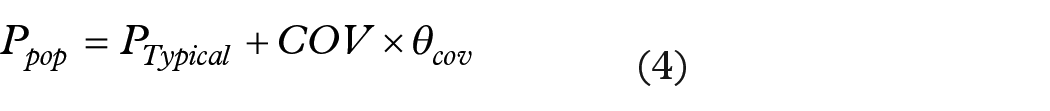Abstract
Objective:
This study aimed to quantitatively compare the efficacy and safety of long-acting β2-agonist (LABA)/long-acting muscarinic antagonist (LAMA) fixed-dose combinations (FDCs) for the treatment of stable chronic obstructive pulmonary disease (COPD), especially in terms of their loss of efficacy in lung function.
Methods:
Randomized controlled clinical trials of LABA/LAMA FDCs for the treatment of stable COPD were comprehensively searched for in public databases. Pharmacodynamic models were established to describe the time course of the primary outcome [trough forced expiratory volume in the first second (FEV1)]. Secondary outcomes [COPD exacerbations, St. George’s Respiratory Questionnaire (SGRQ), Transition Dyspnoea Index (TDI), and rescue medication use] and safety outcomes [mortality, serious adverse events (SAEs), and withdrawals due to adverse events (AEs)] were also compared via a meta-analysis.
Results:
A total of 22 studies involving 16,486 participants were included in this study. The results showed that in terms of primary outcome (change from baseline in trough FEV1), the efficacy of vilanterol/umeclidinium was the highest, while the efficacy of formoterol/aclidinium was the lowest, with a maximum effect value (Emax) of 0.185 L [95% confidence interval (CI): 0.173–0.197 L] and 0.119 L (95% CI: 0.103–0.135 L), respectively. The efficacy of other drugs, such as formoterol/glycopyrronium, indacaterol/glycopyrronium, and olodaterol/tiotropium, were comparable, and their Emax values were 0.150–0.177 L. Except for vilanterol/umeclidinium, the other four LABA/LAMA FDCs showed a certain degree of loss of efficacy. Compared with the efficacy at 2 days, the trough FEV1 (L) relative to baseline at 24 weeks decreased by 0.029–0.041 L. In terms of secondary outcomes, the efficacy of different LABA/LAMA FDCs was similar in TDI and rescue medication use. However, formoterol/aclidinium was better in preventing the COPD exacerbations, while vilanterol/umeclidinium was the best in terms of SGRQ. In addition, different LABA/LAMA FDCs and placebo had similar safety outcomes.
Conclusion:
The present findings may provide necessary quantitative information for COPD medication guidelines.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is one of the most common respiratory diseases worldwide. According to a prediction by the World Health Organization (WHO), the prevalence of COPD will continue to rise in the next 40 years, and the annual number of deaths caused by this disease and associated diseases will exceed 5.4 million by 2060.1,2 The goals in the management of COPD are to prevent deterioration of lung function and to reduce symptoms as well as the frequency and severity of exacerbations. 3
The current guidelines recommend the use of long-acting bigeminal therapy, mainly LABA/LAMA fixed-dose combinations (FDCs) and LABA/ICS FDCs, for the treatment of COPD. Long-acting bronchodilator combinations (LAMA/LABA FDCs) have been identified as the initial drugs of choice for patients with more severe dyspnea, airflow obstruction, and hyperinflation.4 –6 Compared with monotherapy, the combination of two inhaled drugs with different pharmacological categories in the same device provides greater treatment benefits and has the additional advantage of improving patient compliance. 4
A published network meta-analysis conducted by Calzetta et al. 7 indirectly compared the efficacy of five types of LABA/LAMA FDCs for the treatment of COPD. However, uncorrected heterogeneities between trials, such as different treatment durations (6–52 weeks) and patient demographics (such as baseline prebronchodilator forced expiratory volume in the first second (FEV1), proportion of male patients, and age), resulted in ambiguous conclusions.
Through analysis of preliminary data, we found that improvement of lung function (trough FEV1) in patients with COPD exhibited a declining trend after a period of LABA/LAMA FDCs treatment.8 –12 However, previous studies have not analyzed this trend.
Model-based meta-analysis (MBMA) is a method that combines quantitative pharmacology with meta-analysis. Through modeling, MBMA can combine and analyze the heterogeneity data of different treatment durations and different populations as well as quantify the time effects and influencing factors of drugs to predict their efficacy or safety characteristics at different treatment durations and levels of covariates.13,14 In this study, MBMA was used to establish a pharmacodynamic model to quantitatively analyze the efficacy characteristics of different LABA/LAMA FDCs and placebo in improving lung function (trough FEV1) in stable COPD patients. In addition, indicators such as COPD exacerbations and adverse events were also comprehensively analyzed, so as to provide necessary quantitative information for guidelines of COPD treatment.
Methods
Search strategy
The Cochrane Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline for meta-analysis was used to collate data and report results accordingly. 15 A comprehensive search was conducted using PubMed and EMBASE from the inception dates of the databases to 11 November 2019. The search keywords included drug names (arformoterol, formoterol, indacaterol, olodaterol, and salmeterol) and indications (chronic obstructive pulmonary disease). The terms in the same category were connected by ‘or’, while the terms in different categories were connected by ‘and’. Clinical trials were searched accordingly, and the language was limited to English. The details of the search strategies are presented in Supplementary Table S1.
The inclusion criteria were as follows: (1) randomized, controlled, and double-blind clinical trials; (2) adults ⩾40 years of age with a diagnosis of stable COPD; (3) if the change from baseline in trough FEV1 (L) was reported; (4) and sample size was ⩾ 50 patients to avoid sampling errors.
Data extraction
Microsoft Excel software (version 2019) was used for data collection. The following information was extracted from the included studies: literature characteristics (author, year of publication, and country), study design (treatment drugs, dosage, sample size, and treatment duration), patient characteristics [baseline prebronchodilator FEV1, age, proportion of male patients, proportion of current smokers, proportion of ICS users, and postbronchodilator FEV1 (% predicted)], the primary outcome (change from baseline in trough FEV1), secondary outcomes [COPD exacerbations, St. George’s Respiratory Questionnaire (SGRQ) total score, Transition Dyspnoea Index (TDI), and rescue medication use], and the safety outcome [mortality, serious adverse events (SAEs) and withdrawals due to adverse events (AEs)]. If data were presented as a graph, the digitizing software Engauge Digitizer (Mark Mitchell, USA) was used to extract data. All data were independently extracted by two researchers, and any discrepancies were resolved by a third researcher. We ensured that data extraction errors between the two researchers did not exceed 2%; otherwise, the graph was reread, and the mean values were used for the final results.
Risk of bias assessment
Two investigators independently extracted the relevant information and assessed the risk of bias using the Cochrane risk of bias tool. Any disagreements were resolved through discussion with a third investigator. The evaluation items included random sequence generation, allocation concealment, blinding of participants and personnel, blinding in the outcome assessment, incomplete outcome data, selective reporting, and other biases. 16 Other biases were defined as trials in which baseline characteristics were not comparable between the different treatment groups.
Modeling and simulation of primary outcome
The trough FEV1 is most commonly used in clinical trials that evaluate the efficacy of bronchodilators. 17 In this study, the change from baseline in trough FEV1 (L) was used as the primary outcome to establish a pharmacodynamic model. Exploratory analysis of the data showed that the change in trough FEV1 (L) could reach maximum efficacy value at the beginning of drug treatment. However, the efficacy of some drugs gradually decreased over time, thus resulting in a loss of efficacy in lung function. This pharmacodynamic characteristic can be described using the following equation: 18
In equation (1), Ei, j is the observed change from baseline in pre-dose (trough) FEV1 (L) at the time point j in the ith arm, and Emax,i represents the maximal change from baseline in trough FEV1 (L) in the ith arm. τi is the loss of efficacy rate in the ith arm, while εi, j is the residual error at the time point j in the ith arm, which is weighted by the inverse of the square root of the sample size (Ni, j), assumed to be normally distributed, with a mean of 0 and variance of σ 2 . The residual errors are smaller in the arm with larger sample sizes. In equations (2) and (3), Emax, typical and τtypical are typical values for the parameters Emax and τ, respectively. η1,i and η2,i represent the interstudy variability of Emax and τ, respectively. Emax and τ are assumed to be normally distributed, with means of 0 and variances of ω1 2 and ω2 2 .
After the pharmacodynamic model was established, correlation analyses were performed to explore the impact of the factors on the model parameters. Factors such as age, proportion of male patients, proportion of current smokers, proportion of ICS users, and postbronchodilator FEV1 (%predicted) were accordingly tested. For a factor with a missing proportion of <30%, the missing information was imputed using the median value of the factor. Factors with a missing proportion of >30% were not considered during covariate evaluation (such as baseline prebronchodilator FEV1). If the p value of Pearson correlation was <0.05, this factor was considered to be significantly related to the model parameters and was selected for subsequent covariate analyses. 19
The influence of covariates on the parameters of the selected base model was then tested using the forward inclusion–backward elimination method. Upon introduction of a covariate, a decrease in objective function value (OFV) of 3.84 (χ2, α = 0.05, df = 1) was considered statistically significant in the forward inclusion process. During the backward elimination procedure, a covariate was removed from the model if the OFV increased to <6.63 (χ2, α = 0.01, df = 1) during the exclusion. For the introduction of 2-category covariates, see equation (4), and for the introduction of continuous covariates, see equations (5) and (6):
In equations (4)–(6), Ppop is the population value of the pharmacodynamic parameters corresponding to the different levels of covariates; PTypical is the typical value of the pharmacodynamic parameters when the covariate value is equal to the median value of the covariate; COV is the covariate value; COVmedian is the median value of the covariate; θcov is the correction coefficient of the covariates of the pharmacodynamic parameters.
The performance of the final model was evaluated using a diagnostic goodness-of-fit plot. A visual predictive check was then conducted by comparing the 95% CIs of the predicted values with the observed values to assess the predictive capacity of the final model. The robustness of the model was assessed using a nonparametric bootstrap, which was performed using 1000 NONMEM repetitions of the final model. The bootstrap median parameter values and 95% CIs were compared with the respective values estimated from the final model. If these values were close, it indicated that the estimated parameters were stable and less affected by any individual study.
Based on the final model, the typical efficacies and 95% CIs of each drug and placebo at different time points were obtained using Monte Carlo simulations (10,000 times).
Meta-analysis for secondary outcomes and safety outcomes
In this study, COPD exacerbations, TDI, the change from baseline in SGRQ, and the change from baseline in rescue medication use (puffs/day) were evaluated as secondary outcomes. The mortality, SAEs, and withdrawals due to AEs were analyzed as safety outcomes. Since most of the included studies only reported these indicators at the endpoint, we could not establish the time course models for these indicators. Therefore, a meta-analysis was conducted to briefly summarize the characteristics of different LABA/LAMA FDCs relative to placebo on these indicators. Specifically, the mean difference (MD) and risk ratio (RR) with 95% CIs of each drug group relative to the placebo group were calculated for continuous and dichotomous data, respectively.
Software
Model establishment and simulations were performed using NONMEM 7.3 (ICON Development Solutions, USA). Statistical analysis and generation of plots were performed using R4.0.1 (The R Foundation of Statistical Computing). The meta-analysis and quality assessment of the literature was performed using Review Manager 5.3.5 (Cochrane Collaboration, London, England).
Results
Characteristics of included studies
A total of 22 studies with 44 arms consisting of 16,486 participants were included in the analysis (Figure 1).8 –12,20 –36 Among the included 22 studies, 8 studies focused on vilanterol/umeclidinium (N = 3797), 5 studies focused on formoterol/glycopyrronium (N = 2910), 5 studies focused on indacaterol/glycopyrronium (N = 2154), 3 studies focused on formoterol/aclidinium (N = 1826), 2 studies focused on olodaterol/tiotropium (N = 3192), and 11 studies focused on placebo (N = 2607).

Flow chart for study identification and selection.
The mean age of the patients ranged from 61.9 to 67.3 years, and the mean proportion of male patients was 49.2%–96.4%. The mean proportion of current smokers was 25%–59%, and the mean postbronchodilator FEV1 (%predicted) was 37%–59.4%. Treatment duration ranged from 12 to 64 weeks (Table 1). Detailed information on the included studies is shown in Supplementary Table S2.
Brief characteristics of included studies, median (min, max).

COPD, chronic obstructive pulmonary disease; FDC, fixed-dose combinations; FEV1, forced expiratory volume in the first second.
The risk-of-bias items presented as percentage and summary for each included study are shown in Supplementary Figure S1. All the included studies had a low risk of blinding of participants and personnel (performance bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other bias. However, an unclear risk of random sequence generation (selection bias) was found in eight studies due to not mentioning the method used to generate the allocation sequence, and allocation concealment (selection bias) may have occurred in 16 studies due to not stating the method of allocation of subjects. In addition, there was unclear risk of blinding in the outcome assessment (detection bias) in 10 studies, as these studies have not described the blinding of outcome assessments.
Model establishment and assessment for primary outcome
Most of the efficacy data in trough FEV1 included in the analysis were measured within 26 weeks. Thus, to avoid estimation bias in terms of model parameters, this study only analyzed the efficacy data in trough FEV1 within 26 weeks. The final model parameters are presented in Table 2. The diagnostic graphs of the final model showed a relatively good fit of the observed data (Supplementary Figure S2). In addition, the individual fit graph also showed that the predicted data were close to the observed data and showed no obvious bias (Supplementary Figure S3). The observed change from baseline in trough FEV1 (L) in each drug group and placebo group were almost within the 95% CI of the predicted data (Figure 2). This indicated that the model had good predictability. The distribution of model parameters obtained by the bootstrap method was close to the estimated values of the model parameters obtained from the original data set, indicating that the estimation of model parameters was robust and was less affected by individual studies (Table 2). In the covariate screening process, no factors were found to be related to the model parameters. Therefore, the base model was used as the final model.
Parameter estimations of the final model.

CI, confidence interval; Emax, theoretical maximal change from baseline in predose(trough) FEV1 (L); FEV1, forced expiratory volume in the first second; τ, rate of loss of efficacy; η, interstudy variability of pharmacodynamic parameter; ε, residual error.
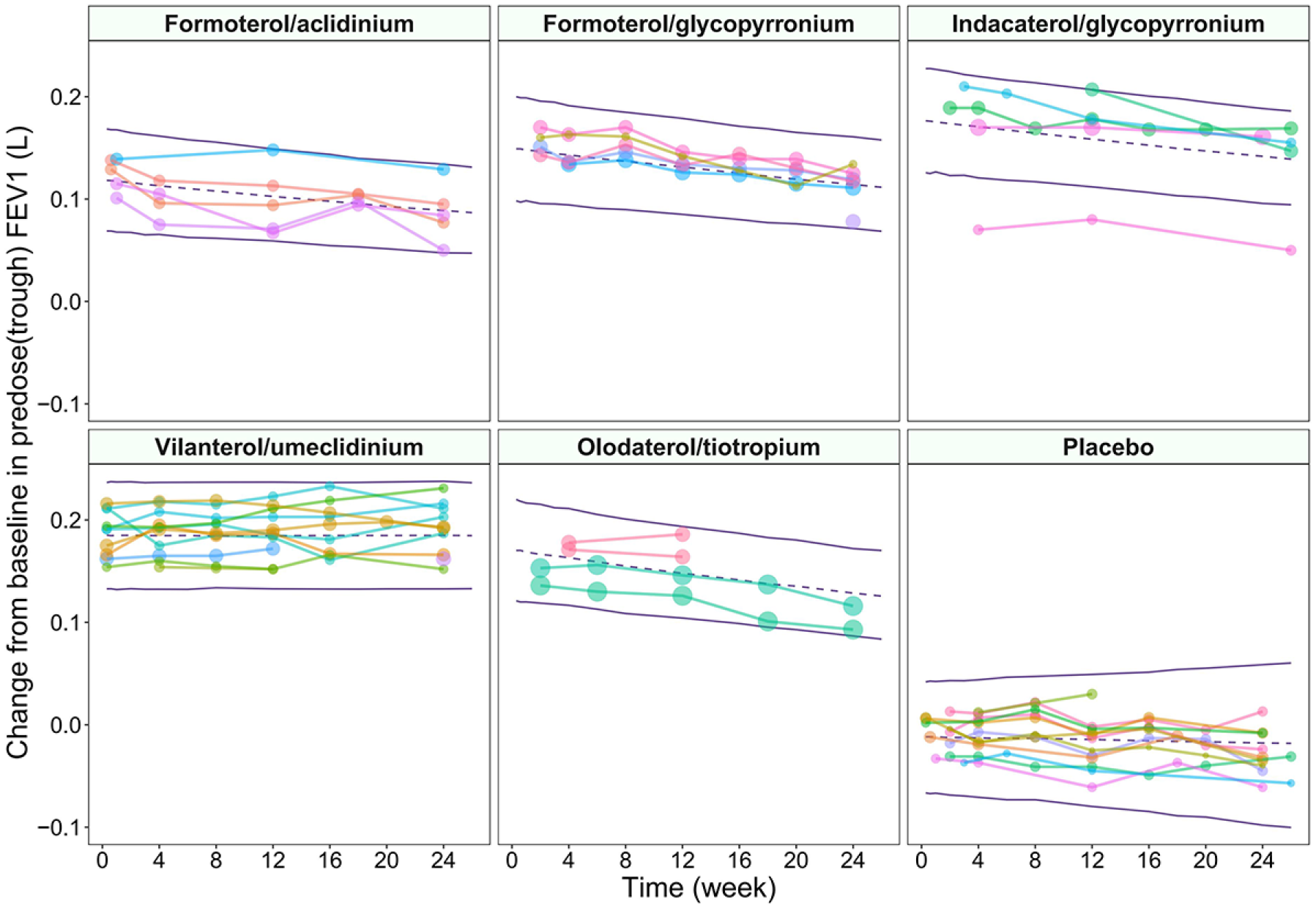
Visual predictive check of the final model of the change from baseline in predose(trough) FEV1 (L). The points represent observed change from baseline in predose(trough) FEV1 (L), and symbol size is proportional to sample size. Points linked by a line are from the same arm. Purple lines are the model-predicted fifth, 50th, and 95th percentiles of the change from baseline in predose(trough) FEV1 (L).
Typical efficacy of drug and placebo for primary outcome
Based on the final model, we simulated the typical efficacy distribution of each drug and placebo at different time points (Table 3). The results showed that in terms of trough FEV1, the efficacy of vilanterol/umeclidinium was the highest and that there was no loss of efficacy; in other words, the efficacy at 24 weeks was the same as that at 2 days. However, the efficacy of formoterol/aclidinium was the lowest, and its efficacy was approximately 75% as that at 2 days; in other words, its efficacy loss was approximately 25% at 24 weeks. The efficacy of the other three drugs was between that of vilanterol/umeclidinium and formoterol/aclidinium, and their rates of efficacy loss were similar. Their efficacy at 24 weeks was approximately 76%–81% of that at 2 days.
Predicted change from baseline in predose(trough) FEV1 (L) of each drug and placebo.
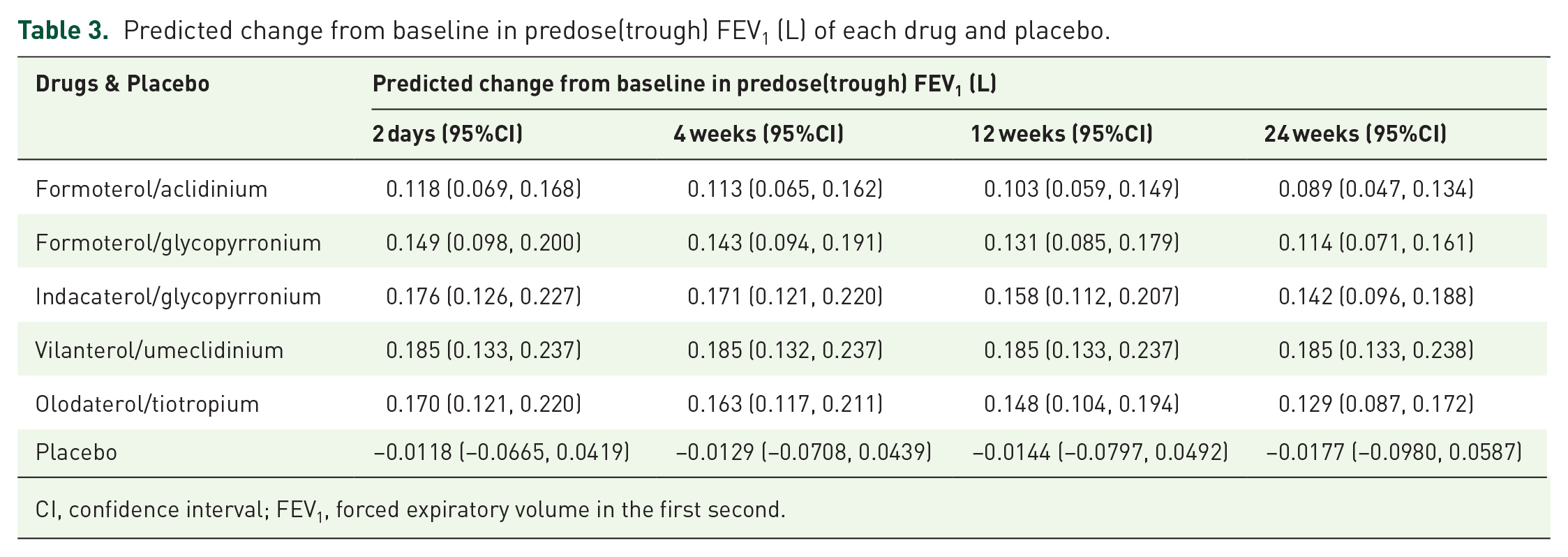
CI, confidence interval; FEV1, forced expiratory volume in the first second.
In addition, we found that the placebo response of COPD was not obvious and that the placebo response at 24 weeks was similar to that at 2 days. The 95% CI of the placebo response at each time point was 0, indicating that the placebo could not effectively improve the trough FEV1 (L) (Figure 3).

Predicted typical time course of the change from baseline in predose(trough) FEV1 (L) of each drug and placebo.
Secondary outcomes
Since the secondary outcomes related to olodaterol/tiotropium were not reported in the included literature, this study only analyzed the secondary outcomes of the other four LABA/LAMA FDCs. The results (Table 4 and supplementary Figures S4–S7) showed that the risk of COPD exacerbations in formoterol/aclidinium was significantly lower than that in placebo, while the risk of COPD exacerbations in formoterol/glycopyrronium, indacaterol/glycopyrronium, and vilanterol/umeclidinium was comparable to that of the placebo. In terms of reducing SGRQ, vilanterol/umeclidinium was the best, and it was significantly better than formoterol/aclidinium. In addition, the efficacy of formoterol/aclidinium, formoterol/glycopyrronium, indacaterol/glycopyrronium, and vilanterol/umeclidinium on TDI was comparable, and in terms of rescue medication use, formoterol/glycopyrronium, indacaterol/glycopyrronium, and vilanterol/umeclidinium had the similar efficacy.
Results of meta-analysis of secondary outcomes.
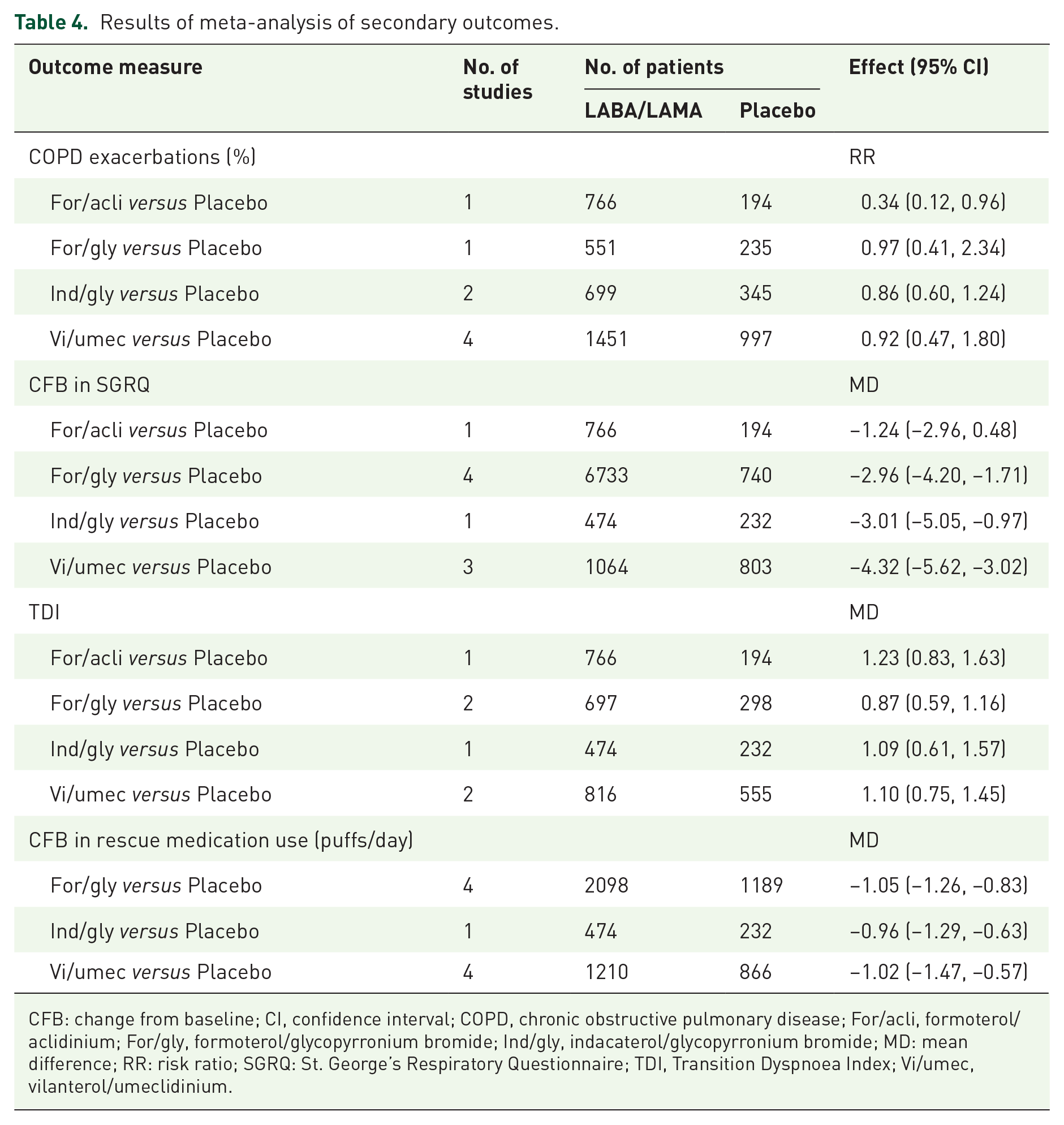
CFB: change from baseline; CI, confidence interval; COPD, chronic obstructive pulmonary disease; For/acli, formoterol/aclidinium; For/gly, formoterol/glycopyrronium bromide; Ind/gly, indacaterol/glycopyrronium bromide; MD: mean difference; RR: risk ratio; SGRQ: St. George’s Respiratory Questionnaire; TDI, Transition Dyspnoea Index; Vi/umec, vilanterol/umeclidinium.
Safety outcomes
Similarly, due to the lack of data on olodaterol/tiotropium, this study only analyzed the safety outcomes of the other four LABA/LAMA FDCs. The results (Table 5 and supplementary Figures S8–S10) showed that the incidences of formoterol/aclidinium, formoterol/glycopyrronium, indacaterol/glycopyrronium, and vilanterol/umeclidinium were similar in mortality, SAEs, and withdrawals due to AEs, and there was no significant difference between those four LABA/LAMA FDCs and placebo.
Results of meta-analysis of safety outcomes.

AEs, adverse events; CI, confidence interval; For/acli, formoterol/aclidinium; For/gly, formoterol/glycopyrronium bromide; Ind/gly, indacaterol/glycopyrronium bromide; RR: risk ratio; SAEs, serious adverse events; Vi/umec, vilanterol/umeclidinium.
Discussion
In this study, we quantitatively evaluated the effectiveness of five LABA/LAMA FDCs in trough FEV1 by establishing a pharmacodynamic model. The results showed that vilanterol/umeclidinium had the highest efficacy on trough FEV1, with an Emax value of 0.185 L (95% CI: 0.173–0.197 L). However, the efficacy of formoterol/aclidinium was the lowest, with an Emax value of 0.119 L (95% CI: 0.103–0.135 L), accounting for only 64% of that of vilanterol/umeclidinium. The efficiencies of other drugs, such as formoterol/glycopyrronium, indacaterol/glycopyrronium, and olodaterol/tiotropium were similar, and their Emax values were 0.150–0.177 L. Formoterol/aclidinium, formoterol/glycopyrronium, indacaterol/glycopyrronium, and olodaterol/tiotropium showed a certain degree of loss of efficacy on trough FEV1. Compared with the efficacy at 2 days, the trough FEV1 (L) relative to baseline at 24 weeks decreased by 0.029, 0.035, 0.034, and 0.041 L, respectively. However, the efficacy of vilanterol/umeclidinium was not lost, and its efficacy at 24 weeks was the same as that at 2 days. The above results clearly reflect the differences in the efficacy of different LABA/LAMA FDCs on trough FEV1, but whether the difference is of clinical significance or not needs to be judged by clinicians.
Vilanterol/umeclidinium was approved by the US Food and Drug Administration in 2013 as a daily long-term maintenance treatment for COPD patients with airflow limitation. 37 This study showed that the efficacy of vilanterol/umeclidinium on trough FEV1 was significantly better than that of the other four LABA/LAMA FDCs, and that its efficacy on long-term use was not lost, thus showing better efficacy in lung function. A published network meta-analysis conducted by Calzetta et al. 7 also suggested that the efficacy of vilanterol/umeclidinium on trough FEV1 was better than that of other LABA/LAMA FDCs. This study combined and analyzed the efficacy of different treatment durations, thus ignoring the influence of heterogeneity of treatment durations on the results. The study also revealed that since the efficacy of vilanterol/umeclidinium on trough FEV1 was not affected by the treatment duration, maximum efficacy was achieved as soon as the drug was used and could be maintained continuously. Therefore, the estimation of the efficacy of vilanterol/umeclidinium on trough FEV1 by the network meta-analysis was accurate, but the efficacy of the other four LABA/LAMA FDCs showed an obvious downward trend over time. The combined analysis of the efficacy of different treatment durations hence introduced a bias.
In addition to drug effects, this study also quantitatively analyzed for placebo effects on trough FEV1 in COPD clinical trials. The results showed that the placebo efficacy at 24 weeks was almost the same as that at 2 days, with no obvious time-effect relationship observed. The 95% CI of the typical value of placebo efficacy at different time points was 0, suggesting that placebo could not significantly improve trough FEV1 (L). Thus, the effects observed in the drug group were mainly due to the drugs.
The median duration of clinical trials included in this study was approximately 24 weeks, and the data showed that the efficacy of LABA/LAMA FDCs on trough FEV1 reached a maximum at the treatment initiation; thus, the maximum efficacy of drugs could be evaluated in the early stages of clinical trials (for example, within 1 week). The main purpose of the later stage of the clinical trial was to evaluate the efficacy maintenance and safety of these drugs. 38 However, this study showed that the efficacy of some LABA/LAMA FDCs decreased linearly over time within 26 weeks; thus, the efficacy loss rate of drugs on trough FEV1 could be predicted in a short treatment duration. The above results suggest that for COPD clinical trials whose main purpose was to evaluate the improvement of airflow limitation as reflected by the lung function indicator [trough FEV1], the evaluation of the efficacy of LABA/LAMA FDCs could be completed in a short treatment duration. 38
In this study, the change from baseline in trough FEV1 (L) was used as the primary outcome, which is commonly used to evaluate the efficacy of COPD maintenance therapy in improvement in lung function. This indicator has been reported in most COPD clinical trials, which have large amounts of data and, thus, are conducive for use in modeling and analysis. 38 However, clinical trials of COPD usually include other efficacy indicators, such as COPD exacerbations, which reflect the condition of exacerbations; SGRQ and TDI, which reflect the quality of life and dyspnea, respectively; and the use of rescue medication (e.g. β2 agonist and reliever inhaler), which reflect effects on symptoms. 38 Since these indicators were not universally reported in the literature, and most of them were only reported at the end point, this study did not perform modeling of these indicators. Therefore, this study only summarized these indicators through a traditional meta-analysis. The results showed that the efficacy of different LABA/LAMA FDCs were similar in terms of TDI and rescue medication use. However, formoterol/aclidinium is better in preventing the COPD exacerbations, while vilanterol/umeclidinium is the best in terms of SGRQ. The result suggests that patients with greater improvements in trough FEV1 had higher decrease in SGRQ score. However, it should be pointed out that due to the small amount of data included in the analysis, the conclusions of the secondary outcomes are not robust and need to be supported by further data.
A network meta-analysis by Schlueter et al. 39 comparing the safety of four LABA/LAMA FDCs except formoterol/glycopyrronium and a randomized controlled trial by Maltais et al. 23 comparing the safety of formoterol/glycopyrronium with that of vilanterol/umeclidinium showed that the safety of the five types of LABA/LAMA FDCs was similar. The results also showed that different LABA/LAMA FDCs and placebo had similar safety outcomes.
Because the current study is limited by the included data, it only included the change from baseline in trough FEV1 (L) within 26 weeks for modeling and analysis. Although the pharmacodynamic model established in this study can predict efficacy characteristics during long-term drug treatment, the reliability of the extrapolation results after 26 weeks requires further verification through clinical trials. Previous studies have shown that age, smoking history, and frequency of previous exacerbations are factors that affect the efficacy of drugs.40 –42 In this study, age, proportion of male patients, proportion of current smokers, proportion of ICS users, and postbronchodilator FEV1 (%predicted) did not have a significant influence on the efficacy of drugs on trough FEV1. The main reason was that this study was based on summary level data from the literature rather than individual data. Since the covariates of the data at the summary level were usually reported as mean or median values, the distribution range of covariates was narrow and the detection power of covariates was low, which was also a limitation of this study. In addition, some LABA/LAMA FDCs have multiple dose combinations; however, due to the small number of studies included and the large variation among trials, it was impossible to analyze the dose-effect relationship of each drug in this study. Since there was no widely accepted standard to evaluate the results of modeling analysis, we did not use a method similar to GRADE methodology to rate the evidence obtained by the pharmacodynamic model. In addition to the above limitations, only English literature was included in this study, which may have led to publication bias.
Conclusion
This study quantitatively evaluated the features of the change from baseline in trough FEV1 (L) associated with five LABA/LAMA FDCs for the treatment of COPD in improving lung function. The results showed that in terms of trough FEV1, the efficacy of vilanterol/umeclidinium was the highest, while that of formoterol/aclidinium was the lowest. Formoterol/aclidinium, formoterol/glycopyrronium, indacaterol/glycopyrronium, and olodaterol/tiotropium showed efficacy loss in trough FEV1 after administration. There was no significant efficacy loss after administration of vilanterol/umeclidinium. In addition, the efficacy of different LABA/LAMA FDCs was similar in TDI and rescue medication use. However, formoterol/aclidinium was better in preventing the COPD exacerbations, while vilanterol/umeclidinium was the best in terms of SGRQ. In terms of safety, different LABA/LAMA FDCs and placebo had similar mortality, SAEs, and withdrawals due to AEs. The above information provided the necessary quantitative supplements for the improvement of COPD treatment guidelines.
Supplemental Material
sj-docx-1-tar-10.1177_17534666211066068 – Supplemental material for Quantitative analysis of efficacy and safety of LABA/LAMA fixed-dose combinations in the treatment of stable COPD
Supplemental material, sj-docx-1-tar-10.1177_17534666211066068 for Quantitative analysis of efficacy and safety of LABA/LAMA fixed-dose combinations in the treatment of stable COPD by Yiwen Gong, Yinghua Lv, Hongxia Liu, Qingshan Zheng and Lujin Li in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors thank all staffs of the Center for Drug Clinical Evaluation, Shanghai University of Traditional Chinese Medicine.
Author contributions
GYW collected the data, performed the analyses, interpreted the data, and drafted the manuscript. ZQS, LLJ, LHX, and LYH supervised the analyses and reviewed the manuscript. All authors contributed to manuscript revision and have read and approved the submitted version.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was not an industry supported study. This work was received financial support from the project of Shanghai Municipal Health Planning Commission (2018YQ48), Shanghai S&T Innovation Plan (17401970900) and The National Natural Science Foundation of China (82174229).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.