Abstract
Given the high proportion of patients with asthma who remain uncontrolled despite controller treatment, there remains a need for the development of more effective treatment options with a proven safety and tolerability profile. Recently, asthma guidelines have evolved to incorporate new therapies, including long-acting muscarinic antagonists (LAMAs) and biologics. Here we focus on the safety profile of tiotropium, a LAMA, using data from the large-scale UniTinA-asthma® clinical trial program, which investigated the use of tiotropium in over 6000 patients with asthma who remained symptomatic despite receiving inhaled corticosteroids maintenance therapy, with or without other adjunct therapies. The large number of patients included allows robust analysis of safety and tolerability. Overall, a similar incidence of patients reporting any adverse event (AE) was observed in the tiotropium (5 µg and 2.5 µg) and placebo groups. Asthma worsening, decreased peak expiratory flow, and upper respiratory tract infections were the most frequently reported AEs. Serious AEs (SAEs) and investigator-defined drug-related AEs were infrequently reported across all treatment groups, including the placebo group, and there were no deaths in any study. Reports of side effects typically associated with anticholinergic drugs, such as dry mouth and urinary retention, were either infrequent or not reported in children, adolescents or adults. The similar proportions of tiotropium-
Introduction
Asthma is a chronic disease of the airways, estimated to affect approximately 235 million children and adults worldwide. 1 In fact, asthma is the most common chronic disease observed in children, affecting approximately 10% of children in the United Kingdom (UK) and the United States of America (USA).2,3
The long-term goals of asthma management include achieving good symptom control, minimizing the risk of asthma exacerbations, reducing hospitalizations, use of rescue medication, airflow limitation and side effects, as well as allowing normal activity levels. 4 Yet achieving these goals remains a major challenge to healthcare systems. Indeed, about 45% of adults and up to 50% of adolescents and younger children have inadequately controlled disease.2,5 The large proportion of patients who have difficulty in gaining and maintaining disease control represents a group at urgent need of effective and acceptable treatment options that have a proven safety and tolerability profile.
In line with Global Initiative for Asthma (GINA) guidelines, asthma management is based on a backbone of inhaled corticosteroid (ICS) therapy, supplemented with add-on therapies for those with unsatisfactory or deteriorating disease control. 4 More recently, international treatment guidelines for asthma have evolved to include new adjunct therapies such as long-acting muscarinic antagonists (LAMAs), including tiotropium bromide and biologics. Tiotropium Respimat® is the only LAMA available for use in asthma; it has been approved for use in many countries, including for adults in Australia, New Zealand, Canada and Singapore (aged 18 years and over), in Japan (aged 15 years and over) and in patients aged 6 years and over in Europe and the USA. While the safety considerations regarding ICSs and long-acting β2-agonists (LABAs) have been widely discussed, the evidence base for newer treatment options is less well known in asthma. However, the safety and tolerability profile of tiotropium bromide has been established in patients with chronic obstructive pulmonary disease (COPD), with over 10 years of experience. 6
The objective of this narrative review was to ascertain the safety and tolerability of tiotropium as an add-on therapy to ICSs, with or without additional controllers, in adults, adolescents and children with symptomatic asthma. Specifically, the review assesses the incidence of patients reporting adverse events (AEs), including serious AEs (SAEs) and treatment-related AEs, in both the general population and more specific ones, such as African-American, Japanese and elderly patients, who may have increased risk of experiencing AEs. The pooling of data from our research program, providing a large number of patients, allows robust ascertainment of the occurrence and type of any occurring AEs, including potentially rare ones.
Methodology
We review here the safety and tolerability outcomes from the large-scale UniTinA-asthma® clinical trial program with tiotropium Respimat®. Included in this analysis are 11 phase III and phase II/III, randomized, double-blind, placebo-controlled studies in adults, adolescents and children with asthma who remained symptomatic despite receiving ICS maintenance therapy, with or without adjunct therapies. In all studies, tiotropium 5 µg or 2.5 µg was delivered as two puffs once daily
Summary of clinical trials of tiotropium in patients with asthma.

Treated population. Tiotropium 5 µg or 2.5 µg was delivered as two puffs once daily
Safety was a primary objective, assessed by the proportion of patients reporting AEs or serious AEs, in addition to documented changes in vital signs, changes in physical examination reported as AEs and vital status information, as described in methodology section.
ACQ-7, seven-question asthma control questionnaire; AE, adverse event; FEV1, forced expiratory volume in 1 s; FEV1(0–3 h), FEV1 within 3 h post-dose; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; NCT, ClinicalTrials.gov identifier; Tio, tiotropium.
Summary of selected inclusion criteria in clinical trials of tiotropium in adults, adolescents and children with asthma.

FEV1 reversibility ⩾12% and ⩾200 mL 15–30 min after 400 μg salbutamol (albuterol).
ACQ-IA, interviewer-administered asthma control questionnaire; ACQ-7, seven-question asthma control questionnaire; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; GINA, Global Initiative for Asthma; ICS, inhaled corticosteroid; LABA, long-acting β2 agonist; LTRA, leukotriene receptor antagonist; N/A, not applicable; NCT, ClinicalTrials.gov identifier.
Recording and identification of AEs
In each trial, patients were instructed to report any AEs to the investigator or other site personnel and investigators, who systematically documented their observations during medical visits. The evaluation of safety and tolerability was thus based on the proportion of patients reporting AEs, as well as any documented changes in vital signs (pulse rate, systolic, and diastolic blood pressure), physical examination or vital status. Vital status information was to be collected on withdrawn patients who had taken at least one dose of trial medication.
For the safety analysis, all AEs (including SAEs) that occurred during the randomized study period between the initial drug intake until 30 days after the last dose of trial medication were collected and documented by the investigator. AEs were coded using the
An AE was defined as any untoward medical occurrence, including an exacerbation of a pre-existing condition, in a patient who received one of the trial medications; the event did not necessarily have a causal relationship with the trial medication. A drug-related AE was defined as an AE for which there was a reasonable causal relationship between the randomized trial medications (tiotropium or placebo) and the AE. The medical judgement of the investigator was used to determine the causal relationship after considering all relevant factors (such as the temporal relationship between treatment administration and the AE) and confounding factors (such as concomitant medications and diseases). An SAE was defined as any AE which met the following criteria: resulted in death or was immediately life-threatening; resulted in persistent or significant disability or incapacity; required or prolonged patient hospitalization; or was a congenital anomaly or birth defect. Any other important medical event which may have jeopardized the patient, or which may have required medical or surgical intervention to prevent one of the outcomes listed above (based on appropriate medical judgement), was also regarded as an SAE.
Results
Overview of efficacy of tiotropium add-on therapy in adults, adolescents and children
A total of six phase III studies of between 12–52 weeks’ duration were conducted to assess the efficacy and safety of once-daily tiotropium 5 μg or 2.5 μg add-on therapy to low-to-high-dose ICSs, with or without additional controllers,
Overall, four phase III studies and one phase II/III study were conducted to assess the efficacy and safety of once-daily tiotropium Respimat® 5 µg and 2.5 µg
Safety and tolerability of tiotropium in adults, adolescents and children with asthma
To date, the safety and tolerability of tiotropium has been investigated in over 6000 patients with asthma, including adults,7–10,16 adolescents11,12 and children.13–15 Overall, findings demonstrate a similar proportion of patients reporting any AEs between tiotropium (5 µg and 2.5 µg) and placebo treatment groups (Table 3). Similarly, the reporting of SAEs and investigator-defined drug-related AEs was low and comparable across treatment groups, and no deaths were reported in any trial.
Summary of proportion of patients reporting AEs in clinical trials of tiotropium in adults, adolescents and children with asthma.

Data are presented as
Treated set. Tiotropium 5 µg or 2.5 µg was delivered as two puffs once daily
SAE was defined as any AE that resulted in death, was immediately life-threatening, resulted in persistent or significant disability/incapacity, required or prolonged patient hospitalization, was a congenital anomaly/birth defect, or was to be deemed serious for any other reason that might have jeopardized the patient, and might have required medical or surgical intervention to prevent one of the other outcomes listed in the above definitions.
AE, adverse event; NCT, ClinicalTrials.gov identifier; SAE, serious adverse event; Tio, tiotropium.
Adults with symptomatic mild, moderate or severe asthma
Overall, the number of patients reporting AEs was comparable between treatment groups within each study7–9 (Table 3). The reported incidence of patients with investigator-defined treatment-related AEs was low and balanced across treatment groups in patients with mild-to-moderate (tiotropium 5 µg, 1.3%; tiotropium 2.5 µg, 1.3%; placebo, 1.3%), 9 moderate (tiotropium 5 µg, 7%; tiotropium 2.5 µg, 7%; placebo, 5%) 8 and severe (tiotropium 5 µg, 5.7%; placebo, 4.6%) 7 asthma. A small and comparable number of patients reporting AEs led to treatment discontinuation in each of the treatment groups (Table 3).
The incidence of adult patients reporting SAEs was generally low and largely comparable between treatment groups in each study; these SAEs are described in detail below.7–9
In the GraziaTinA-asthma® trial, two patients reported an SAE: one with a life-threatening asthma exacerbation in the placebo group and one with breast cancer in the tiotropium-treated group. Neither were considered to be related to treatment. 9 In the MezzoTinA-asthma® trials, three patients had life-threatening SAEs: one myocardial infarction, one organophosphorus pesticide poisoning, and one anaphylactic reaction that occurred in tiotropium-treated patients, none of which were regarded as drug-related by the principal investigator. 8 In the two PrimoTinA-asthma ® trials, involving 912 patients, SAEs were reported in 77 patients: 37 (8.1%) in the tiotropium group and 40 (8.8%) in the placebo group. The majority of SAEs were attributed to asthma (17 patients in the tiotropium group and 21 in the placebo group). SAEs considered to be life-threatening occurred in three patients, all in the tiotropium group; these included an asthma exacerbation in two patients and cerebral infarction in one patient, 7 none of which were fatal. In the 52-week CadenTinA-asthma ® trial involving 285 patients, SAEs were reported by four patients (3.5%) receiving tiotropium 5 μg and four (3.5%) receiving 2.5 μg, compared with nine (15.5%) in the placebo group. None were fatal, and only one, in the placebo group, was considered to be related to treatment. 10
Adolescents and children with symptomatic moderate or severe asthma
In adolescents and children with symptomatic moderate asthma (RubaTinA-asthma® and CanoTinA-asthma ® ), the overall proportion of patients reporting AEs was comparable between tiotropium and placebo groups, with a low number of patients reporting investigator-defined treatment-related AEs in both studies12,14 (Table 3).
In RubaTinA-asthma®, SAEs were reported in seven adolescents: three patients receiving tiotropium 5 µg (asthma exacerbation, upper abdominal pain and anaphylactic reaction following a plant sting), two patients receiving tiotropium 2.5 µg (appendicitis and multiple injuries following an accident) and two patients receiving placebo (gastroenteritis and teratoma), none of which were considered to be related to treatment. 12 The proportion of children with symptomatic moderate asthma in CanoTinA-asthma® reporting SAEs was low, with events being reported in one patient receiving tiotropium 5 µg (appendicitis and paralytic ileus), three patients receiving tiotropium 2.5 µg (two patients with asthma worsening and one with appendicitis) and six patients receiving placebo (two reports of asthma worsening, one of paranasal sinus hematoma, one of gastroenteritis, one of renal abscess, one of anaphylactic shock, and one of concussion, fall and skull fracture). 14
In adolescents and children with symptomatic severe asthma (PensieTinA-asthma® and VivaTinA-asthma®), the overall proportion of patients reporting AEs was comparable between the tiotropium and placebo groups, with a low incidence of investigator-defined treatment-related AEs being reported (Table 3); most AEs were mild or moderate in intensity.11,13
In 12- to 17-year-old children with symptomatic severe asthma (PensieTinA-asthma®), one patient in the placebo group experienced a treatment-related AE (palpitations) that led to discontinuation of treatment. Overall, three patients reported SAEs: two patients receiving tiotropium 5 µg (ligament sprain and asthma) and one patient receiving tiotropium 2.5 µg (atopic dermatitis and pyoderma); however, these were not considered to be related to tiotropium treatment. 11
In children with symptomatic severe asthma (VivaTinA-asthma®), investigator-defined drug-related AEs were reported for three participants: one patient receiving tiotropium 5 μg experienced dizziness; one patient receiving placebo experienced cough; and another patient also receiving placebo experienced asthma, cough, decreased appetite, fatigue and metabolic cardiomyopathy. AEs leading to discontinuation were reported for four patients: two receiving tiotropium 5 μg (asthma in two patients) and two receiving placebo (cough and metabolic cardiomyopathy). A total of eight patients reported SAEs: asthma in three patients and appendicitis in one patient (tiotropium 5 μg); asthma and epilepsy (each occurring in one patient receiving tiotropium 2.5 μg); and asthma and asthmatic crisis in one patient each (placebo), none of which were considered to be related to the study drug. 13
Younger children with symptomatic asthma (aged 1–5 years)
In the single trial involving 101 patients aged 1–5 years with persistent asthmatic symptoms, the proportion of children reporting any AE was lower in the tiotropium groups than in the placebo group (55.6%, 58.1% and 73.5% in the tiotropium 5 μg, 2.5 μg and placebo groups, respectively; Table 3); the main AE reported in all groups was asthma-related symptoms. No AEs leading to discontinuation of trial treatment were reported. SAEs were reported for three children: all were in the placebo group and required hospitalization. 15
Overview of most frequently occurring AEs
The most frequently reported AEs (⩾5%) in placebo-controlled trials with tiotropium of 12–52 weeks’ duration in adults, adolescents and children with symptomatic asthma were asthma worsening, decreased peak expiratory flow (PEF) rate (not measured in children 1–5 years) and nasopharyngitis (Table 4).
Pooled analysis of the most frequently occurring AEs reported.
(a). Adults with symptomatic mild, moderate and severe asthma (AEs reported in ⩾2% of patients in any treatment group, by preferred term * ) 17 .

Treated set. Adapted from Dahl and Kaplan (2016). 17
Pooled data from seven trials: NCT00350207 (phase II,16-week trial of tiotropium 5 µg and twice-daily salmeterol 50 µg in patients receiving ICSs), 18 PrimoTinA-asthma®, MezzoTinA-asthma®, GraziaTinA-asthma® and CadenTinA-asthma®.
Placebo Respimat® 5 µg pool: all seven trials; placebo Respimat® 2.5 mg pool: MezzoTinA-asthma®, GraziaTinA-asthma® and CadenTinA-asthma®.
Asthma worsening or exacerbation.
AE, adverse event; NCT, ClinicalTrials.gov identifier.
(b). Adolescents and children, symptomatic moderate and severe asthma (AEs reported in ⩾2% patients in any treatment group, by preferred term * ) 19 .
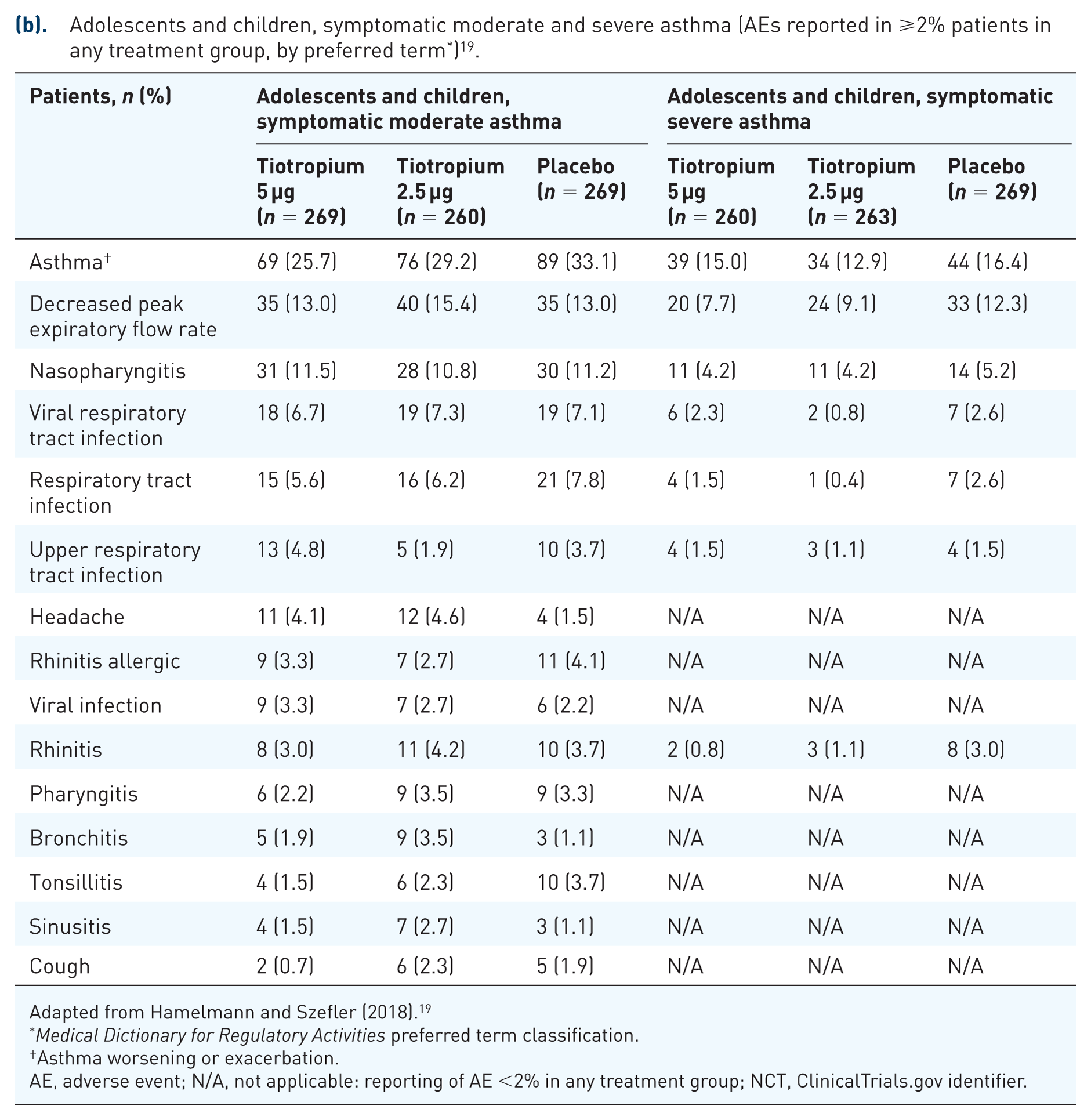
Adapted from Hamelmann and Szefler (2018). 19
Asthma worsening or exacerbation.
AE, adverse event; N/A, not applicable: reporting of AE <2% in any treatment group; NCT, ClinicalTrials.gov identifier.
(c). Children 1–5 years with persistent asthmatic symptoms (AEs reported in ⩾5% of patients in any treatment group by preferred term * ) 15 .
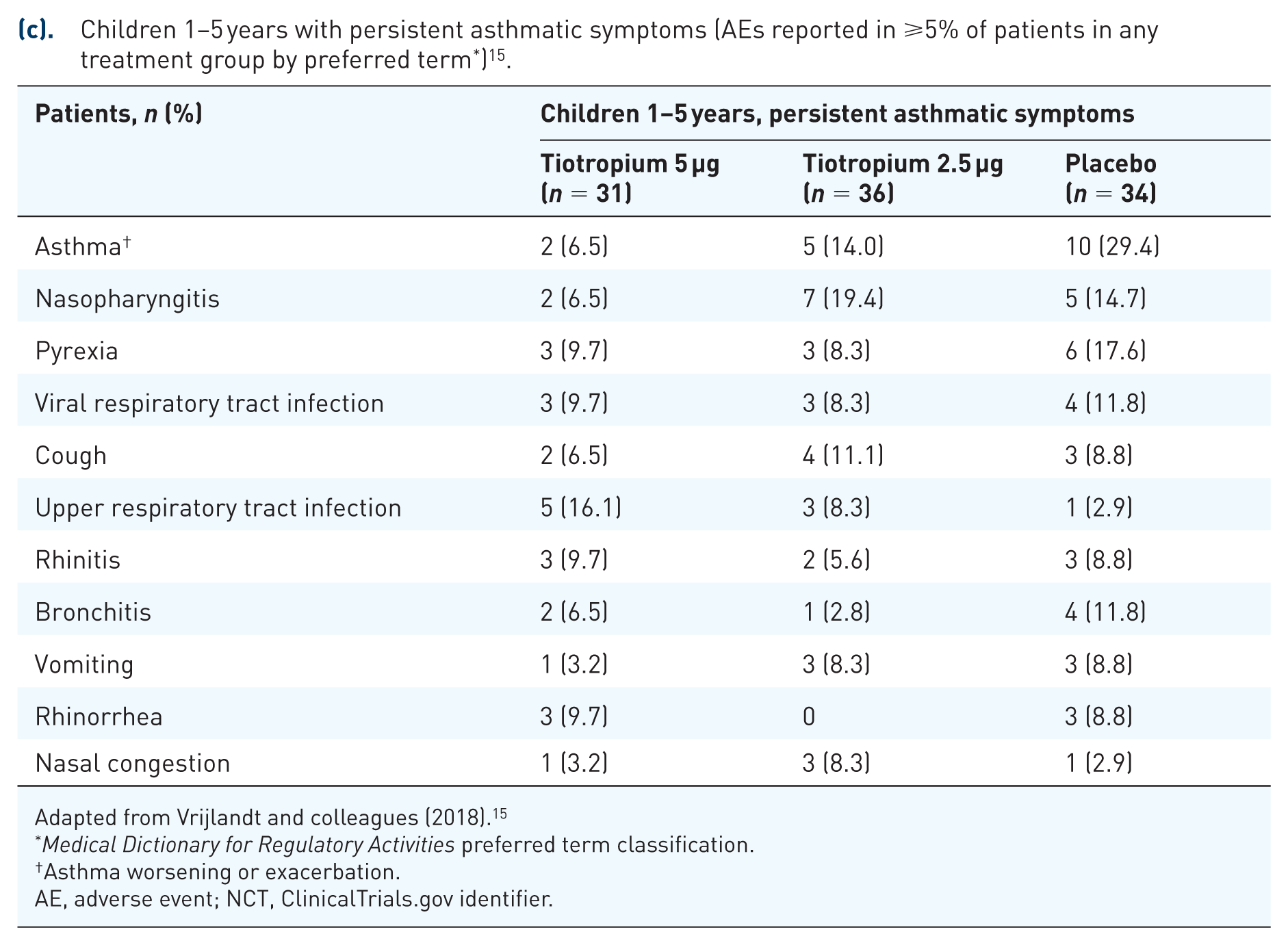
Adapted from Vrijlandt and colleagues (2018). 15
Asthma worsening or exacerbation.
AE, adverse event; NCT, ClinicalTrials.gov identifier.
In children aged 1–5 years with persistent asthmatic symptoms, the most common AEs (occurring in ⩾5% of patients in any group) were asthma worsening, nasopharyngitis, viral respiratory tract infections, upper respiratory tract infections, cough, rhinitis, bronchitis, pyrexia, vomiting, rhinorrhea and nasal congestion. There were no deaths in this study. 15
AEs of special interest with LAMAs in asthma management
Reported class effects of anticholinergics have been highlighted in previous reviews of anticholinergic drugs; these include cardiac events such as tachycardia, dry mouth and gastrointestinal complications such as constipation, as well as urinary retention and urinary tract infections.20–23 These were specifically examined in this review.
Adults with asthma
A pooled safety analysis of 3474 adults with symptomatic asthma from seven phase II and III clinical trials has previously summarized the AEs of special interest in adults, with dry mouth reported by 1% of patients in the tiotropium 5 µg group and 0.5% of patients in the placebo group.
24
Findings were similar for the tiotropium 2.5 µg pooled group
Adolescents and children with asthma
There was no specific reporting of AEs of special interest in adolescents or children with symptomatic moderate or severe asthma.11–14 Moreover, in CanoTinA-asthma®, there were no reports of dry mouth, an AE commonly associated with anticholinergic drugs.
In the NinoTinA-asthma® study, which involved preschool children with persistent asthmatic symptoms, one child reported cough and one reported dry mouth in each of the tiotropium 5 µg and placebo groups. 15 However, the relatively small sample size precludes firm conclusions on safety either for specific age groups, such as preschool children, or rare side effects.
Safety of tiotropium in specific population groups
Studies with tiotropium for the management of asthma have mainly been conducted in the white population; however, the large database collated across the UniTinA-asthma® program does provide limited additional evidence in specific groups, including African-American and Japanese patients, and the elderly. This is of interest because ethnic differences may drive different tolerabilities, and older patients have a potential for an increasing number of comorbidities that can also affect safety responses.
African-American patients with asthma
Graham and colleagues
25
recently conducted a secondary analysis of the safety and tolerability data from tiotropium Respimat® studies, comparing AE reporting in the population of African-American patients aged 1–75 years with symptomatic mild, moderate or severe asthma treated with tiotropium
Japanese adults with asthma
CadenTinA-asthma® was a 52-week study in 285 Japanese patients with symptomatic moderate or severe asthma. 10 The primary objective of this study was to assess the long-term safety of tiotropium Respimat® 5 μg and 2.5 μg. The incidence of AEs, drug-related AEs and AEs leading to discontinuation was comparable between active treatments and placebo (Table 3). The most commonly reported AEs were nasopharyngitis, decreased PEF rate, asthma worsening, bronchitis, pharyngitis and gastroenteritis. Bronchitis was reported by 9.6%, 13.2% and 7.0% in the tiotropium 5 μg, 2.5 μg and placebo groups, respectively, and asthma worsening by 28.9%, 29.8% and 38.6% in the tiotropium 5 μg, 2.5 μg and placebo groups, respectively. Overall, the safety and tolerability profiles of tiotropium were comparable with that of placebo in patients from Japan, and similar to findings from studies conducted in other regions involving patients of largely white ethnicity as part of the tiotropium clinical development program 10 (Table 3).
Elderly patients
In a subgroup analysis of the pooled studies PrimoTinA-asthma® and MezzoTinA-asthma® in patients aged <65 and ⩾65 years with symptomatic severe or moderate asthma (from PrimoTinA-asthma® and MezzoTinA-asthma®), safety and tolerability of once-daily tiotropium Respimat® were comparable to those of placebo and similar between subgroups.
26
A pooled safety analysis involving seven phase II and III clinical trials investigating tiotropium Respimat® compared with placebo investigated the incidence of AEs in patients with asthma who were <65 and ⩾65 years old.
24
In individuals aged <65 years, the frequencies of patients with AEs (tiotropium 5 µg, 60.1%
Pregnancy and breastfeeding
As pregnant or nursing women and female patients of child-bearing potential who did not use a highly effective method of birth control were excluded from the clinical trials described, there are a very limited amount of data from the use of tiotropium in pregnant women. The SmPC states that while animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity at clinically relevant doses, as a precautionary measure, it is preferable to avoid the use of tiotropium during pregnancy. Similarly, it is unknown whether tiotropium is excreted in human breast milk. Despite studies in rodents that have demonstrated that excretion of tiotropium bromide in breast milk occurs only in small amounts, use of tiotropium is not recommended during breastfeeding.
Discussion
This review of the robust safety and tolerability of tiotropium focuses on data from the large UniTinA-asthma® clinical trial program in patients with asthma who remained symptomatic despite receiving ICS maintenance therapy, with or without other therapies. Overall, a similarly low incidence of patients reporting any AE was observed in tiotropium (5 µg and 2.5 µg) and placebo groups. Asthma worsening, decreased PEF, nasopharyngitis and upper respiratory tract infections are generally the most frequently reported AEs. The reporting of AEs associated with class effects of anticholinergic drugs was low and similar across age groups. SAEs and investigator-defined drug-related AEs were infrequently reported across treatment groups, and there were no deaths in any study. Overall, the reporting of AEs and SAEs was low across age groups, severity of disease or ethnic groups. Some years ago, a meta-analysis concluded that anticholinergics were associated with increased cardiovascular risk in COPD patients. 22 Since then, studies such as UPLIFT have provided contrary evidence showing that the relative risks for serious cardiovascular events, heart failure, and myocardial infarction were all significantly lower with tiotropium than placebo.27,28 The studies described here, in asthma patients, are of shorter duration than UPLIFT, but nevertheless provide no indication of any increased cardiovascular risk with tiotropium.
As for other comparative drugs, only one study in the UniTinA-asthma® program included a comparison of tiotropium with a LABA: the replicate MezzoTinA-asthma® study of the efficacy and safety of tiotropium in adult patients with symptomatic moderate asthma included salmeterol as an active comparator
The findings of the current narrative review are in line with previous systematic analyses of the safety of tiotropium in pooled data from the adult studies 24 and in children aged 6–11 years with symptomatic moderate and severe asthma. 29 These analyses concluded that tiotropium was well tolerated across all doses, with no life-threatening or fatal AEs reported.
Given the increasing risk of comorbidities with ageing, it is generally accepted that treatment regimens should be as simple as possible for elderly asthmatic patients, so as to help avoid potential contraindications with different treatments. Evidently, there is already a depth of experience with tiotropium in older patients with COPD from trials and real-world use. 6 A pooled analysis of nearly 13,000 patients with COPD treated with tiotropium demonstrated a good safety profile: 62.6% and 65.5% of patients treated with tiotropium and placebo, respectively, reported at least one AE during the trial period and 21.7% of tiotropium- and 22.8% of placebo-treated patients reported SAEs. The risk of an AE was significantly reduced with tiotropium [rate ratio (RR) (95% CI): 0.90 (0.87, 0.93)], as was the risk of an SAE [RR (95% CI): 0.94 (0.89, 0.99)]. 30 These data, combined with the more limited experience in the asthma trials described here, provides reassurance for its use in older patients with asthma.
An interesting observation from this current review is that, despite similar dosing with tiotropium (5 µg and 2.5 µg) in trials performed in adults, adolescents or children, the overall proportion of reported AEs and SAEs appeared lower in the younger population (aged 1–17 years), for both the intervention and control groups, than in adults with moderate or severe asthma (Table 3). Similarly, the apparent lower numbers of reported SAEs observed in adults with asthma in this review compared with that previously reported in COPD studies6,30 may perhaps be attributed in part to the older median age of patients in COPD compared with that of asthma trials. Alternatively, the observed differences in AE reporting may be a consequence of the studies involving more patients with a longer treatment duration and more comorbidities and co-interventions in the adult groups compared with the pediatric groups.
Conclusion
In asthma, tiotropium is an effective treatment option when added to ICSs, with or without additional controllers, in adults, adolescents and children across a range of asthma severities. Tiotropium displays an excellent safety profile, with comparable proportions of patients reporting AEs and SAEs to those treated with placebo. Side effects typical of the anticholinergic therapeutic class, such as dry mouth and urinary retention, were low or absent in all age groups. Tiotropium therefore provides an additional well-tolerated and effective therapeutic option for the treatment of asthma in patients whose disease remains poorly controlled despite ICSs plus adjunct therapy, or as an alternative therapeutic option in patients where LABAs or other add-on options are unsuitable or ineffective. Moreover, the evidence supports tiotropium Respimat® as a well-tolerated add-on option for the management of symptomatic asthma, irrespective of age and severity and across ethnic groups.
Footnotes
Acknowledgements
The authors take full responsibility for the scope, direction, content of, and editorial decisions relating to the manuscript, were involved at all stages of development, and have approved the submitted manuscript. Medical writing assistance, in the form of the preparation and revision of the draft manuscript, was supported financially by Boehringer Ingelheim and provided by Louise Brady, PhD, of MediTech Media under the authors’ conceptual direction and based on feedback from the authors. Boehringer Ingelheim was given the opportunity to review the manuscript for factual accuracy only.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
DD reports receiving consulting fees, lecture fees and payment for the development of educational activities from Boehringer Ingelheim, Chiesi, Dey Pharma, Novartis, Nycomed and Pfizer.
FMD reports receiving unrestricted research funds from AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Merck Canada, Novartis and Trudell Medical; honorarium for consultancy work from Boehringer Ingelheim and Sanofi; and honorarium as speaker from Boehringer Ingelheim and AstraZeneca (China).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
