Abstract
Background:
Information about angiotensin II (Ang II), angiotensin-converting enzyme 2 (ACE2), and Ang-(1–7) levels in patients with COVID-19 is scarce.
Objective:
To characterize the Ang II–ACE2–Ang-(1–7) axis in patients with SARS-CoV-2 infection to understand its role in pathogenesis and prognosis.
Methods:
Patients greater than 18 years diagnosed with COVID-19, based on clinical findings and positive RT-PCR test, who required hospitalization and treatment were included. We compared Ang II, aldosterone, Ang-(1–7), and Ang-(1–9) concentrations and ACE2 concentration and activity between COVID-19 patients and historic controls. We compared baseline demographics, laboratory results (enzyme, peptide, and inflammatory marker levels), and outcome (patients who survived versus those who died).
Results:
Serum from 74 patients [age: 58 (48–67.2) years; 68% men] with moderate (20%) or severe (80%) COVID-19 were analyzed. During 13 (10–21) days of hospitalization, 25 patients died from COVID-19 and 49 patients survived. Compared with controls, Ang II concentration was higher and Ang-(1–7) concentration was lower, despite significantly higher ACE2 activity in patients. Ang II concentration was higher and Ang-(1–7) concentration was lower in patients who died. The Ang II/Ang-(1–7) ratio was significantly higher in patients who died. In multivariate analysis, Ang II/Ang-(1–7) ratio greater than 3.45 (OR = 5.87) and lymphocyte count ⩽0.65 × 103/µl (OR = 8.43) were independent predictors of mortality from COVID-19.
Conclusion:
In patients with severe SARS-CoV-2 infection, imbalance in the Ang II–ACE2–Ang-(1–7) axis may reflect deleterious effects of Ang II and may indicate a worse outcome.
Introduction
Angiotensin-converting enzyme 2 (ACE2) is an enzyme (carboxypeptidase) that exists both as a membrane-associated form and as a secreted form. ACE2 was first discovered in 2000, and it is homologous to ACE1 being 42% sequence identity and 61% sequence similarity. ACE2 contains as ACE a single zinc-binding domain HEXXH in the active site, and it is not inhibited by ACE inhibitors.1–3
ACE2 cleaves angiotensin (Ang) I and II into angiotensin 1–9 [Ang-(1–9)] and angiotensin 1–7 [Ang-(1–7)], respectively. These peptides were described as cardiovascular protectors, able to regulate vascular tone, blood pressure, electrolyte balance, and water intake.1,4,5 The enzyme has an important role in heart failure, in diabetic, 6 and in inflammatory lung disease. 7 When bound to its Mas receptor, Ang-(1–7) induces vasodilation and exerts anti-inflammatory, anti-hypertrophic, and anti-fibrotic activities.3,4,8
Given its interaction with the viral S protein of SARS-CoV-2, ACE2 has been identified as the receptor for SARS-CoV-2, the infectious agent responsible for the COVID-19 pandemic. This finding established a critical link between ACE2, immunity, inflammation, and lung and cardiovascular diseases.4,8,9 By binding to ACE2, the virus decreases the tissue activity of ACE2, which aggravates SARS-CoV-2-induced inflammation in various organs, notably the lung.9,10 The role of the Ang II–ACE2–Ang-(1–7) axis in the physiopathology of SARS-CoV-2 infection, however, remains speculative.3,4,8,9,11 High levels of Ang II have been found to correlate with viral load. 12 The clinical findings for patients with COVID-19 point mainly to an increase in Ang II concentration and a lack of activity of Ang-(1–7), which suggest that blockage of ACE2 is involved in the pathogenesis of COVID-19, as our group has reported for patients with pulmonary arterial hypertension. 13
Information about Ang II and Ang-(1–7) levels and ACE2 activity in patients with COVID-19 is scarce and controversial.14–24 In this study, we aimed to characterize the Ang II–ACE2–Ang-(1–7) axis in patients with SARS-CoV-2 infection to understand better its role in pathogenesis. We hypothesized that, compared with healthy people, patients with COVID-19 will have higher levels of Ang II, lower ACE2 activity, and therefore lower Ang-(1–7) levels and that the values will correlate with the severity and outcome of the disease. We measured the serum concentrations of other protective angiotensins such as Ang-(1–9) and explored the interactions of these factors with the markers of inflammation described in this population.12,25,26
Methods
Study design
This was a single-center cohort study to assess the role of the Ang II–ACE2–Ang-(1–7) axis in the pathogenesis of SARS-CoV-2 infection. This study was performed at the National Institute of Cardiology of Mexico, an academic center for tertiary care devoted to the study and management of cardiovascular diseases and allied conditions. As the COVID-19 pandemic evolved, our hospital converted the emergency department and cardiovascular critical care unit into areas dedicated to the critical care of COVID-19 patients.
Study participants
The included patients were older than 18 years and had been diagnosed with COVID-19, based on clinical and radiological findings and a positive diagnostic test for SARS-CoV-2 infection [reverse transcription polymerase chain reaction (RT-PCR)] from a nasopharyngeal swab at admission and who required hospitalization and treatment at our institution between April 12 and July 20, 2020. Our hospital received only seriously ill patients, and patients with milder forms of the disease were transferred to less specialized medical centers and therefore were not included in the present analysis. A negative rapid influenza test was obtained for all patients.
Blood samples obtained from all patients upon admission were sent to the immunology laboratory where they were immediately processed for the measurement of inflammatory markers. 26 Only patients from whom we received blood samples for the measurement of the levels of interleukin 6 (IL-6) and other inflammatory markers were included. The serum of these samples was used for the measurement of different components of the Ang II–ACE2–Ang-(1–7) axis. Techniques for the measurements of enzymes and peptides are described in the supplemental material. Briefly, Ang II and Ang-(1–7) levels were measured by capillary zone electrophoresis as described by Tenorio and colleagues. 27 Aldosterone concentration was measured by ELISA [Aldosterone ELISA Kit (Cat. ADI-900-173, Enzo Life Sciences, Lausen, Switzerland)], Ang-(1–9) concentration was determined by using a commercial kit [Angiotensin-(Ang 1–9) ELISA Kit, MYBioSource, San Diego, CA, USA, Cat. MBS2022456], ACE2 concentration was determined by using a commercial kit (ACE2 human ELISA Kit, AdipoGen International, Palo Alto, IL, USA/Cat. AG-45A-0022EK-KIO1), and ACE2 activity was determined enzymatically by using a commercial kit (ACE2-human ELISA Kit, Sensolyte, AnasPecInc., Seraing, Belgium/Cat. 72086). For all these measurements, the sera were processed according to the evaluation protocol established by the manufacturers. For the measurement of antibodies directed against ACE2, we used a modification of the method described by Takahashi and colleagues 28 (see supplemental material).
For comparison, we used the normal levels of the Ang II–ACE2–Ang-(1–7) axis obtained before the pandemic started from carefully selected healthy blood donors who were free of cardiovascular disease from our institutional blood transfusion biobank and reported in a previous study. 13
Demographic data, in-hospital evolution, and outcome were obtained from each patient’s electronic record. Clinical and laboratory data were obtained independently from the electronic medical record by two investigators (JG-F, CG-A), and the resulting databases were compared and reviewed by a third investigator (LMA-G). Discrepancies were resolved by reviewing each discordant medical record again. At hospital admission, patients were classified according to the World Health Organization (WHO) guidelines as having moderate or severe disease. Moderate disease was defined as clinical signs of pneumonia such as fever, cough, dyspnea, and tachypnea but no sign of severe pneumonia, in particular arterial oxygen saturation (SaO2) ⩾90% on room air. Severe disease was defined as clinical signs of pneumonia plus one of the following: respiratory rate greater than 30 breaths/min, severe respiratory distress, or SaO2 less than 90% on room air. 29
All treatments, imaging and laboratory studies, admission to the intensive care unit, and indications for mechanical ventilatory support were performed at the discretion of each of the treating physicians. Similarly, the decision to discharge to home was made solely by the treating physician according to the clinical status of each patient. In their admission to the hospital, patients signed an informed consent form, and the investigation and ethics committees of the Ignacio Chávez National Institute of Cardiology (Mexico City, Mexico) approved the study (project number 20-1186).
Statistical analysis
We compared the concentrations of Ang II, aldosterone, Ang-(1–7), Ang-(1–9), Ang II/Ang-(1–7) ratio, and ACE2 concentration and activity between COVID-19 patients and the values from historic controls. For patients, we compared baseline demographic characteristics, laboratory results (levels of enzymes, peptides, and inflammatory markers) according to severity, and outcome (patients who survived versus those who died from COVID-19).
All categorical variables are presented as frequencies and percentages. All continuous variables were tested using the Kolmogorov–Smirnov test to determine whether they had a normal or nonnormal distribution. Continuous variables with a nonnormal distribution are reported as medians and 25th and 75th percentiles (interquartile range). Significant differences between groups were identified using chi-square or Fisher exact probability tests for categorical variables and the Mann–Whitney U test for continuous variables.
To determine the optimal cutoff values for the Ang II/Ang ratio for predicting in-hospital mortality, receiver operating characteristic (ROC) curve analysis was performed, and the exact value was determined using Youden’s index (sensitivity [1 − specificity]), which is a discriminatory cutoff method for diverse biomarkers. 30
A multivariable regression model with backward selection was then used to adjust for potential confounders based on established associations between clinical and laboratory markers and in-hospital mortality. Candidate covariates included in the multivariate analysis were selected from clinical variables and laboratory markers. These included the values for variables in the Ang II–ACE2–Ang-(1–7) axis at the time of admission that were associated with mortality in a univariate analysis with p ⩽ 0.05. Treatment strategies and outcome variables, such as mechanical ventilation or thrombosis, were not introduced in the analysis. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated. All analyses were two-tailed, and a value of p < 0.05 was accepted as significant. GraphPad Prism statistical software (v. 9; GraphPad Software, La Jolla, CA) and IBM SPSS Statistics (v. 20; IBM Corp., Armonk, NY) were used for the analyses.
Results
Serum samples from 74 patients [age: 58 (48–67.2) years; 68% men] with moderate (20%) or severe (80%) COVID-19 were analyzed. During their hospital stay of 13 (10–21) days, 25 (34%) patients died as a consequence of the disease and 49 (66%) patients survived. The demographic, clinical, and laboratory data; treatment trends; and outcomes of the total group as well as those who died or survived are shown in Table 1. Prior history of disease included hypertension (50%), diabetes (34%), and chronic kidney disease (18%). Before admission, some patients had been treated with ACE inhibitors (18%) or angiotensin receptor blockers (ARBs) (24%). At admission, most patients were tachycardic, tachypneic, and febrile and had low SaO2 [84% (73–87); the normal value for Mexico City is 93 ± 2%]. Leukocytosis and lymphopenia, which are characteristics of COVID-19, were observed in these patients as were abnormalities in other serum markers such as C-reactive protein, D-dimer, ferritin, and albumin.25,26 The creatinine level was also elevated.
Demographic, clinical, functional, and laboratory variables and outcome characteristics of patients with COVID-19.
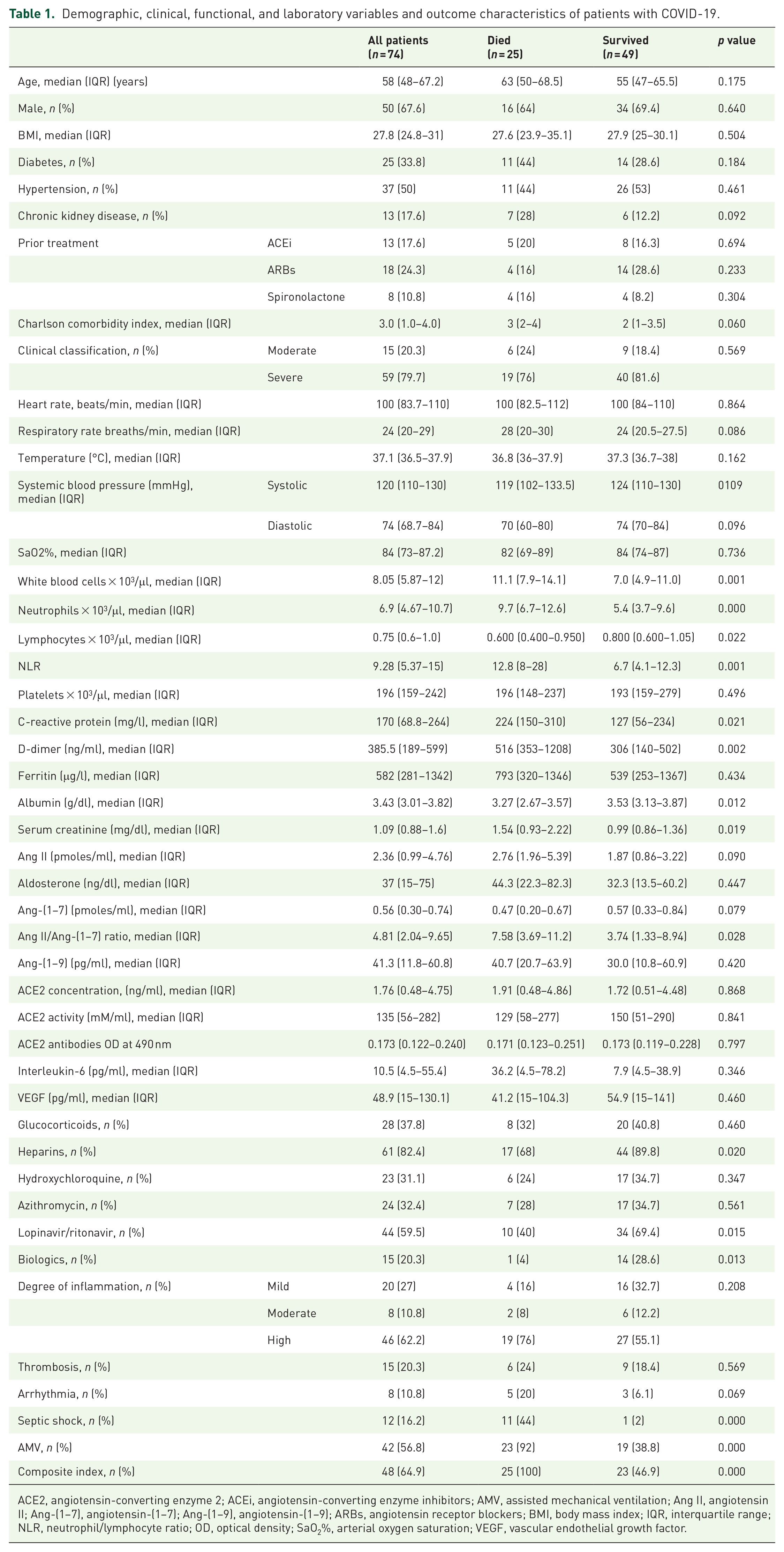
ACE2, angiotensin-converting enzyme 2; ACEi, angiotensin-converting enzyme inhibitors; AMV, assisted mechanical ventilation; Ang II, angiotensin II; Ang-(1–7), angiotensin-(1–7); Ang-(1–9), angiotensin-(1–9); ARBs, angiotensin receptor blockers; BMI, body mass index; IQR, interquartile range; NLR, neutrophil/lymphocyte ratio; OD, optical density; SaO2%, arterial oxygen saturation; VEGF, vascular endothelial growth factor.
The Ang II levels were elevated and Ang-(1–7) levels were low, despite a significant increase in ACE2 activity in COVID-19 patients compared with our previous determinations in healthy people. 13 Antibodies against ACE2 were also higher in COVID-19 patients (Table 2).
Blood serum level measurements of angiotensin (Ang) II, aldosterone, Ang-(1–7), Ang-(1–9), angiotensin-converting enzyme 2 (ACE2) concentration, and ACE2 activity in COVID-19 patients and control subjects.
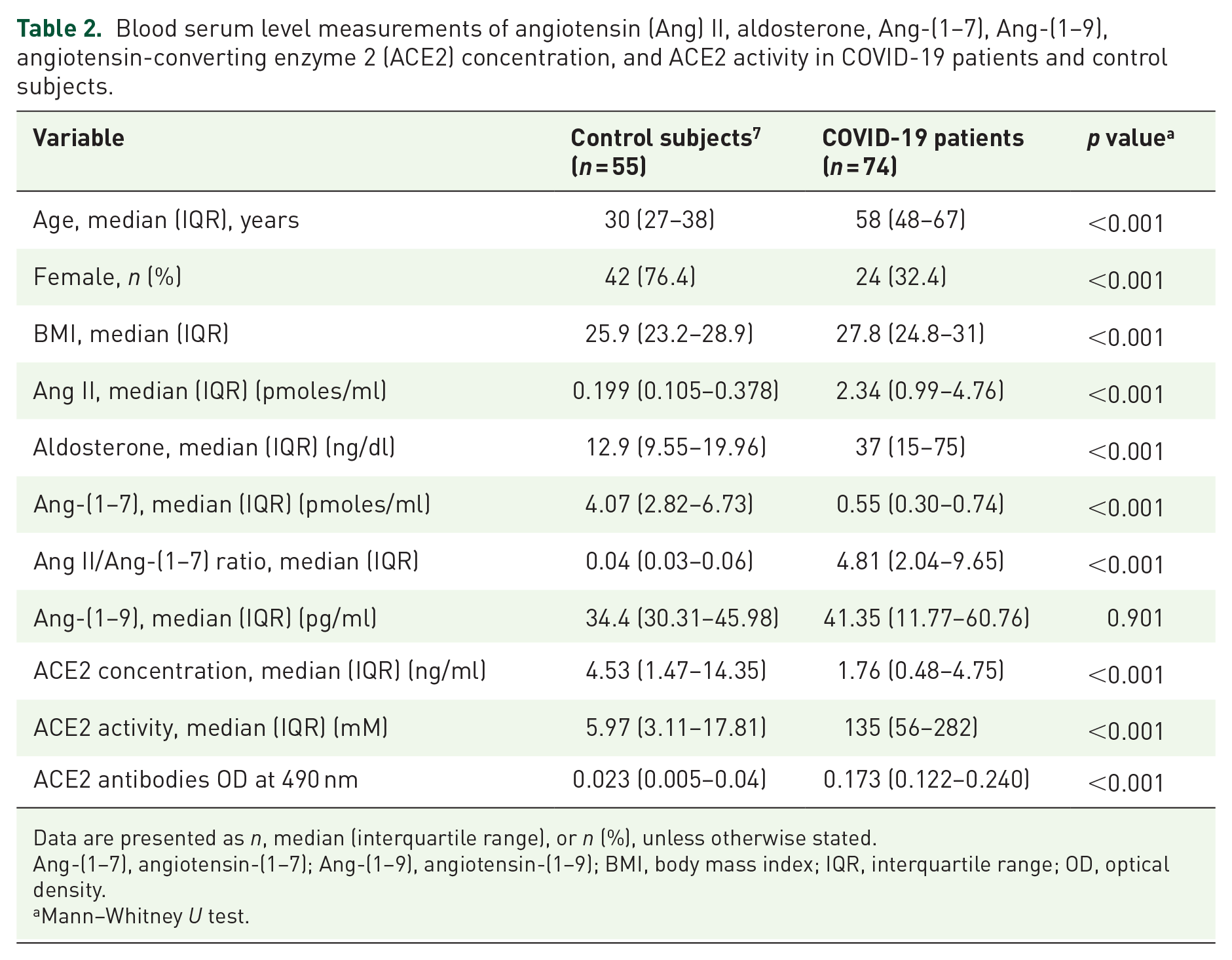
Data are presented as n, median (interquartile range), or n (%), unless otherwise stated.
Ang-(1–7), angiotensin-(1–7); Ang-(1–9), angiotensin-(1–9); BMI, body mass index; IQR, interquartile range; OD, optical density.
Mann–Whitney U test.
Because of the severity of the disease, 57% of patients required mechanical ventilation, 20% had evidence of thrombosis, 16% developed shock, 13.5% developed arrhythmias, and, as mentioned above, 34% died during hospitalization.
Differences between patients who died and who survived are shown in Table 1. The Charlson comorbidity index and leukocyte count were higher and lymphocyte count was lower in patients who died. The neutrophil/lymphocyte ratio (NLR) and levels of C-reactive protein, D-dimer, and creatinine were significantly higher and serum albumin concentration was significantly lower in patients who died. Markers of inflammation and proliferation, such as IL-6 and vascular endothelial growth factor concentrations, did not differ between patients who survived and those who died. The percentages of patients who developed septic shock and needed mechanical ventilation were significantly higher in patients who died.
Ang II concentration tended to be higher and Ang-(1–7) concentration tended to be lower in patients who died. The Ang II/Ang-(1–7) ratio was significantly higher in patients who died. Ang-(1–9) and ACE2 concentrations, ACE2 activity, ACE2 antibody levels, and aldosterone concentration did not differ significantly between patients who died and those who survived (Table 1 and Figure 1). These results were not related to the previous use of ACE inhibitors, ARBs, or spironolactone. Regarding ACE inhibitors, the results were [Ang II: 2.028 (0.989–4.107) versus 2.428 (0.998–4.788), p = 0.665; Ang-(1–7): 0.608 (0.431–0.725) versus 0.497 (0.251–0.791), p = 0.394; ratio: 3.53 (2.15–7.66) versus 4.99 (2.01–10.37), p = 0.375; ACE2 concentration: 2.15 (0.567–4.704) versus 1.72 (0.487–4.797), p = 0.881; ACE2 activity: 171.1 (44.27–265.9) versus 132 (58.2–322.3), p = 0.717] in patients with and without ACE inhibitors, respectively.
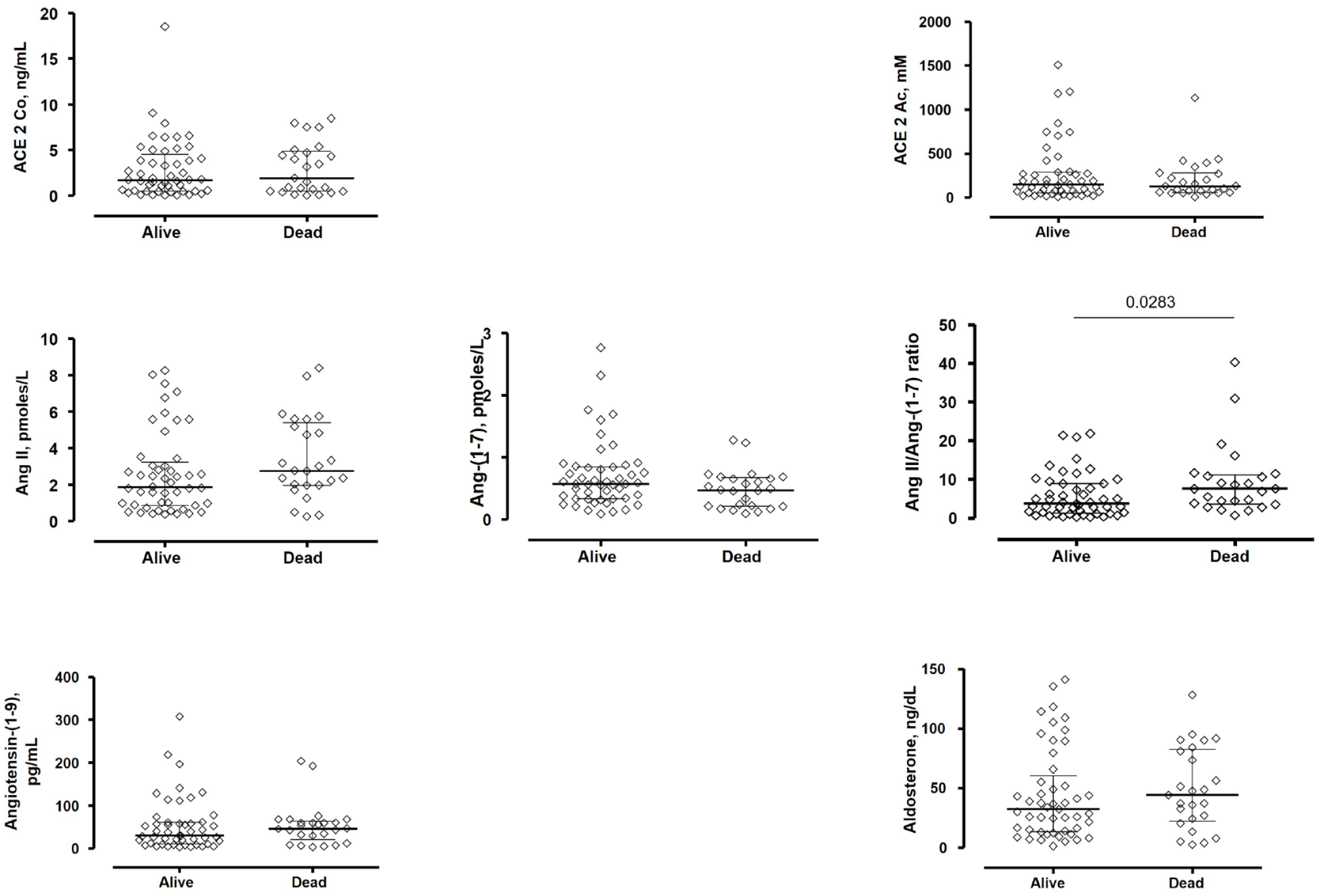
Peptide and enzyme differences between patients who survived and died from SARS-CoV-2 infection. Serum concentration of angiotensin-converting enzyme 2 (ACE2), ACE 2 concentration (ACE 2 Co), ACE 2 activity (ACE 2 Ac), angiotensin II (Ang II), Ang-(1–7), Ang II/Ang-(1–7) ratio, concentration of Ang-(1–9), and aldosterone. The difference in the Ang II/Ang-(1–7) ratio was significant.
To explore further the role of the Ang II/Ang-(1–7) ratio, we performed ROC curve analysis to determine the optimal cutoff for this ratio for predicting in-hospital mortality. The value of 3.45 (C statistic = 0.65) was determined using Youden’s index (Figure 2). ACE2 activity differed significantly between patients with an Ang II/Ang-(1–7) ratio above and below 3.45 [0.294 mM/ml (0.225–0.745) versus 0.078 mM/ml (0.052–0.150), respectively; p < 0.001]. The correlation between ACE2 activity and Ang II/Ang-(1–7) ratio was also significant (Figure 3).
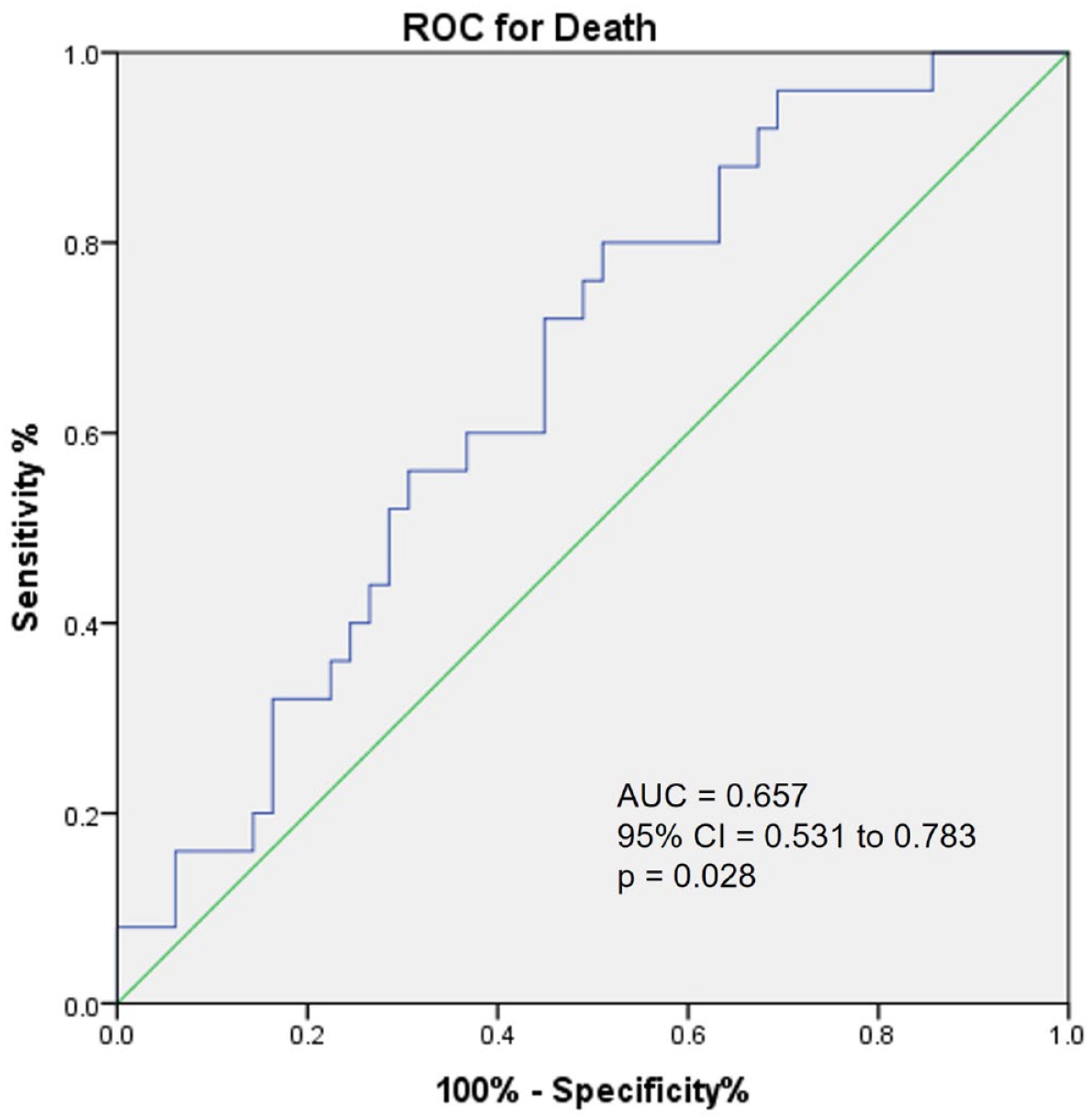
Receiver operating characteristic (ROC) curve showed only a fair but significant performance (AUC = 0.657) for the accuracy of the Ang II/Ang-(1–7) ratio > 3.45 for predicting death during hospitalization of patients with COVID-19.

Pearson correlation between ACE 2 activity and Ang II/Ang-(1–7) ratio. The Ang II/Ang-(1–7) ratio decreased with increasing ACE 2 activity.
The results of the univariate and multivariate analyses are shown in Tables 3 and 4. After adjusting for confounding variables, patients with a value of ⩾3.45 for the Ang II/Ang-(1–7) ratio had a fivefold higher risk of in-hospital mortality (95% CI = 1.224–20.889; p = 0.025). Another factor that was independently associated with increased in-hospital mortality was lymphopenia or a cell count ⩽0.65 × 103/µl (OR = 8.432, 95% CI = 1.384–51.385; p = 0.021) (Table 4).
Independent predictors of mortality in patients with COVID-19: univariate analysis.
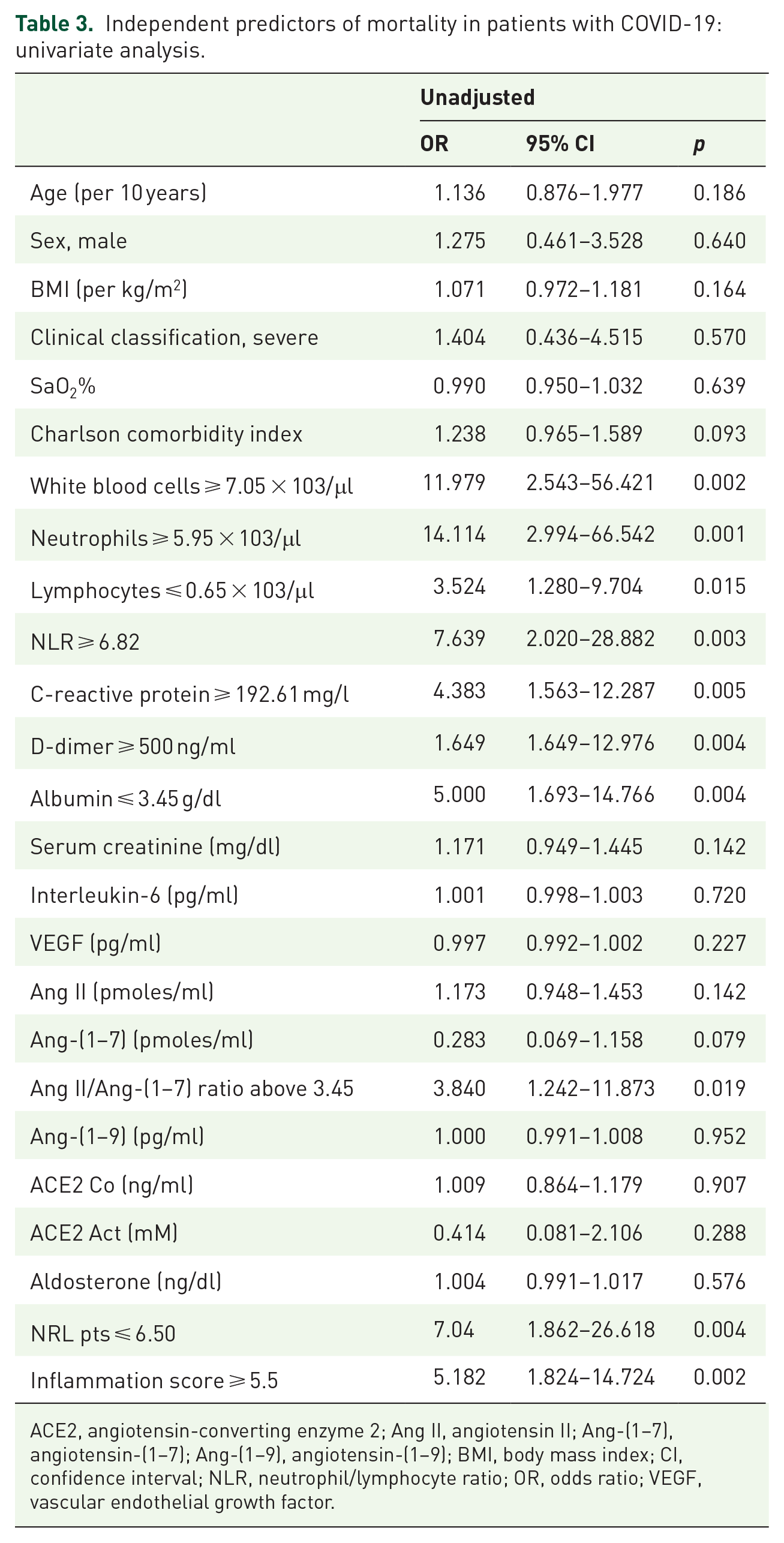
ACE2, angiotensin-converting enzyme 2; Ang II, angiotensin II; Ang-(1–7), angiotensin-(1–7); Ang-(1–9), angiotensin-(1–9); BMI, body mass index; CI, confidence interval; NLR, neutrophil/lymphocyte ratio; OR, odds ratio; VEGF, vascular endothelial growth factor.
Independent predictors of mortality in patients with COVID-19: multivariate analysis.

CI, confidence interval; OR, odds ratio.
Discussion
In this study, our analysis included the most important components of the Ang II–ACE2–Ang-(1–7) axis in patients with SARS-CoV-2 infection. We found an imbalance in this axis in favor of the deleterious effects of Ang II, as reflected by a higher mortality in patients with an Ang II/Ang-(1–7) ratio higher than the cutoff value of 3.45 [OR = 5.87 (1.224–20.889); p = 0.025]. Another factor that was independently associated with increased in-hospital mortality was lymphopenia or a lymphocyte count ⩽0.65 × 103/ml (OR = 8.432, 95% CI = 1.384–51.385; p = 0.021).
Other studies have also explored the Ang II–ACE2–Ang-(1–7) axis in COVID-19.14–24 They have focused mainly on the isolated ACE2 concentration or activity and their potential contributions to the severity of the disease and have produced conflicting results. Some studies have found that an elevated ACE2 concentration or activity correlates with a worst outcome,14–19 whereas others have not found elevated concentrations compared with controls or differences in terms of severity or outcome.20–24
The recent study of Reindl-Schwaighofer and colleagues 14 is interesting. These authors measured Ang II and Ang-(1–7) concentrations and ACE2 activity in patients with COVID-19 at admission and up to 20 days of hospitalization. They found that the concentration of Ang II decreased and the concentration of Ang-(1–7) increased over time and that these changes paralleled the increase in ACE2 activity, particularly in those with severe disease. These findings suggested a shift toward a potentially protective alternative renin-angiotensin-aldosterone system (RAAS) given that, at the peak of ACE2 concentration (day 10), 29 of 32 patients with severe COVID-19 remained alive. The Ang-(1–7)/Ang II ratio increased from 7% (early) to 31% (late) in patients with severe COVID-19, which suggests a beneficial increase in the formation of Ang-(1–7) from Ang II. In patients with no severe COVID-19, no statistically significant increase in the concentration/activity of alternative RAAS metabolites was observed. Another recent study 15 also reported an elevated ACE2 activity in COVID-19 and a greater increase in patients with severe disease and that ACE2 activity remained elevated during the disease evolution. These two studies and others18,19 emphasize the importance of the follow-up of these variables during the evolution of the disease to define better the pathophysiological role of ACE2 in SARS-CoV-2 infection.14,15,18,19
The results in our study are in contrast to those of Valle-Martins 31 who found an increase in Ang-(1–7) and a decrease in Ang II and Ang-(1–5) as compared to controls. In this study, peptides were measured by an appropriate technique in the arterial blood, but the sample size (n = 19) and the absence in the measurement of ACE2 concentration and activity precludes its interpretation. In another article from the same group of investigators, Pucci and colleagues 32 performed a manual meta-analysis of the reported series performed until then and found that indeed Ang-(1–7) was elevated at baseline compared to controls. They also found that soluble ACE2 was also elevated, but its activity remained constant. In their analysis, Ang II concentration was found deceased in half of the series and elevated in the other half. In these two articles, alternate pathways such as prolyl oligopeptidase (POP) 33 are used to explain the increase in Ang-(1–7). It is recognized, however, that in an article by Files and colleagues, 23 no difference was observed between POP activity in COVID-19 patients and controls.
Although not statistically different, Ang II concentration tended to be higher and Ang-(1–7) concentration tended to be lower in our patients who died. The Ang II/Ang-(1–7) ratio was significantly higher in patients who died. The enzymatic/peptide profile favoring Ang II pathway in our study corresponds to the unbalanced renin-angiotensin system (RAS) as a possible dysregulated RAS scenario proposed by Pucci and colleagues. 32 It is important to establish that in our study, these two important effectors of the axis were measured by capillary zone electrophoresis in plasma and not by ELISA because of the established limitations of this technique. 34
In our study, we found no differences in Ang II, Ang-(1–7), Ang-(1–9), and ACE2 concentrations or activity at the baseline between patients who survived and those who died (Figure 2). Interestingly, however, ACE2 activity differed significantly according to the cutoff value of 3.45 for the Ang II/Ang-(1–7) ratio. This finding suggests that ACE2 is related to the final Ang II/Ang-(1–7) ratio. The role of ACE2 activity is also emphasized by the significant correlation between ACE2 activity and Ang II/Ang-(1–7) ratio, as shown in Figure 3. It should be acknowledged that, in the metabolism of Ang II and its conversion to Ang-(1–7), it takes a significant amount of ACE2 to convert Ang II to Ang-(1–7) and that such levels are present in the tissue but usually not in the blood where activity is low.35,36 Most recent studies in COVID-19, however, have assessed ACE2 concentration and activity in blood but not directly in tissue.14–16,18–24
Although our study does not contribute to a more complete understanding of the role of the Ang II–ACE2–Ang-(1–7) axis in the physiopathology of COVID-19, it underlines the importance of a more complete evaluation of the RAAS metabolites on admission as an indicator of a patient’s prognosis. This knowledge may be useful in evaluating new therapeutic options.37–42
Limitations
Our study is limited by the retrospective nature of the analysis. The sampling of most analytes, however, was performed carefully and simultaneously at admission irrespective of the patient outcomes. Other limitations include the lack of appropriate controls including subjects with similar comorbidities or inclusion of patients with mild or asymptomatic COVID-19, lack of serial determinations of analytes during disease evolution, lack of measurements of other important components of the Ang II–ACE2–Ang-(1–7) axis, and, finally, lack of information regarding viral-related variables such as the viral load.
Conclusion
The Ang II–ACE2–Ang-(1–7) axis is abnormally balanced toward a predominant and deleterious role of Ang II and may be associated with disease severity and outcome in patients with COVID-19.
Supplemental Material
sj-docx-1-tar-10.1177_17534666221122544 – Supplemental material for The prognostic importance of the angiotensin II/angiotensin-(1–7) ratio in patients with SARS-CoV-2 infection
Supplemental material, sj-docx-1-tar-10.1177_17534666221122544 for The prognostic importance of the angiotensin II/angiotensin-(1–7) ratio in patients with SARS-CoV-2 infection by Luis M Amezcua-Guerra, Leonardo del Valle, Héctor González-Pacheco, Rashidi Springall, Ricardo Márquez-Velasco, Felipe Massó, Malinalli Brianza-Padilla, Daniel Manzur-Sandoval, Julieta González-Flores, Carlos García-Ávila, Yaneli Juárez-Vicuña, Fausto Sánchez-Muñoz, Martha A Ballinas-Verdugo, Edna Basilio-Gálvez, Araceli Paez-Arenas, Mauricio Castillo-Salazar, Sergio Cásares-Alvarado, Adrián Hernández-Diazcouder, José L Sánchez-Gloria, Claudia Tavera-Alonso, Rodrigo Gopar-Nieto and Julio Sandoval in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
