Abstract
Aims:
Given the variability of previously reported results, this systematic review aims to determine the clinical effectiveness of convalescent plasma employed in the treatment of hospitalized patients diagnosed with COVID-19.
Methods:
We conducted a systematic review of controlled clinical trials assessing treatment with convalescent plasma for hospitalized patients diagnosed with SARS-CoV-2 infection. The outcomes were mortality, clinical improvement, and ventilation requirement.
Results:
A total of 51 studies were retrieved from the databases. Five articles were finally included in the data extraction and qualitative and quantitative synthesis of results. The overall risk of bias in the reviewed articles was established at low-risk only in two trials. The meta-analysis suggests that there is no benefit of convalescent plasma compared with standard care or placebo in reducing the overall mortality and the ventilation requirement. However, there could be a benefit for the clinical improvement in patients treated with plasma.
Conclusion:
Current results led to assume that the convalescent plasma transfusion cannot reduce the mortality or ventilation requirement in hospitalized patients diagnosed with SARS-CoV-2 infection. More controlled clinical trials conducted with methodologies that ensure a low risk of bias are still needed.
Introduction
The SARS-CoV-2 virus, first detected in Wuhan, China, has caused a global pandemic. 1 What is known about the microorganism is established by genomic analysis as the disease spreads.2,3 The pandemic still represents a global health threat; in mid-January 2021, the total number of COVID-19 cases reported by the World Health Organization (WHO) is close to 100 million, while deaths have exceeded two million. 4 One year after the onset of the disease, it is known that there are cases with different degrees of severity ranging from asymptomatic cases to critical patients, in whom respiratory failure, septic shock, or multi-organ failure occurs, requiring various hospital care and supportive treatment. 5 WHO points out that there are more than 200 vaccines under investigation; only some in phase III and IV are being distributed currently.6–9
The distribution of these vaccines is subject to each country’s production, acquisition, storage, and distribution capacities. As of January 2021, 90% of the vaccines produced are concentrated in nine countries. 10 These problems limit a large percentage of the world population to be vaccinated as soon as possible and therefore reach herd protection, this limitation being even more significant in low-income countries, which will have to wait a considerable time longer; this is because of the pre-order manufacturing contracts for vaccines to 13% of the population mainly in the European Union. 6 All this implies that people will continue to be infected and will continue to die from this infection in low-income countries. Therefore, to find a treatment that reduces the severity of the disease and reduces the incidence of fatality is still a significant public health concern.
On the other hand, besides the limitations of access to the vaccine, their efficacy has been established in a range from 50% to 95%.8,9 Therefore, in countries where there will not be prompt protection of the population, it is necessary to use treatments to recover hospitalized patients when the effectiveness of the vaccines is not as expected.
Among the repurposed treatments, the use of passive immunity has been suggested as an alternative since the beginning of the pandemic. 11 The use of convalescent plasma as a treatment against COVID-19 was approved in March 2020 by the Food and Drug Administration. 12 The treatment uses the administration of antibodies collected from people recently infected and recovered from COVID-19. The results of the use of plasma are variable, reporting efficacy if its use is in the first 16 days of illness, which was associated with an improvement in the first days after treatment and lower requirements for ventilatory support. On the other hand, some studies show no evidence of clinical effectiveness of plasma in severely or critically ill patients for preventing disease progression.13–19 Given the variability of previously reported results, the present work aims to determine the clinical effectiveness of convalescent plasma employed in the treatment of hospitalized patients with diagnosis of COVID-19.
Methods
A systematic review was conducted adhering to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines for conducting systematic reviews. 20 The question in this review was: What is the clinical effectiveness of convalescent plasma employed in the treatment of hospitalized patients with diagnosis of COVID-19?
To conduct the review, the PICOS structure was followed according to these points:
Patients: adults hospitalized with a diagnosis of SARS-CoV-2 infection;
Intervention: treatment with convalescent plasma;
Comparison: placebo or standard care;
Outcomes: overall mortality, clinical improvement at 7 days, clinical improvement at 14 days, clinical improvement at 28 days, ventilation requirement, duration of hospitalization (days), virological clearance, and severe adverse events;
Studies (type of): clinical trials published in peer-reviewed journals.
The search was carried out in
Studies that met the following criteria were included: (i) controlled clinical trials, (ii) studies that included hospitalized patients with SARS-CoV-2 infection, (iii) published in 2020 and 2021, (iv) published in English, Chinese, Spanish, or Portuguese. The exclusion criteria were: (i) not being a clinical controlled trial, (ii) not treating hospitalized patients, and (iii) not using convalescent plasma.
All references were managed with Mendeley® software. The selection of the articles began with the removal of duplicate articles and proceeded with the reading of the title and abstract, carried out independently by reviewers 1, 2, and 3. The final decision in cases of disagreement was based on the criteria of a fourth reviewer. In the second phase, the same reviewers read the full text of the studies to define which would be included for the extraction and synthesis of data. The data were stored in Microsoft Office Excel spreadsheets and organized in an instrument constructed by the authors considering: characteristics of the study (author, year, country), sample, study design, and characteristics of the results.
The risk of bias of the studies was evaluated using the ROB2 tool. 21 The included studies were independently assessed by reviewers 1 and 5 (see Supplemental file).
The qualitative synthesis was developed following the assessed outcomes: overall mortality, clinical improvement at 7 days, clinical improvement at 14 days, clinical improvement at 28 days, ventilation requirement, hospital stay (days), virological clearance, and severe adverse events.
Statistical analysis
Meta-analyses of inverse variance were conducted for three outcomes: clinical improvement at day 7, ventilation requirement, and overall mortality. Meta-analyses were conducted with Revman v5.4 using pooled fixed effects odds ratios. The significance and the magnitude of heterogeneity across studies were calculated using the Q and
The review protocol was registered on the PROSPERO platform (CRD42020184436).
Results
Following the described PICOS structure, this systematic review retrieved 51 studies from the databases. After the removal of four duplicates, 47 articles were read in title and abstract. Forty-two were eliminated, resulting in five articles for full-text reading. Four articles were finally included in the data extraction and qualitative and quantitative synthesis of results (Figure 1).

Preferred Reporting Items for Systematic Reviews and Meta-Analyses flowchart of the inclusion process in the systematic review.
The overall risk of bias in the reviewed articles was established at low-risk only in two randomized, double-blind clinical trials.17,22 The remaining three studies were established at high risk of bias due to issues in the randomization process.16,18,19 More details can be seen in the Supplemental file.
Patient samples ranged from 103 (the study with the fewest patients) to 464 (the study with the most patients); four clinical trials included adult patients,16,18,19,22 while the fifth study was focused only on older adult patients. 17 The retrieved results were: mortality, clinical improvement at 7, 14, and 28 days (defined by clinical scales), ventilation requirement, the mean duration of hospitalization (in days), virological clearance (by laboratory tests), progression to severe disease, and severe adverse events (Table 1).
Main characteristics of the included studies.

The five studies reported using convalescent plasma at different dosages. Li
All studies quantified anti-COVID-19 antibody titers, but only Li
Outcomes assessed
The primary outcome assessed by this systematic review was the mortality in hospitalized patients diagnosed with SARS-CoV-2 infection. Two clinical trials assessed the mortality of hospitalized patients at day 28,18,19 and three studies reported mortality at any time from allocation16,17,22 (Table 1).
Three studies reported clinical improvement. Li
The progression to severe disease was assessed by Agarwal
Two studies18,19 reported the virological clearance using different criteria, and all studies reported adverse events identified in patients.16–19,22 The study published by Li
Table 2 shows the main results from the four articles included in the qualitative synthesis. The reduction in overall mortality is supported by Li
Reported outcomes in the included studies.

IQR, interquartile range.
Li
Meta-analysis
The result of two studies was integrated into the fixed-effects meta-analysis for comparing convalescent plasma

Forest plot of convalescent plasma transfusion for hospitalized patients with SARS-CoV-2 infection. Comparison: convalescent plasma
Four studies reporting ventilation requirement outcomes16–18,22 were compared to test the overall effect of convalescent plasma. The random-effects meta-analysis results show no association with ventilation requirement (OR: 1.23; CI: 0.63–2.42) (Figure 3).
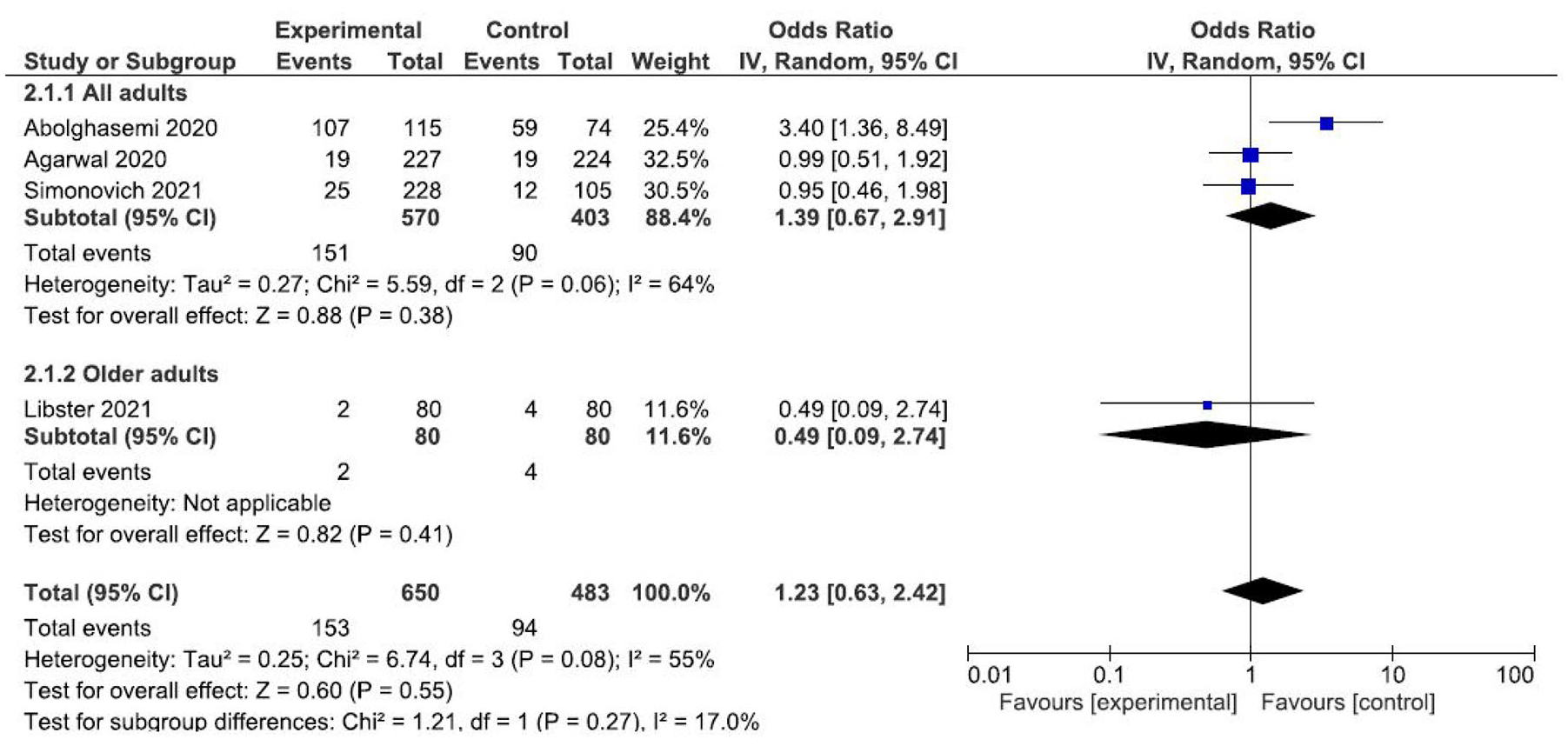
Forest plot of convalescent plasma transfusion for hospitalized patients with SARS-CoV-2 infection. Comparison: convalescent plasma
Finally, the results of four studies16,18,19,22 were meta-analyzed to establish comparisons on the overall mortality. The meta-analysis of fixed effects suggests no benefits using the convalescent plasma transfusion for reducing the risk of overall mortality (OR: 0.83; CI: 0.59–1.18) (Figure 4).
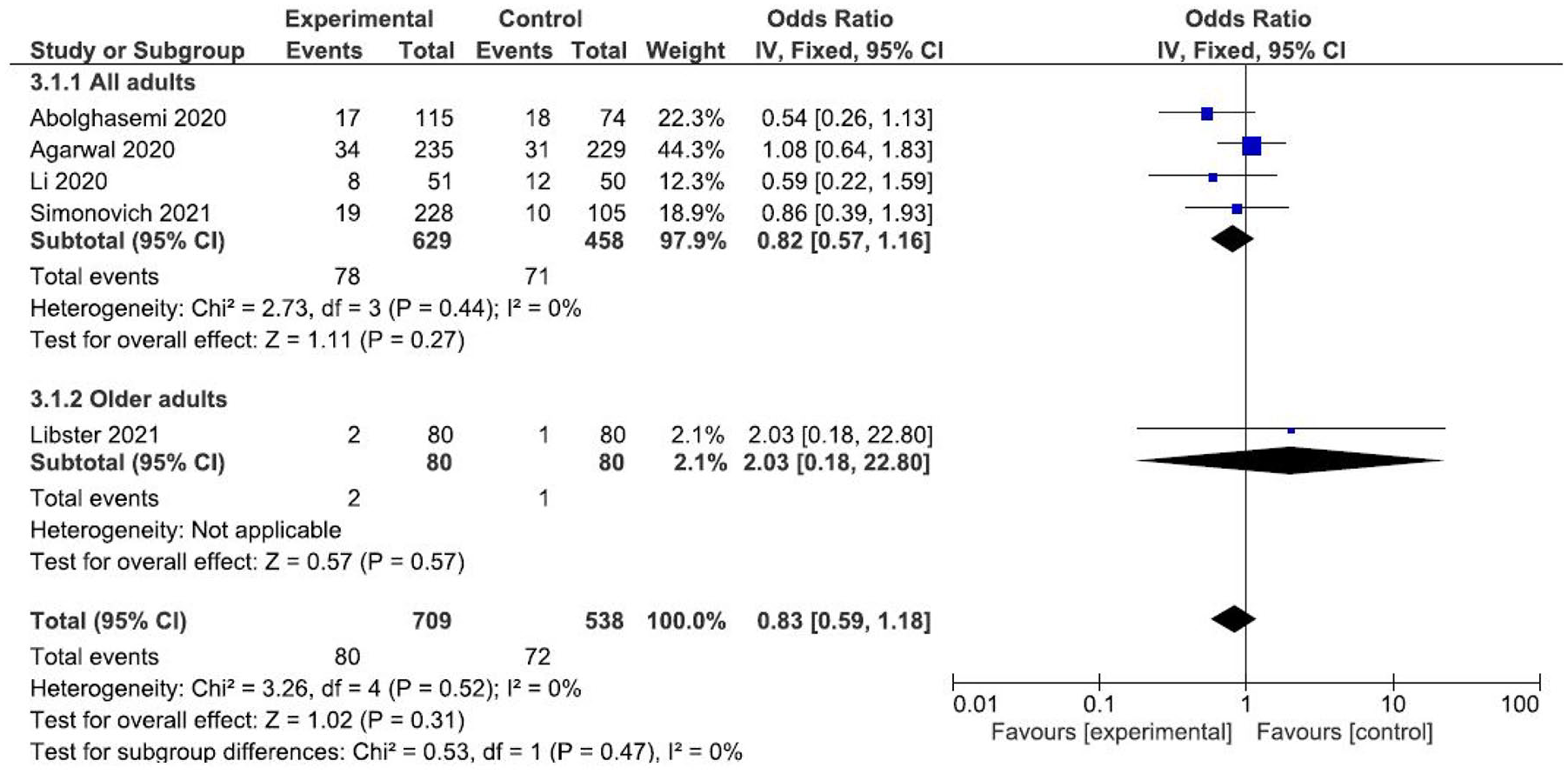
Forest plot of convalescent plasma transfusion for hospitalized patients with SARS-CoV-2 infection. Comparison: convalescent plasma
Discussion
This systematic review was focused on adult hospitalized patients diagnosed with SARS-CoV-2 infection, treated with convalescent plasma transfusion. The studies included in this review were quite heterogeneous regarding the doses of plasma administered, the co-treatment with repositioned antiviral drugs in the experimental group and the control group, and the results obtained by each clinical trial. Considering only controlled clinical trials published in peer-reviewed journals, 1249 patients were included in hospitals from China, India, Iran, and Argentina.
Regarding the overall quality of the studies, three out of five clinical trials were considered at high risk of bias due to the lack of blinding; this is a common characteristic of many clinical studies that started to run under emergency conditions due to the persistent health crisis, with recurring waves in some countries in Latin America, and other countries. 25
Four published systematic reviews on convalescent plasma have shown that this treatment could reduce the mortality,26–29 but did not include only controlled trials and did not include the last studies produced in Argentina by Libster
The use of convalescent plasma was associated with clinical improvement, which is consistent with other previously published studies;26,29,31 however, Li
This early-stage plasma therapy is also supported by a study conducted in the Netherlands, 38 which had to be halted prematurely. The authors showed that late-administered plasma therapy did not benefit patients because they already had high virus-neutralizing antibody titers on the day of study inclusion with titers comparable to the recovered donors’.
The use of neutralizing antibody tests 19 is strongly recommended to ensure the quality of the plasma transfused to patients, without forgetting that the host’s response depends on particular phenomena, such as original antigenic sin. This phenomenon shows that previous exposure to a similar virus triggers a strong but not specifically neutralizing immune response, so having high antibody titers or a strong immune response does not indicate that the virus is being neutralized.
Still, the optimal dosage and the best time point for the convalescent plasma transfusion need to be determined in well-designed clinical trials. 39 Additionally, the genetic variability of the virus and the patients should be analyzed since the treatment may not have the specific immunoglobulins to resolve the infection. 2
Regarding the influence of antiviral drugs used in the articles included in this systematic review, the study by Li
There is enough evidence proving that convalescent plasma administration does not have many severe adverse events in transfusion.16–19,29,33 In contrast, more research is needed on the synergistic effect that plasma could have with other repositioning drugs, as has been demonstrated, for example, with the use of remdesivir, as has been published in other studies.19,29,42
Among the limitations of this study, the rapid generation of new knowledge in times of the pandemic can potentially affect the timeliness of this review in a few months. The second limitation is the heterogeneity and high risk of bias in the studies. In this review, we chose not to issue recommendations with the GRADE methodology due to heterogeneity and the high risk of bias. Another limitation is that not all studies have used the same dosage of convalescent plasma in infected patients. The fourth limitation that must be considered is the use of antiviral drugs in the control groups or both groups of patients in three out of four clinical trials included in this review.
In times of recurring waves of the COVID-19 pandemic, the analysis of potential treatments proposed for hospitalized patients is still necessary since vaccines’ procurement and logistics are still seen within a complex scenario for many low-income countries. In many low-income countries, vaccination is likely to occur six to twelve months after high-income countries, in part due to logistical problems, as stated by the World Bank. 43 so the search for a clinically effective treatment is still a major concern globally.
In the ClinicalTrials.gov platform dozens of clinical trials are currently registered that assess the treatment with plasma, so the addition of new evidence in the coming months could change the direction of the analyses in this review.
Conclusion
The current evidence suggests that transfusion with convalescent plasma cannot reduce the mortality or ventilation requirement in hospitalized patients diagnosed with SARS-CoV-2 infection. More controlled clinical trials conducted with methodologies that ensure a low risk of bias with neutralizing antibody tests to ensure a good quality of plasma are still needed.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666211028077 – Supplemental material for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-tar-10.1177_17534666211028077 for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis by Roberto Ariel Abeldaño Zuñiga, Ruth Ana María González-Villoria, María Vanesa Elizondo, Anel Yaneli Nicolás Osorio, David Gómez Martínez and Silvia Mercedes Coca in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-2-tar-10.1177_17534666211028077 – Supplemental material for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis
Supplemental material, sj-pdf-2-tar-10.1177_17534666211028077 for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis by Roberto Ariel Abeldaño Zuñiga, Ruth Ana María González-Villoria, María Vanesa Elizondo, Anel Yaneli Nicolás Osorio, David Gómez Martínez and Silvia Mercedes Coca in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_17534666211028077 – Supplemental material for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis
Supplemental material, sj-pdf-3-tar-10.1177_17534666211028077 for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis by Roberto Ariel Abeldaño Zuñiga, Ruth Ana María González-Villoria, María Vanesa Elizondo, Anel Yaneli Nicolás Osorio, David Gómez Martínez and Silvia Mercedes Coca in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-4-tar-10.1177_17534666211028077 – Supplemental material for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis
Supplemental material, sj-pdf-4-tar-10.1177_17534666211028077 for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis by Roberto Ariel Abeldaño Zuñiga, Ruth Ana María González-Villoria, María Vanesa Elizondo, Anel Yaneli Nicolás Osorio, David Gómez Martínez and Silvia Mercedes Coca in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-5-tar-10.1177_17534666211028077 – Supplemental material for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis
Supplemental material, sj-pdf-5-tar-10.1177_17534666211028077 for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis by Roberto Ariel Abeldaño Zuñiga, Ruth Ana María González-Villoria, María Vanesa Elizondo, Anel Yaneli Nicolás Osorio, David Gómez Martínez and Silvia Mercedes Coca in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-6-tar-10.1177_17534666211028077 – Supplemental material for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis
Supplemental material, sj-pdf-6-tar-10.1177_17534666211028077 for Clinical effectiveness of convalescent plasma in hospitalized patients with COVID-19: a systematic review and meta-analysis by Roberto Ariel Abeldaño Zuñiga, Ruth Ana María González-Villoria, María Vanesa Elizondo, Anel Yaneli Nicolás Osorio, David Gómez Martínez and Silvia Mercedes Coca in Therapeutic Advances in Respiratory Disease
Footnotes
Author contributions
RAAZ contributed to the development of the research project.
RAAZ, MVE, SMC, AYNO, DGM, and RAMGV performed data collection, analyzed and interpreted the results.
RAAZ, SCM, and RAMGV wrote the article. All authors reviewed and approved the final version.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Data accessibility statement
All data are available upon request to the corresponding author.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
