Abstract
Dasatinib, a tyrosine kinase inhibitor, is usually prescribed for chronic myeloid leukemia and Philadelphia chromosome–positive acute lymphoblastic leukemia. However, some patients may develop an intolerance to this drug over the years. Among various toxicities related to dasatinib, dasatinib-associated interstitial pneumonitis is not reported frequently in the literature yet. Moreover, published studies have reported only few cases of dasatinib-associated pneumonitis, almost exclusively in chronic myeloid leukemia. In this study, we describe three cases of dasatinib-associated interstitial pneumonitis in patients with chronic myeloid leukemia or Philadelphia chromosome–positive acute lymphoblastic leukemia (a 56-year-old man, a 34-year-old man, and a 46-year-old woman) at our institution. In all three patients, the time from the initiation of dasatinib therapy to the onset of interstitial pneumonitis varied greatly. Among them, one patient underwent a surgical lung biopsy, which revealed chronic granulomatous inflammation without any causative pathogen. In all patients, dasatinib was discontinued after the diagnosis of interstitial pneumonitis, and two patients were treated with systemic steroids. Although infrequent, dasatinib-induced pneumonitis should be considered a possible diagnosis in dasatinib-treated patients with fever and respiratory symptoms. In addition, hematologists and pulmonologists should be aware of this rare but critical toxicity.
Keywords
Background
Dasatinib is a breakpoint cluster region-Abelson (BCR-ABL) tyrosine kinase inhibitor (TKI), and it has been used as a standard treatment for chronic myeloid leukemia (CML) and Philadelphia chromosome–positive acute lymphoblastic leukemia (ALL). In the era of TKIs, wherein a considerable proportion of patients with CML can expect long-term survival with TKIs, some patients have developed intolerance, rather than resistance, to TKIs, which has become a more frequent issue in drug maintenance. Therefore, prevention, early detection, and proper management of adverse events during treatment are important for optimal management of CML.
Among the various dasatinib-associated adverse events, pulmonary toxicities, including pleural effusion and pulmonary arterial hypertension, have been relatively well described.1,2 In the DASISION trial, the enrolled patients were treated with dasatinib, and among them, 28% of patients experienced pleural effusion. 3 However, in most cases, pleural effusion could be effectively controlled with dasatinib dose reduction and supportive care, and only 6% of patients discontinued dasatinib because of pleural effusion. In some cases, although rare, it is known that dasatinib should be discontinued permanently when pulmonary arterial hypertension develops. In contrast to these adverse events, interstitial pneumonitis associated with dasatinib has not been thoroughly investigated. To date, only a small number of cases have been reported, almost exclusively in patients with CML, and only a few studies have described dasatinib-associated pneumonitis in patients with Philadelphia chromosome–positive ALL.4–7 In this study, we describe three cases of dasatinib-associated interstitial pneumonitis encountered in our tertiary institution and review related published studies.
Case presentation
Case 1
A 56-year-old man diagnosed with CML has been treated with dasatinib since July 2012. Although there were no significant toxicities with dasatinib, he failed to achieve a complete molecular response; therefore, dasatinib was switched to nilotinib after 10 months of treatment at the physician’s discretion. He achieved a deep molecular response (DMR) with nilotinib, but because he developed a skin rash after 6 months of treatment, the TKI had to be changed to imatinib. Unfortunately, after 5 months of imatinib treatment, the patient experienced facial edema, and the TKI was again changed to dasatinib. Subsequently, he did not have any significant toxicity when 100 mg of dasatinib was readministered for more than 4 years.
In December 2019, the patient was referred to our clinic because of dyspnea, cough, and grade 3 myalgia according to the Common Terminology Criteria for Adverse Events (CTCAE). When he visited our hospital, he was already being treated with antibiotics and low-dose corticosteroids, considering the possibility of pneumonia, but the symptoms did not improve. His laboratory findings indicated a white blood cell (WBC) count of 7.90 × 103/µl (neutrophils, 26.8%; lymphocytes, 58.2%; monocytes, 9.1%; eosinophils, 5.3%) and C-reactive protein (CRP) level of 0.77 mg/dl. Arterial blood gas analysis revealed hypoxia with a pH of 7.393, PO2 of 58.1 mmHg, and O2 saturation of 89.3% at room air. Chest radiography showed reticular opacity and ground-glass opacity (GGO), and chest computed tomography (CT) confirmed diffuse GGO, interlobular septal thickening, and reticular opacity, mainly in the bilateral lower lobes (shown in Figure 1(a)). There were no abnormal findings on echocardiography. Autoimmune markers, including rheumatoid factor, antineutrophil cytoplasmic antibody, and anticyclic citrullinated peptide antibodies, were negative. The antinuclear antibody titer was 1:320 with a mitotic pattern; however, extractable nuclear antigens were not detected. Moreover, the patient had no symptoms suggestive of an autoimmune disease.

Chest computed tomography (CT) findings of the patients before and after the treatment. In case 1, chest CT at presentation revealed diffuse ground-glass opacity (GGO), interlobular septal thickening, and reticular opacity in bilateral lower lobes (a). After steroid treatment, marked improvement was seen after 4 months (b). In case 2, ill-defined GGO was noted, mainly in the left lower lung field (c). Three months after discontinuation of dasatinib, GGO was decreased (d). In case 3, chest CT showed diffuse GGOs and peribronchial ill-defined nodules in the bilateral upper lobes, with a small amount of bilateral pleural effusion (e). With steroid treatment, follow-up chest CT after 1 month showed resolution of diffuse GGOs in both lungs (f).
Dasatinib was discontinued because of a suspected diagnosis of dasatinib-associated interstitial pneumonitis. The differential count of WBC was normalized after TKI discontinuation, but the symptoms did not improve 1 week after drug discontinuation. Considering the severity of pneumonitis and lack of clinical recovery, video-assisted thoracoscopic (VATS) lung biopsy was performed for an accurate diagnosis. Histopathologic results showed multifocal interstitial lymphocytic infiltration, and there was no evidence of infection. Histologically, the alveolar walls were thickened by a uniform, cellular, and inflammatory interstitial infiltrate including lymphocytes, plasma cells, and histiocytes without architectural distortion (shown in Figure 2). Peribronchiolar lymphohistiocytic aggregates and noncaseating granulomatous inflammation were identified in part.
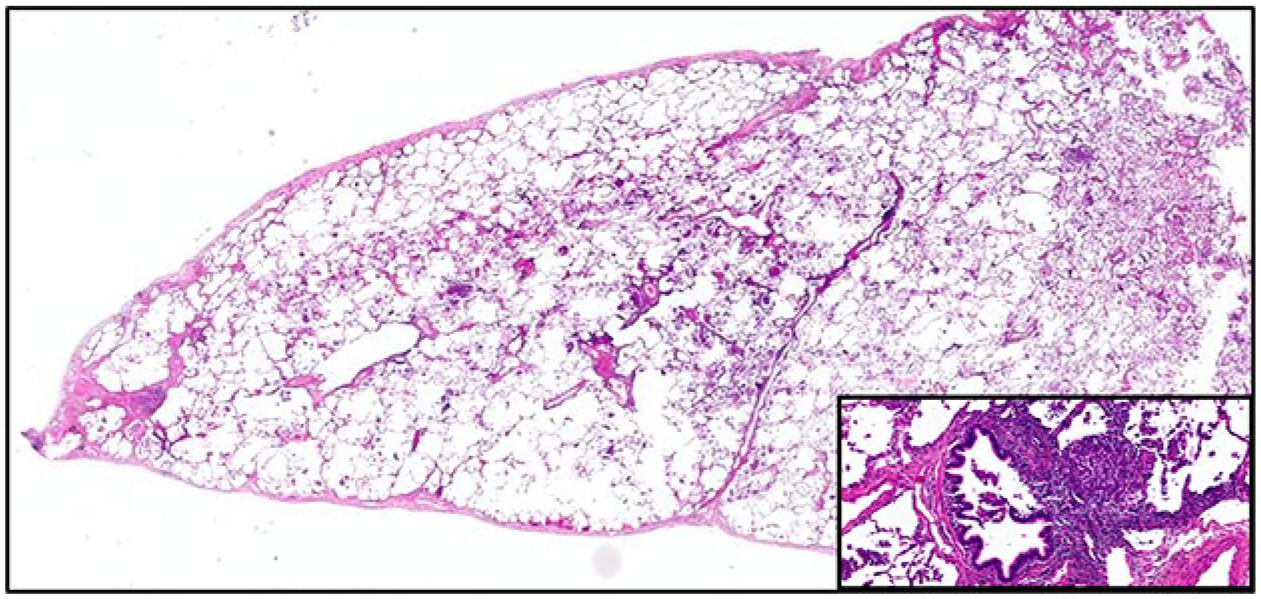
Pathologic features of the video-assisted thoracoscopic lung biopsy. The alveolar walls are thickened by a uniform, cellular, inflammatory interstitial infiltrate including lymphocytes, plasma cells, and histiocytes without architectural distortion (hematoxylin and eosin stain, 1.4×). Inset: Peribronchiolar lymphohistiocytic aggregates and noncaseating granulomatous inflammation (hematoxylin and eosin stain, 20×).
In January 2020, which was 3 weeks after hospitalization, prednisolone (0.5 mg/kg) was started. Serial chest radiography showed gradual improvement of opacities in both lungs, and complete resolution of lung lesions was observed on follow-up chest CT scan after steroid tapering over 4 months (shown in Figure 1(b)). As the patient showed continued DMR and experienced the toxicities of various TKIs, TKIs were discontinued, and the patient was closely followed up with BCR-ABL quantitative reverse transcription polymerase chain reaction (RT-qPCR). Currently, sustained DMR is maintained for more than 24 months after TKI discontinuation.
Case 2
A 34-year-old man with no history of pulmonary disease was diagnosed with CML in September 2019 and was treated with 100 mg of dasatinib for 13 months. Although the dose of dasatinib was reduced to 80 mg because of cytopenia, he achieved an early molecular response at 3 months and maintained a major molecular response.
He visited the emergency department in October 2020 with complaints of CTCAE grade 1 fever and grade 2 headache persisting for 1 week. On examination, he was normotensive but showed fever and tachycardia of 38.3°C and 103 beats per minute, respectively. His respiratory rate was 19 breaths per minute, and oxygen saturation was 96% on room air. Physical examination results, including chest auscultation, were unremarkable. The WBC count was 4.05 × 103/µl, with 59.1% of segmented neutrophils. Except for elevations in grade 1 liver enzymes (aspartate transferase, 53 IU/L; alanine transferase, 51 IU/L) and mild elevation in CRP (1.52 mg/dl; reference range, 0–0.5 mg/dl), there were no significant abnormalities in other laboratory parameters. Chest radiography performed 1 week later showed no abnormalities. However, the fever persisted despite prescribing antipyretics; therefore, a chest CT scan was performed on the 11th day of fever. Chest CT findings revealed ill-defined GGO and consolidation, especially in the left lower lung field (shown in Figure 1(c)). Bronchoalveolar lavage (BAL) fluid analysis showed a lymphocyte-dominant pattern with no evidence of infection, including pneumocystis pneumonia (PCP), cytomegalovirus (CMV), respiratory virus, tuberculosis, or fungus. Echocardiography performed to exclude cardiac problems did not reveal any significant abnormal findings. Because of a suspected diagnosis of dasatinib-associated interstitial pneumonitis, dasatinib was discontinued. The fever resolved 3 days after the discontinuation of dasatinib, and TKI therapy was resumed 16 days after the discontinuation of the TKI, changing to imatinib. Follow-up chest CT performed after 3 months showed a decreased extent of the previously noted GGO (shown in Figure 1(d)). With regular outpatient department visits, the patient maintained molecular response 4.0, without significant toxicity to imatinib.
Case 3
A 46-year-old woman was diagnosed with Philadelphia chromosome–positive ALL with a complex karyotype in February 2021. She received imatinib 400 mg/day with induction combination chemotherapy (daunorubicin, cyclophosphamide, vincristine, and dexamethasone) and achieved complete remission. During subsequent consolidation chemotherapy (methotrexate, cytarabine, and cyclophosphamide), the imatinib dose was reduced to 300 mg/day because of drug-related skin rash and generalized edema. Unfortunately, she developed a molecular relapse detected by BCR-ABL RT-qPCR after 6 months of imatinib treatment. The TKI was changed to dasatinib (140 mg/day).
One week after the start of dasatinib therapy, she developed CTCAE grade 2 fever (39.1°C) and dry cough. At the time of symptom onset, the respiratory rate was 24 breaths per minute and oxygen saturation in room air was 89.0%. The laboratory findings on admission were as follows: WBC count, 13.41 × 103/µl with 59.0% neutrophils; CRP, 2.84 mg/dl; procalcitonin, 0.13 ng/ml; aspartate transferase, 47 IU/L; and alanine transferase, 71 IU/L. Chest CT revealed diffuse GGO and peribronchial ill-defined nodules in the bilateral upper lobes, with a small amount of bilateral pleural effusion (shown in Figure 1(e)). BAL fluid analysis showed a lymphocyte-dominant pattern, and no microorganisms were identified, including PCP, CMV, respiratory virus, tuberculosis, or fungus. Empirical antibiotics were stopped, and considering that the patient was planning an allogeneic stem cell transplantation the next month, steroids were used without delay. Prednisolone at a dose of 1 mg/kg was started, and the dose was tapered off for 3 weeks. She showed symptomatic improvement, and follow-up chest CT showed resolution of diffuse GGOs in both lungs (shown in Figure 1(f)). The patient underwent haploidentical allogeneic hematopoietic stem cell transplantation from his son without significant toxicity.
Discussion and conclusions
Since the first case of alveolar pneumonia with dasatinib in a CML patient was reported in 2006, several cases of dasatinib-associated interstitial lung diseases (ILDs) have been reported (Table 1). 4 In contrast to dasatinib-associated pleural effusion, which has been well described in phase III trials, the incidence and characteristic features of dasatinib-associated pulmonary ILDs are not sufficiently understood.8,9 In 2007, Bergeron et al. 5 reported that seven patients (17.5%) developed parenchymal lung disease among 40 patients who used dasatinib for 13 months. In 2019, another study investigating the incidence of dasatinib-associated interstitial pneumonitis was published, and this case series reported that two patients (8.7%) were diagnosed with interstitial pneumonitis out of 23 patients who had used dasatinib for more than 6 years. 6 In a national postmarketing study conducted by the Food and Drug Administration (FDA), 24 cases of dasatinib-associated lung infiltration and 19 cases of dasatinib-associated pneumonitis were reported to the FDA adverse event reporting system in 2012, whereas in Japan, dasatinib-associated pneumonitis was reported in 3.7% of patients in postmarketing surveillance.1,7 Because dasatinib became available in Korea in 2008, a total of 113 patients used dasatinib in our hospital, and interstitial pneumonitis was identified in three patients (2.7%). These heterogeneous data suggest that more patients may experience dasatinib-associated pneumonitis than expected by the clinician, and hematologists and pulmonologists should be aware of this rare but critical toxicity.
Summary of the literature review of cases of interstitial pneumonitis in patients treated with dasatinib.

BAL, bronchoalveolar lavage; CML, chronic myeloid leukemia; CT, computed tomography; F, female; GGO, ground-glass opacity; LD, lymphocytic cellular pattern; M, male; mPd, methylprednisolone; NA, not assessed; ND, neutrophilic cellular pattern; Pd, prednisolone; ST, septal thickening; TBLB, transbronchial lung biopsy; TKI, tyrosine kinase inhibitor.
Pathological findings consistent with cellular nonspecific interstitial pneumonia.
According to previous reports, the time from the initiation of dasatinib therapy to the onset of interstitial pneumonitis varies from patient to patient. 6 Our patients also developed pulmonary disease at various time points, ranging from the first week to the fourth year of dasatinib treatment. Furthermore, the pattern and distribution of lung parenchymal abnormalities on chest CT also vary, making it difficult to make an accurate differential diagnosis based on radiological findings alone. In addition to chest CT scans, bronchoscopy and echocardiography were performed in most patients described in case reports to exclude the possibility of infection or cardiogenic problems. By contrast, only a few cases have attempted pathologic diagnosis through lung biopsy, including some cases reported by French and Japanese groups.5,7,10 Interestingly, in the study by Takekoshi et al., although their patient showed pathological findings generally consistent with cellular nonspecific interstitial pneumonia, various pathological findings also coexisted in one patient. One of our patients who underwent VATS lung parenchymal biopsy showed chronic granulomatous inflammation, but the infectious etiology, such as tuberculosis or fungus, was not identified. Although there are still not enough cases, given the various pathologic findings currently being reported, the possibility of dasatinib-related changes should always be considered when performing a biopsy in these clinical scenarios.
A considerable proportion of patients showed clinical improvement immediately after cessation of the drug, and some patients who required additional treatment also showed a favorable response to steroids without requiring additional immunosuppressive therapy or leaving unacceptable sequelae. However, if a proper diagnosis is not made while maintaining the causative drug without appropriate treatment, then the patient may be exposed to prolonged inflammation, which can lead to chronic hypoxia.
Indeed, our experience with the first patient allowed us to make a suspected diagnosis of this adverse event at an early stage in other patients using dasatinib, allowing early and appropriate management of patients without further invasive diagnostic procedures. Fortunately, our patients also showed a clinical course similar to that of previous reports, with one patient recovering only after the discontinuation of dasatinib and another patient showing rapid resolution of the symptoms and disappearance of radiologic abnormalities after steroid administration. Furthermore, previous reports have suggested that dasatinib can be safely used after clinical improvement. 6 None of our patients resumed dasatinib again, but with the aforementioned data, readministration of dasatinib could be an option if no alternative treatment is available.12,13
Although the mechanism of pulmonary toxicities with BCR-ABL TKIs has not yet been fully understood, off-target effects related to different kinase inhibitors have been suggested as one of the major mechanisms like other TKI-associated toxicities. 14 Notably, dasatinib is known to bind favorably to the SRC family compared with other BCR-ABL TKIs, which is related to an autoimmune mechanism. 15 In this regard, some studies, although limited, have suggested that lung toxicities of dasatinib are associated with SRC targeting, mimicking autoimmune ILDs.6,7 From this viewpoint, treatment for dasatinib-associated interstitial pneumonitis has mainly focused on avoiding the causative drug and empirical steroid administration, similar to the usual initial approach to autoimmune ILDs. However, the mechanism of dasatinib-associated pulmonary toxicity requires further elucidation. In conclusion, we describe three cases of dasatinib-associated interstitial pneumonitis in patients with CML and Philadelphia chromosome–positive ALL. The clinical features and response to medical management in these patients generally coincide with those mentioned in other reports. It is important for clinicians to be aware of the side effects and detect them without delay. Moreover, dasatinib-induced pneumonitis should be considered a possible diagnosis when patients treated with dasatinib have fever and respiratory symptoms. Although it is rare and not fatal in most cases, additional research is required on the pathogenesis, risk factors, and optimal management of interstitial pneumonitis associated with dasatinib.
