxygenation index; STPF, standardize P/F ratio.
History of . . . .
During hospitalization, 47.1% of patients required noninvasive mechanical respiratory strategies. Severe ARDS (P/F < 100 mmHg) at admission was present in 7.8% of patients and was more common in patients who died during hospital stay (19.6%).
Main results
At admission, 14.6% of patients had increased lactate levels ⩾2.0 mmol/l. Patients’ characteristics and ABG parameters associated with increased lactate levels at admission are reported in e-Tables 1 and 2. Age and P/F were independently associated with elevated lactate levels. In addition to P/F, standardized P/F, respiratory index, and respiratory rate-oxygenation index were inversely correlated with elevated lactate levels at univariate analysis. Lactate assessment on the second day from admission was available for 89 patients, in which Δ Lac was calculated. In 52 patients, lactate levels decreased, while in 37 patients, lactate levels increased.
In-hospital death at 30 days occurred in 57 patients (27.7%) (Table 1). Death occurred in 22.3% and 56.7% of patients with normal and increased lactate at admission, respectively, and these data accounted for a 56.7% positive predictive value and a 77.3% negative predictive value. The median lactate level was 1.0 (IQR 0.8–1.3) mmol/l in survivors at 30 days and 1.3 (IQR 1.0–2.1) mmol/l in patients who died during hospital stay (p-value < 0.001). In particular, 17 out of 57 (29.8%) and 13 out of 149 (8.7%) patients who died and who survived respectively had increased lactate levels at admission (sensitivity 29.8%, specificity 91.3%). Of note, all patients with lactate ⩾4 mmol/l at admission died during the hospital stay. e-Table 3 shows factors associated with death at 30 days at univariate analysis. The risk for death associated with lactate and pH values at admission is shown in Figure 2 and e-Figure 1. At multivariate analysis, lactate levels ⩾2 or ⩾4 mmol/l at admission were independently associated with in-hospital death at 30 days (HR 2.53, 95% CI 1.29–4.95, p-value 0.0066; HR 8.10, 95% CI 1.88–34.87, p-value 0.0050, respectively), as it was the case for P/F <100, chronic obstructive pulmonary disease and age (Table 2; Figure 3; and e-Figure 2). Adjusted survival curves based on Cox proportional hazard model are shown in e-Figure 3. Lactate increase at 24 h was not associated with death (HR 1.37, 95% CI 0.42–4.49, p-value 0.6045).

Risk of death at 30 days associated with arterial lactate level at admission.
Risk factor for in-hospital death at 30 days (Cox proportional hazard model–multivariate).

CI, confidence interval; HR, hazard ratio; P/F, P/F ratio.

Kaplan–Meier survival curves based on cutoff lactate level of 2 mmol/l for 30-days in-hospital death.
Of note, the AUC for lactate levels at admission to predict 30-day in-hospital death was 0.69 (95% CI 0.61–0.77) (Figure 4).
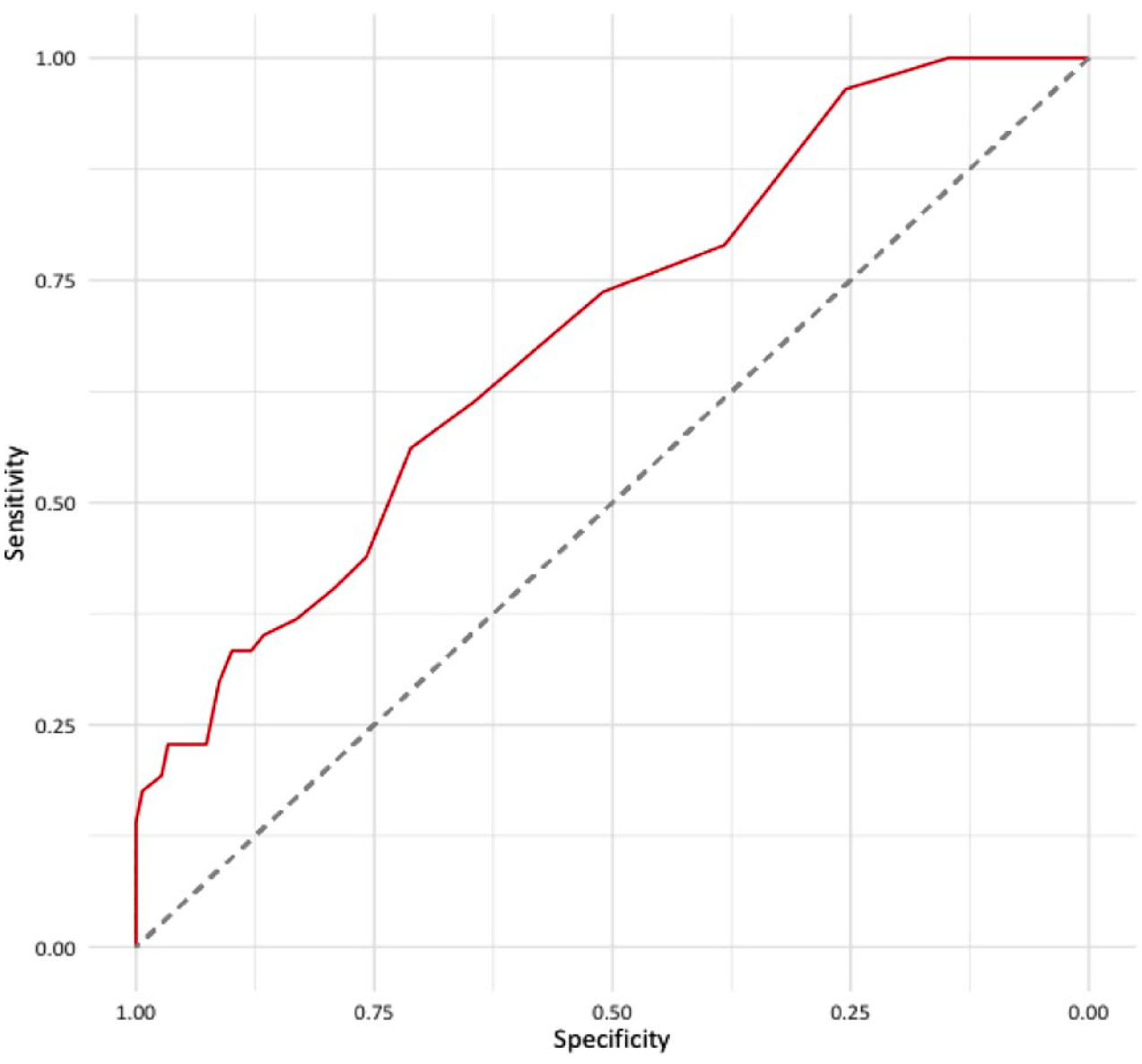
Discriminatory ability of lactate levels at admission for in-hospital death at 30 days.
The association between lactate ⩾2 or ⩾4 mmol/l at admission and in-hospital death at 30 days was confirmed even in patients having a P/F ratio ⩽300 mmHg and in patients with pCO2 ⩽45 mmHg at admission (e-Tables 4 and 5). A positive nonsignificant association was found in the subgroup of patients with pH ⩽7.45, but the sample size was small (85 patients) (e-Table 6).
Discussion
Our study shows that in non-ICU patients admitted for COVID-19-related respiratory failure, elevated lactate levels at admission are independently associated with in-hospital death at 30 days. The association is confirmed in patients with P/F ⩽300 mmHg at admission. Lactate clearance, considered as a variation of lactate levels at 24 h, was not significantly associated with in-hospital death.
Lactate is a reversible product of glucose metabolism generated in anaerobic metabolism, and it is associated with the metabolic response to stress. Hyperlactatemia may originate from several causes: tissue hypoxia or deficit in oxygen delivery, impaired oxygen extraction, peripheral shunting, stress, and increased adrenergic stimulation. 18 Overall, hyperlactatemia should be regarded as an adaptive response to an injury rather than a simple marker of tissue hypoxia. 22 In fact, lactate may play a role as an alternative energy supply in shock patients.22,24,36,37 Overall, high lactate levels may be seen as a stress response with activation of the sympathetic nervous system, increased glycolysis, and a modified bioenergetic supply.18,22,38 The role of lactate as a predictor of death in critically ill patients is established, mainly in sepsis.21,39,40 Updated guidelines from the Surviving Sepsis Campaign recommend assessing lactate and lactate kinetics to manage critically ill COVID-19 patients, with a low level of evidence. 41 Studies in COVID-19 patients admitted to ICU suggested a positive association between hyperlactatemia or lactate clearance and mortality.28–30 Limited data exist on the potential association of arterial or venous lactate levels with mortality in non-ICU COVID-19 patients.31,32 In our study of non-ICU patients, hyperlactatemia at admission was independently associated with in-hospital death at 30 days. Moreover, increased lactate levels were associated with the severity of respiratory failure as defined according to different respiratory indexes. However, in our study lactate clearance at 24 h was not significantly associated with in-hospital death. Whether the lack of association could have been influenced by the timing of assessment (24 h) or whether this analysis would have required a larger sample size is debatable. Indeed, the mean time to death in our study was 10 ± 7 days. With this time lag, the role of lactate clearance at 24 h could be of limited clinical value.
The discriminatory power for lactate levels at admission to predict in-hospital death was modest. However, hyperlactatemia (⩾2 mmol/l) showed a high specificity to predict death. In particular, all patients with lactate ⩾4 mmol/l died at 30 days. Future studies are required to confirm these thresholds and results.
In a retrospective cohort study of critically ill COVID-19 patients treated in the ICU, lower blood pH was a strong prognostic factor for fatal outcomes. 42 In our study, elevated lactate levels were associated with in-hospital death regardless of acidemia or impaired kidney function, a condition linked to acidemia in hyperlactatemia. 18 The increase in lactate levels is a marker of prolonged and severe hypoxia at the tissue level that recognizes a wide range of causes. An increase in lactates may allow emergency physicians to rapidly identify critically ill patients before the compensation mechanisms are exhausted. At various stages of the pandemic, hospital overcrowding resulted in guidelines that suggested postponing access to the ED in favor of home management. These strategies have often proved unsuccessful, while better results in terms of survival may be obtained with early (and possibly noninvasive) treatments. Our data, showing higher mortality in patients with elevated lactate at ED access, confirm this interpretation.
Like other studies, we considered in-hospital death as the study outcome. 11 Death is a hard clinical event, independent of physician judgment. Differently, the decision to admit a patient to the ICU is physician-dependent, and death in the ICU is subjective to other non-COVID-related complications, such as bacterial superinfections. 43
Our study has some limits. First, one of the study cohorts was retrospective. Second, the low proportion of patients with a second assessment at 24 h could have limited the analysis to demonstrate a significant association between Δ Lac and adverse outcome, so further studies with a larger sample size would be required. Moreover, cutoff values for lactate levels of 2 and 4 mmol/l were considered according to previous studies and guidelines in septic patients, while more accurate values could be validated in further studies of COVID-19 patients.
Our study also presents some strengths. In particular, it is among the first to have demonstrated an association between elevated lactate levels and adverse outcomes in non-ICU COVID-19 patients, providing an adequate sample size for the credibility of the results.
Conclusion
In conclusion, our study shows that elevated arterial lactate is independently associated with in-hospital death at 30 days in non-ICU patients admitted for COVID-19-related respiratory failure. Prospective studies are required to confirm the prognostic role of arterial lactate in this particular setting.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231186730 – Supplemental material for Arterial lactate as a risk factor for death in respiratory failure related to coronavirus disease 2019: an observational study
Supplemental material, sj-docx-1-tar-10.1177_17534666231186730 for Arterial lactate as a risk factor for death in respiratory failure related to coronavirus disease 2019: an observational study by Giorgio Maraziti, Laura Marchini, Greta Barbieri, Marco Falcone, Francesco Corradi, Mara Graziani, Lorenzo Ghiadoni and Cecilia Becattini in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We gratefully acknowledge the critical revision of the manuscript by Prof. Stefano Nava, Università Alma Mater Studiorum Bologna, IRCCS Ospedale di Sant’Orsola, Bologna.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
