Abstract
Background:
The TORRACTO® study evaluated the effects of tiotropium/olodaterol versus placebo on endurance time during constant work-rate cycling and constant speed shuttle walking in patients with chronic obstructive pulmonary disease (COPD) after 12 weeks of treatment.
Methods:
The effects of once-daily tiotropium/olodaterol (2.5/5 and 5/5 μg) on endurance time during constant work-rate cycle ergometry (CWRCE) after 6 and 12 weeks of treatment were compared with placebo in patients with COPD in a randomized, double-blind, placebo-controlled, parallel-group clinical trial. Endurance time during the endurance shuttle walk test (ESWT) after 6 and 12 weeks of treatment was also evaluated in a subset of patients.
Results:
A total of 404 patients received treatment, with 165 participating in the ESWT substudy. A statistically significant improvement in endurance time during CWRCE was observed after 12 weeks (primary endpoint) with tiotropium/olodaterol 5/5 µg [14% (p = 0.02)] but not with tiotropium/olodaterol 2.5/5 µg [9% (p = 0.14)] versus placebo. In the ESWT substudy, a trend to improvement in endurance time during ESWT after 12 weeks (key secondary endpoint) was observed with tiotropium/olodaterol 5/5 µg [21% (p = 0.055)] and tiotropium/olodaterol 2.5/5 µg [21% (p = 0.056)] versus placebo.
Conclusion:
Tiotropium/olodaterol 5/5 µg improved endurance time during cycle ergometry versus placebo, with a strong tendency to also improve walking endurance time.
[ClinicalTrials.gov identifier: NCT01525615.]
Introduction
Activity-related dyspnea and exercise intolerance are important features of chronic obstructive pulmonary disease (COPD) that negatively impact on ability to perform activities of daily living. As such, evaluating exercise tolerance is now considered to be an essential component of disease assessment. 1 Of available exercise-testing protocols, constant work-rate exercise tests, such as cycle and treadmill endurance tests and the endurance shuttle walk test (ESWT), are the most sensitive for detecting change in exercise capacity following intervention (pharmacologic and nonpharmacologic). 2
Over the past several years, constant work-rate cycle ergometry (CWRCE) has been used extensively in clinical trials evaluating the efficacy of long-acting bronchodilators in patients with COPD,2,3 and is often included in clinical development programs as a complementary assessment of efficacy, alongside traditional assessments of lung function. There is, however, a debate regarding the most appropriate endurance exercise testing protocol.2,4–7 In this regard, there has been recent interest in the ESWT,7–11 in some part due to a view that field walking tests are more reflective of activities performed by patients in everyday life. 12
The TORRACTO® study was part of a clinical program evaluating the efficacy and safety of tiotropium/olodaterol in patients with COPD. TORRACTO® was designed to evaluate two doses of tiotropium/olodaterol (2.5/5 and 5/5 µg) compared with placebo on exercise endurance time (EET) during CWRCE in patients with COPD in a parallel-group design after 12 weeks of treatment. Secondary objectives were to evaluate EET during the ESWT after 6 and 12 weeks of treatment with tiotropium/olodaterol in a subset of patients.
Methods
TORRACTO® [ClinicalTrials.gov identifier: NCT01525615] was a multicenter, multinational, randomized, double-blind, placebo-controlled, parallel-group trial to evaluate the effects of once-daily tiotropium/olodaterol (2.5/5 and 5/5 μg) compared with placebo on exercise tolerance after 12 weeks of treatment in patients with COPD (5/5 μg is the approved tiotropium/olodaterol dose). The primary evaluation of exercise tolerance was based on EET during CWRCE, while evaluation of EET during ESWT was conducted in a subset of patients (ESWT subgroup).
The study was conducted at 58 centers in 10 countries. The ESWT substudy was conducted at a subset of 26 sites with experience in the ESWT; all patients at the selected sites were asked to participate in the substudy.
Patients
Patients aged 40–75 years with a clinical diagnosis of COPD and stable airway obstruction were included if they had postbronchodilator forced expiratory volume in 1 s (FEV1)/forced vital capacity (FVC) <70% and postbronchodilator FEV1 <80% and ⩾30% predicted normal. Patients were current or exsmokers with a smoking history of >10 pack-years. Exclusion criteria are detailed in the online supplement.
Study design
During screening, patients performed spirometry to determine study eligibility. Eligible patients were randomized to receive one of three treatments: tiotropium/olodaterol 2.5/5 μg, tiotropium/olodaterol 5/5 μg, or placebo, all delivered once daily via the Respimat® (Boehringer Ingelheim International GmbH, Ingelheim am Rhein, Germany) inhaler (Figure 1).

Study design.
Throughout the 12-week treatment period, trial medication was self-administered by the patient once daily between 7 a.m. and 10 a.m.; on test days, administration of trial medication was performed under research staff supervision. Patients continued with inhaled corticosteroids if taken at baseline. Open-label salbutamol (albuterol) was provided as rescue medication throughout the study.
The study was conducted in accordance with the Declaration of Helsinki and the International Conference on Harmonization Tripartite Guideline for Good Clinical Practice. Before the study started, the protocol was reviewed and approved by the Institutional Review Boards, and all patients provided written, informed consent.
Pulmonary function testing
Spirometry (FEV1, FVC) was performed using standard methodology. 13 FEV1 and FVC measured immediately before the first dose of trial medication at the randomization visit was a priori defined as the pretreatment baseline. Spirometry was also performed 1 h postdose at the randomization visit, and predose and 1 h postdose after 6 and 12 weeks of treatment.
Incremental cycle ergometry and constant work-rate cycle ergometry
The specific details of both incremental cycle ergometry and CWRCE have been described previously.14–16 During run-in, patients performed incremental cycle ergometry to symptom limitation to determine peak work rate (Wpeak), the highest work rate maintained for ⩾30 s. Two CWRCE tests to symptom limitation at 75% Wpeak were performed during run-in: the first test familiarized the patient with the exercise protocol, while the second test ⩾4 days later was a priori defined as the pretreatment baseline. To avoid excessively long endurance times that may be limited by motivation rather than physiology/symptoms, patients with an endurance time of >25 min in training or baseline CWRCE were not eligible for randomization. CWRCE at weeks 6 and 12 was performed 2 h postdose. Additional information is provided in the online supplement.
Incremental and endurance shuttle walking (endurance shuttle walk test substudy)
Patients included in the ESWT substudy performed an incremental shuttle walk test (ISWT) 2–4 days after the initial screening visit. 17 At 2–4 days after the baseline and training CWRCE tests, training and baseline ESWTs were performed at a walking speed corresponding to 85% of the peak oxygen consumption estimated from the ISWT. 8 The ESWT was repeated at the same walking speed after 6 and 12 weeks of treatment, 2 h postdose, again with a 2–4-day separation between the CWRCE and ESWT. Both the ISWT and ESWT were performed in an enclosed corridor on a flat 10-meter-long course. Patients who completed the highest level in the ISWT were not eligible for the substudy because a walking speed for the ESWT could not be established for these patients. Patients with an endurance time during the training or baseline ESWT of >15 min were excluded from the ESWT substudy due to the limited (20-min) duration of the audio used for pacing the ESWT. These patients did continue in the main part of the study (CWRCE). Further details are provided in the online supplement.
Breathing discomfort, leg discomfort, and inspiratory capacity
During CWRCE and ESWT, patients rated intensity of breathing discomfort and leg discomfort using the modified Borg scale 18 at rest, during each 2-min interval during exercise, and at the end of exercise. During CWRCE (but not ESWT) patients also performed an inspiratory capacity (IC) maneuver after each rating of breathing and leg discomfort. Immediately after completing exercise, patients were asked to identify the primary reason for stopping exercise using a previously described questionnaire. 19
Safety assessments
At each visit, adverse events (AEs) reported by the patient were recorded irrespective of causality. Pulse rate and systolic and diastolic blood pressure were measured at each clinic visit immediately prior to spirometry. Clinical laboratory testing (hematology, blood chemistry, and urinalysis) and a resting 12-lead electrocardiogram were performed at screening and at week 12.
Efficacy endpoints
The primary endpoint was EET during CWRCE after 12 weeks of treatment. Owing to the skewness of the EET data during CWRCE, 20 the primary analysis was prespecified to be based on the logarithm to the base 10 of EET during CWRCE. The key secondary endpoint was EET during ESWT after 12 weeks of treatment (ESWT subgroup), also based on log10-transformed data.
Other exercise-related efficacy endpoints included pre-exercise, isotime, and end-exercise IC during CWRCE, and intensity of breathing and leg discomfort during CWRCE and ESWT.
Isotime for each patient was defined as the minimum EET among the pretreatment baseline, week 6, and week 12 CWRCE tests. Isovalue was defined as the value of a specific parameter at isotime. This isovalue could be an observed value (e.g. when it occurred at the end of exercise) or it could be determined by interpolation between the values at the two time points immediately above and below the isotime.
FEV1 and FVC (trough and 1 h postdose) were also measured to confirm bronchodilator efficacy.
Statistical analysis
Mean log10 EETs from CWRCE and ESWT were analyzed using a restricted maximum likelihood-based mixed-effects model repeated measure (MMRM) approach. Analyses included the fixed, categorical effect of treatment, test day, and treatment-by-test-day interaction, as well as the continuous, fixed covariates of baseline and baseline-by-test-day interaction. The baseline covariate term also used the logarithm to the base 10. The primary analysis was performed on the full analysis set (i.e. all randomized patients who received at least one dose of treatment, and who had both baseline and at least one postbaseline measurement at or before 12 weeks for the primary efficacy endpoints).
Based on a previous trial, 21 an appropriate estimate of the standard deviation for log10 EET from CWRCE was 0.206; with this standard deviation, a sample size of 130 patients per group provided 90% power to detect a 21% improvement in EET during CWRCE.
Analyses for EET during CWRCE at 12 weeks (primary endpoint), EET during ESWT at 12 weeks (key secondary endpoint), and pre-exercise IC during CWRCE at 12 weeks (secondary endpoint) were included in a step-down hierarchical testing strategy. Testing strategy details are provided in the online supplement (Figure S1).
The same MMRM approach was adopted for all other continuous secondary endpoints. Adjusted mean values as well as treatment contrasts were presented, together with 95% confidence intervals. Secondary endpoints (i.e. other than EET during ESWT and pre-exercise IC at 12 weeks) were not included in the step-down hypothesis-testing hierarchy and are considered descriptive (nominal p values).
All treated patients were included in the safety analyses, which were descriptive in nature.
Results
Patient disposition and baseline characteristics
A total of 404 patients were randomized and received treatment (Figure 2), with 385 included in the full analysis set.
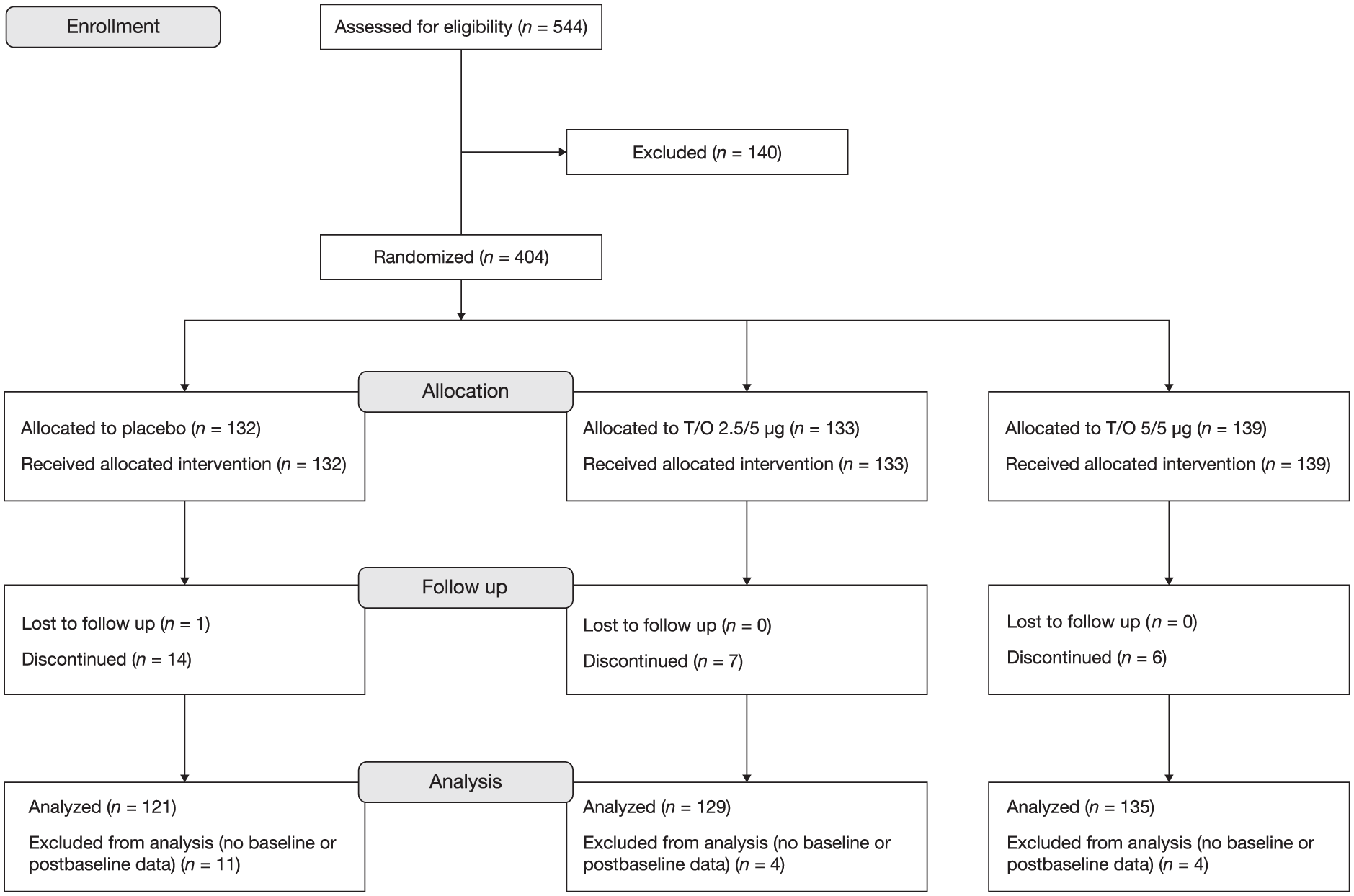
CONSORT diagram.
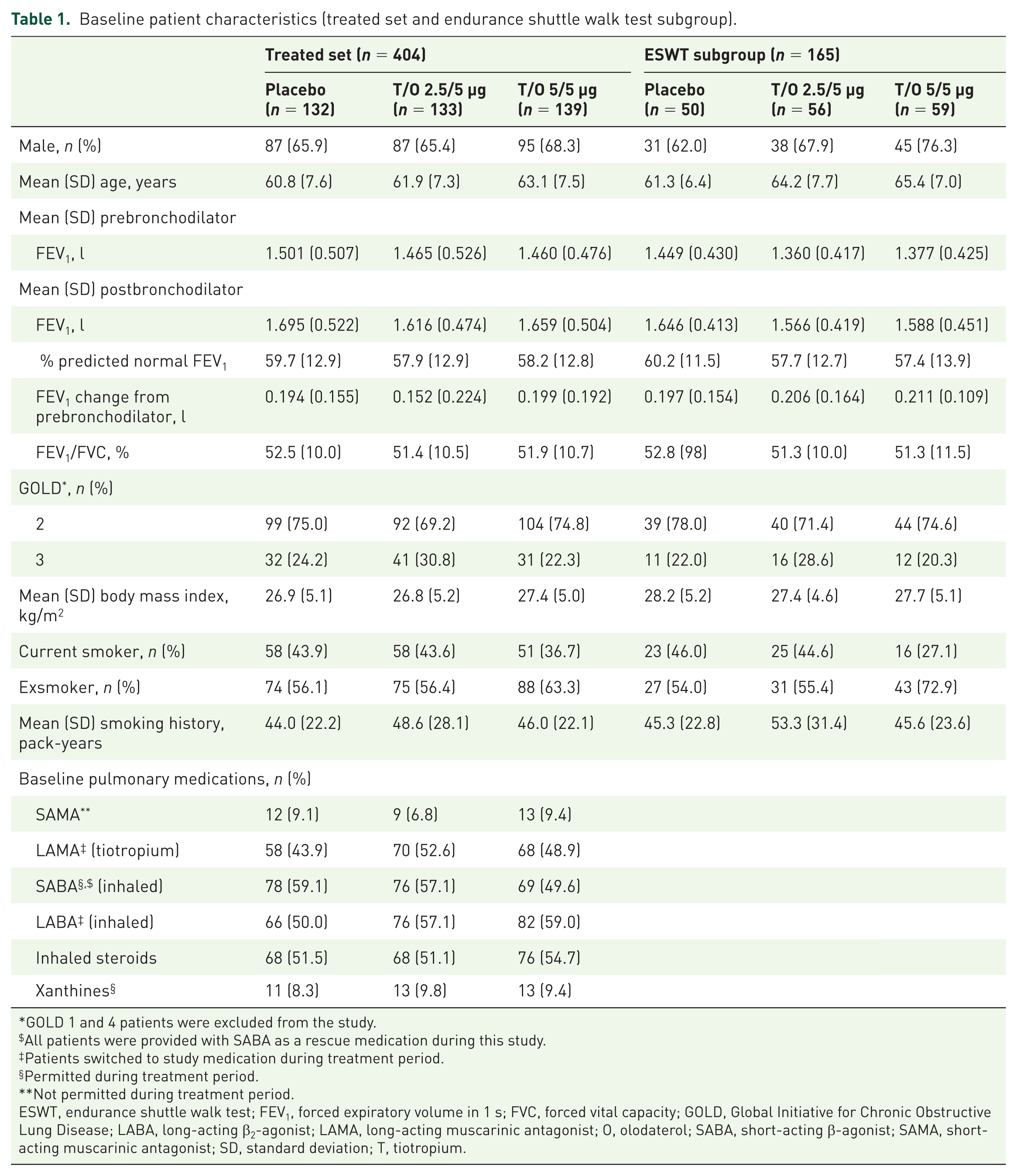
Baseline patient characteristics for the treated set are shown in Table 1. Overall, 73% were Global Initiative for Chronic Obstructive Lung Disease stage 2, with mean predicted normal FEV1 of 58.6%.
Baseline patient characteristics (treated set and endurance shuttle walk test subgroup).
GOLD 1 and 4 patients were excluded from the study.
All patients were provided with SABA as a rescue medication during this study.
Patients switched to study medication during treatment period.
Permitted during treatment period.
Not permitted during treatment period.
ESWT, endurance shuttle walk test; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; GOLD, Global Initiative for Chronic Obstructive Lung Disease; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist; O, olodaterol; SABA, short-acting β-agonist; SAMA, short-acting muscarinic antagonist; SD, standard deviation; T, tiotropium.
Baseline exercise-specific patient characteristics for the treated set are shown in Table 2. Baseline characteristics were generally well balanced across treatment groups.
Baseline incremental and constant work-rate cycle ergometry characteristics (treated set).

CWRCE, constant work-rate cycle ergometry; EET, exercise endurance time; IC, inspiratory capacity; O, olodaterol; SD, standard deviation; T, tiotropium.
A total of 165 patients were included in the ESWT subgroup. Baseline patient characteristics and exercise-specific characteristics for the ESWT subgroup are shown in Tables 1 and 3, respectively. Baseline characteristics were generally well balanced across treatment groups.
Baseline endurance shuttle walk test characteristics (endurance shuttle walk test subgroup, n = 165).

EET, exercise endurance time; O, olodaterol; SD, standard deviation; T, tiotropium.
Two patients had an EET during CWRCE at baseline of >25 min (i.e. screen failure); 11 patients had an EET during ESWT at baseline of >15 min (i.e. not included in the ESWT substudy, but continued in the main study).
Constant work-rate cycle ergometry (full analysis set)
Common baseline geometric mean EET [standard error (SE)] during CWRCE was 443.0 (12.4) s. Geometric mean (SE) EET during CWRCE at week 12 (primary endpoint) was 463.6 (18.8) s for placebo, 503.6 (19.6) s for tiotropium/olodaterol 2.5/5 µg, and 527.5 (20.2) s for tiotropium/olodaterol 5/5 µg, with a statistically significant increase of 13.8% compared with placebo for tiotropium/olodaterol 5/5 µg (p = 0.02) and an increase of 8.6% compared with placebo for tiotropium/olodaterol 2.5/5 µg (p = 0.14) (Figure 3). Since the comparison for tiotropium/olodaterol 2.5/5 µg compared with placebo did not reach statistical significance, subsequent hypothesis tests within the step-down hypothesis-testing strategy are considered descriptive rather than confirmatory (nominal p values).

Geometric mean exercise endurance time during constant work-rate cycle ergometry after 6 and 12 weeks.
Geometric mean (SE) EET at week 6 was 427.7 (17.1) s for placebo, 522.3 (20.2) s for tiotropium/olodaterol 2.5/5 µg, and 525.6 (20.0) s for tiotropium/olodaterol 5/5 µg, with a 22.9% increase compared with placebo for tiotropium/olodaterol 5/5 µg (nominal p = 0.0002) and a 22.1% increase compared with placebo for tiotropium/olodaterol 2.5/5 µg (nominal p = 0.0004) (Figure 3).
The common mean IC values at baseline were: pre-exercise, 2397 ml; isotime, 2131 ml; and end-exercise, 2111 ml. Mean IC increased at pre-exercise, isotime, and end-exercise for both tiotropium/olodaterol doses compared with placebo at weeks 6 and 12 (Figure 4a) (nominal p < 0.05 at all time points for tiotropium/olodaterol 2.5/5 and 5/5 µg).

Mean inspiratory capacity (a) and slope of intensity (b) of breathing discomfort after 12 weeks.
The rate of increase in intensity of breathing discomfort (Borg scale) during exercise [(end of exercise Borg score minus pre-exercise Borg score)/EET] was lower for both tiotropium/olodaterol doses compared with placebo at weeks 6 and 12 (Figure 4b) (nominal p = 0.0218 for tiotropium/olodaterol 2.5/5 µg and p = 0.0598 for tiotropium/olodaterol 5/5 µg at week 12).
For both tiotropium/olodaterol 2.5/5 and 5/5 µg, increases in FEV1 and FVC were observed 1 h after the first dose, and predose and 1 h postdose after 6 and 12 weeks.
Endurance shuttle walking (ESWT substudy)
Common baseline geometric mean EET (SE) during ESWT was 311.2 (13.7) s. Geometric mean (SE) EET during ESWT at week 12 (key secondary endpoint) was 311.4 (22.5) s for placebo, 377.2 (25.9) s for tiotropium/olodaterol 2.5/5 µg, and 376.4 (25.0) s for tiotropium/olodaterol 5/5 µg, with an increase of 20.9% compared with placebo for tiotropium/olodaterol 5/5 µg (nominal p = 0.055) and an increase of 21.1% compared with placebo for tiotropium/olodaterol 2.5/5 µg (nominal p = 0.056) (Figure 5). One patient on placebo, four patients on tiotropium/olodaterol 2.5/5 µg, and two patients on tiotropium/olodaterol 5/5 µg reached test termination (i.e. 20-min duration).

Geometric mean endurance shuttle walk test endurance time after 6 and 12 weeks in the endurance shuttle walk test subgroup.
Geometric mean (SE) ESWT EET at week 6 was 312.0 (22.5) s for placebo, 377.9 (25.8) s for tiotropium/olodaterol 2.5/5 µg, and 376.2 (24.9) s for tiotropium/olodaterol 5/5 µg, with an increase of 20.6% compared with placebo for tiotropium/olodaterol 5/5 µg (nominal p = 0.058) and an increase of 21.1% compared with placebo for tiotropium/olodaterol 2.5/5 µg (nominal p = 0.055) (Figure 5). No patients on placebo, three patients on tiotropium/olodaterol 2.5/5 µg, and two patients on tiotropium 5/5 µg reached test termination (i.e. 20-min duration).
Decreases in the rate of increase in intensity of breathing discomfort (Borg scale) were observed with both tiotropium/olodaterol doses compared with placebo at weeks 6 and 12 (online supplement Figure S2).
Arithmetic versus log10-transformed treatment effects
To facilitate the interpretation and comparisons across studies, the more ‘traditional’ approach based on arithmetic EET data is provided for comparison to the log10-transformed data [online supplement Tables S2 and S3 (CWRCE) and Tables S4 and S5 (ESWT)]. Using the arithmetic data, there were 25.0 and 14.4% increases in EET during CWRCE with tiotropium/olodaterol 5/5 µg over placebo at 6 and 12 weeks, respectively.
Safety (treated set)
Incidences of AEs, serious AEs, and AEs leading to discontinuation with tiotropium/olodaterol 2.5/5 and 5/5 µg were similar to placebo (online supplement Table S6). Most AEs were mild to moderate in severity and not considered related to study medication by the investigator. The most common AE was COPD exacerbation, which occurred in 12.1%, 11.3%, and 7.2% of patients receiving placebo, tiotropium/olodaterol 2.5/5 µg, and tiotropium/olodaterol 5/5 µg, respectively. Two deaths occurred during the study, both in patients receiving tiotropium/olodaterol 2.5/5 µg: one patient died from lung cancer and the other from cardio-respiratory arrest (this event did not occur during the exercise testing). Neither was considered related to study drug by the investigator.
Discussion
In this study, tiotropium/olodaterol 5/5 µg increased EET during CWRCE by 13.8% compared with placebo after 12 weeks in patients with moderate to severe COPD (primary endpoint). In the ESWT substudy, a 20.9% increase in EET for tiotropium/olodaterol 5/5 µg compared with placebo after 12 weeks did not reach statistical significance (p = 0.055).
The effects of tiotropium/olodaterol on EET during CWRCE in moderate to very severe COPD have also been investigated in two replicate, double-blind, crossover studies of 6-week duration (MORACTO®). 22 Similar to the present study, the MORACTO® studies evaluated two doses of tiotropium/olodaterol (2.5/5 and 5/5 µg), and included placebo, tiotropium 5 µg alone, and olodaterol 5 µg alone as comparator arms to evaluate the efficacy of tiotropium/olodaterol compared with placebo and its added benefit over the individual monotherapies. The TORRACTO® study was designed to complement the MORACTO® studies by determining whether the effects of tiotropium/olodaterol compared with placebo on EET during CWRCE were maintained up to 12 weeks to document the duration of effect.
EET during CWRCE for tiotropium/olodaterol 5/5 µg was similar at 6 weeks (geometric mean: 525.6 s) and 12 weeks (geometric mean: 527.5 s), representing 18.6 and 19.1% improvements, respectively, compared with a baseline geometric mean EET of 443.0 s. Statistically significant improvements in EET for tiotropium/olodaterol 5/5 µg compared with placebo were observed after both 6 and 12 weeks, with differences in response magnitude (22.9% improvement after 6 weeks, 13.8% improvement after 12 weeks) explained by variability in EET in the placebo arm (geometric means of 427.7 and 463.6 s after 6 and 12 weeks, respectively).
Tiotropium/olodaterol 2.5/5 µg did not achieve statistical significance for the primary endpoint (EET during CWRCE after 12 weeks). However, several lines of evidence point to an effect of the lower dose on exercise endurance: (i) tiotropium/olodaterol 2.5/5 µg significantly improved EET during CWRCE compared with placebo in both MORACTO® studies [26.5% (p < 0.0001) and 12.1% (p = 0.0003)]; (ii) there was a nominally significant improvement in EET during CWRCE after 6 weeks (22.1%, nominal p = 0.0004) in the present study; (iii) in post hoc analysis, change from baseline in EET during CWRCE after 6 and 12 weeks was nominally significant.
Endurance time during ESWT has been used as an outcome measure in pulmonary rehabilitation studies for many years. 23 Recently, there has been increased attention on ESWT as a methodology to evaluate bronchodilator efficacy. Early single-center studies provided promising results, showing that endurance time during ESWT offered a sensitive test to assess bronchodilation in patients with COPD.7,9–11 However, multicenter, multinational studies have value in supporting generalizability, as has been done for CWRCE.21,24–32 At the time of study initiation in March 2012, there were no published studies using a multicenter trial with ESWT to evaluate the effects of pharmacotherapy in COPD. The first multicenter study was conducted in 2010–2011, with results published in 2013. 33 Two replicate, multicenter studies were conducted in 2011–2012, with results published in 2014. 34
The present study adds to the knowledge about the use of ESWT to assess the effects of bronchodilation on exercise capacity in patients with COPD in a multicenter setting. Based on the methodologic concerns regarding the transition from a single-center to a multicenter setting, a high degree of attention was placed on trial oversight with respect to the training and monitoring of the ESWT. Details of these quality control efforts are described in the online supplement as reference for other researchers interested in conducting multicenter studies using ESWT. The ESWT performed well in this trial; the lack of a learning effect in the placebo arm (i.e. similar EET in placebo arm at baseline and after 6 and 12 weeks) suggests that study participants were able to produce reproducible exercise tests. Future efforts to determine the minimal quality control criteria in multicenter ESWT studies are warranted.
Although statistical significance was just missed (p = 0.055), the observed 20% improvement in EET in ESWT at 6 and 12 weeks with both doses of tiotropium/olodaterol is beyond the proposed minimally important difference for this parameter. 35 With the recognition that the ESWT has a fixed test termination of 20 min, 8 study eligibility criteria included a baseline EET of <15 min to allow for sufficient post-treatment endurance times within the 20-min test. This was the case for the majority of patients, but one patient in the placebo group, four patients in the tiotropium/olodaterol 2.5/5 µg group, and two patients in the tiotropium/olodaterol 5/5 µg group reached the 20-min test termination time point during ESWT after 12 weeks (i.e. did not reach symptom limitation). This may have contributed to reducing the statistical power of the study. A more recent study (PHYSACTO®), with a greater sample size than the present study (65–72 patients per treatment arm), has shown significant improvements in EET during ESWT after 8 weeks of treatment with tiotropium/olodaterol compared with placebo in patients who participated in a self-management behavior-change program (29% increase, p < 0.01). 23
An important methodologic consideration in relation to the current trial is that statistical analyses were performed on log10-transformed endurance time data. Although these analyses are justified in that endurance time during constant work-rate exercise is asymmetrically distributed around the mean, with a significant skew towards long EETs,20,36 this approach complicates comparisons with previous trials.
For the purpose of facilitating comparisons across studies, we also report arithmetic mean treatment effects of tiotropium/olodaterol versus placebo, which ranged from 118 s to 124 s, or ~25% increase over placebo for CWRCE at week 6. Despite expected differences in adjusted mean values between untransformed and log10-transformed approaches, relative (%) increases in EET for active treatment arms versus placebo and the respective statistical significances were very similar for the two statistical approaches, supporting the robustness of the findings.
The improvements in EET versus placebo are consistent with what is expected from single and dual long-acting bronchodilators.21,25,28,32,36,37 Also, increases in EET in CWRCE with active treatments in the present study were beyond the estimated range for a clinically meaningful difference, compared with placebo, of 46–105 s for endurance time, as proposed by the European Respiratory Society task force on outcomes in COPD. 38 Despite not reaching formal statistical significance, the increase in EET during ESWT, averaging 81–88 s or 21% at week 6, compared advantageously with previous publications (121 s, 31% at 3 weeks; 39 47 s, 15% at 12 weeks 34 ). The increase in EET during ESWT with tiotropium/olodaterol was also beyond the proposed clinically meaningful difference. 35
The primary evidence of clinical efficacy to support the health authority approval of tiotropium/olodaterol 5/5 µg was based on the observed effects on lung function (FEV1) and health-related quality of life (St. George’s Respiratory Questionnaire) in the 52-week TONADO® studies. However, exercise capacity has been recommended as an important additional clinical outcome in COPD pharmacologic trials. 40 The results from the present TORRACTO® study, as well as from the previously reported MORACTO® studies, extend the clinical evaluation of tiotropium/olodaterol 5/5 µg, and provide a more complete characterization of therapeutic benefit at the impairment-disability interface. The results show that the primary effects of tiotropium/olodaterol 5/5 µg on lung function lead to clinically meaningful downstream improvements in the capacity to perform different types of exercise (cycling, walking), which is arguably a more meaningful outcome to patients with COPD, who struggle to perform daily activities due to exertional breathlessness.
Conclusion
Tiotropium/olodaterol 5/5 µg improved endurance time during cycle ergometry versus placebo, with a strong tendency to also improve walking endurance.
Footnotes
Acknowledgements
The authors meet the criteria for authorship as recommended by the International Committee of Medical Journal Editors. They take full responsibility for the scope, direction, content of, and editorial decisions relating to, the manuscript; they were involved at all stages of development, and have approved the submitted manuscript. The authors received no compensation related to the development of the manuscript. This work was supported by Boehringer Ingelheim International GmbH. Medical writing assistance was provided by Rob Kite, BSc, of Complete HealthVizion, which was contracted and compensated by Boehringer Ingelheim Pharma GmbH & Co. KG.
Funding
This work was funded by Boehringer Ingelheim Pharma GmbH & Co. KG.
Conflict of interest statement
FM has received research support and speaker fees from AstraZeneca, Novartis, and Grifols, and research support, advisory board participation, and speaker fees from Boehringer Ingelheim and GlaxoSmithKline. DO’D has received research grants via his institution from AstraZeneca, Boehringer Ingelheim, and GlaxoSmithKline, and has served on speaker bureaus, consultation panels, and advisory boards for Almirall, AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Novartis, and Pfizer. JBGI has nothing to disclose. A-MK has received payment for lectures from Boehringer Ingelheim, AstraZeneca, and Novartis, and her institution has received compensation for the conduct of clinical studies from AstraZeneca, Boehringer Ingelheim, Roche, GlaxoSmithKline, Novartis, Chiesi, Takeda, Bayer, Sterna Biologicals, Johnson & Johnson, and Sanofi. DS has received personal fees from Apellis, Cipla, Genentech, Peptinnovate, and SkyePharma, and grants and personal fees from AstraZeneca, Boehringer Ingelheim, Chiesi, Glenmark, Merck, Mundipharma, Novartis, Pfizer, Pulmatrix, Teva, Theravance, and Verona. RC has received personal fees from Sunovion and Theravance, and grants and personal fees from Boehringer Ingelheim, AstraZeneca, and Astellas. AH, KT, and YZ are employees of Boehringer Ingelheim.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
