Abstract
Background:
Selection of the most appropriate device for a switch from one inhaler to an equivalent product is known to have a major impact on clinical outcomes in patients with asthma or chronic obstructive pulmonary disease (COPD). Salmeterol/fluticasone propionate (S/F) Easyhaler® has been demonstrated to be therapeutically equivalent with a reference product. However, no data on real-life effectiveness are currently available for patients switching to S/F Easyhaler from another S/F inhaler.
Methods:
The aim of this prospective, open, multicenter, non-interventional study was to assess clinical effectiveness of propionate S/F Easyhaler in adult asthma and COPD patients switched from another inhaler. The primary endpoints were Asthma Control Test (ACT) and COPD Assessment Test (CAT). Secondary endpoints included assessments of patient satisfaction and preference and physician/nurse perception on S/F Easyhaler use. The study included three visits during a 12-week follow-up.
Results:
A total of 211 patients (160 with asthma; 51 with COPD) were included in the analyses. In patients with asthma, there was a statistically significant increase in the mean ACT score at week 12 (20.2 ± 3.9) compared with the baseline (18.6 ± 4.1), with a mean increase of 1.6 (±3.5) points (p < 0.0001). In patients with COPD, CAT score persisted from baseline (19.9 ± 8.6) to week 12 (19.6 ± 7.0). Patients were significantly more satisfied with Easyhaler and most patients preferred Easyhaler over their previous inhaler. The physicians/nurses reported that it was ‘very easy’ to teach the use of Easyhaler and the training took less than 5 minutes in most cases.
Conclusion:
The results from this prospective real-life clinical study indicate better or at least similar treatment control of asthma and COPD after switching to S/F Easyhaler from another S/F inhaler. This study also shows that S/F Easyhaler was favored by the patients and that it is easy to teach, learn and use in a real-life setting.
The reviews of this paper are available via the supplemental material section.
Keywords
Introduction
Asthma and chronic obstructive pulmonary disease (COPD) are common respiratory diseases which are characterized by chronic inflammation of the airways and associated with high level of morbidity, mortality, and socioeconomic costs.1,2 Asthma affects more than 330 million people worldwide and causes approximately 400,000 deaths annually.3–5 COPD has a comparatively lower prevalence, affecting 251 million people, but a significantly higher mortality, causing 5% of annual global deaths (more than 3 million people).6,7
The long-term goals of asthma management aim at achieving good symptom control and maintaining normal activity levels, as well as minimize the risk of exacerbations, fixed airflow limitation, and treatment side effects. 1 Although asthma in most patients can be controlled with available standard therapies, many patients still experience persistent symptoms and frequent exacerbations.1,8 COPD is not curable, but treatment can significantly relieve and reduce impact of symptoms, improve quality of life, and lower the risk of death. 2
Inhalation is recommended as the primary route of administration for medication used to manage asthma and COPD.1,2 Despite the proven efficacy of inhaled medication, several reports indicate that compliance with asthma/COPD treatments remains poor.9,10 It is estimated that 50% of adults and children on long-term therapy fail to adhere to their treatment regimen. 11 Furthermore, it has been shown that from 20% to 82% of the patients do not use inhalers correctly, which may have clinical implications for treatment efficacy and subsequent disease control.12–15 Correct inhaler training, ease and correctness of use, patient preference, and adherence can play a key role in increasing treatment effectiveness. 1
Salmeterol/fluticasone propionate (S/F) Easyhaler (Orion Pharma, Espoo, Finland) is a multidose dry powder inhaler (DPI), which has been approved in most European countries for the administration of salmeterol and fluticasone propionate, in combination, for the treatment of adolescents (12–17 years of age) and adults with asthma and adults with COPD. The combination of S/F propionate is widely used in management of asthma and COPD with proven efficacy and favorable tolerability.16,17 The combination has been shown to be more effective than fluticasone propionate alone in both indications.16–19 Furthermore, a combination of an inhaled corticosteroid (ICS) and long-acting β2-adrenoceptor agonist (LABA) in a single inhaler is more convenient to use than two separate inhalers, and may therefore improve compliance and lead to greater rates of disease control. 20
There are several reasons for switching a patient from one inhaler to another equivalent product, such as suboptimal treatment response, daily dosing frequency or economic reasons.21–24 In addition, there have been calls to replace pressurized metered-dose inhalers (pMDIs) with the more sustainable DPIs in order to combat climate change.25,26 It is well established that selection of the most appropriate device for a switch has a major impact on clinical outcomes. 22 Recent evidence indicates that patients may be switched from other inhalers to Easyhaler with no reduction in clinical effectiveness or cost increase. 27 Most patients learn to use Easyhaler in less than 5 minutes and it has high patient preference.28,29 However, there are currently no data available on the real-life effectiveness of S/F Easyhaler after switching from another S/F inhaler.
Randomized controlled trials are based on highly selected patient populations and controlled settings, which differ from the realities of clinical or home settings. 30 Therefore, the results from randomized controlled trials may not predict the value of switching patients to a different inhaler in real-life clinical practice, where inhaler technique and device characteristics can influence effectiveness. 21 The primary objective of this study was to assess the clinical effectiveness of S/F Easyhaler in achieving and maintaining asthma or COPD control after a switch from another S/F inhaler. In addition, satisfaction and preference of inhalers, quality of life (asthma), dyspnea (COPD), perception of Easyhaler inhaler, and healthcare resource utilization were assessed. This is the first study evaluating S/F Easyhaler in a real-life clinical setting.
Methods
Study participants
Patients were recruited by general practitioners and pneumologists from outpatient clinics in Germany and Sweden. Adult (⩾18 years of age) patients with diagnosed asthma or COPD who had been using S/F combination treatment for at least 3 months before the beginning of the study, and for whom the decision had already been made to switch to S/F Easyhaler, were eligible for enrollment. Patients were excluded from the study if they were pregnant or breastfeeding, were participating in a clinical trial, had hypersensitivity to any ingredient of the medication, or had any significant medical disease, condition, or other factor that, in the physician’s opinion, might interfere with study assessments or study participation.
All patients gave written informed consent. Verbal and written information about the study was given to the study subject candidates before recruitment. Adequate time and opportunity were given to inquire about details of the study and to decide whether or not to participate before signing the informed consent.
Study design
This was a prospective, open, multicenter, non-interventional study in adult patients with asthma or COPD to assess the clinical effectiveness of S/F Easyhaler to achieve and maintain asthma or COPD control. The data collection was conducted between January 2019 and February 2020.
The study included three visits during a 12-week follow-up: (1) a screening and baseline visit to assess patient eligibility, to train inhaler handling, and to collect baseline data; (2) an interim visit to check the inhalation technique at week 2 (±1 week); and (3) a final assessment visit at week 12 (+4 weeks). S/F Easyhaler 50/250 or 50/500 µg/dose was prescribed according to the local SmPC and routine clinical practice. As in a real-life situation the daily dose of S/F Easyhaler was decided by the treating physician. The study nurse or physician trained the patients to use the Easyhaler according to the instructions.
Primary endpoints
In patients with asthma, the primary endpoint was change in the Asthma Control Test (ACT; QualityMetric, Lincoln, RI, USA) total score from baseline to week 12 follow-up. ACT is a validated self-assessment questionnaire, which is based on a 4-week recall of five items of asthma control. Each item is rated from 1 to 5, resulting in a total score of 5–25, with higher scores reflecting better asthma control. 31 A change of 3 points is considered the threshold for a minimal clinically important difference (MCID). 32
In patients with COPD, the primary endpoint was change in the COPD Assessment Test (CAT) total score from baseline to 12-week follow-up. CAT is a validated, eight-item self-administered questionnaire developed to measure health status impairment in patients with COPD. 33 A CAT score <10 indicates low, 10–20 medium, 21–30 high, and ˃30 very high impact of disease on patient’s wellbeing and daily life.
Secondary endpoints
The investigator assessed asthma symptom control according to Global Initiative for Asthma (GINA) guidelines and the patient was classified to have well-controlled, partly controlled, or uncontrolled asthma. 1 Change in asthma-related quality of life was assessed using the self-administered mini-Asthma Quality of Life Questionnaire (mini-AQLQ). 34 In patients with COPD, dyspnea in daily living was evaluated by the Modified Medical Research Council (mMRC) dyspnea scale, which quantifies disability attributable to breathlessness.2,35 Lung function, evaluated by forced expiratory volume in 1 s (FEV1) and forced vital capacity (FVC), was assessed as part of the routine clinical examination 36 and the sites followed their normal practice for monitoring lung function with serial spirometry.
Patient satisfaction and inhaler preference (previous inhaler at baseline and S/F Easyhaler after 12 weeks of treatment) was assessed using the Feeling of Satisfaction with Inhaler (FSI-10) questionnaire. 37 In addition, preference regarding Easyhaler and the previous S/F inhaler was asked at the final visit. The physician or nurse rated his/her perception of Easyhaler overall use for each patient with a predefined questions and answer categories, at baseline and at the week 12 visit. At baseline, the Easyhaler training activities were evaluated and the reason to switch to Easyhaler from the previous inhaler was recorded. At week 12, the physician or nurse rated his/her opinion on integration of the inhaler in patient’s everyday life and patient’s compliance and capability to use Easyhaler.
Healthcare resource utilization within the previous 3 months was assessed at baseline (treatment with previous inhaler) and at week 12 (treatment with S/F Easyhaler) using a questionnaire which recorded: number of outpatient clinic visits due to respiratory symptoms, number of hospitalizations for respiratory symptoms, number of missed working days due to respiratory symptoms, and respiratory medications used.
Adverse events (AEs) and serious AEs (SAEs) were recorded from enrollment until the patient left the study or the final visit. AEs were summarized and the severity was categorized. In addition, discontinuations due to AEs were listed.
Statistical analyses
The changes in the ACT and CAT total score from baseline to final visit at week 12 were evaluated by analysis of variance for repeated measures (RM-ANOVA) (primary endpoint). The estimated difference and standard deviation of the differences were calculated for exploratory purposes. Of the secondary endpoints, the FEV1 and FVC and their predicted values, FEV1/FVC, patient satisfaction/preference, physician/nurse perception, mini-AQLQ, and mMRC were evaluated in a similar manner to the primary endpoints. If any of the endpoints did not follow the assumptions of RM-ANOVA, a Wilcoxon signed-rank test was applied. Asthma symptom control was tabulated by frequency tables. All the parameters that were common across patients with asthma and COPD were reported both separately in patients with asthma and COPD and in the total population. Changes in the categorized ACT, CAT, and asthma symptom control were tabulated by visit. The healthcare utilization variables and AEs were evaluated by frequency tables and summary statistics.
Analyses with the full analysis set (FAS) are reported. FAS refers to all patients who had taken at least one dose of study treatment and for whom baseline and at least one post-baseline evaluation of the primary endpoint was available.
It was estimated that a total of 230 patients was required for the study. The estimate was based on the assumption that to detect non-inferiority in COPD primary endpoint between baseline and final visits, a sample size of 62 was required to provide an approximate power of 80% at a one-sided significance level of 0.05. This was based on the assumptions that there was no worsening in CAT score after switching, the standard deviation of the CAT was 7.5, and there was a correlation of 0.65 between baseline and final visits in the CAT score. The total sample size was based on the 2:1 ratio of asthma and COPD patients in the study (accounting for an expected dropout rate of 18%).
All statistical analyses, tables, and subject data listings were performed with SAS® (for Windows) version 9.4. (SAS Institute Inc., Cary, NC, USA).
Ethical considerations
The study was performed in accordance with the Declaration of Helsinki and the Standards of Good Clinical Practice, Good Pharmacoepidemiology Practice, the European Network of Centres of Pharmacoepidemiology and Pharmacovigilance, and Good Pharmacovigilance Practice. The study was approved by the Ethik-Kommission der Ärztekammer Hamburg (Weidestr. 122 b, 22083 Hamburg, Germany; PV5902) and local ethics committees as appropriate. The study is registered at ClinicalTrials.gov (identifier NCT03755544).
Results
Patient characteristics
A total of 231 patients (178 with asthma; 53 with COPD) were enrolled in the study (Table 1). All the enrolled patients were White (n = 231), the majority of them were female (n = 141; 61.0%), and the mean age was 57.6 (±15.8) years. A majority of the patients (n = 135; 58.4%) had never smoked, 61 patients (26.4%) were former smokers, and 35 patients (15.2%) were current smokers. The mean pack years for asthma and COPD patients were 16.0 and 33.7, respectively. Altogether, 20 patients with asthma and 2 patients with COPD discontinued the study. Reasons for the treatment discontinuation were adverse event (n = 9), personal reason (n = 7), lost to follow-up (n = 4), and other reasons (n = 2). A total of 211 patients (160 with asthma; 51 with COPD) fulfilled the criteria for FAS and were included in the analyses of the primary and secondary endpoints (Figure 1).
Baseline characteristics for the enrolled study population.

Based on analysis of full analysis set.
COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; SD, standard deviation.
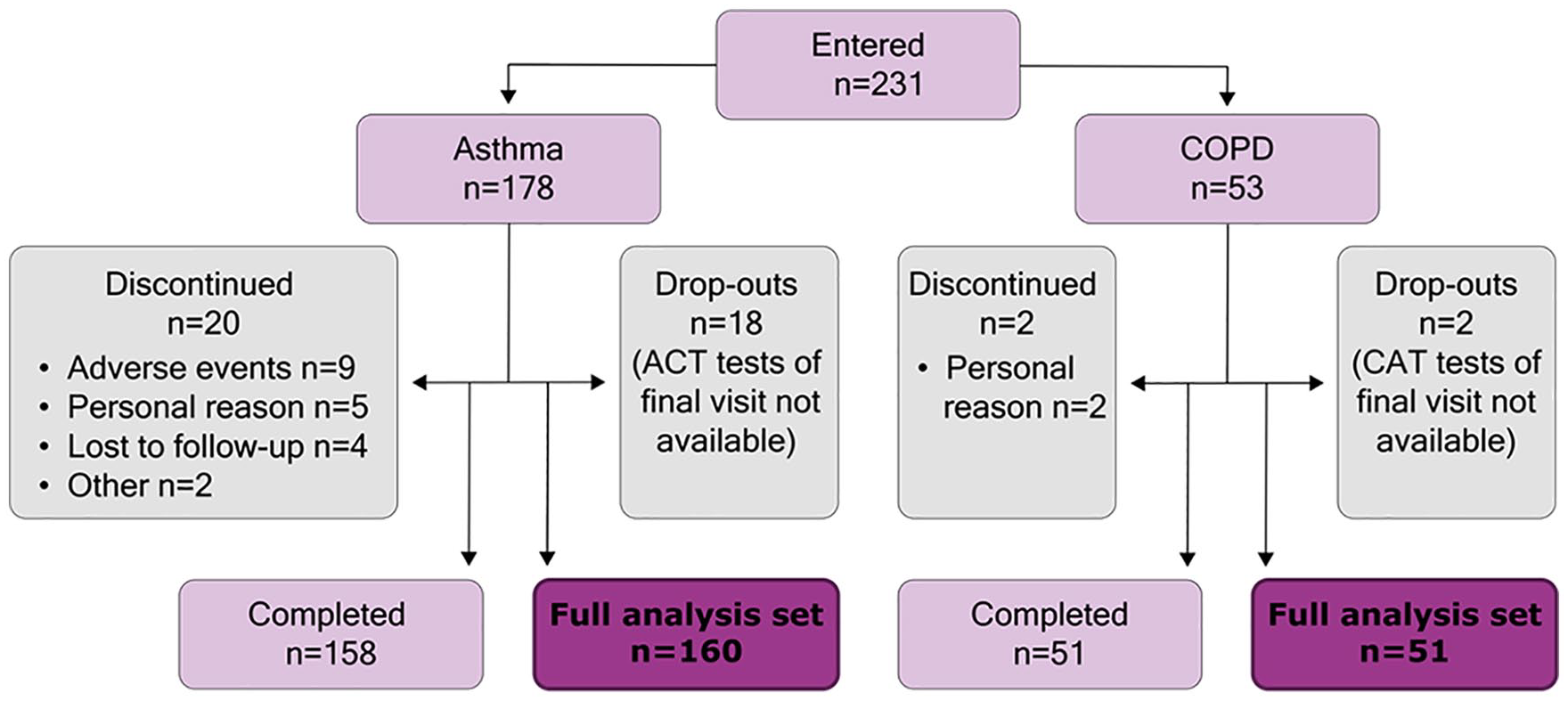
Patient disposition. Analyses were performed using the full analysis set.
For 12 patients with asthma the dose was higher and for seven patients lower after the switch, whereas 4 patients with COPD started with higher dose after the switch. During the study there were only five changes in the daily dose of S/F Easyhaler. For three subjects the dose was changed to lower dose and for one subject a higher dose was introduced for a week and then changed back to the earlier level.
Changes in ACT score, asthma symptom control, and asthma-related quality of life
In patients with asthma, there was a statistically significant increase in the mean ACT score at week 12 (20.2 ± 3.9) compared with the baseline (18.6 ± 4.1), with a mean increase of 1.6 (±3.5) points (p < 0.0001) [Figure 2(a)]. Improvement of asthma symptoms was seen in all five ACT items during the study. This primary endpoint result indicates at least the same or somewhat better disease control after 12 weeks treatment with S/F Easyhaler compared with previous inhaler, considering that the MCID criteria was not exceeded in the total asthma population but statistical significance was reached. ACT score increased by ⩾3 points (MCID) in 58 (36.3%) patients and decreased by ⩾3 points in 13 (8.1%) patients.

(a) Change in mean ACT total score from baseline to final visit and (b) proportion (%) of patients with well-controlled, partly controlled or uncontrolled asthma at baseline and final visits. Categories were defined according to 2018 GINA guideline. 1 Analyses were performed with FAS of asthma patients (n = 160) and the p-value was analyzed by using RM-ANOVA.
The number of subjects with well-controlled asthma according to GINA classification increased significantly from the baseline to the final visit. A total of 113 patients (70.6%) had partly or well-controlled asthma at the baseline compared with 143 patients (89.4%) at week 12 (p < 0.00001) [Figure 2(b)]. When compared with baseline, asthma patients also experienced a statistically significant improvement in mini-AQLQ score after 12 weeks of S/F Easyhaler treatment. The mini-AQLQ score increased from 4.87 (±1.11) at baseline to 5.32 (±1.04) at the final visit, which corresponds to an improvement of 0.45 (±0.90) points (p < 0.0001).
Changes in the CAT score, symptom burden, and dyspnea in COPD
CAT scores remained at the same level from baseline (19.9 ± 8.6) to week 12 (19.6 ± 7.0) in patients with COPD (−0.3 ± 6.3; p > 0.05) [Figure 3(a)]. No significant difference was observed in the symptom burden between the baseline and final visits [Figure 3(b)]. CAT score decreased by ⩾2 points in 20 patients (39.2%) and increased by ⩾2 points in 19 patients (37.3%). In addition, no significant change was observed in the mean mMRC dyspnea score from baseline (1.4 ± 1.0) to the final visit (1.3 ± 1.1; change from the baseline −0.1 ± 0.9, p > 0.05).

(a) Change in mean CAT total score from baseline to final visit and (b) proportion (%) of patients with low, medium, high, and very high impact of COPD at baseline and final visits. Categories were based on CAT scores. 32 Analyses were performed with FAS of COPD patients (n = 51) and the p-value was analyzed by using RM-ANOVA.
Spirometry
Lung function parameters, including FEV1, FVC, FEV1/FVC and all percentage predicted values, remained stable from baseline to final visit (Annex Table 1).
Patient satisfaction and preference and physician/nurse perception on S/F Easyhaler use
Patients were more satisfied with Easyhaler than with their previous inhaler (Table 2). The mean increase in the FSI-10 total score during the study was 5.9 and 6.2 for patients with asthma and COPD, respectively. The differences were statistically significant in both asthma and COPD groups (p < 0.0001). Altogether, among those who answered, 76.2% of patients with asthma (n = 115/151; p < 0.0001) and 73.5% of patients with COPD (n = 36/49; p = 0.001) preferred S/F Easyhaler over their previous S/F inhaler.
Patient’s FSI-10 score at baseline (3-month period with previous inhaler) and at final visit (after 12 weeks treatment with S/F Easyhaler).

COPD, chronic obstructive pulmonary disease; FSI-10, Feeling of Satisfaction with Inhaler; SD, standard deviation; S/F, salmeterol/fluticasone.
The most common reason for switching to Easyhaler from a previous inhaler was ease of use (45.9% of patients). The physicians and nurses reported that it was ‘very easy’ to teach the use of Easyhaler to a majority of the patients (67.5%; n = 137/203), and in most cases (82.1%; n = 165/201) the training took less than 5 minutes (Table 3). Only seven patients (3.3%) needed additional training for inhalation technique during the interim visit at week 2. At the final visit, the physicians and nurses reported that 28.4% (n = 59/208) of the asthma and COPD patients had a ‘good’ and 63.9% (n = 133/208) ‘very good’ integration of Easyhaler usage in everyday life. 29.6% (n = 61/206) of the patients had a ‘good’ and 66.0% (n = 136/206) ‘very good’ treatment compliance. Altogether, 87.0% (n = 180/207) of the patients had no difficulties in handling Easyhaler and 80.7% (n = 167/207) of the patients reported that they will continue to use Easyhaler.
Physician/nurse perception on S/F Easyhaler use.
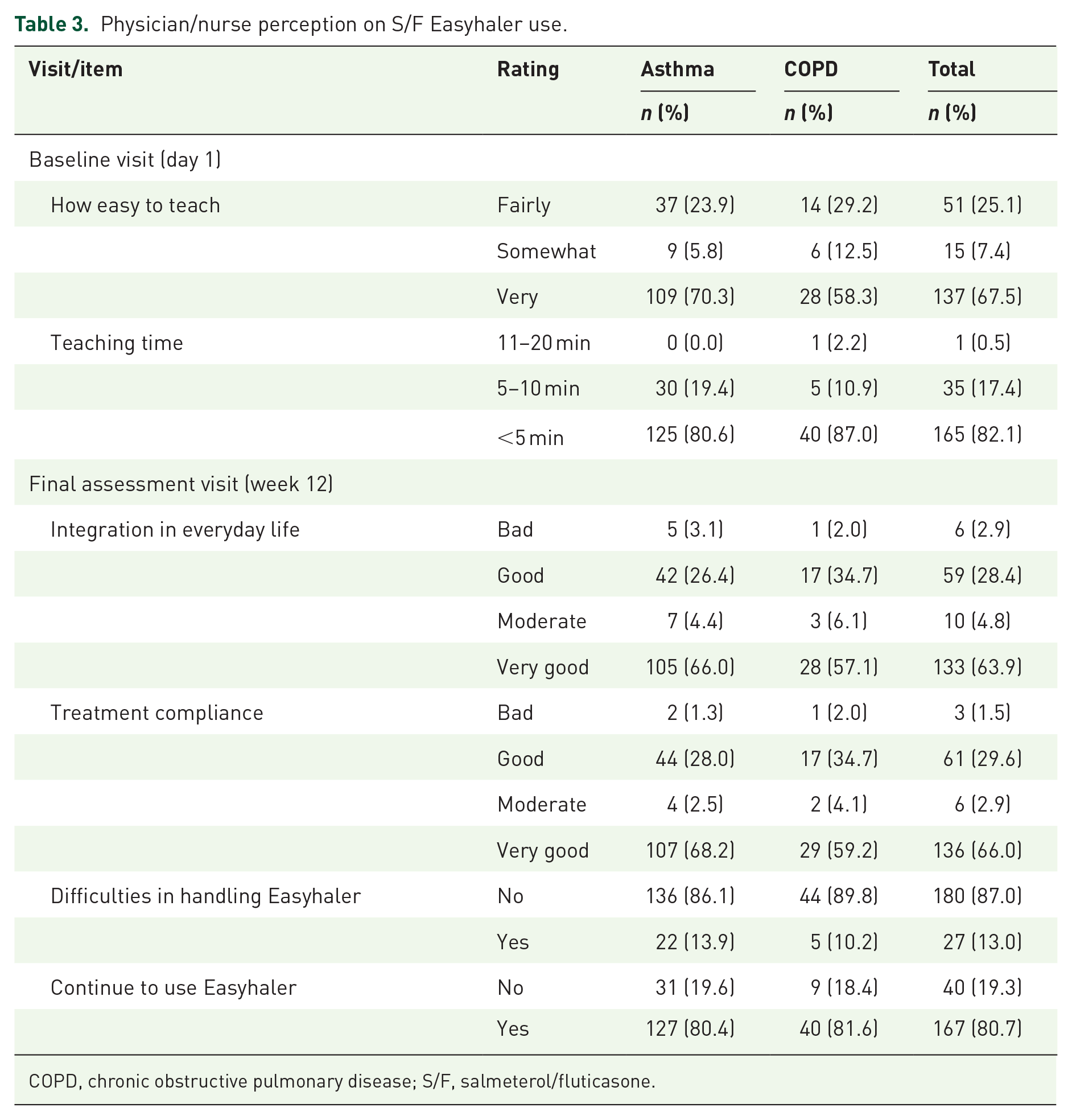
COPD, chronic obstructive pulmonary disease; S/F, salmeterol/fluticasone.
Healthcare resource utilization
Decreases in patient-reported healthcare resource utilization due to respiratory symptoms were observed between the 3-month period prior to the switch and the 3-month period after it. In the asthma group, there were 13 (8.1%) patients hospitalized prior to switch versus 2 (1.3%) after the switch, 32 (20.0%) patients with visits to outpatient clinics prior to switch versus 18 (11.3%) after the switch, and 92 (58.2%) patients with visits to physicians prior to switch versus 54 (33.8%) after the switch (Annex Table 2). The corresponding results for the COPD patients were 4 (7.8%) versus 1 (2.0%) patients hospitalized, 9 (17.6%) versus 5 (10.0%) patients with outpatient clinic visits, and 34 (66.7%) versus 21 (42.9%) patients with visits to physician. Among patients with asthma, 25 (15.8%) patients reported missed working days in the 3-month period prior to the switch compared to 9 (5.7%) after the switch. Among patients with COPD, the corresponding figures were five (9.8%) and four (8.3%), respectively.
Safety
A total of 70 AEs were reported by 47 patients after the start of study treatment. A total of 21 AEs in 17 patients were considered to be related to the study treatment; 17 were reported in patients with asthma and 4 in patients with COPD. Three subjects reported three SAEs. The most common AEs during the study were cough in eight patients and nasopharyngitis in four patients.
Discussion
This prospective, open, multicenter, non-interventional study demonstrated the real-life clinical effectiveness of S/F Easyhaler among patients with asthma and COPD who have been switched to S/F Easyhaler from another S/F inhaler. The results reported here also showed that Easyhaler is highly preferred by patients and easy to teach and use from the healthcare professional perspective.
In this study, statistically significant improvement of ACT score was observed in patients with asthma during the 12-week follow-up. In addition, there was a statistically significant improvement in the number of patients with well-controlled asthma and in asthma-related quality of life. These results imply better or at least similar treatment control in asthma after switching to S/F Easyhaler from the previous inhaler. Similar real-life evidence has recently been reported in patients with asthma who switched from budesonide/formoterol Turbuhaler® (AstraZeneca, London, UK) to budesonide/formoterol Easyhaler. 38 In the current study the ATC score increased, while FEV1 remained stable from baseline to final visit. This discrepancy may at least partly be explained by high FEV1 at baseline (82.4% of predicted) and high proportion of female (62.9%). 39
In patients with COPD, persistent CAT score and level of symptom burden consistent with the baseline was observed during the 12-week follow-up, suggesting that the treatment control remained after the switch to S/F Easyhaler.
The clinical development program of S/F Easyhaler has involved in vitro and in vivo comparisons of S/F Easyhaler with a reference product, Seretide® Diskus®/Accuhaler® (GlaxoSmithKline, London, UK).40,41 These studies have shown that S/F Easyhaler is therapeutically equivalent with the reference product. This study brings important additions to the existing evidence of S/F Easyhaler by providing data on real-life effectiveness and safety.
The selection of the most appropriate inhaler requires consideration of a patient’s ability to use the device correctly, preference, and satisfaction with the device. 42 Greater patient satisfaction is known to lead to improved treatment compliance and greater rates of disease control. 29 In consistency with earlier Easyhaler studies, high patient preference for Easyhaler was observed in this study.43–46 Approximately three out of four patients with asthma and COPD preferred to use Easyhaler over their previous S/F inhaler. There were also statistically significant increases in FSI-10 scores at final visit compared to baseline in both asthma and COPD groups. In addition, our results show that in the majority of cases, it took less than 5 minutes for a healthcare professional to teach the use of Easyhaler to the patient. This result is in line with a recent study of more than 1000 Hungarian patients with asthma or COPD, which showed that most patients were able to learn the correct use of Easyhaler in less than 5 minutes.28,29 In this real-life study it was not possible to evaluate the time needed to train the previous inhaler.
Concurrent use of salmeterol and fluticasone propionate does not result in untoward interaction that would affect the pharmacodynamic or pharmacokinetic profiles of the individual drugs or their adverse effect profiles. 47 The safety and tolerability profile of S/F Easyhaler has shown to be comparable to other ICS/LABA combinations. 40 This study suggests that S/F Easyhaler has a favorable safety profile also in real-life setting. Altogether, 21 treatment-related AEs were reported by 17 patients. Nine patients discontinued treatment due to AEs, but all of these events were assessed as mild, with the exception of moderate cough in one patient.
Besides clinical effectiveness, safety, and ease of use, there has been an increasing emphasis on overall treatment costs in asthma and COPD, as well as the environmental effects of inhalers. In an earlier study, the patients who switched from other ICS devices to Easyhaler were more likely to achieve overall asthma control and have comparable or lower asthma-related healthcare costs than patients, who switched to any other inhaler. 27 The healthcare resource utilization reported here are consistent with these earlier results. There was a decrease in the healthcare resource use during the 12-week study period compared to the resource use 3 months before the study initiation, and the overall resource use was low. Easyhaler is also an environmentally sustainable treatment option: in terms of carbon footprint, DPIs are propellant-free and have less than 10% of the climate impact of hydrofluorocarbon-containing pMDIs. 26
The main strength of this study is that it provides evidence on the real-life effectiveness of S/F Easyhaler for the first time. The decision to switch to S/F Easyhaler had already been made before patients were enrolled into the study. Study participation was proposed consecutively to all patients eligible for the study to minimize selection bias, and the prescription was made according to routine clinical practices. Therefore, the study population is considered to be representative of real-life S/F Easyhaler patients in the participating clinics. The primary endpoints of the study are utilized in usual practice and they are meaningful to patients and, in overall, the data collection and instruments were based on well-established and validated methods. The study setting used here is well-suited for understanding real-life effectiveness of the product and provides evidence that can guide real-world use in clinics.
The key limitations include open study design and lack of comparator arm. In addition, 10% and 21% of patients with asthma were current or ex-smokers, respectively, so potential COPD complications among them cannot be ruled out. It is important to note also, that most analyses were based on patient-reported outcomes. Participation in a study may, as such, increase patients’ adherence to treatment or lead to better compliance with instructions from their doctor. Therefore, the results from this study should be considered with some caution.
The results from this prospective, real-life clinical study indicate better or at least similar treatment control of asthma and COPD after switching to S/F Easyhaler from another S/F inhaler. This study shows also that S/F Easyhaler is favored by the patients and that it is easy to teach, learn, and use in real-life setting.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666211027787 – Supplemental material for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice
Supplemental material, sj-pdf-1-tar-10.1177_17534666211027787 for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice by Ines Vinge, Jörgen Syk, Athanasios Xanthopoulos, Hendrik Laßmann, Mikko Vahteristo, Ulla Sairanen, Satu Lähelmä, Rudolf Hennig and Matthias Müller in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-2-tar-10.1177_17534666211027787 – Supplemental material for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice
Supplemental material, sj-pdf-2-tar-10.1177_17534666211027787 for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice by Ines Vinge, Jörgen Syk, Athanasios Xanthopoulos, Hendrik Laßmann, Mikko Vahteristo, Ulla Sairanen, Satu Lähelmä, Rudolf Hennig and Matthias Müller in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_17534666211027787 – Supplemental material for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice
Supplemental material, sj-pdf-3-tar-10.1177_17534666211027787 for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice by Ines Vinge, Jörgen Syk, Athanasios Xanthopoulos, Hendrik Laßmann, Mikko Vahteristo, Ulla Sairanen, Satu Lähelmä, Rudolf Hennig and Matthias Müller in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-4-tar-10.1177_17534666211027787 – Supplemental material for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice
Supplemental material, sj-pdf-4-tar-10.1177_17534666211027787 for A non-interventional switch study in adult patients with asthma or COPD on clinical effectiveness of salmeterol/fluticasone Easyhaler® in routine clinical practice by Ines Vinge, Jörgen Syk, Athanasios Xanthopoulos, Hendrik Laßmann, Mikko Vahteristo, Ulla Sairanen, Satu Lähelmä, Rudolf Hennig and Matthias Müller in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors thank the investigators and patients taking part in the study. Medical writing support was provided by Tero Ylisaukko-oja, from MedEngine Oy, funded by Orion Corporation. Harlan Barker, from MedEngine Oy performed the language review.
Conflict of interest statement
IV reports no conflicts of interest. JS reports personal fees from Orion Corporation during the conduct of the study. MM received consulting fees, and AX, HL and RH received fees for preparation of the manuscript from Orion Corporation during the conduct of this study. SL, MV and US are employees of Orion Corporation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was funded by Orion Corporation.
Data-sharing statement
The data is available upon reasonable request
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
