Abstract
Objectives:
The IMPACT trial has compared the benefit in the reduction of moderate/severe exacerbations of single inhaler triple therapy (SITT) with fluticasone furoate (FF)/umeclidinium (UMEC)/vilanterol (VI) versus dual therapy with FF/VI (ICS/LABA) and UMEC/VI (LAMA/LABA) in the treatment of patients with chronic obstructive disease (COPD). This study performs a subgroup analysis of the cohort from Spain in the IMPACT study.
Materials and Methods:
In IMPACT, a 52-week randomized, double-blind, parallel-group, multicenter study (N = 10,355), patients ⩾40 years of age with COPD and ⩾1 moderate/severe exacerbations in the previous year were randomized 2:2:1 to once-daily FF/UMEC/VI 100/62.5/25 µg, FF/VI 100/25 µg or UMEC/VI 62.5/25 µg administered via the Ellipta inhaler. Here, we present a subgroup analysis of the 499 patients from Spain, included in the intent-to-treat (ITT) population in the study. Endpoint assessed included exposure-adjusted rate of moderate and severe exacerbations.
Results:
In the Spain cohort, the exposure-adjusted rate of on-treatment moderate/severe COPD exacerbations per year for FF/UMEC/VI was 1.31 versus 1.43 and 1.57 for FF/VI and UMEC/VI, respectively. No new adverse events were identified. The results are consistent with those observed in the overall ITT study population.
Conclusion:
In the Spain cohort of the IMPACT study, patients receiving triple therapy with FF/UMEC/VI had a lower exposure-adjusted rate of exacerbations compared with FF/VI and UMEC/VI, similar to the overall population.
The reviews of this paper are available via the supplemental material section.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is the third-leading cause of death worldwide, with an estimated 3.2 million deaths in 2017. 1 Declining COPD mortality rates are observed in a majority of countries, but the number of COPD deaths has actually been increasing, due to population growth and ageing. 2
According to clinical practice guidelines, the following three general objectives of treatment of COPD can be summarized: reduce chronic symptoms, reduce the frequency and severity of exacerbations and improve the prognosis of the disease. 3 In its 2019 update, the Global Initiative for Chronic Obstructive Pulmonary Disease (GOLD) made therapeutic recommendations based on the treatable trait of the patient, dyspnea, or exacerbations.
The triple therapy comprises an inhaled glucocorticoid (ICS) and two bronchodilators: a long-acting muscarinic antagonist (LAMA) and a long-acting beta-2 agonist (LABA). Triple therapy is recommended by GOLD in COPD patients with clinically significant symptoms and at risk of frequent exacerbations despite being treated with a combination of ICS/LABA or LAMA/LABA. 4
The triple ICS/LAMA/LABA inhaled therapy, administered through different devices, has demonstrated its efficacy in improving lung function, symptoms, health status and the reduction of moderate/severe COPD exacerbations compared with ICS, LABA, or LAMA monotherapy and against LAMA/LABA and ICS/LABA combinations.5,6 In addition, several studies have shown their positive effects on many of the patient’s clinical and functional parameters, and their contribution to a marked improvement in HRQoL. 7 However, up until quite recently, treatment with the triple ICS/LAMA/LABA therapy has required patients to use multiple inhalers several times a day, which can negatively influence the use of the devices and adherence to treatment.8,9
Recently, combinations containing an ICS, a LABA and a LAMA have been developed in a single device; these combinations offer, among others, potential advantages in practicality and adherence to treatment.
The results of the IMPACT study in the evaluation of the efficacy and safety of the triple ICS/LAMA/LABA therapy (formed by fluticasone furoate/umeclidinium/vilanterol (FF/UMEC/VI) compared with dual therapies (ICS/LABA combination formed by FF/VI or the LAMA/LABA combination formed by UMEC/VI), in symptomatic patients with COPD and at least one exacerbation in the previous year, reported a lower modelled rate of moderate or severe exacerbations per year with FF/UMEC/VI (0.91) compared with FF/VI (1.07) or UMEC/VI (1.21) combination in this population. 10 In addition, the annual modelled rate of severe exacerbations that resulted in hospitalization was also lower in the FF/UMEC/VI group, with a figure of 0.13 compared with 0.19 in the UMEC/VI group.
Of patients in the intent-to-treat (ITT) population, 5% were from Spain, which allowed us to obtain results applicable to the specific characteristics of our patients and to Spanish clinical practice. The present study presents the results of the IMPACT study in the population recruited in Spanish centers.
Methods
IMPACT is a phase III, multicenter, randomized, double-blind, parallel-group study whose design and main results have been previously published. 10 The main objective was to compare the effect of triple inhaled therapy FF/UMEC/VI (100 μg/62.5 μg/25 μg), compared with combinations of FF 100 μg/VI 25 μg and UMEC 62.5 μg/VI 25 μg on the rate of moderate and/or severe exacerbations at 52 weeks of treatment.
Worldwide, a total of 10,355 patients with COPD of 40 years or older and relevant symptomatology were recruited (CAT questionnaire score ⩾10), with a forced expiratory volume during the first second (FEV1) less than 50% of the predicted value and history of at least one moderate or severe exacerbation in the previous year, or an FEV1 of 50–80% of the predicted value and at least two moderate exacerbations or a severe exacerbation in the previous year. 10 The general description of the most relevant inclusion and exclusion criteria of the IMPACT study was previously published.10,11 Once enrolled in the study and before randomization, patients continued taking their own medication, which could include a LAMA, LABA, or ICS alone or in combination, over a period of 2 weeks. The study was carried out in 37 countries from June 2014 to July 2017. The determination of the sample size, the randomization as well as the statistical analysis were carried out as previously described in the study protocol. 11
The study lasted a total of 55 weeks, with an initial period of 2 weeks in which the patients maintained their usual treatment, a treatment phase of 52 weeks, and a follow-up period of 1 week. In the treatment phase, patients were randomized in a 2: 2: 1 ratio to one of the three treatment groups in the following order: FF/UMEC/VI (100/62.5/25 μg), FF/VI (100/25 μg), and UMEC/VI (62.5/25 μg), respectively. Each treatment was administered in a single dry powder inhaler (Ellipta, GlaxoSmithKline).
This publication presents the results of the IMPACT study in the 499 patients recruited in Spanish centers. 11 It has not been determined if the differences found between the treatment groups were statistically significant, since formal analyses were not performed due to the small sample size in this population. The objective of the analysis is to know the baseline characteristics of the patients with COPD who participated in the IMPACT study in Spain, the incidence and exposure adjusted rate of moderate/severe exacerbations and the incidence of adverse events (AEs) after 52 weeks of treatment in the three arms of the study.
Results
Demographic characteristics of the study population
A total of 59 Spanish centers participated in the IMPACT study, 55 of which randomized 499 patients who were included in the ITT population. Of the national sample, 194 patients received FF/UMEC/VI, 207 FF/VI, and 98 UMEC/VI. Most patients (79%) were men, the mean age (standard deviation, SD) was 66.9 (8.06) years and the highest percentage of patients (46%) was concentrated in the group of 65–74 years. There were no clinically significant differences between the three arms of treatment with respect to the initial demographic characteristics, the COPD exacerbations in the previous year or lung function (Table 1). The body mass index (BMI) in the Spain cohort population was 28.2 (5.1) kg/m2. Most patients had moderate or severe airflow limitation (defined as post-bronchodilator FEV1 ⩾30% or <80% predicted), with a mean value (SD) of 49.4 (14.6)% predicted. The bronchodilator reversibility test showed a mean (SD) increase in FEV1 of 94.8 (120.5) ml following salbutamol administration, with a mean (SD) percent reversibility of pre-bronchodilator FEV1 of 8.99 (11.2)% (Table 1).
Baseline characteristics of the patients (ITT population).*
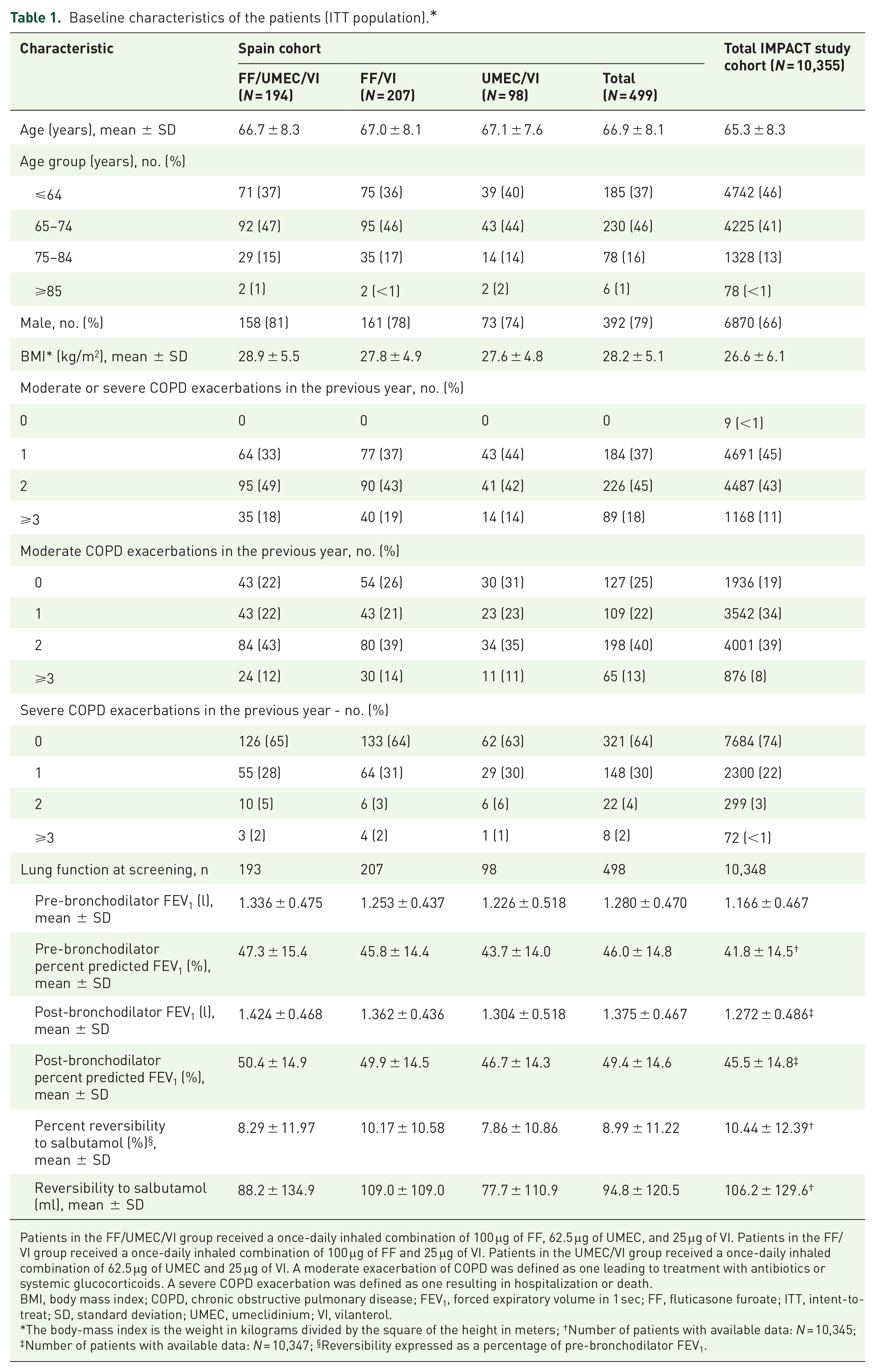
Patients in the FF/UMEC/VI group received a once-daily inhaled combination of 100 μg of FF, 62.5 μg of UMEC, and 25 μg of VI. Patients in the FF/VI group received a once-daily inhaled combination of 100 μg of FF and 25 μg of VI. Patients in the UMEC/VI group received a once-daily inhaled combination of 62.5 μg of UMEC and 25 μg of VI. A moderate exacerbation of COPD was defined as one leading to treatment with antibiotics or systemic glucocorticoids. A severe COPD exacerbation was defined as one resulting in hospitalization or death.
BMI, body mass index; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 sec; FF, fluticasone furoate; ITT, intent-to-treat; SD, standard deviation; UMEC, umeclidinium; VI, vilanterol.
The body-mass index is the weight in kilograms divided by the square of the height in meters; †Number of patients with available data: N = 10,345; ‡Number of patients with available data: N = 10,347; §Reversibility expressed as a percentage of pre-bronchodilator FEV1.
Main outcome: COPD exacerbations
The exposure-adjusted rate of COPD moderate/severe exacerbations during treatment in the FF/UMEC/VI group was 1.31 per patient/year, compared with 1.43 and 1.57 among those assigned to FF/VI and UMEC/VI, respectively (Table 2). These results are consistent with the IMPACT study, where the modelled estimated annual rate of exacerbations for FF/UMEC/VI was also lower, of 0.91, compared with 1.07 in the FF/VI group [rate ratio 0.85; 95% confidence interval (CI), 0.80 to 0.90, percentage reduction 15%, p < 0.001] and 1.21 in the UMEC/VI group (rate ratio 0.75; 95% CI, 0.70 to 0.81, percentage reduction 25%, p < 0.001). The results are consistent when moderate and severe exacerbations have been assessed independently, with rates lower in all cases for FF/UMEC/VI.
Moderate or severe COPD exacerbations (Spain ITT Population of the IMPACT study).
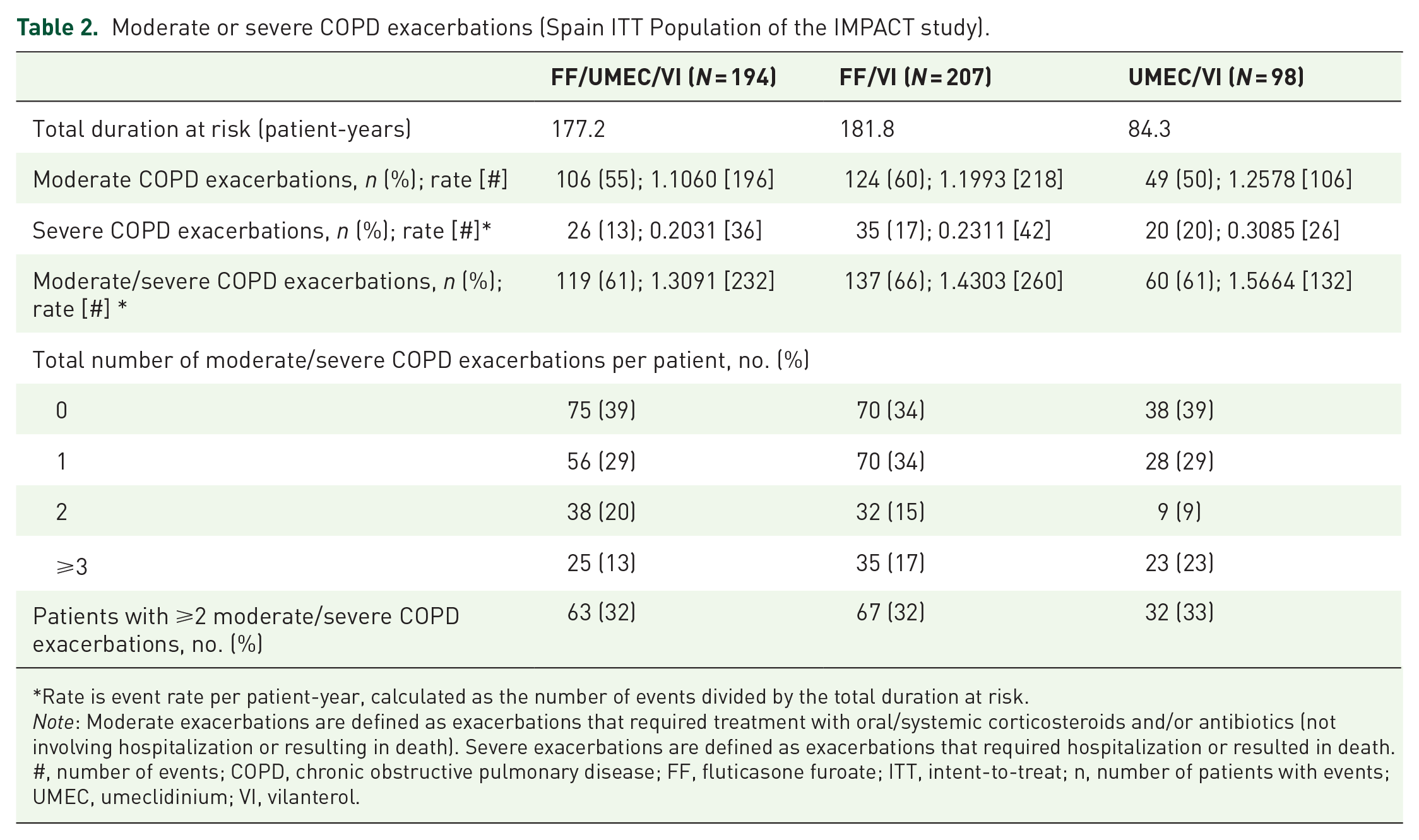
Rate is event rate per patient-year, calculated as the number of events divided by the total duration at risk.
Note: Moderate exacerbations are defined as exacerbations that required treatment with oral/systemic corticosteroids and/or antibiotics (not involving hospitalization or resulting in death). Severe exacerbations are defined as exacerbations that required hospitalization or resulted in death.
#, number of events; COPD, chronic obstructive pulmonary disease; FF, fluticasone furoate; ITT, intent-to-treat; n, number of patients with events; UMEC, umeclidinium; VI, vilanterol.
The exposure-adjusted rates obtained for those patients in Spain were 1.11, 1.20, and 1.26 for moderate exacerbations and 0.20, 0.23, and 0.31 for severe exacerbations, in patients assigned to FF/UMEC/VI, FF/VI, and UMEC/VI, respectively.
The exposure-adjusted rate of moderate/severe exacerbations in the Spain cohort over 52 weeks, although higher, follows the same trend as the global population of the IMPACT study (Figure 1).

Comparison of the annual exposure-adjusted rate of moderate/severe exacerbations per patient at 52 weeks for each treatment arm for Spain cohort versus the global cohort included in the IMPACT study.
Safety and adverse events
In general, the FF/UMEC/VI AEs profile was similar to that of the dual therapy comparators; 69% of the patients assigned to FF/UMEC/VI suffered any AE, compared with 71% for the FF/VI group and 76% for the UMEC/VI, with an exposure-adjusted rate of 2.45 compared with 2.58 events per patient-year with FF/VI and 2.87 events per patient-year with UMEC/VI. The post hoc exposure-adjusted rate of pneumonia adverse event of special interest was low in all three treatment arms (FF/UMEC/VI: 0.10; FF/VI: 0.08; UMEC/VI: 0.07 events per patient-year) The post hoc exposure-adjusted rate of cardiovascular adverse events of special interest was similar in the three treatment arms, 0.12 for FF/UMEC/VI 0.10 for FF/VI and 0.11 for UMEC/VI (Table 3). As in the case of exacerbations previously described, no statistical significance has been determined, since comparisons cannot be made due to such a low number of events.
Adverse events of special interest during the study period for each of the treatment arms in the Spain cohort.

Analysis of AESI was conducted post hoc. Note: Rate is event rate per patient-year, calculated as the number of events divided by the total duration at risk. AESI were AEs which have specified areas of interest for ICS, LAMA, or LABA, or for patients with COPD.
AE, adverse event; AESI, adverse event of special interest; COPD, chronic obstructive pulmonary disease; FF, fluticasone furoate; ITT, intent-to-treat; N, number of patients or events; SD, standard deviation; SMQ, Standardized MedDRA (Medical Dictionary for Regulatory Activities) query; UMEC, umeclidinium; VI, vilanterol.
Discussion
The results of the population recruited in Spanish centers of the IMPACT study are similar to those described in the overall sample of the study where a reduction of moderate/severe COPD exacerbations was observed in the FF/UMEC/VI group compared with both dual therapies (UMEC/VI and FF/VI) despite being a cohort of patients with specific characteristics.
The baseline demographics of patients included in the Spain cohort of the IMPACT study show some similarities and differences with respect to the global sample of the overall ITT population of the IMPACT study. The mean age (67 in the Spain cohort versus 65 years in the global ITT population) and BMI (28.2 versus 26.6 kg/m2) were similar; however, the Spain cohort had a greater proportion of males (79% versus 66%) and a higher post-bronchodilator FEV1 % predicted (49.4% versus 45.5%). Both the Spanish subset cohort and the total IMPACT cohort had important variation regarding baseline reversibility to salbutamol as reflected by the standard deviation (120.5 ml vs 129.6 ml, respectively), the worst 25% of the Spanish cohort achieved less than 30 mL vs the best 25% of them achieved more than 160 mL.
The rate of moderate/severe COPD exacerbations in the treatment phase was higher in Spanish centers than in the overall sample in the IMPACT study. A possible explanation could be the baseline higher rate of severe COPD exacerbations in the previous year, higher age and BMI and lower reversibility to salbutamol observed in the Spanish subset. Consistent with the results obtained in the IMPACT study, the group of patients treated with FF/UMEC/VI presented a lower annual rate of moderate/severe COPD exacerbations, compared with FF/VI and UMEC/VI: 1.31, 1.43 and 1.57 events per patient/year, respectively.
In line with these results, a recently published meta-analysis demonstrated a significant decrease in the risk of moderate/severe COPD exacerbations with triple ICS/LAMA/LABA therapy compared with dual therapy (ICS/LABA, or LAMA/LABA) or LAMA monotherapy, in addition to a reduction in the rate of moderate/severe exacerbations, a reduction in the rate of severe exacerbations, and improvements in lung function and quality of life, at the same time that the safety profile was similar, which would confirm the clinical benefit of FF/UMEC/VI in COPD patients with relevant symptomatology and at least two exacerbations during the previous year. 6
There is some controversy regarding the use of ICS in COPD and the relative benefits of FF/UMEC/VI compared with dual bronchodilation (LAMA plus LABA) in patients with a history of exacerbations. However, in this study a reduction of moderate/severe exacerbations COPD with FF/UMEC/VI compared with UMEC/VI has been observed, strengthening the efficacy of FF/UMEC/VI in patients with previous history of exacerbation. The results limited to the Spain cohort of IMPACT have also shown that FF/VI was better than UMEC/VI with respect to the reduction of the rates of moderate/severe COPD exacerbations (1.43 and 1.56, respectively), as it has also been observed in the global population of the IMPACT study. 10
The prevention of severe exacerbation is perhaps one of the most important aspects of the management of the disease, being largely responsible for the morbidity and mortality of COPD. 12 In 2011, the GOLD initiative recognized that the identification of patients at risk of exacerbations is a key factor in guiding maintenance treatment alternatives. 13 Likewise, the update of GOLD 2019 differentiates for the first time the maintenance treatment of COPD patients based on tractable features, dyspnea, and exacerbations. 14
Regarding safety, and in line with the global population of the IMPACT study, the results in the Spain cohort showed a higher incidence of pneumonia in patients treated with FF/UMEC/VI. However, the pneumonia rate observed in this study was relatively low for the three treatment groups, FF/UMEC/VI, FF/VI, and UMEC/VI. Although there is evidence to support the possibility of an increased risk of pneumonia associated with prolonged use of ICS in patients with COPD, 15 it should be taken into account as an important bias in virtually all studies that pneumonia was not a predetermined AE, so no objective definition of pneumonia or radiological confirmation was required. This fact could lead to an erroneous diagnosis, due mainly to the similarities in the clinical presentation between pneumonia and AE derived from COPD. However, since there is still no evidence of a statistically significant increase in mortality related with pneumonia in patients treated with ICS, the risk-benefit equation remains significantly in favor of the treatment of patients with COPD with ICS. 15 Indeed, the global population of the IMPACT trial showed that on-treatment all-cause mortality was significantly lower in regimens that included ICS (FF/UMEC/VI and FF/VI) than with UMEC/VI, which is additional evidence of the favorable risk–benefit profile. 10 In this analysis of the Spain cohort of IMPACT FF/UMEC/VI also did not increase the risk of cardiovascular AEs, which was consistent with both results in the overall population of the IMPACT study and previous meta-analyses suggesting that dual LAMA/LABA therapy does not increase the risk of fatal cardiovascular events in patients with COPD. 6
Among the strengths of the IMPACT study is the high number of patients evaluated worldwide, which allows for regional subanalyses, and the design of the study like “real life” in the sense that all patients were randomized without a previous washout period, as is done in the daily clinic when therapies are changed in patients with COPD. Obviously, the limitations in the interpretation of these results is that the sample size of the Spain cohort does not allow differentiated conclusions outside the context of the global IMPACT study.
Conclusion
In line with the results obtained in the IMPACT study, in the Spain cohort the combination of FF, UMEC, and VI reduced the rate of moderate/severe COPD exacerbations compared with FF/VI or UMEC/VI. In addition, FF/UMEC/VI safety profile was similar to that of the dual therapy comparators and in line with the global ITT. The results obtained show the benefit of FF/UMEC/VI in COPD patients in Spain with at least one moderate/severe exacerbation in the previous year.
Supplemental Material
Author_Response – Supplemental material for Efficacy of FF/UMEC/VI compared with FF/VI and UMEC/VI in patients with COPD: subgroup analysis of the Spain cohort in IMPACT
Supplemental material, Author_Response for Efficacy of FF/UMEC/VI compared with FF/VI and UMEC/VI in patients with COPD: subgroup analysis of the Spain cohort in IMPACT by José M. Marín, Luis Mateos, Juan Roldán, José M. Echave-Sustaeta, Sergi Pascual-Guardia, Maria V. Pardo, Beatriz Velasco, C. Elaine Jones, Sally Kilbride and David A. Lipson in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_1_v.1 – Supplemental material for Efficacy of FF/UMEC/VI compared with FF/VI and UMEC/VI in patients with COPD: subgroup analysis of the Spain cohort in IMPACT
Supplemental material, Reviewer_1_v.1 for Efficacy of FF/UMEC/VI compared with FF/VI and UMEC/VI in patients with COPD: subgroup analysis of the Spain cohort in IMPACT by José M. Marín, Luis Mateos, Juan Roldán, José M. Echave-Sustaeta, Sergi Pascual-Guardia, Maria V. Pardo, Beatriz Velasco, C. Elaine Jones, Sally Kilbride and David A. Lipson in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v.1 – Supplemental material for Efficacy of FF/UMEC/VI compared with FF/VI and UMEC/VI in patients with COPD: subgroup analysis of the Spain cohort in IMPACT
Supplemental material, Reviewer_2_v.1 for Efficacy of FF/UMEC/VI compared with FF/VI and UMEC/VI in patients with COPD: subgroup analysis of the Spain cohort in IMPACT by José M. Marín, Luis Mateos, Juan Roldán, José M. Echave-Sustaeta, Sergi Pascual-Guardia, Maria V. Pardo, Beatriz Velasco, C. Elaine Jones, Sally Kilbride and David A. Lipson in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors would like to thank all researchers who contributed to the study in Spain for their participation in the study: Ramón Agüero Balbín, Ignacio Arriaga, Ferrán Barbé Illa, Miguel Barrueco Ferrero, Ana Bustamante Ruiz, Jesús Camino Buey, Pere Casán Clará, Luis De Teresa Parreño, Félix Del Campo Matías, José Pablo Díaz Jiménez, Adolfo Domenech del Río, José María Echave-Sustaeta, Juan Luis García Rivero, Daniel Geat Argañaraz, Javier Gómez De Terreros Caro, Elia Gómez Merino, Javier Hueto Pérez de Heredia, Jose Maria Ignacio García, Milagros Iriberri Pascual, Eva López Antolín, Pedro Jorge Marcos Rodríguez, Alicia Marín, José Mª Marín Trigo, Encarnación Martínez Navarro, Pyrene Martínez Piera, Luis Mateos, Asunción Mira Vicedo, Fernando Molina, Eduard Monsó Molas, Josep Morera Prat, Elsa Naval, Francisco Ortega Ruiz, Juan Ortiz de Saracho, Mercedes Pallero Castillo, Sergi Pascual Guardia, Germán Peces-Barba Romero, Mercé Pérez Vera, Pedro Plaza Valía, Francesc Pont Barrio, Ana Pueyo Bastida, Mª Teresa Ramírez Prieto, David Ramos Barbón, Ana Rañó, Juan Roldán Sánchez, Mª Ángeles Ruiz Cobos, Ernest Sala Llinàs, Fernando José Sánchez-Toril López, Salud Santos Pérez, Alberto Saura Vinuesa, Salvador Sitjar Martínez de Sas, Néstor Soler Porcar, Jesús Suárez Martinez, Alexis Tena Domingo, Pere Torán Montserrat, Luis Valdés Cuadrado, Emma Vázquez Espinosa y Sara Vilá Valls.
Author contribution(s)
Data-sharing statement
Declaration of Conflicting Interests
José M. Marín reports personal fees from GlaxoSmithKline (GSK), Chiesi and Astra-Zeneca, non-financial support from Menarini.
Luis Mateos reports personal fees from GSK, Novartis, Astra Zeneca, Boehringer-Ingelheim, Chiesi, Teva, Rovi and Ferrer.
Juan Roldán declared no conflicts of interest.
Sergio Pascual reports personal fees from GSK, Boehringer-Ingelheim and TEVA.
José M. Echave-Sustaeta received fees as a speaker and advisor for GSK and as a researcher in clinical studies sponsored by GSK, Novartis, Astra Zeneca, Boehringer Ingelheim, Sanofi and Bayer.
María V. Pardo, Beatriz Velasco, C. Elaine Jones, Sally Kilbride and David A. Lipson are GSK employees and hold stock/shares in GSK.
Ethics statement
This study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice Guidelines.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by GSK (study number: CTT116855). The funders of the study had a role in study design, data analysis, data interpretation, and writing of the report. The corresponding author had full access to all the data and the final responsibility to submit for publication.
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
