Abstract
Background:
Numerous studies have shown that dipeptidyl peptidase-4 inhibitors (DPP-4i) may regulate immunological pathways implicated in asthma. The association between DPP-4i use and risk of asthma development is limited, however.
Aim:
We aimed to evaluate if DPP-4i treatment in individuals with type 2 diabetes mellitus (T2DM) is associated with a lower risk and severity of asthma.
Methods:
We performed a population-based retrospective cohort study using the Longitudinal National Health Insurance Research database between 2008 and 2015. After one-to-four propensity score matching from 1,914,201 patients with defined criteria, we enrolled 3001 patients who were on DPP-4i (DPP-4i group) for a diagnosis of T2DM but without a diagnosis of asthma for further analysis. Cox proportional hazards regression analysis was performed to estimate and compare the risk of developing and severity of asthma, including no acute exacerbations event (No-AE), acute exacerbations (AEs), status asthmaticus (Status), and required endotracheal intubation (ET-tube intubated), between the two groups.
Results:
The participants had a mean age of 66.05 ± 17.23 years and the mean follow-up time was 4.96 ± 4.39 years. The risk of asthma development was significantly lower in the DPP-4i group than in the non-DPP-4i group [adjusted hazard ratio (HR) = 0.65; 95% confidence interval (CI) = 0.29–0.83;
Conclusion:
The use of DPP-4i decreased the risk and severity of asthma with a class effect among No-AE, AE, status of asthma events, but not in ET-tube intubated events. Our report suggests that DPP-4i may play a role in attenuating the impact of asthma on incidence in the future and on more severe forms of disease exacerbation in T2DM patients.
Introduction
According to the Nutrition and Health Survey, the prevalence of diabetes mellitus (DM) was 11.6% (men = 12.9%; women = 10.5%) in Taiwan during 2013–2016.
1
Jiang
As the first-line treatment for type 2 DM, metformin has been reported to be associated with a lower risk of asthma exacerbation, asthma-related emergency department visits, and hospitalization. 14 Another previous study in a Taiwan population reported that among patients with concurrent asthma and diabetes, metformin users had a lower risk of asthma-related hospitalization and asthma exacerbation. 15 In addition, a meta-analysis indicated that metformin decreased the risk of asthma-related emergency room visits for patients with concurrent asthma and diabetes but was not statistically significant for decreasing asthma-related hospitalization and exacerbation. 16
The benefits of second-line with treatment agents for type 2 DM, including sulfonylureas (SUs), sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide-1 receptor (GLP-1R) agonists, and dipeptidyl peptidase-4 inhibitors (DPP-4is), when treating asthma were limited and inconclusive, however.17–20
DPP-4, also known as adenosine deaminase complexing protein 2 or cluster of differentiation 26 (CD 26), is encoded by the
Materials and methods
Data source and participants
A single-payer NHI program was launched by the Taiwanese government in March 1995. We used data from the NHIRD and 1,914,201 patients included in the Longitudinal Health Insurance Database in Taiwan (2008–2015). The annual coverage rate of the NHI program has ranged from 96.1% to 99.6%, with more than 20 million Taiwanese citizens enrolled since 1997. In the NHIRD, all diseases before 2015 were coded according to the
Ethical approval
This retrospective cohort study was approved by the Institutional Review Board of Tri-Service General Hospital (IRB No. B-110–47), and informed consent was waived as the database used in this study was deidentified.
Study design and sampled participants
Sitagliptin was the first marketed DPP-4i in Taiwan since 2008. We choose the time between 1 January 2008 and 31 December 2015 for a cumulative analysis of the occurrence of asthma development or exacerbation.
We recruited patients aged ⩾18 years with ICD-9-CM diagnosis codes for T2DM (ICD-9-CM: 250.XX), who had at least three outpatient visits, and used DPP-4i for >90 days (Figure 1). Treatment of patients in non-DPP-4i group included metformin, SUs, meglitinides, thiazolidinediones, sodium-glucose cotransporter 2 inhibitors, α-glucosidase inhibitors, and insulin. We excluded patients who were identified on the index date as (1) being aged 18 years or younger; (2) those previously diagnosed with asthma; (3) those with comorbid disorders (COPD, respiratory tract cancer, bronchiectasis); (4) those who were on DPP-4i before the index date; and (5) those who received GLP-1 agonist before the tracking (Figure 1).
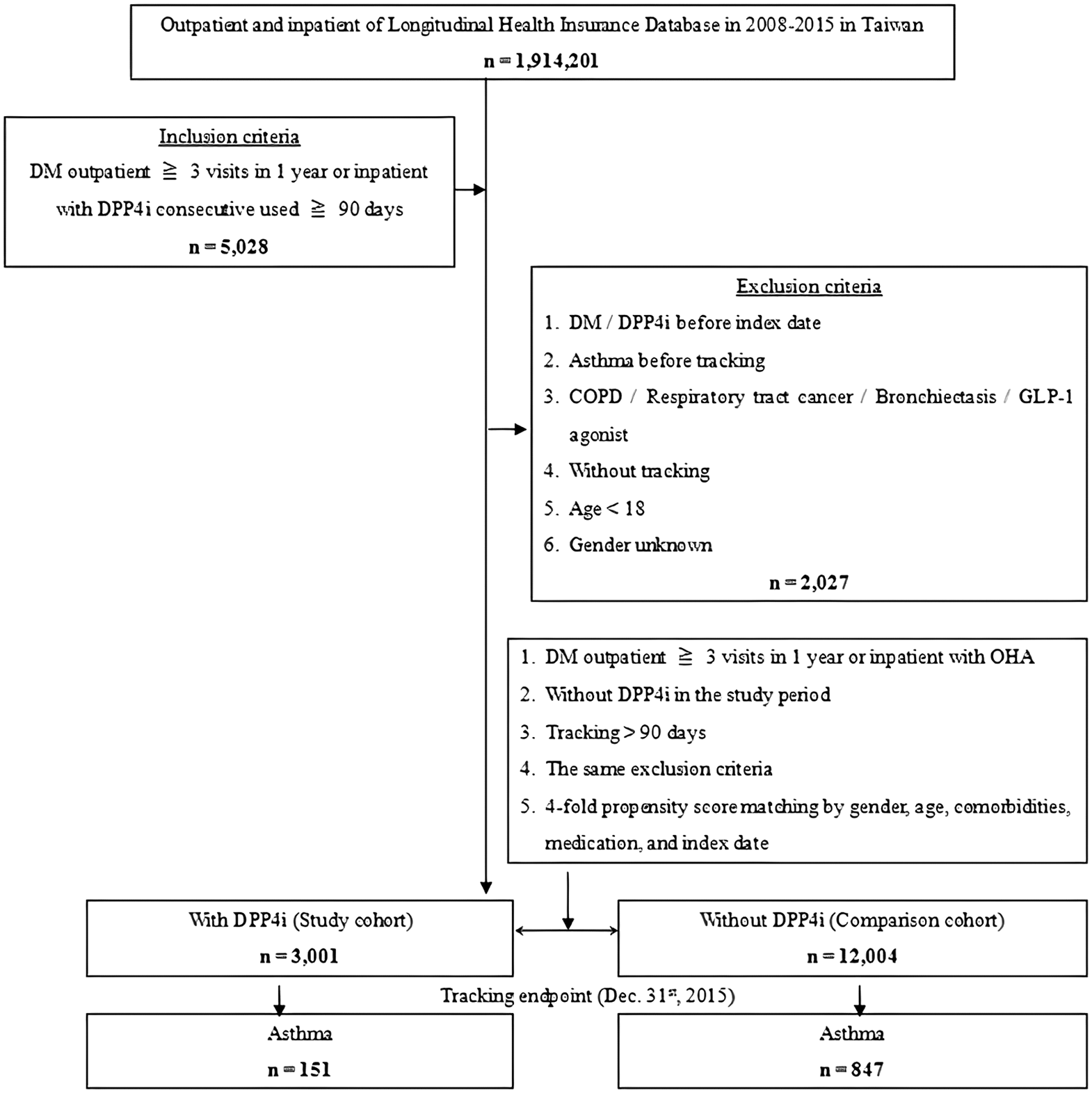
The flow chart of study sample selection.
Propensity score matched comparison cohort
To reduce demographic differences between the DPP-4i group and the non-DPP-4i group, we performed 1:4 propensity score matching by age, sex, race/ethnicity, history of smoking, medical comorbidities, complications of T2DM, and urbanization level. After 1:4 propensity score matching from 1,914,201 patients with defined criteria, we finally enrolled 3001 patients in DPP-4i group and 12,004 patients in non-DPP-4i group (Figure 1).
Covariates and outcome measurements
All covariates were measured during the pre-index period. They were age, sex, race/ethnicity, history of smoking, hypertension (HTN), hyperlipidemia, obesity, macrovascular or microvascular complications of T2DM, chronic kidney disease, obstructive sleep apnea syndrome, and urbanization level. The urbanization level of residence was defined according to the population and various indicators of the level of development. Level 1 was defined as a population >1,250,000, and a specific designation as political, economic, cultural, and metropolitan development. Level 2 was defined as a population between 500,000 and 1,249,999, and as playing an important role in the political system, economy, and culture. Urbanization Levels 3 and 4 were defined as a population between 149,999 and 499,999, and <149,999, respectively. 26 The medical comorbidities and T2DM vascular complications were selected by corresponding ICD-9-CM codes in outpatient medical claims previous to the index date including HTN (ICD-9-CM: 401.1, 401.9, 402.10, 402.90, 404.10, 404.90, 405.1, 405.9), hyperlipidemia (ICD-9-CM: 272), obesity (ICD-9-CM: 278), heart failure (ICD-9-CM: 428), obstructive sleep apnea (ICD-9-CM: 780.51, 780.53, 780.57), microvascular complications of T2DM (ICD-9-CM: 250.52, 362.0 for retinopathy; 250.6, 357.2 for neuropathy; 250.4, 250.42 for nephropathy), and macrovascular complications of T2DM (ICD-9-CM: 430-438 for stroke; 440.2–440.3, 440.8–440.9, 443, 444.22, 444.8, 447.8–447.9 for peripheral arterial disease; 410-41 for coronary artery disease). We adjusted the variables in relation to the Charlson comorbidity index scores to better control the confounding effect from comorbidity. The primary outcomes in this study were events of asthma development but no acute exacerbations event (No-AE), acute exacerbation of asthma (AE), status asthmaticus (Status), and requiring endotracheal intubation (ET-tube intubated). No-AE was defined as a patient who was diagnosed with asthma and received asthma therapy without an asthma exacerbation event (ICD-9-CM: 493.00, 493.10, 493.20, 493.90). AE was defined as a patient who used a systemic corticosteroid plus an emergency room visit during the follow-up period (ICD-9-CM: 493.02, 493.12, 493.22, 493.92). Status was defined as severe asthma unresponsive to repeated courses of systemic corticosteroid or beta-agonist therapy in an emergency room visit (ICD-9-CM: 493.01, 493.11, 493.21, 493.91). ET-tube intubated was as defined as AE or Status and ET-tube intubated (AE/Status with ICD-9-CM: OP96.04).
Statistical analysis
Statistical analysis was performed using the Statistical Product and Service Solutions, version 22 (SPSS Inc., Chicago, IL, USA). Demographic characteristics were compared using the
Results
Study sample characteristics
The baseline characteristics of the study sample are presented in Table 1. There were 3001 patients in the DPP-4i group and 12,004 patients in the non-DPP-4i group after fourfold propensity score matching. The participants had a mean age of 66.05 ± 17.23 years. The prevalence of comorbidities, listed in Table 1, showed no significant differences (all
Characteristics of study in the baseline.

CCI_R, Charlson comorbidity index without dementia; DM, diabetes mellitus.
Categorical variables were compared using the Chi-square or Fisher’s exact test and expressed as
Continuous variables were compared using Student’s
Table 2 shows that asthma developed in 151 (5.03%) patients in the DPP-4i group and 847 (7.06%) patients in the matched controls (
Characteristics of study in the endpoint.

CCI_R, Charlson comorbidity index without dementia; DM, diabetes mellitus.
Categorical variables were compared using the Chi-square or Fisher’s exact test and expressed as
Continuous variables were compared using Student’s

Kaplan–Meier for cumulative risk of asthma among DM patients aged 18 years and over stratified by DPP-4i with the log-rank test.
Table 3 shows the Cox regression analysis of the association between DPP-4i use and the risk of developing asthma. The HR of overall asthma was 0.57 (95% CI = 0.27–0.79;
Factors of asthma using the Cox regression.

CCI_R, Charlson comorbidity index without dementia; CI, confidence interval; DM, diabetes mellitus; HR, hazard ratio.
Adjustment for age, sex, race/ethnicity, ever smoking, HTN, hyperlipidemia, obesity, macrovascular or microvascular complications of DM, chronic kidney disease, obstructive sleep apnea syndrome, the Charlson comorbidity index score, and urbanization level.
Table 4 reveals that compared with the non-DPP-4i group, the HRs of the DPP-4i group for severity of asthma as No-AE, AE, Status, and ET-tube intubated were 0.55 (95% CI = 0.24–0.70;
HRs of asthma severity categorized by with or without DPP-4i.

AE, acute exacerbation; CI, confidence interval; ET-tube intubated, required endotracheal intubation; HR, hazard ratio; No-AE, no acute exacerbations event; PYs, person-years; Status, Status asthmaticus.
Without DPP-4i as reference group.
Status asthmaticus does not respond to standard treatments of bronchodilators and corticosteroids.
In adjusted Hazard ratio, HR was adjusted according to the variables listed in the baseline characteristics.
Table 5 shows the comparison of the risk of asthma among different DPP-4i types. All types of DPP-4i had significant protective effects against asthma development when the duration of therapy was greater than 1 year (all
Comparison of the risk for asthma according to the use of DPP-4i.
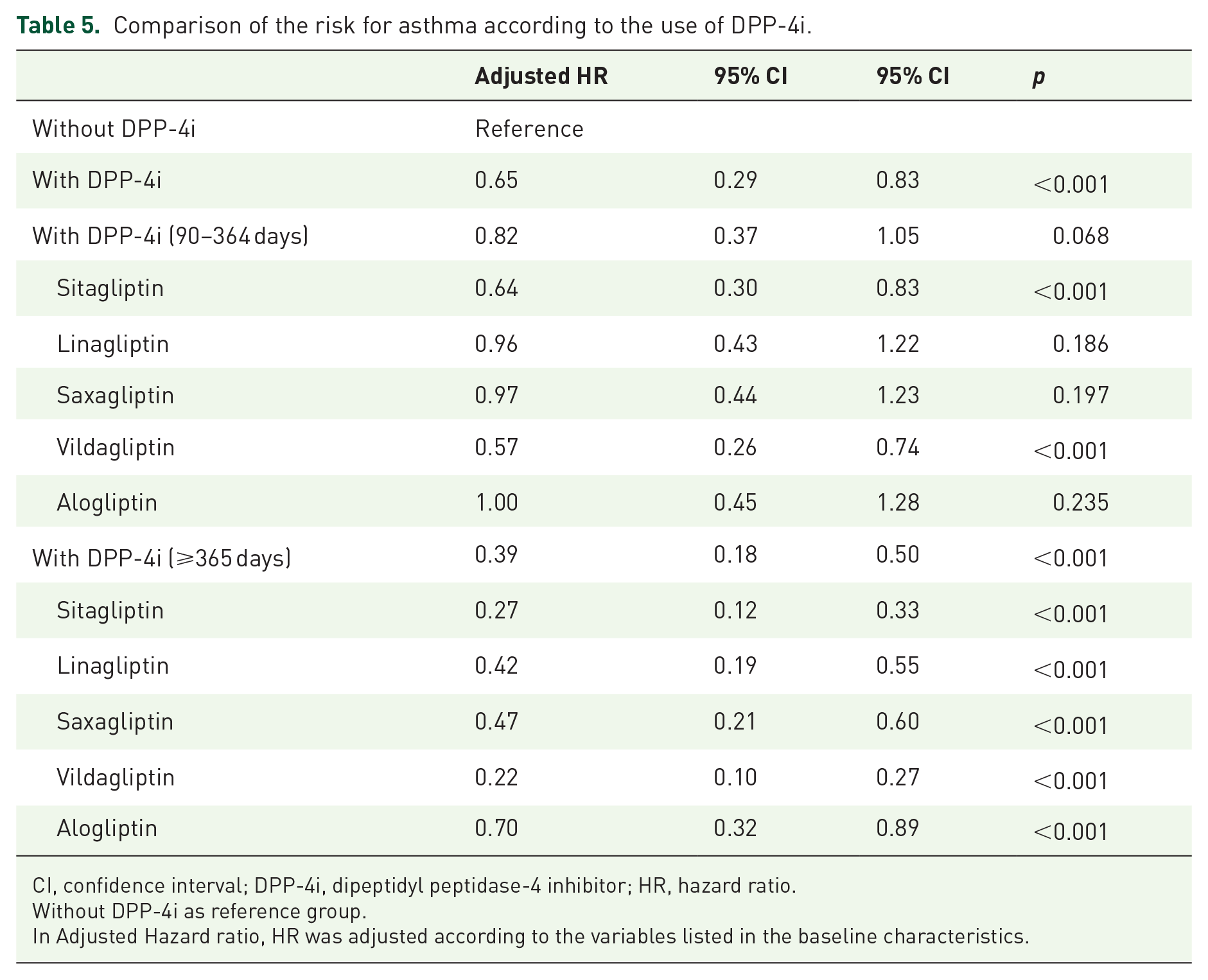
CI, confidence interval; DPP-4i, dipeptidyl peptidase-4 inhibitor; HR, hazard ratio.
Without DPP-4i as reference group.
In Adjusted Hazard ratio, HR was adjusted according to the variables listed in the baseline characteristics.
Discussion
This study indicated that the DPP-4i group was associated with a 34% lower risk of asthma development compared with that of the non-DPP-4i group. In addition, compared with the non-DPP-4i group, the DPP-4i group showed associated decreased risks for developing asthma, including Non-AE, AE, and status of asthma but excluding ET-tube intubated. Endotracheal intubation may be required when a patient presented with respiratory distress. This study, however, defined ET-tube intubated as AE or Status and ET-tube intubated (AE/Status with ICD-9-CM: OP96.04). There are many reasons affect the results such as conservative physicians or conservative family members will choose bilevel positive airway pressure machine for respiratory failure patients instead of ET-tube intubation. This study also demonstrated that all types of DPP-4i may decrease the incidence of asthma in the future in T2DM patients when the duration of therapy was greater than 1 year. It seems that DPP-4i use has cumulative effect against asthma development.
Shiobara
Wu
Furthermore, our data show that microvascular complications of T2DM and CCI_R are better in the non-DPP-4i group. In clinical practice (during AD 2008–2015), the first choice of oral antidiabetic drugs (OADs) after metformin were SU or thiazolidinediones (TZDs) because of the more powerful lowering HbA1c ability.36,37Optimize glucose control can reduce the risk or slow the progression of microvascular complications of DM. Moreover, there is an emerging role of SGLT2i in reducing the risk of kidney disease progression and preventing worsening of albuminuria.38,39 Thus, if the treatment strategy for DM including SGLT2i, the microvascular complications of DM will decrease in the future. The Taiwan NHI benefits only offer one drug between DPP-4i and SGLT2i, however. This might be the reason for why microvascular complications of DM, and CCI_R are better in the non-DPP-4i group.
Contrary to this study results, Colice G.
Strengths of this study include the very large sample size and T2DM, and asthma diagnoses were confirmed by a trained physician. In addition, this study included patients who were diagnosed with T2DM but without asthma and were recruited for cumulative analysis of occurrences of asthma development or exacerbation. Based on this study design, we could minimize the impact of patients with poorly controlled asthma before they were recruited.
Limitations
There were still some limitations to this study, however. First, patients with asthma or T2DM could be identified using insurance claims data. Furthermore, data on the severity and duration of T2DM and impact on diabetes control, such as HbA1c levels and body mass index (BMI) were not available. In addition, it was inability to classify endotypes (T2 high/T2 low) and phenotypes (such as atopic, late onset, smoke-related, and obesity-related) of asthma based on our database. Besides, we could only estimate treatment durations of each DPP-4i by dividing the cumulative doses of individual medications by defined daily doses. Moreover, there was no patient had used the biological therapies, including anti-immunoglobulin E (IgE): omalizumab; anti-IL-5: mepolizumab, reslizumab, and benralizumab, and anti-IL-4R: dupilumab during this study (2008–2015). Finally, a population-based study cannot clarify the actual mechanism of the association between DPP-4i use and asthma in patients with T2DM, and further efforts in this direction are required.
Conclusion
In conclusion, DPP-4i use was associated with a 34% lower risk of asthma development in a Taiwanese population with T2DM compared with that in the non-DPP-4i use group. In addition, DPP-4i use has also associated with protected against the development of asthma of different severities with a class effect among No-AE, AE, status of asthma events, but not in ET-tube intubated events. In brief, our report suggests that DPP-4i may play a role in attenuating the impact of asthma on incidence in the future and on more severe forms of disease exacerbation in T2DM patients.
