Abstract
Background and aims:
Post-tuberculosis bronchial stenosis (PTBS) is one of the most common complications of tracheobronchial tuberculosis. Silicone stent serves as a major treatment for maintaining airway patency. However, silicone stent placement remains a large challenge in patients with severe cicatricial PTBS. Our objective was to evaluate the efficacy and safety of covered, self-expanding, metallic stents (SEMSs) as a transition to silicone stent implantation for treating severe PTBS.
Methods:
We retrospectively reviewed the data of patients with severe PTBS who received airway stenting in the First Affiliated Hospital of Guangdong Medical University between September 2015 and May 2019. The types of the stent, intervention procedures, bronchoscopic findings, clinical outcomes and related complications were collected and analyzed.
Results:
Fifty-eight cases with severe PTBS were included in this study. Thirteen (22.4%) of the patients received bronchial silicone stent implantation immediately after dilations. For the remaining 45 (77.6%) patients, silicone stents could not be deployed after dilations and SEMSs implantation was implemented as a bridge to silicone stenting. The SEMSs were placed for an interval of 28.4 ± 11.1 days. All of the silicone stents were inserted successfully following the removal of SEMSs. No SEMS-related complication occurred. The subgroup analysis showed that patients who received transitional SEMSs had less luminal caliber but fewer transbronchial dilations before silicone stent implantation (
Conclusion:
Covered SEMS placement as a transition to silicone stenting could serve as a feasible procedure to reduce complications and improve the success rate of silicone stent implantation in patients with severe PTBS.
Keywords
Introduction
Tuberculosis (TB) is the world’s leading cause of death by an infectious pathogen. A total of 1.4 million people died from TB in 2019. 1 Tracheobronchial tuberculosis (TBTB) is defined as a tuberculous infection of the center airway, reported incidence around 10–54.3% among patients with active pulmonary TB.2–7 Tracheobronchial stenosis reportedly occurred in 59.7% TBTB patients, 7 commonly inducing obstructive pneumonia, pulmonary atelectasis, or even respiratory failure.
Interventional bronchoscopic procedures, including laser, electrocautery, cryotherapy and balloon dilation, are widely used for treating patients with airway stenosis, relieving symptoms and improving lung function. Silicone stent, which serves as the ‘gold standard’ for the management of benign airway stenosis,8–10 could contribute to re-establishing the luminal patency of the stenotic airway. However, TBTB might induce development of granulation proliferation, severe cicatricial constriction and airway distortion, making it a challenge to place silicone stent in the stenotic airway, with a failure rate of 27.2% in patients with TBTB-related tracheobronchial stenosis. 11
Self-expanding metallic stent (SEMS) is controversial in the management of benign airway stenosis for its related complications and difficulty in removal of the stents. 12 Nevertheless, the thin wall woven with flexible nitinol wire mesh and the self-expanding characteristic of SEMS are conducive for deploying in the complex stenotic airway and providing a sustained tracheo-bronchodilation effect. Hence, the readily movable covered SEMS could act as a transition to the silicone stenting in the management of severe post-TB bronchial stenosis (PTBS).
Materials and methods
Patients
We reviewed the clinical data of patients with PTBS who were treated with silicone stent at the First Affiliated Hospital of Guangzhou Medical University between September 2015 and May 2019. We recruited patients based on the following selection criteria: (1) severe PTBS with the luminal diameter reduced ⩾75% and the length of stenotic segment >2 cm); (2) recurrent attacks of breathlessness due to airway restenosis after balloon dilation; (3) complex stenosis with cartilage involvement and circumferential contraction scarring (length >1 cm).13,14 The exclusion criteria were as follows: (1) active pulmonary TB; (2) severe bronchiectasis or destructive pulmonary disease; (3) simple stenosis without contracted scarring;13,14 or (4) absence of follow-up data.
The demographics, clinical characteristics, interventional bronchoscopic procedures and clinical outcomes were collected and analyzed. All patients were constantly informed and the study protocol was approved by the Ethics Committee of the First Affiliated Hospital of Guangdong Medical University (ethics review No.: 2018-16).
Intervention procedure
Three-dimensional computed tomography (CT) image reconstruction and bronchoscopy (BF-260, Olympus Corporation, Japan) were performed to evaluate the airway stenosis before the interventional treatment. All procedures were performed through rigid bronchoscope (Storz Medical AG, Germany) under general anesthesia. Holmium laser (VersaPluse PowerSuite, Lumenis, USA), Nd: YAG laser (MY100C, Ligenesis, China), electrosurgical unit (VIO200D, Erbe, Germany) combined with ballooning were employed to resect fibrous bands and dilate the airway for the access of airway stent. A silicone stent would be implanted directly if the rigid bronchoscope could pass through the airway stenotic segment after the bronchoscopic treatment. Otherwise, a covered SEMS (Nanjing Eco, Micro-Tech, China; or Ultraflex, Boston Scientific, USA) with outer diameter of 10–12 mm would be placed for 2–4 weeks prior to silicone stenting. A silicone stent (Dumon, Novatech, France) with outer diameter of 9–11 mm was implanted after dilation treatment or removal of SEMS (see the flow chart in Figure 1). The type and size of silicone stents were determined based on the luminal diameter and length of the stenotic segment post interventional treatment. The silicone stent would be placed for at least 3 months; prolonged placement or removal of the stents was based on the physician’s decision according to the bronchoscopic follow-ups and the airway condition.
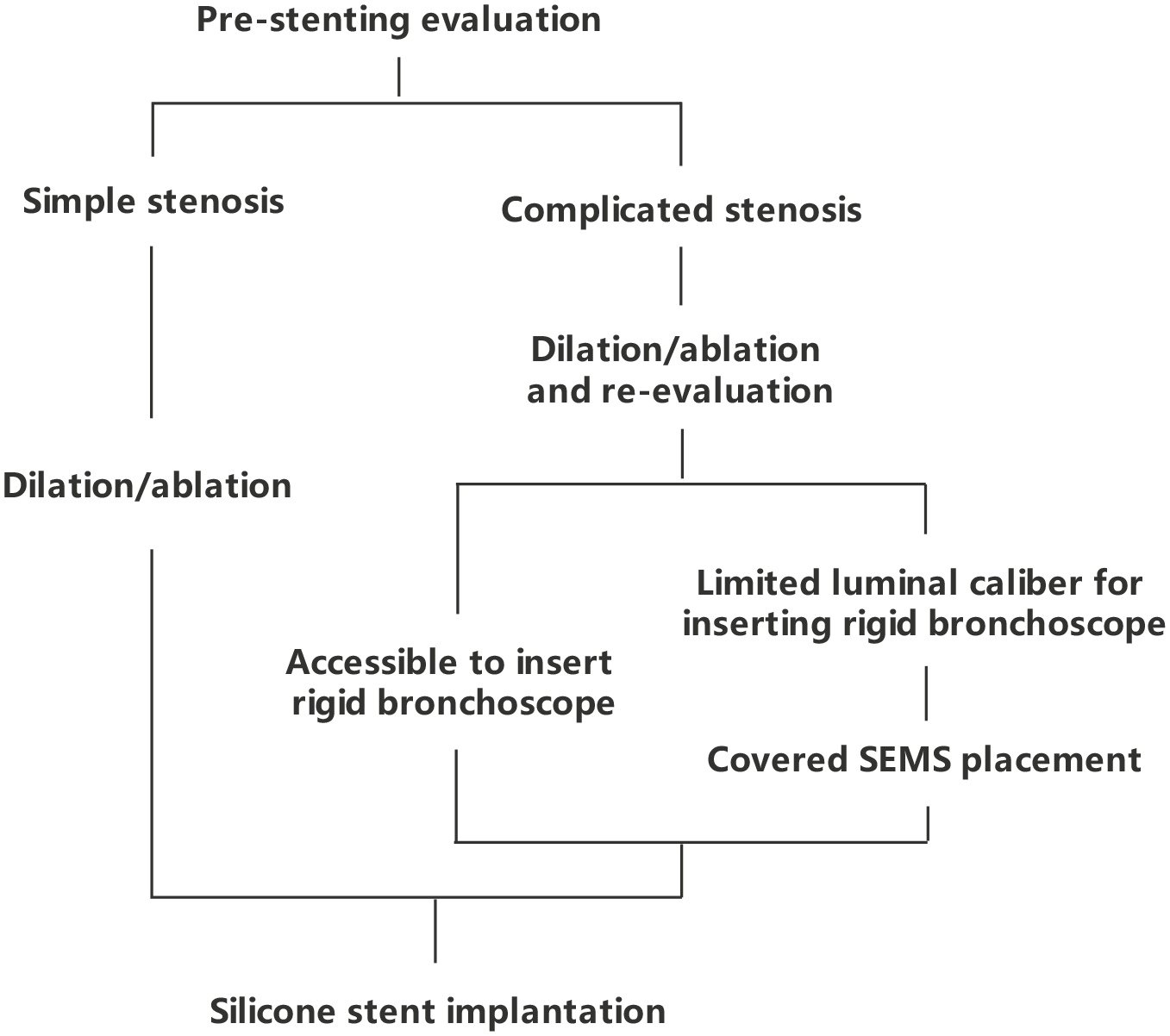
Flow chart of the silicone stent placement for severe post-tuberculosis bronchial stenosis.
Efficacy and safety evaluation
The bronchoscopic follow-ups were conducted on the second day and every 2 weeks after the placement of SEMS, and at the first, second, third, six, ninth and 12th months after silicone stenting for monitoring the related complications, including pneumothorax, mediastinal emphysema, airway rupture, secretion retention, stent migration and granulation tissue formation. Data on lung function test and the dyspnea index [Modified Medical Research Council dyspnea scale (mMRC)] were collected before and after treatment to assess the clinical effects of the airway stenting. All the patients were followed up for 1 year after the removal of silicone stent.
Statistical analysis
Statistical analysis was performed using SPSS version 22.0 (SPSS Inc., Chicago, USA). Data were expressed as median (range) values for continuous variables or as number (percentage). Paired
Results
Patients’ characteristics
A total of 121 TBTB patients with severe bronchial stenosis were treated with silicone stents between September 2015 and May 2019. Twenty-eight patients with simple stenosis and 35 patients who transferred to a local hospital after airway stenting were excluded. Finally, 58 out of 121 (47.9%) patients with complex PTBS were enrolled in this study. All patients were diagnosed of TBTB-related airway stenosis without active pulmonary TB. The clinical characteristics of patients are summarized in Table 1 and the flowchart of bronchoscopic interventional management is shown in Figure 2.
Baseline characteristics of the study population.

Data are presented as
CT, computed tomography; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity.

Flow chart of bronchoscopic interventional management in post-tuberculosis bronchial stenosis patients.
Bronchoscopy findings and outcomes of SEMS
In 58 enrolled patients, the median (range) luminal diameter increased from 2 (0–3) mm to 6 (5–7) mm, average 30 mm (20–40) in length, after conventional bronchoscopic procedures (laser, electrocautery or balloon dilation). Covered SEMSs implantation was required in 45 out of 58 (77.6%) patients for the limited airway caliber to deploy a silicone stent even after bronchoscopic treatment. All patients had immediate symptoms alleviated after stenting, the mMRC scale improved from 1.79 ± 0.60 to 0.23 ± 0.43 (
Granulation tissue formation (23.3%, 10/43) and secretion retention (11.6%, 5/43) occurred after SEMSs implantation, which were simple to manage. No severe complication occurred and the stents were well tolerated. All covered SEMSs stents could be removed successfully without distinct airway injury or stent removal-related complications (e.g. airway laceration or bleeding). The median (range) luminal diameter of stenotic airways had been increased to 9 (8–12) mm after stenting. The bronchoscopic findings and the outcomes of SEMS are summarized in Table 2. 15
Bronchoscopy findings and the outcome of self-expanding metallic stent implantation (
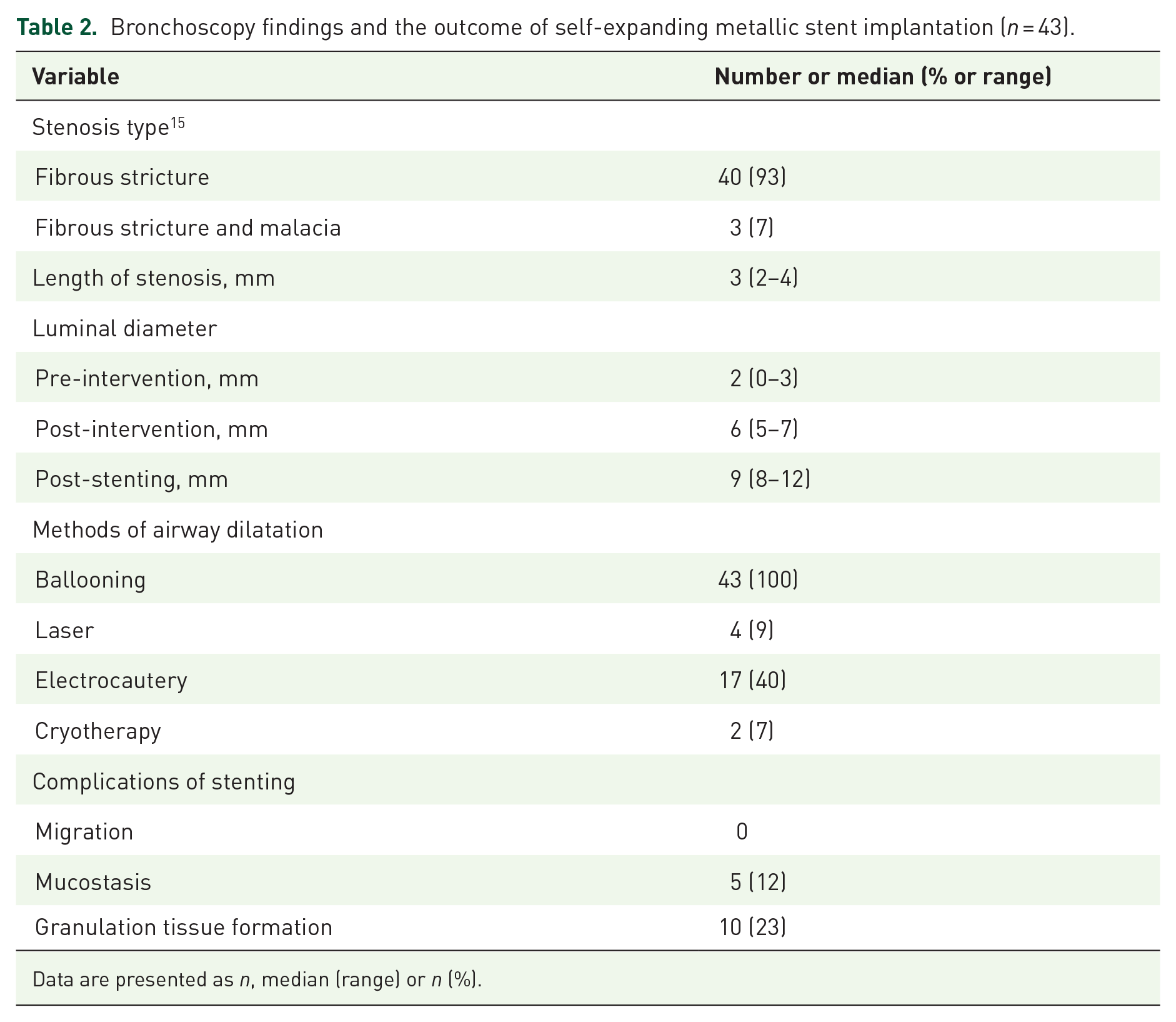
Data are presented as
Outcomes of silicone stents implantation
Silicone stents were implanted immediately after the removal of SEMSs in 45 out of 58 patients. All of the silicone stents were implanted successfully without any procedure-related complications. The chest CT and the bronchoscopic images of a typical case before and after stenting are shown in Figure 3. The other 15 patients received silicone stents implantation directly after the conventional bronchoscopic procedures; however, two of them failed in treating with silicone stent directly because the stents could not be deployed fully.

Chest computed tomography (CT) and the bronchoscope images before and after stenting in a 36 year-old patient with post-tuberculosis bronchial stenosis. (a) Left main bronchus severe stenosis. (b) A covered self-expanding metallic stent (SEMS) (Micro-Tech, 10 mm × 40 mm) was placed for 1 month. (c) The SEMS was transitioned to a straight silicone stent (Dumon, BD, 12 mm × 40 mm). (d) Chest CT before stenting treatment. (e) Chest CT before the removal of covered SEMS.
Overall, 70.7% (41 out of 58) patients presented a stable clinical condition during the follow-up period of 12 months, 36 patients had the silicone stents removed after a median of 14 months (7–32), five patients with airway malacia experienced a prolonged stent placement of an interval of 24 (18–32) months. Repeat interventional procedures were required in 25.9% (50 out of 58) patients due to granulation tissue overgrowth. Stents replacement occurred in two patients due to the angulation and migration of the stents in the third and eighth months. A total of 32.2% (18 out of 58) patients experienced minor complications, including cough, secretion retention and granulation proliferation.
Subgroup analysis
Patients were divided into two groups for analysis of the outcomes of the stenting treatment: Group 1, patients underwent a silicone stent placement after bronchoscopic intervention; Group 2, patients received a SEMS as a transition to silicone stent implantation. There was no significant difference in age, sex, duration of silicone stents placement, symptoms and stents-related complications between these two groups. Subgroup analysis showed that, compared with Group 2, patients in Group 1 had greater luminal diameter before and post bronchoscopic dilation. Further, patients in Group 1 had larger numbers of bronchoscopic dilatations with a lower success rate of silicone stents placement compared with those in Group 2 (Table 3).
Subgroup analysis in patients who received bronchoscopic interventions.

Data are presented as
Data in bold indicated the comparison between Group 1 and Group 2 with statistical significance.
Discussion
Silicone stent implantation remains a great challenge for patients with severe airway stenosis. In this study, we were the first to employ the SEMS as a bridge to silicone stent placement in the management of severe PTBS. The results demonstrated that the short-term placement of SEMS could dilate the stenotic airway with easy removal for reducing related complications, allowing the successful implantation of silicone stent. It is a novel and safe bronchoscopic procedure with considerable feasibility, contributing to the management of severe post-TB airway stenosis and increase of the success rate of silicone stenting.
Interventional bronchoscopic techniques have been widely used in the treatment of airway stenosis, instead of surgical operation with potential related complications.8,16 However, airway restenosis remains one of the most common complications after bronchoscopic dilation (e.g. laser, high-frequency electrosurgery, cryotherapy or balloon dilation).8,17 Silicone stent, regarded as the “gold standard” for treating benign stenosis,
10
has limitations in deployment and implantation in the tortuous and severely stenotic airways. Low
Previous studies recommended that silicone stents should be placed for 6–18 months in benign airway stenosis.8,24,25 In the current study, silicone stents could be implanted and deployed successfully after the continuous dilation by SEMS, and were placed for a median of 14 months, achieving a stable clinical status in 70.7% patients. Subgroup analysis illustrated that efficacity and late complications were similar between the two groups, whereas patients in Group 2 had greater severity of airway stenosis, characterized by smaller pre- and post-intervention luminal diameters. SEMS placement might be conducive to minimizing the bronchoscopic interventional procedures and achieving a high success rate of silicone stenting. In brief, temporary placement of SEMS in severe post-TB stenosis could help to dilate the complex stenotic airway effectively and increase the success rate of silicone stent implantation.
All patients enrolled in this study had severe PTBS. The average luminal diameter was 6 (5–7) mm after mechanical dilation, which was much smaller than that reported in previous studies,8,26 making it difficult to deploy a silicone stent in the stenotic airway. Hence, the post-dilation airway caliber might serve as the key factor to determine the treatment with temporary SEMS implantation as a bridge to silicone stenting. The following indications are recommended to be considered for patients who need a temporary SEMS prior to silicone stent placement: (1) severe post-TB stenosis with luminal diameter less than 25% of the normal caliber; (2) severe airway stenosis with fibrotic stricture and distortion; (3) inaccessibility for the rigid bronchoscope to the stenotic airway and difficulty in deploying a silicone stent even after transbronchial dilation and/or ablation.
Some caveats should be considered in this study. First, this was a single center retrospective study with a small sample size. The optimum duration of SEMS has not been studied in the present study. In our experience, 2–4 weeks of SEMS placement could be safe and effective for patients with severe PTBS. Second, underlying diseases and disease duration have not been recorded and analyzed in this study. Third, how many interventions should be performed before stents implantation and whether the interventional procedures could influence the overall prognosis have not been fully elucidated in this study.
In conclusion, temporary SEMS implantation could serve as a bridge to silicone stenting for patients with severe post-TB airway stenosis, particularly in cases of inaccessibility to advance the rigid bronchoscope or silicone stent even after the bronchoscopic treatments (e.g. thermal ablation, balloon dilation). This novel interventional procedure was found safe and feasible to re-establish the patency of stenotic airway with a temporary SEMS with continuous dilation effect against the fibrotic airway stricture and, eventually, to implant a silicone stent in patients with severe airway stenosis.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666211019564 – Supplemental material for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis
Supplemental material, sj-pdf-1-tar-10.1177_17534666211019564 for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis by Zi-Qing Zhou, Jia-Xin Feng, Yu Chen, Zhu-Quan Su, Chang-Hao Zhong, Xiao-Bo Chen, Chun-Li Tang, Jie-Rong Huang and Shi-Yue Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-2-tar-10.1177_17534666211019564 – Supplemental material for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis
Supplemental material, sj-pdf-2-tar-10.1177_17534666211019564 for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis by Zi-Qing Zhou, Jia-Xin Feng, Yu Chen, Zhu-Quan Su, Chang-Hao Zhong, Xiao-Bo Chen, Chun-Li Tang, Jie-Rong Huang and Shi-Yue Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_17534666211019564 – Supplemental material for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis
Supplemental material, sj-pdf-3-tar-10.1177_17534666211019564 for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis by Zi-Qing Zhou, Jia-Xin Feng, Yu Chen, Zhu-Quan Su, Chang-Hao Zhong, Xiao-Bo Chen, Chun-Li Tang, Jie-Rong Huang and Shi-Yue Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-4-tar-10.1177_17534666211019564 – Supplemental material for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis
Supplemental material, sj-pdf-4-tar-10.1177_17534666211019564 for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis by Zi-Qing Zhou, Jia-Xin Feng, Yu Chen, Zhu-Quan Su, Chang-Hao Zhong, Xiao-Bo Chen, Chun-Li Tang, Jie-Rong Huang and Shi-Yue Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-5-tar-10.1177_17534666211019564 – Supplemental material for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis
Supplemental material, sj-pdf-5-tar-10.1177_17534666211019564 for Self-expanding covered metallic stents as a transition to silicone stent implantation in management of severe post-tuberculosis bronchial stenosis by Zi-Qing Zhou, Jia-Xin Feng, Yu Chen, Zhu-Quan Su, Chang-Hao Zhong, Xiao-Bo Chen, Chun-Li Tang, Jie-Rong Huang and Shi-Yue Li in Therapeutic Advances in Respiratory Disease
Footnotes
Author contributions
ZQZ, JXF, YC and ZQS performed the literature search and drafted the manuscript; ZQZ, JXF, YC, ZQS, XBC, CHZ, JRH and CLT were responsible for subject recruitment; ZQZ, JXF, YC and ZQS contributed to the data collection, data analysis and data interpretation; ZQZ, JXF, YC, ZQS and SYL contributed to study conception; SYL provided critical review of the manuscript and approved the final submission.
Conflict of interest statement
SYL declares that he has received the Clinical Innovation Research Program of Bioland Laboratory (No. 2018GZR0201002) and Collaborative Innovation Research Program of the First Affiliated Hospital of Guangzhou Medical University (No. 201505-gyfyy). ZQS declares that he has received the Foundation for Youth Innovation Talent in Higher Education of Guangdong, China (No. B195001307). All other authors declare no potential conflict of interest. None of the funding sources had any role in the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Clinical Innovation Research Program of Bioland Laboratory (No. 2018GZR0201002). Collaborative Innovation Research Program of the First Affiliated Hospital of Guangzhou Medical University (No. 201505-gyfyy). Foundation for Youth Innovation Talent in Higher Education of Guangdong, China (No. B195001307)
Ethics statement
All the patients involved in this study provided informed consent. The study protocol was approved by the Ethics Committee of the First Affiliated Hospital of Guangdong Medical University (ethics review No.: 2018-16).
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
