Abstract
Background:
Infection due to nontuberculous mycobacteria (NTM) is an emerging issue worldwide, and we aimed to address the epidemiology and mortality association of NTM infection requiring treatment in Taiwan.
Methods:
We used the 2003–2018 data of 2 million representative individuals in Taiwan’s National Health Insurance Research Database. We identified patients with newly diagnosed NTM infection and received treatment as NTM cases. Age- and sex-matched (1:40) as well as propensity score-matched (PSM) (1:2) non-NTM individuals were selected as non-NTM controls. We used a Cox proportional hazard model to determine hazard ratios (HRs) with 95% confidence intervals (CIs).
Results:
We identified 558 patients with NTM infection requiring treatment. The mean age was 62.5 ± 15.4 years, and 57.5% of them were male. The incidence increased from 0.54 per 100,000 person-year in 2003 to 3.35 per 100,000 person-year in 2018. The overall mortality was 35.2%, with a mean follow-up duration of 4.1 ± 3.6 years. We found that NTM infection was independently associated with a greater risk of mortality (HR: 1.71; 95% CI: 1.47–1.98) compared with age- and sex-matched controls, and the association remained consistent (HR: 1.44; 95% CI: 1.19–1.75) compared with propensity-matched controls. We also found that old age, male, high Charlson comorbidity index, and the use of steroids or anti-neoplastic agents/immunosuppressants were associated with mortality risk.
Conclusion:
In conclusion, we found a steady increase in patients with NTM infection requiring treatment in Taiwan and further demonstrated that NTM infection was associated with greater risk of mortality using two comparable non-NTM control subjects.
Background
Nontuberculous mycobacteria (NTM) infection is currently an emerging health issue with a steady increase in both the incidence and prevalence worldwide.1–3 A number of studies have shown a globally increased burden of NTM infection in the past two decades, particularly in the elderly and immunocompromised population; however, the factors associated with mortality among patients with NTM infection are not completely understood, mainly due to the lack of comparable control subjects in most previous studies.4–7
Environmental NTMs are ubiquitous pathogens and have been isolated from water supplies, soils, and healthcare facilities worldwide. 8 The global increased incidence and prevalence of NTM infection may be attributed to the increase in susceptible hosts, including the elderly as well as immunocompromised patients, improved awareness of NTM infection, advanced detection methods, and changing environmental exposures.9,10 Furthermore, it has been estimated that NTM infectious may continue to rise due to the continuous increase of the elderly population in countries including Taiwan.11,12
NTM infection can be classified as positive NTM isolates and NTM disease, defined by fulfilling the diagnostic criteria, which consist of both clinical and microbiologic evidence. 13 However, the reported NTM disease among those with positive NTM isolates ranged from 20% to 60%, and the underlying comorbidities have been implicated with NTM disease, indicating the crucial need to investigate the clinical impact of NTM infection in patients with underlying comorbidities.14–16 Indeed, the start of treatment for NTM infection is a clinical dilemma due to the need for prolonged and combinational anti-mycobacterial agents as well as drug–drug interaction in the elderly with multiple comorbidities; however, data regarding long-term mortality among patients requiring treatment for NTM infection remain scarce.11,17 Therefore, there is a crucial need to address the long-term outcomes and to clarify the associations between mortality and NTM infection requiring treatment in aged patients with comorbidities.
In this study, we used a population-based claim data set to investigate the epidemiological trends, to address the long-term mortality, and to identify risk factors of long-term mortality among patients with NTM infection requiring treatment between 2003 and 2018 in Taiwan.
Materials and methods
Study design and data source
Taiwan launched a single-payer National Health Insurance (NHI) program on 1 March 1995. As of 2018, 99.82% of Taiwan’s population were enrolled. 18 The National Health Insurance Research Data (NHIRD) serves as the database of the NHI and contains original claims data for reimbursement. The Bureau of NHI (BNHI) manages the NHIRD and releases the data for research purposes. Notably, the NHIRD provide a representative database of 2,000,000 individuals randomly selected from all NHI enrollees who received medical services in 2000 (Longitudinal Health Insurance Database, LHID2000). In this study, the ambulatory, inpatient, and enrollment data from the 2003–2013 NHIRD were used to identify all patients with new NTM infection among the 2,000,000 individuals within the LHID2000.
Definitions of NTM infection
We included adult patients with NTM infection, defined by both International Classification of Diseases: 9th Revision – Clinical Modification (ICD-9-CM) codes for NTM: 031.0, 031.1, 031.2, 031.8, and 031.9 and a concurrent prescription of at least two anti-mycobacterial drugs for at least 28 days within 12 months of the diagnosis. Anti-mycobacterial antibiotics consisted of conventional anti-mycobacterial medications (isoniazid, ethambutol, rifampicin, and pyrazinamide), new macrolides (clarithromycin and azithromycin), fluoroquinolones (ofloxacin, ciprofloxacin, levofloxacin, and moxifloxacin), aminoglycosides (streptomycin, amikacin, and kanamycin), tetracyclines (minocycline, doxycycline, and tigecycline), carbapenem (imipenem and meropenem), cefoxitin, and linezolid. 19 To avoid the confounding effect of co-existence or post-tuberculous NTM on post-NTM mortality, we excluded patients who had a diagnosis of tuberculosis infection.
Selection of comparable non-NTM subjects
Figure 1 illustrates the enrollment of NTM cases and comparable non-NTM control subjects. We enrolled adult individuals who had at least one ambulatory visit during 2003–2018 and applied the same exclusion criteria as those used for NTM cases, as follows: (1) a diagnosis of tuberculosis (n = 155,308) and (2) age lower than 20 years (53,777). A total of 1,646,706 individuals were eligible for inclusion in the control group. We then used age-sex matching and propensity matching in this study. We matched the NTM cases and non-NTM controls at a ratio of 1:40 for sex, age, and year of the index date. After matching, there were 558 patients in the NTM group and 22,320 individuals in the non-NTM group. Moreover, we aimed to reduce the impact of bias and confounding variables on mortality through propensity-score matching (PSM), which was conducted at a ratio of 1:2 for sex, age, index date, and comorbidities. In the propensity-matched subjects, we identified 558 NTM patients and 1116 control individuals without NTM.
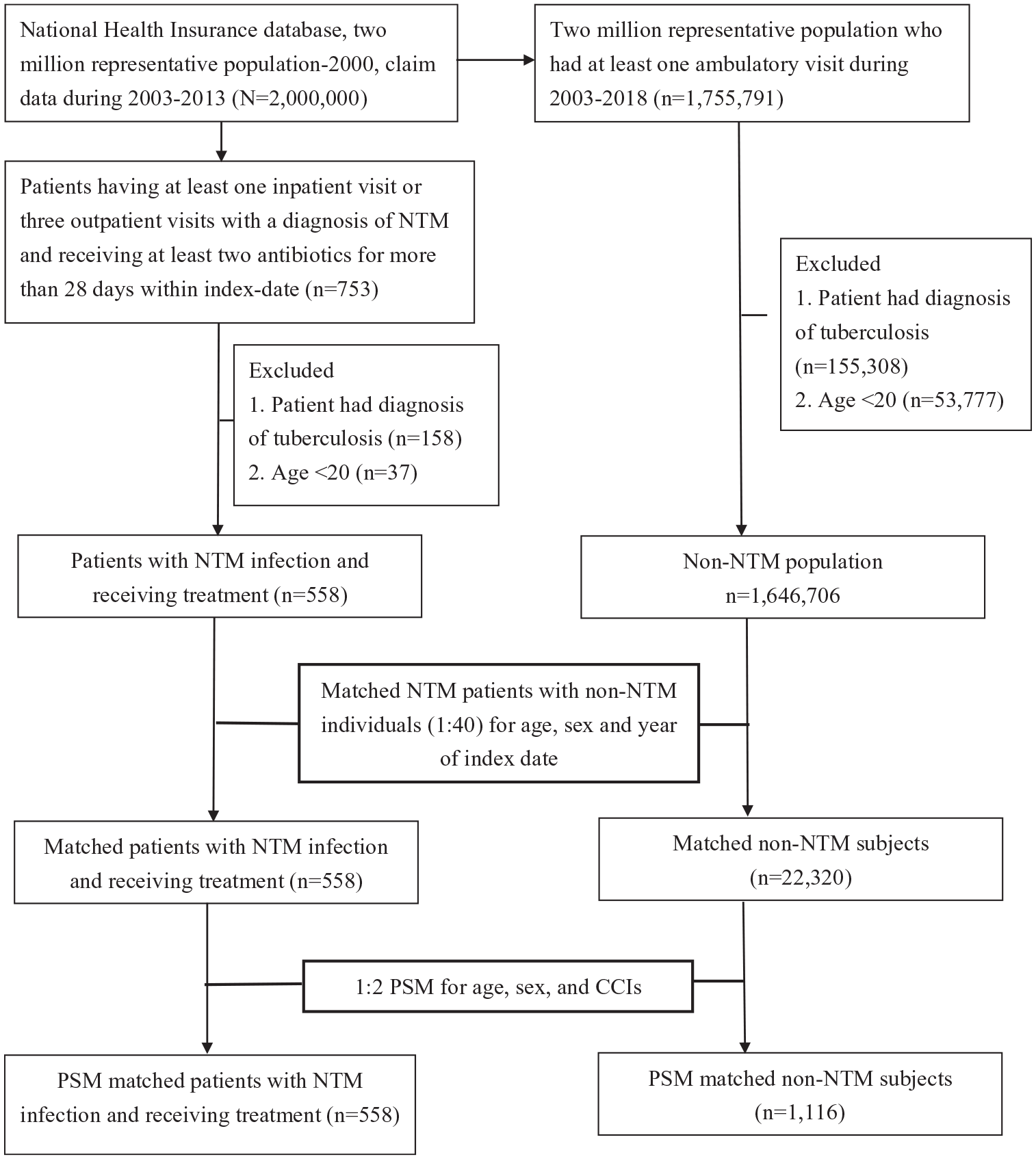
Flow chart of the study design for investigating the impact of NTM infection on mortality.
Potential confounders
Potential confounders that were adjusted for in the logistical regression model included age, sex, and Charlson comorbidity index (CCI). The presence of comorbidity was defined as having at least three ambulatory visits or one inpatient visit with a corresponding ICD-9-CM code within 1 year before the index date. The CCI, as adapted by Deyo et al., 20 was used to evaluate the level of general comorbid medical conditions.
Statistical analysis
The demographic data are presented as a mean ± standard deviation for continuous variables and as a percentage of patients in categorical variables. The differences were analyzed using Student’s t-test for continuous variables and Pearson’s χ2 test for categorical variables. We calculated the annual incidence, new cases divided by the population in a given year, between 2003 and 2018, and further calculated age- and sex-specific incidence. The survival time curve was plotted to illustrate the trend of long-term survival among patients with NTM infection requiring treatment. A Cox proportional hazard model was conducted to estimate the hazard ratio (HR) of mortality in patients with NTM infection after adjustment for sex. All the data were analyzed using Statistical Analysis Software version 9.3 (SAS Institute, Inc., Cary, NC, USA). A p value < 0.05 was considered statistically significant.
Results
A steady increased number of patients with NTM infection requiring treatment in Taiwan
A total of 558 subjects with NTM infection requiring treatment were identified (Table 1). Their mean age was 62.5 ± 15.4 years, and 57.5% of them were male. The majority of patients had comorbidities, and the proportion of CCI = 0, 1, 2, 3 and >3 were 22.9%, 32.4%, 18.8%, 9.9%, and 15.9%, respectively (see the detailed comorbidity in Supplemental Table 1). Moreover, the proportion of patients receiving steroid, anti-neoplastic agents, and immunosuppressants were 53.4%, 6.5%, and 4.5%, respectively. Figure 2 illustrates the annual standardized incidence rate of NTM infection requiring treatment in Taiwan between 2003 and 2018, and a steadily increased number of NTM infections requiring treatment was found in both males and females (Figure 2). The incidence increased from 0.54 per 100,000 person-year in 2003 to 3.35 per 100,000 person-year in 2018 (Table 2) (see details in supplemental Table 2). The prevalence also increased steadily from 0.68 per 100,000 person-year in 2003 to 7.17 per 100,000 person-year in 2018.
Baseline characteristics in patients with NTM infection receiving treatment and non-NTM control subjects.

CCI: Charlson comorbidity index; NTM: nontuberculous mycobacteria; PSM: propensity score matching.
Prednisolone equivalent.
Incidence rate and prevalence of patients with NTM requiring treatment during 2003–2018.

NTM: nontuberculous mycobacteria.

Incidence rate of patients with NTM infection and receiving treatment categorized by sex.
High incidence and mortality of NTM infection requiring treatment in the elderly and males
The growth of the elderly population is a major public health issue worldwide, and the continuous rise in the elderly population has been one of key issues of NTM infection in Taiwan.11,21 Hence, we were interested in investigating the age-associated incidence and mortality rate of NTM infection requiring treatment. Figure 3 shows not only high incidence but also high mortality of NTM infection in those 65 years or older, particularly in males. Taken together, these data showed a high incidence of NTM infection in the elderly in Taiwan, particularly among males.

Incidence rate (a) and mortality rate (b) of patients with NTM infection and receiving treatment categorized by sex and age groups.
Age-specific impact on the long-term outcome of NTM infection
In addition to the incidence and prevalence of NTM infection, we also aimed to address the long-term mortality of patients with NTM infection compared with the two comparable non-NTM subjects. We found that 35.1% of enrolled patients with NTM infection died, with a mean follow-up duration of 4.1 ± 3.6 years (Table 3). In the age- and sex-matched subjects, we found a higher incidence rate ratio (IRR) of mortality (IRR: 2.32, 95% CI: 2.01–2.68) in NTM cases compared with those in non-NTM control subjects. The IRR was 1.63 (95% CI: 1.36–1.96) when compared with the propensity-matched subjects. In addition to the higher risk seen in males (HR: 1.56; 95% CI: 1.47–1.66), we found that old age and high CCI categories were independently associated with the risk of mortality in a dose-dependent manner. Moreover, those who received prednisolone with a dose higher than 5 mg/kg (HR: 1.96; 95% CI: 1.71–2.25) or who took anti-neoplastic agents/immunosuppressants (HR: 2.37; 95% CI: 2.06–2.73) had a higher mortality risk. After adjustment for the potential confounders, including age, sex, comorbidities, and the use of steroids, as well as anti-neoplastic agents/immunosuppressants, we found that NTM infection was independently associated with mortality (HR: 1.71; 95% CI: 1.47–1.98; Table 4). In the propensity-matched subjects, the association between NTM and mortality remained consistent (HR: 1.44; 95% CI: 1.19–1.75).
Incident mortality in patients with NTM infection and non-NTM control subjects before and after PSM.

IRR: incidence rate ratio; NA: not applicable; NTM: nontuberculous mycobacteria; PSM: propensity score matching.
Crude and adjusted hazard ratios for the mortality in patients with NTM infection and non-NTM control subjects before and after PSM.
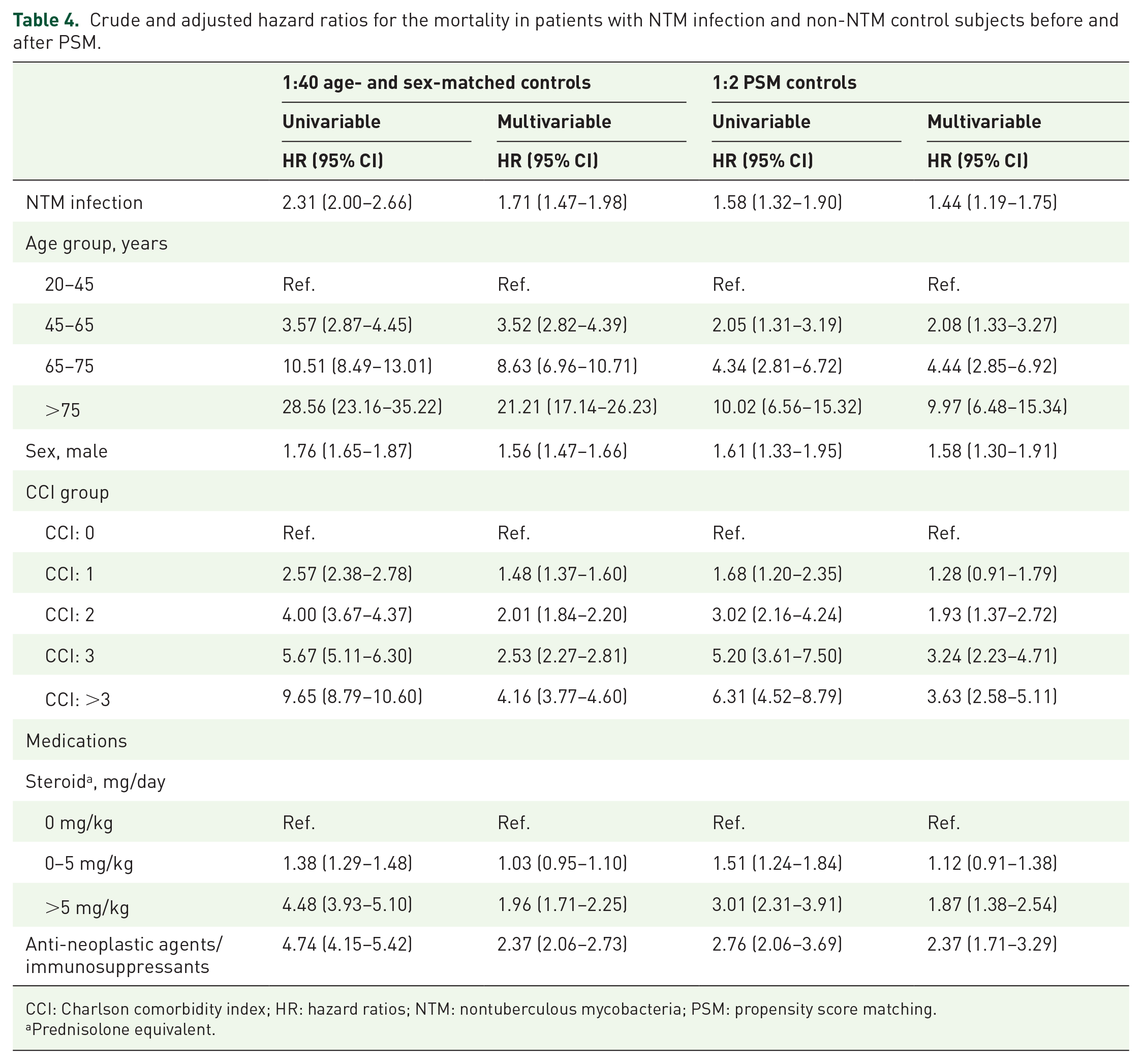
CCI: Charlson comorbidity index; HR: hazard ratios; NTM: nontuberculous mycobacteria; PSM: propensity score matching.
Prednisolone equivalent.
Discussion
NTM infection is currently a global health concern, and in this study, we investigated its incidence, prevalence, and mortality association among patients with NTM infection requiring treatment in Taiwan using a nationwide, population-based data set. We found a steady increase of patients with NTM infection requiring treatment in Taiwan during 2003–2018, particularly in the elderly, in males, and in the immunocompromised population. Notably, the overall mortality was 35.1% among patients with NTM requiring treatment with follow-up for approximately 4 years, and we further used two groups of comparable control subjects to delineate the independent impact of NTM infection on mortality. We believe that these findings provide crucial epidemiological evidence with respect to the increasing threat of NTM infection, particularly in countries with aging populations and growing numbers of immunocompromised patients.
Owing to the relatively high mortality in patients with NTM infection, there is a need to clarify the factors that affect the risk of mortality in NTM infection. Hayashi et al. investigated the prognostic factors among 634 patients with Mycobacterium avium complex (MAC) lung disease with a median follow-up of 4.9 years at the Saitama Cardiovascular and Respiratory Center in Japan and found that the overall mortality was 23.9%. 22 Similarly, Fleshner et al. analyzed 106 patients with NTM lung disease at the Clinical Center of the National Institutes of Health in the United States and found that the overall mortality was 25% (27/106) with a median follow-up of 4.9 years. 23 Moreover, Diel et al. 5 conducted a meta-analysis including 9035 patients with MAC lung disease from 17 studies and found that the 5-year mortality was approximately 27% (95% CI: 21.3–37.8%).
There is a wide range of characteristics among patients with NTM infection worldwide. Two recently published studies reported that patients with NTM infection in Korea appear to be young with a relatively low mortality. Jhun et al. 24 analyzed the data of 1445 patients with NTM lung disease in the NTM Registry of Samsung Medical Center and found that the 5-, 10-, and 15-year cumulative mortality rates for NTM patients were 12.4%, 24.0%, and 36.4%, respectively. The majority of the enrolled patients in the study by Jhun et al. 24 tended to be young, with 64.8% (936/1,445) younger than 65 years. Similarly, Lee et al. 7 conducted a population-based Korean study with comparable control subjects to determine the impact of NTM infection in the Korean population and reported that the 6-, 10-, and 14-year mortality rates of NTM infection were 13.7%, 19.2%, and 22.9%, respectively. It is interesting to note that the subjects with NTM infection enrolled by Lee et al. 7 were also young, with a mean age of 39.2 ± 20.9 years. Unlike the aforementioned Korean studies in which the 5-year mortality was approximately 12% among young patients with NTM pulmonary infection, Andrejak et al. 17 reported a 5-year mortality rate of 40.1% in a nationwide study conducted in Denmark which included 1282 patients with NTM infection whose age was approximately 60 years. The population of patients with NTM infection enrolled by Andrejak et al. appears to be similar to the studied population in this study, and the mortality was also consistent with long-term mortality, as we have shown in this study. The aforementioned studies in distinct countries have revealed the apparently high mortality in patients with NTM infection; however, the studies to clarify mortality impact of NTM infection is still lacking, given that the lack of comparable control subjects, particularly age-matched control subjects. In this study, we included two comparable control subjects matched for age, sex, as well as CCI, and determined the impact of NTM infection on mortality.
NTMs are highly pervasive microbes in the environment with distinct rates depending on the geographic regions. 8 Yano et al. investigated the genetic linkage between global Mycobacterium avium subsp. hominissuis (MAH) strains obtained from humans, animals, and the environment and demonstrated extensive mutual homologous recombination among subspecies of MAH, thereby providing molecular evidence of the linkage between NTM infection and environment. 25 The allelic variations among MAH strains across distinct countries reflect the geographic diversity of NTM infection and highlight the crucial need for region-specific epidemiological data. 25 In this Taiwanese population-based study, we found a steady increased incidence and prevalence of NTM infection requiring treatment in Taiwan, indicating the need for vigilance of NTM infection among the high-risk population in Taiwan.
A number of studies have explored the main NTM species in Taiwan, particularly among the elderly.26,27 Lai et al., 26 investigating 3175 NTM isolates among 1633 elderly Taiwanese patients, reported that merely 19.6% (326/1633) of patients met the criteria for NTM diseases, and in the majority (90.2%, 294/326) of elderly patients with NTM diseases, the diagnosis was NTM lung disease. The aforementioned finding is consistent with our data that the majority (92.2%) of enrolled patients with NTM infection requiring treatment in this study was NTM infection with lung involvement, including both pulmonary NTM infection (53.8%) as well as mixed pulmonary and extra-pulmonary NTM infection (38.4%) (Supplemental Table 3). With regard to NTM species, Lai et al. 26 reported that MAC (35.2%) was the most frequently isolated species, followed by rapid-growing mycobacteria, including M. abscessus (17.2%), M. fortuitum complex (14.5%), and M. chelonae complex 16 (12.7%) among the 3175 NTM isolates obtained from elderly patients in Taiwan. Lee et al. 26 recently analyzed relevant investigations of NTM in Taiwan, which were mainly hospital-based studies, and reported that the incidence of NTM disease has increased in the past decade, and MAC as well as M. abscessus were the main NTM species in Taiwan. 27 Lee et al. 26 concluded that there is a crucial need to address NTM disease burden in Taiwan by analyzing nationwide, population-based data. 27 This study provides evidence of increasing numbers of patients with NTM infection requiring treatment in Taiwan and our findings provide insights into the factors affecting the risk of mortality in patients with NTM infection by performing comparisons with two matched non-NTM populations.
In this study, enrollment of patients was restricted to those with NTM infection and receiving anti-mycobacterial treatment. Thus, the diagnosis of NTM infection was likely more accurate using these stringent criteria. Currently, the diagnosis of NTM lung disease consists of clinical criteria including the image survey and microbiologic evidence.28,29 Notably, one key statement of the guideline states that making the diagnosis of NTM lung disease does not necessitate the administration of therapy given that treatment for NTM infection should be a decision based on potential risks and benefits of therapy for individual patients with NTM disease. 13 In this study, the stringent definition we used to enroll those receiving treatment for NTM infection might have only underestimated, rather than overestimated, the prevalence of NTM disease in Taiwan.
As was performed in this study, a number of studies have used ICD codes to identify patients with NTM infection in claims databases.30,31 Ku et al. 31 recently reported the validity of the diagnosis of NTM using ICD codes in the United States’ Medicare database (2006–2014) using the microbiology-based diagnosis of NTM infection in the Bronchiectasis and NTM Research Registry as the reference standard. Ku et al. found that an ICD code used once by a clinician has a moderate validity for identifying NTM infection, and the positive predictive value (PPV) was 63.2% (95% CI, 57.1–69.4%). The PPV increased to 72.1% (95% CI, 63.3–79.9%) with the additional restriction of a second visit between 30–365 days after the first visit with the same ICD code. Moreover, a further increased PPV was found when restricting the diagnosing physicians to infection specialists or pulmonologists. 31 In this study, we used at least three outpatient or one inpatient visit with an ICD code of NTM infection, as well as concomitant administration of anti-mycobacterial medication, which is generally prescribed by an infection specialist or pulmonologist in Taiwan, to define patients with NTM infection; therefore, in this study, the PPV of NTM infection was likely high using such a stringent diagnosis of NTM infection.
There were limitations in this study that merit discussion. First, there was a lack of information on NTM species in this claim data set; therefore, we could not assess the species-specific effect in this study. Second, given that NTM infection is not a notifiable disease in Taiwan, we might have underestimated, rather than overestimated, the prevalence of NTM infection, particularly as we used stringent criteria to define NTM infection. Third, the study design was retrospective, as with most research on the epidemiology of NTM; therefore, the causal inference could not be drawn. Fourth, the potential unmeasured confounders, such as cystic fibrosis, although cystic fibrosis is a rare disease in the Asian population.32,33
In conclusion, using a nationwide, population-based data set, we found a steady increased incidence and prevalence of NTM infection requiring treatment in Taiwan, particularly among the elderly, males, and immunocompromised patients. We also clarified the impact of NTM infection on mortality using age-, sex-, and comorbidity-matched comparable non-NTM subjects. These epidemiological data indicate that NTM infection poses a growing health threat and further highlight the crucial need for strategies to improve outcomes among patients with NTM requiring treatment.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666221103213 – Supplemental material for Mortality association of nontuberculous mycobacterial infection requiring treatment in Taiwan: a population-based study
Supplemental material, sj-pdf-1-tar-10.1177_17534666221103213 for Mortality association of nontuberculous mycobacterial infection requiring treatment in Taiwan: a population-based study by Hsin-Hua Chen, Ching-Heng Lin and Wen-Cheng Chao in Therapeutic Advances in Respiratory Disease
Footnotes
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of Taichung Veterans General Hospital Taiwan (IRB no: CE20295A). Informed consent was waived as all of the data obtained were anonymized.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from Taichung Veterans General Hospital (grant nos: TCVGH-1107307D and TCVGH-1104402D). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The data analyzed in this study are in accordance with the legal restriction of Personal Information Protection Act, which is imposed by the government of Taiwan. Therefore, raw data from NHIRD cannot be made publicly available. Requests for data can be sent as a formal application to the Health and Welfare Data Science Center, Department of Statistics, Ministry of Health and Welfare, Taiwan.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
