Abstract
Background:
There is little information on the optimal storage conditions for recovery of nontuberculous Mycobacterium spp. (NTM) from refrigerated sputum.
Objectives:
We investigated the storage duration that could increase the culture-positive rate of NTM isolates.
Design:
In this prospective study, we collected NTM isolates and clinical data from patients with repeated culture-positive NTM pulmonary disease (NTM-PD).
Methods:
From June 2020 to July 2021, the participants were instructed to randomly collect six sputum samples and immediately store them in a refrigerator at 4°C until the date of their clinic visit. At the outpatient visits, expectorated spot sputum samples were collected.
Results:
A total of 226 sputum samples were collected from 35 patients. The median duration of refrigeration was 6 days (maximum duration: 36 days). The overall culture-positive rate was 81.6%. While there was a trend for a higher culture positivity rate when stored for ⩽3 weeks, this was not significant compared with those stored for >3 weeks (p = 0.610). According to sputum microscopy, smear-positive sputum was 100% isolated, but smear-negative samples had a culture-positive rate of 77.5%. Similarly, there was no significant association between sputum storage duration and culture positivity (p = 0.511). In addition, the recovery rate of the refrigerated sputum was comparable with the collected spot expectorated sputum (82.6% versus 80.6%, p = 0.795), which is suggestive of the long-term viability of NTM in refrigerated sputum.
Conclusion:
Our data demonstrated the long-term viability of refrigerated NTM, and the culture positivity rate of these samples was comparable with the spot expectorated sputum. These results suggest that implementing sputum refrigeration would enhance convenience in diagnosing and following patients with NTM-PD.
Plain language summary
Under usual circumstances, most patients with suspected NTM submit spontaneously expectorated sputum rather than induced sputum for the purpose of testing the causative organism. By collecting and storing sputum specimens for a longer period than before, it is expected that more sufficient and adequate collection of sputum specimens will be possible.
Keywords
Introduction
Nontuberculous mycobacteria (NTM) are ubiquitous microorganisms that affect various human organs, of which the lung is the most commonly involved site. 1 Along with the increasing incidence and prevalence of NTM pulmonary disease (NTM-PD) worldwide,2,3 even surpassing that of tuberculosis (TB) in some countries,4,5 there has been robust evidence that NTM-PD is associated with morbidity and mortality. 6 Therefore, timely diagnosis and treatment are essential when managing NTM-PD.
Sputum acid-fast bacilli (AFB) smear and culture are the cornerstones of diagnosing and evaluating treatment response in patients with NTM-PD. 7 For diagnosis of NTM-PD, the same NTM species should be isolated in at least two expectorated sputum samples. However, in the cases of scanty sputum, AFB results are limited in their interpretation, and sputum induction, bronchoscopy, or culture-independent methods may be required to establish the diagnosis.7–9 Although these procedures are widely used in diagnosing many respiratory diseases, some institutions have limited implementation due to human and material resources. Moreover, it may not be realistic to repeat sputum tests when patients live far from medical institutions and have limited access to health-care services. This can lead to delayed diagnosis and increased medical costs.
According to the clinical guidelines, sputum samples should be processed immediately; otherwise, refrigerated if delays are anticipated. 10 However, there are no specific comments on how long the samples can be stored in the refrigerator. Extrapolating from studies on TB, it can be assumed that the viability of bacilli is associated with several conditions, such as storage temperature, duration, and presence or absence of preservatives.11,12 However, there is scanty information on the effect of sputum storage on NTM recovery. To address an unmet need and practical limitations in the diagnosis of NTM-PD, we designed a prospective study to evaluate the maximal refrigeration duration of sputum and compare the culture recovery rate in patients with NTM-PD.
Materials and methods
Study participants
Patients diagnosed with NTM-PD at Seoul National University Bundang Hospital between June 2020 and July 2021 were eligible for this prospective study. Further, patients with daily sputum amounts of more than 25 ml per day and repeated culture positives were included. The diagnosis of NTM-PD was based on the American Thoracic Society (ATS)/European Respiratory Society (ERS)/European Society of Clinical Microbiology and Infectious Disease Society of America guidelines. 7
Protocol
Participants were provided with six sputum containers (50-ml tubes, BD Falcon, Corning, NY, USA) without adding preservatives and instructed to collect five sputum samples randomly in the early morning from 10 days before the outpatient visit. One remaining container was for long-term storage testing of at least 10 days. After sputum collection, containers were to be immediately stored in a home refrigerator at 4°C until the date of the clinic visits. On the visiting day, participants transported the six containers without ice packs and performed a seventh spot sputum test on-site (Supplemental figure 1).
The specimens were processed with an equal volume of NaOH-citrate-N-acetyl-
NTM identification
The mycobacteria isolated in culture were tested for species identification. The nucleic acid was extracted by heating with 5% Chelex 100 solution (Bio-Rad laboratories, Hercules, CA, USA). Real-time polymerase chain reaction (PCR) of senX3-regX3 and laboratory-developed three-channel multiplex real-time PCR were performed. In addition, melting curve analysis for three regions, including the 16S rRNA general region, the hsp65 region, and the 16S rRNA hypervariable region A was conducted to detect and differentiate NTM and TB. 14 Three-channel multiplex real-time PCR was capable of detecting 17 NTM species and TB and of detecting TB and NTM simultaneously. An rpoB PCR restriction fragment length polymorphism test was performed to confirm NTM identification at the species level as previously reported.14–16 The final identification results were decided on the basis of the real-time PCR, multiplex real-time PCR, and rpoB PCR restriction analysis results.
Data collection
Clinical, microbiological, and radiological data were obtained for all patients. Data on sputum smear grade, culture positivity, and time-to-positivity (TTP) were collected. TTP was defined as the interval between the first day of incubation and a positive signal in the BACTEC MGIT 960 system. The positive signal was monitored automatically every 60 min. Participants’ computed tomography patterns were classified as fibrocavitary, nodular bronchiectatic, and unclassifiable based on the ATS/ERS guidelines. 7
Statistical analysis
Continuous variables were expressed as median (range), and categorical variables were presented as frequency (percentage). Chi-square, Fisher’s exact tests, or linear-by-linear association test were used. In all analyses, p-values <0.05 were considered statistically significant. Statistical analyses were performed using IBM SPSS version 24 (SPSS, Inc., Chicago IL, USA) and Prism version 5 (GraphPad, San Diego, CA, USA).
Results
Baseline characteristics
From June 2020 to July 2021, 35 patients with NTM-PD were enrolled. The median age was 65 years (range, 59.0–71.0 years), and there were 20 females (57.1%). A total of 226 sputum samples were collected from the participants. A total of 31 were sputum samples collected at the outpatient clinic (seventh sample), and 195 were samples that were stored in home refrigerators (first to sixth samples). The most common causative organism was Mycobacterium intracellulare (48.6%), and nodular bronchiectasis (80%) was the prevalent form of the disease. Sputum AFB smear results showed that 48 samples were smear positive: 9 with trace, 13 with 1+, 22 with 2+, 3 with 3+, and 1 with 4+ (Table 1).
Baseline characteristics.

Data are expressed as a median (range) or counts (%).
CT, computed tomography; M, mycobacterium.
Storage duration and sputum results
The median duration of refrigeration was 6 days (range, 3–8 days). The overall culture-positive rate was 81.6%, and only three samples were contaminated. The recovery rates in the outpatient spot sputum sample and refrigerated samples were 80.6% and 82.6%, respectively (p = 0.795). There was no significant difference in the recovery rate among the NTM species (Supplemental figure 2). The most extended storage duration was 36 days, and the maximal refrigeration time for culture positivity was 28 days (Figure 1).

Refrigeration duration and its culture positivity (dots), and rate of sputum culture positivity (bars) of each participant. The point color encodes nontuberculous mycobacteria culture result: positive (black), negative (white).
Sputum storage duration was categorized into four groups at weekly intervals. The majority of specimens (94.2%) were refrigerated for ⩽2 weeks. There was a trend for a higher culture positivity rate until ⩽3 weeks, but no statistical significance was observed (p = 0.610, Figure 2).

Culture positivity rate of outpatient spot (n = 31) and refrigerated samples (n = 195) according to the storage duration categorized by weekly intervals.
According to the sputum smear results, smear-positive specimens were 100% cultured, but smear-negative had a culture-positive rate of 77.5% (Figure 3). Among the smear-negative sputum samples, the culture positivity of refrigerated samples stored longer than 22 days showed a similar trend to the total samples, but no significant difference was observed (p = 0.511, Figure 3).

Culture positivity rate according to sputum smear microscopy results.
The minimal and maximal TTP of cultures were 2 and 40 days, respectively. There was no significant association between TTP and sputum storage duration (Figure 4).
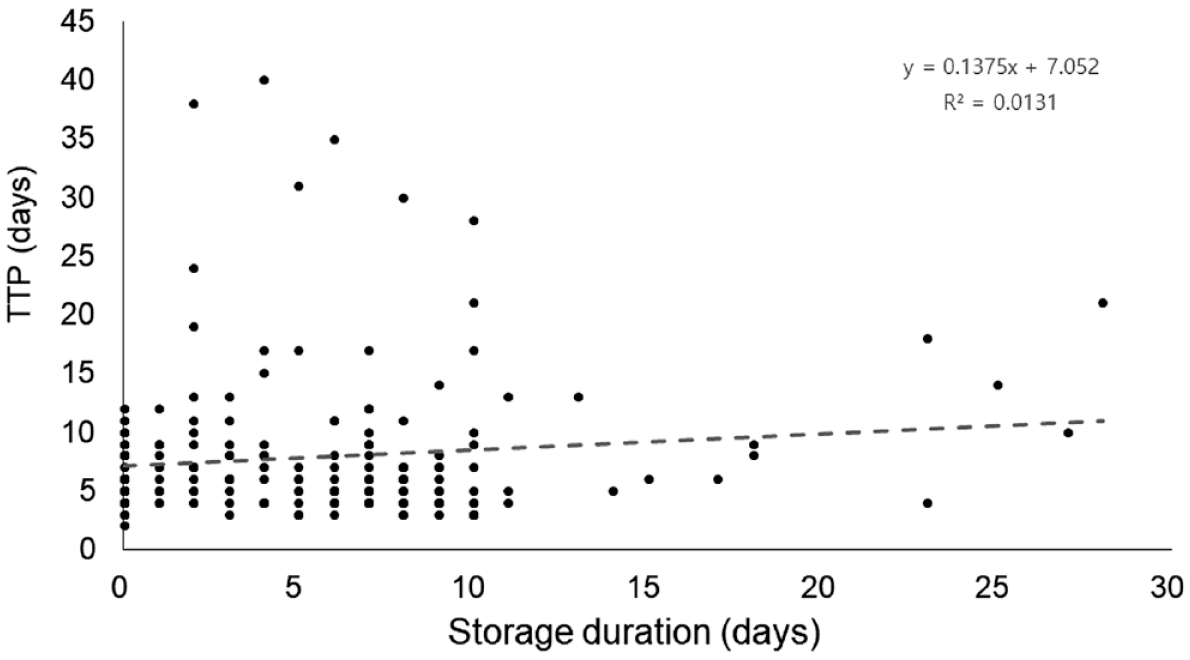
The association between storage duration and time-to-positivity of the culture-positive samples by BACTEC MGIT 960.
Discussion
In this prospective study, we investigated the maximal duration of sputum refrigeration that yields high recovery rates. We found that the overall culture-positive rate was 81.6%, and this trend was sustained up to 3 weeks. In addition, similar results were observed in the smear-negative samples. It is also notable that the positive culture rates of the randomly collected and stored samples were not inferior to those collected at the outpatient clinic. With the increasing prevalence of NTM-PD worldwide, 17 timely diagnosis and decision-making regarding treatment initiation are imperative. In a real-world setting, however, cases that are radiologically and clinically suspicious of NTM-PD but do not meet the microbiologic criteria are frequently encountered. Considering that adequately collected sputum samples are essential to prevent diagnostic delays and provide additive information on antibiotic resistance, our study has laid the groundwork for improving the diagnosis rate while providing a convenience to patients.
Investigations for proper sputum storage have been conducted primarily for TB, particularly in high-burden countries with poor infrastructures. According to the literature, it has been reported that the viability of M. tuberculosis complex was 80–94.4% when sputum was refrigerated at 4°C.12,18 Recently, Pennings et al. 19 compared the culture positivity rate at room temperature and 4°C for 7 days in patients with NTM-PD. The authors reported the overall NTM recovery rate was 78.9%. Although there was no difference in culture positivity between the two storage temperatures, there was less contamination in the samples stored in the refrigerator (32% versus 5%). Our results confirmed these findings with a more extended duration of refrigerated storage.
There have been concerns about the optimal temperature conditions for the storage of sputum samples. According to the World Health Organization, it is recommended to add preservatives, such as 0.6% cetylpyridinium bromide or 1% cetylpyridinium chloride, to sputum specimens that are expected to be exposed to room temperature for more than 2 days. 20 Because the preservatives are crystallized and become inactive when the samples are refrigerated, these samples should be stored after removing the preservatives through centrifugation immediately after transport to the laboratory. In addition, it has been known that these preservatives have a negative effect on the recovery of mycobacteria. 21 In contrast, Tessema et al. evaluated the positive recovery rate of M. tuberculosis from frozen samples. In the study, high culture positivity (78.7–83.1% using different culture media) was shown despite long-storage times (132 days, range 16–180 days). 22 Although the differences in recovery rate have not yet been studied in NTM, freezing at −20°C may be an alternative option for storage as extrapolated from the previous study on TB.
Our study has several limitations. First, this was a single-arm study with no comparators. However, the current study demonstrated an expansion on the concept of the previous study in which sputum refrigeration showed a favorable recovery rate and lower contamination compared with storage at room temperature. 19 Second, because NTM identification was not requested, mixed infection or infection by other strains may exist. To minimize this limitation, we included only patients in whom the same strain was repeatedly cultured. Third, the quality and quantity of expectorated sputum were not measured. Nevertheless, a low contamination rate and high culture positivity might confirm proper sample collection and processing has been achieved. In addition, a high proportion of AFB smear negativity or low-level positivity (trace or 1+) suggests that repetitive sputum collection with refrigeration could be a practical method for the diagnosis of NTM-PD, even in patients with minimal disease activity. Fourth, the number of each NTM species was insufficient for statistical verification. Further studies with a large number of samples are warranted to investigate the differences in recovery rate across the NTM species. Last, we have demonstrated the long-term viability of NTM during refrigeration; however, transportation issues, such as utilizing a cold-chain transport system, are likely to be separate. In other words, our results are not applicable to countries with limited resources or difficult logistics. Given the previous study showing that mycobacteria maintained their viability at room temperature for up to 2 weeks, it may be a reasonable option to store sputum samples in the refrigerator before shipping and delivering them to the laboratory at room temperature.
In conclusion, long-term viability of NTM was observed in refrigerated sputum from patients with NTM-PD, and the culture positivity rate of refrigerated sputum samples was not inferior to the spot sputum samples. Our data suggest that implementing sputum storage in a refrigerator at 4°C would provide convenience for patients and clinicians in diagnosing and following up with the disease.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231162244 – Supplemental material for A prospective study on the long-term storage of sputum and the recovery of nontuberculous mycobacteria
Supplemental material, sj-docx-1-tar-10.1177_17534666231162244 for A prospective study on the long-term storage of sputum and the recovery of nontuberculous mycobacteria by Byoung Soo Kwon, Jeong Su Park, Jung-A Shin, Eun Sun Kim, Sung Yoon Lim, Myung Jin Song, Yeon-Wook Kim, Hyung-Jun Kim, Yeon Joo Lee, Jong Sun Park, Young-Jae Cho, Ho Yoon, Choon-Taek Lee and Jae Ho Lee in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
