Abstract
Objective:
Scant clinical data are available on the effects of current treatments for asthma on different subgroups of patients with this disease. We conducted a prospective, noninterventional, multicenter real-life study in adult patients with persistent asthma, and we specifically analyzed the effects of treatment with extrafine beclometasone dipropionate/formoterol (BDP/F) in asthma patients categorized by phenotypes related to small airways (i.e. smoking habits, disease duration, and air trapping).
Methods:
Patients received BDP/F as a fixed combination (100/6 μg), administered in 1–2 inhalations twice daily over a period of 12 weeks. Peak expiratory flow (PEF), forced expiratory volume in 1 second (FEV1), forced vital capacity (FVC), number of asthma attacks, asthma control, and severity of asthma symptoms were evaluated in the overall population and in different subgroups at three different time points.
Results:
Overall, 213 patients were enrolled. In the overall population the treatment resulted in a significant increase in the proportion of well controlled patients (from 6.1% to 66.3%; p<0.001), and a reduction of uncontrolled subjects (70.3% versus 10.0%; p<0.001). BDP/F was also associated with a reduction in asthma attacks and an improvement of symptoms. These results were confirmed in specific subgroups of patients identified as small airway phenotypes: smokers, elderly patients, those with long duration of disease and air trapping.
Conclusions:
This real-life observational study indicates that extrafine BDP/F in a fixed combination improves asthma control and symptoms in the overall population as well as specific subgroups of patients.
Introduction
Goals for the treatment of asthma, which currently affects over 300 million people worldwide, have changed in recent years and recommendations are now based on the level of effective control of symptoms rather than on disease severity [Global Initiative for Asthma, 2012; Braman, 2006; Masoli et al. 2004; Barnes, 2012]. Although effective therapies and management options for asthma are available, a large proportion of patients with asthma still have poorly controlled symptoms and face long-term respiratory impairment and disability [Partridge et al. 2006]. One of the major difficulties in the control of the disease is that asthma is a complex and highly heterogeneous condition characterized by differences in the pathogenesis, and therefore asthmatic patients present different clinical phenotypes, variable severity of the symptoms, and varying responses to the treatment [Chapman et al. 2008]. In order to devise effective management strategies tailored to patient needs, it is important to understand the various phenotypes and pathological mechanisms of chronic asthma. Traditionally, features such as atopy, trigger factors and severity of exacerbations were used to classify asthma, but more recently important clinical phenotypes such as smoking status, long duration of the disease, those with partially-fixed airway obstruction and those with dependency or resistance to previous treatments have been used [Wenzel, 2006; Holgate, 2013; Chanez et al. 2007]. It is generally agreed that asthma is a disease entity of different phenotypes; however, very little clinical data on the effects of current treatments on different patient subgroups are available.
Asthma is characterized by inflammatory and remodelling processes affecting both large airways (internal diameter ⩾2 mm) and small airways (internal diameter <2 mm) [Van den Berge et al. 2011; van der Wiel et al. 2013]. A recent review has pointed out that impairment of small airways can be of particular relevance in specific phenotypes of asthmatic patients, such as patients with severe asthma, with a long duration of the disease, elderly patients and smoking asthmatics [Contoli et al. 2012]. It is therefore hypothesized that use of extrafine formulations that ensure more uniform distribution throughout the entire bronchial tree can improve small airway function, providing additional clinical benefits in terms of increased asthma control in asthmatic patients as compared to larger-particle formulations.
Treatment of asthma with inhaled corticosteroids (ICSs) and long-acting inhaled β2-agonists (LABAs) has been shown to effectively manage asthma in both clinical trials and clinical practice, and to provide a marked reduction in hospitalization and mortality rates [Barnes, 2012]. The fixed combination of beclometasone dipropionate and formoterol (BDP/F, Foster®, Chiesi Farmaceutici, Italy) 100/6 μg is characterized by extrafine particle size, which results in improved lung deposition and uniform treatment of asthma symptoms throughout the entire bronchial tree [De Backer et al. 2010; Huchon et al. 2009]. This translates into greater asthma control and quality of life when compared with larger-particle ICS/LABA fixed combinations [Terzano et al. 2012; Allegra et al. 2012; Muller et al. 2011]. Recently, extrafine BDP/F was shown to effectively prolong time to first severe exacerbation when used for both maintenance and reliever therapy (MART) in patients with uncontrolled asthma [Papi et al. 2013]. The MART approach also resulted in significant reduction of yearly rates of severe exacerbations, as well as significant decrease in the use of systemic corticosteroids and hospitalizations or emergency visits.
In order to investigate the real-life effectiveness of extrafine BDP/F in routine clinical practice, we conducted a prospective, noninterventional, multicenter real-life study in patients with persistent asthma enrolled through primary care centers in Austria. Unselected adult subjects receiving standard therapy for asthma were switched to extrafine BDP/F for 12 weeks. Clinical outcomes at the end of the study were compared with baseline characteristics in the overall population. As an additional analysis, patients were categorized on the basis of three independent parameters related to small airway impairment (i.e. smoking habit, disease duration, and air trapping) to assess the effects of treatment with extrafine BDP/F in specific asthma phenotypes where small airway abnormalities were reported to have a more prominent role [Contoli et al. 2012].
Methods
Study design and patients
This was a prospective, multicenter, noninterventional study conducted in 55 centers in Austria. The study started in February 2011 and ended in November 2011 (registered at the Austrian Federal Office for Safety in Health Care/Austrian Agency for Health and Food Safety, NIS000544).
Adult patients (⩾18 years) with asthma diagnosed according to GINA guidelines [Global Initiative for Asthma, 2012] and who had an indication for treatment with a corticosteroid and a long-acting β2-agonist according to the same guidelines, were consecutively enrolled in participating centers. Inclusion criteria were bound to the conditions of use outlined in the Summary of Product Characteristics. The aim of this study was to obtain data concerning tolerance, symptoms treatment and asthma control of patients with bronchial asthma treated with extrafine BDP/F.
Patients were enrolled if they had not been treated with extrafine BDP/F before entering the study. Patients received extrafine BDP/F as a fixed combination (100/6 μg), administered at 1–2 inhalations twice daily over a period of 12 weeks. Patients experiencing an exacerbation of symptoms received oral steroids or antibiotics. All treatment and management decisions were taken only according to the clinical judgement of the investigating physicians.
The study was performed according to Austrian law, which does not require an ethics approval and written informed consent for a noninterventional study.
Study evaluations
All patients underwent examinations at three points in time: baseline, 4–8 weeks and 12 weeks after the start of the treatment. According to the standard practice of our center, clinical data, medical history of patients (type of asthma, and pretreatment) and inspiratory flow (IF) data were collected (using a Personal Best® Universal Range Peak Flow Meter, Respironics, Germany) at baseline. In addition, the number of asthma attacks (during daytime and night-time), the number of exacerbations and parameters of pulmonary function (peak expiratory flow [PEF], forced expiratory volume in 1 second [FEV1], and forced vital capacity [FVC]), were evaluated at each visit. FVC data were available only for 141 patients. Asthma control was assessed at baseline and at week 12, according to GINA guidelines (controlled, partly controlled, and uncontrolled) [Global Initiative for Asthma, 2012]. The severity of asthma symptoms (dyspnea, wheezing, chest tightness, and cough) was examined according to a 4-point scale from 0 (not present) to 3 (severe). Information on patients’ symptoms were collected during the directed interview of the physician to patient during every visit. Safety evaluations were performed at each visit.
Data analysis
A total of 213 patients were included in the analysis which was carried out in the overall population and in different subgroups of patients, stratified according to: (a) smoking habits (nonsmoker, exsmoker, smoker); (b) disease duration (⩽10 years versus >10 years), and (c) presence of air trapping (indicated by FVC measurement).
Data were shown as mean and standard deviation of mean and 95% confidence interval. A Student’s t-test for paired data and McNemar test were used to assess differences between baseline and 12 weeks follow up and for categorical variables, respectively.
Results
Baseline characteristics
Overall, 213 patients were enrolled in the study. Table 1 describes the main characteristics of patients at baseline. Patients were evenly distributed between genders (49.1% female) and, although the majority of patients were nonsmokers (53.1%), a significant proportion of current smokers (23.9%) and exsmokers (23.0%) were included. Among smokers, 53.2% of patients consumed ⩾20 pack/years. About 40% of patients reported disease duration >10 years. The proportion of patients with a peak inspiratory flow (PIF) <60 l/min represented more than 60% of the overall studied population.
Patients’ characteristics at baseline.

BUD/F, budesonide/formoterol; DPI, dry powder inhaler; FP/S, fluticasone propionate/salmeterol; ICS, inhaled corticosteroid; LABA, long-acting beta2-agonist; PIF, peak inspiratory flow; pMDI, pressurized metered dose inhaler; SABA, short-acting beta2-agonist.
Overall population
Table 2 compares the clinical outcomes at baseline and the end of the study in the overall population. Treatment with extrafine BDP/F for 12 weeks showed significant improvements in asthma control with an increase of the percentage of patients with well-controlled asthma from 6.1 to 66.3% and a decrease in the proportion of patients with uncontrolled asthma from 70.3% to 10.0% (p < 0.01). The mean number of asthma attacks during daytime decreased from 1.8 to 0.2 per week and the number of night-time attacks decreased from 1.2 to 0.1 per week (p < 0.01). Furthermore, the percentage of patients using their reliever medication more than twice a week was significantly reduced on treatment with extrafine BDP/F, as well as the percentage of patients with exacerbations, defined as acute worsening of persisting symptoms over 24 hours (p < 0.01). Significant improvements were also observed in lung function parameters (see Table 2; p < 0.01 versus baseline for all variables).
Clinical outcomes after 12 weeks of treatment with extrafine BDP/F in the overall population (N = 213).

p < 0.01 versus baseline for all variables quoted (Student’s t-test for paired data).
According to Global Initiative for Asthma Guidelines.
FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PEF, peak expiratory flow; SD, standard deviation.
Analysis by age indicated that elderly patients (>50 years) tended to have poorer lung function parameters although they displayed a similar level of asthma control and no difference in presentation of symptoms. Treatment with extrafine BDP/F was associated with improvements in disease control, symptoms and lung function parameters irrespective of age group (data not shown).
A subgroup of 105 patients was treated with larger particles ICS/LABA fixed combinations before entering the study. Among these patients, 2.9% had controlled asthma at baseline, whereas 25.0% were partially controlled and 72.1% were uncontrolled. The proportion of patients with controlled disease increased to 60.6% after 12 weeks of treatment with extrafine BDP/F, as well as parameters reflecting disease control such as daytime and night-time symptoms, use of rescue medication, limitation of physical activity and lung function.
A trend towards the improvement of lung function and symptoms was already present at 4–8 weeks (FEV1: 2.9% predicted; PEF: 353 l/min; FVC: 3.6% predicted).
Patients categorized by smoking history, disease duration, and air trapping
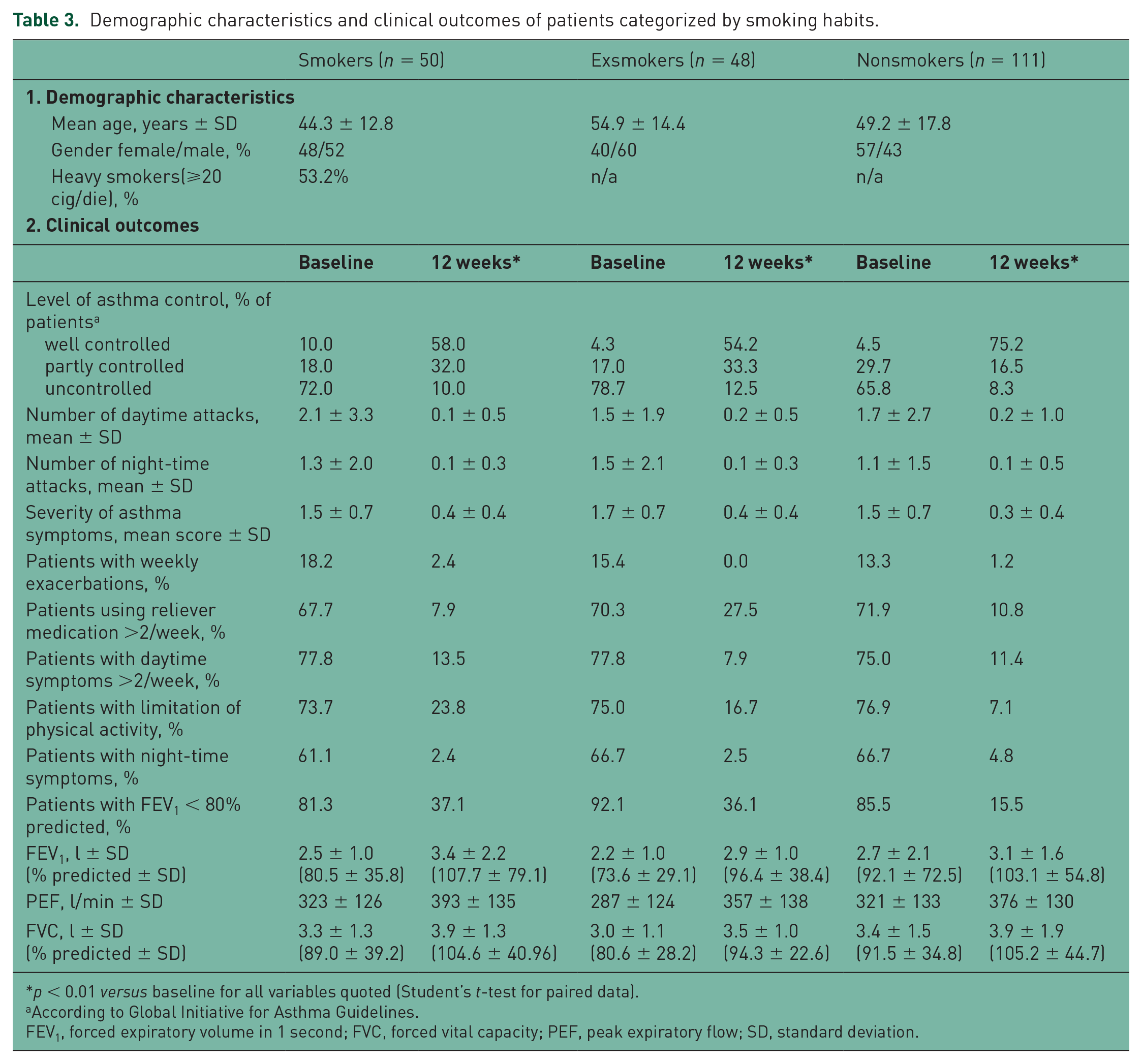
Treatment with extrafine BDP/F resulted in significant improvements of asthma control, symptoms and lung function parameters in patients with asthma phenotypes related to small airways (i.e. smokers, patients with long duration of asthma and air trapping) (Tables 3–5 and Figure 1).
Demographic characteristics and clinical outcomes of patients categorized by smoking habits.
p < 0.01 versus baseline for all variables quoted (Student’s t-test for paired data).
According to Global Initiative for Asthma Guidelines.
FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PEF, peak expiratory flow; SD, standard deviation.
Demographic characteristics and clinical outcomes of patients categorized by duration of disease.

p < 0.01 versus baseline for all variables quoted (Student’s t-test for paired data).
According to Global Initiative for Asthma Guidelines.
FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PEF, peak expiratory flow; SD, standard deviation.
Demographic characteristics and clinical outcomes of patients categorized by forced vital capacity (<80% versus ⩾80%).

p < 0.01 versus baseline for all variables quoted (Student’s t-test for paired data).
According to Global Initiative for Asthma Guidelines.
FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PEF, peak expiratory flow; SD, standard deviation.

Severity of asthma symptoms in specific asthma phenotypes. Score is calculated on a 4-point scale from 0 (not present) to 3 (severe). Data are reported as mean ± standard deviation; comparisons are versus baseline.
Smoking habits
Response to treatment was similar in light and heavy smokers (<20 and ⩾20 pack/years, respectively, data not shown). Lung function parameters at baseline tended to be slightly more impaired in exsmoker patients, but the treatment was equally effective as compared with the other groups. The group of nonsmokers showed a greater improvement in the level of asthma control (75.2% versus 58.0% of smokers and 54.2% of exsmokers); however, ~30% of smokers and exsmokers reached partially-controlled asthma at the end of treatment. After 12 weeks of treatment, compared with the baseline, smokers showed significant improvement of lung function parameters (see Table 3; p < 0.01 for all variables). During the study, smokers showed significant improvement of symptoms such as cough, chest tightness, wheezing and dyspnea (Figure 1). In more detail, cough score decreased from 1.6 at baseline to 0.4 points at week 12, chest tightness from 1.8 to 0.6 points, wheezing from 1.4 to 0.3 points and dyspnea from 1.3 to 0.2 points (p < 0.01 for all comparisons).
Duration of asthma
Patients with longer duration of the disease (>10 years) had poorer lung function, tended to use more reliever medication and to have more daytime symptoms. Significant improvement of all variables was observed after treatment in patients with both short and long duration of asthma. The improvement in the level of asthma control was significant in both groups, with 72.7% of patients with short duration of asthma and 55.6% of patients with long duration of asthma achieving well controlled disease (p < 0.01 versus baseline for both groups). Patients with longer duration of disease showed significant improvement of lung function parameters compared with baseline (see Table 4; p < 0.01 for all variables). Symptoms score changed as follows: cough score decreased from 1.6 at baseline to 0.4 points at week 12, chest tightness from 1.9 to 0.6 points, wheezing from 1.4 to 0.4 points and dyspnea from 1.2 to 0.2 points (p < 0.01 for all comparisons).
Air trapping (FVC < 80%)
Overall, a statistically significant change in FVC from baseline of 520 ml was recorded (see Table 5). Patients with baseline air trapping showed a significant improvement at the end of the study (p < 0.01).
Tolerability
No adverse effects related to the study medication were reported. Treatment was well tolerated with eight withdrawals (change in medication, poor treatment response, and symptom improvement after allergic season). A total of 95.5% of patients continued treatment with extrafine BDP/F after the study period.
Discussion
Our results point out two important aspects. First, the overall population of unselected patients receiving treatment with extrafine BDP/F for the first time showed significant improvements in asthma control and lung function, reduction of symptoms as well as use of reliever medications. Second, these results were consistent also in subgroups of patients identified as small airway phenotypes (i.e. smokers, patients with a long duration of the disease (>10 years) and patients with air trapping [FVC < 80%]). Dyspnea and cough improved in all subgroups, therefore treatment with extrafine BDP/F may improve quality of life of our patients, although this was not measured in this study.
In patients with asthma, the goal of therapy is to control symptoms, prevent exacerbations, and maintain lung function while at the same time allowing the patient to lead a normal active life. Although for many years data from randomized, controlled trials (RCTs) were considered the gold-standard in therapeutic decision-making, the importance of data from real-life experience has recently come to the forefront. One of the major limitations of relying solely on data from RCTs is that inclusion criteria normally specify that patients are homogeneous and as a result large subgroups of patients are excluded (i.e. smokers, elderly patients, and patients with comorbidities). Just a few reports of real-life studies have been recently published indicating the efficacy and safety of asthma treatments in different patient groups not normally included in RCTs but further data are needed [Terzano et al. 2012; Allegra et al. 2012; Muller et al. 2011; Brusselle et al. 2012]. Moreover, the need for examining small airways function is everyday practice, and further investigating the effects of extrafine ICSs have been advocated [Ivancso et al. 2013]. The results of our study give an important impact that real-life clinical trials are a good tool to study different asthma phenotypes.
Cigarette smoking in asthma is associated with poor symptom control and smokers are more likely to be admitted to hospital due to poorly controlled asthma compared with nonsmokers with asthma [Polosa and Thomson, 2013]. The mechanisms proposed for the different response to treatment observed in smoking patients include altered airway inflammation, especially in the small airways, and corticosteroid insensitivity [Polosa and Thomson, 2013; Thomson and Chaudhuri, 2009; Chalmers et al. 2002]. Chalmers and colleagues reported that efficacy of treatment with inhaled ICSs is impaired in active smokers who, after 3 weeks of treatment with fluticasone, showed statistically significantly lower improvement in lung function parameters as compared to nonsmoker asthmatics [Chalmers et al. 2002]. The differential response of smoker asthmatics to ICS treatment might be explained by prominent neutrophilic inflammation or dysregulation of genes involved in response to steroid receptors [Chalmers et al. 2002]. Differently from the study by Chalmers and colleagues, our study indicated that treatment with extrafine BDP/F provides comparable improvement of symptoms and asthma control both in smokers (current and former) and in nonsmokers, with a high proportion of patients showing normalized lung function at the end of the treatment period. Patients included in our study were all treated with large-particle formulations before entering the study, apart from seven patients who received extrafine ciclesonide (three former smokers and four nonsmokers). These results suggest that treatment with an extrafine formulation, that can also effectively target the small airways, provides significant benefit in asthmatic smokers, which have proved to be less responsive to treatment with large-particle corticosteroids. Our results are in agreement with those of Brusselle and colleagues who reported similar benefits in former or current smokers [Brusselle et al. 2012].
Preventing worsening of asthma over time is vital as is the need to ensure efficacy of treatment in patients who have long-term asthma [O’Byrne, 2009]. Only a few studies have assessed the relationship between disease duration and treatment response, indicating that improvement of lung function parameters is significantly higher in patients with shorter duration of symptoms [Selroos et al. 1995; Redington, 2001]. The possible mechanisms underlying this different response are still unknown and it is speculated that they are linked to the persistent uncontrolled inflammation and remodeling of the airways. The patient cohort in our study included patients with different durations of disease. We found that, after treatment with extrafine BDP/F, more than half of patients with a long history of asthma had controlled disease compared to less than 5% at baseline.
Our findings show that small airway-related components of the disease (e.g. nocturnal symptoms and air trapping), are significantly improved after treatment with extrafine BDP/F. Alterations in the small airways can lead to small airway closure or near closure, which in turn results in peripheral air trapping. FVC has been previously suggested as a potential indicator of air trapping [Contoli et al. 2010] and has been used in this study, since, in a real-life setting, body plethysmography or nitrogen washout assessments are not done regularly in primary care centers and therefore it was not possible to evaluate other parameters used for the characterization of air trapping. We observed that patients with air trapping (baseline FVC < 80% of predicted) tended to have lower levels of asthma control. A study has suggested the use of FVC as an indicator of small airway impairment and predictor of response to extrafine treatment, showing that patients with greater improvement of FVC after treatment with extrafine particles also demonstrated a significant decrease of parameters indicative of airway and systemic inflammation [Popov et al. 2001]. Our results indicate that treatment with extrafine BDP/F has a positive effect on the reduction of air trapping as demonstrated by normalization of FVC values from 65% to 83%. This could be consistent with a more peripheral deposition and enhanced treatment of air trapping and small airway inflammation [Papi et al. 2007]. Concerning symptoms, patients with air trapping showed the highest reduction in dyspnea of all studied subgroups (2.2 at baseline to 0.7 after 12 weeks treatment with extrafine BDP/F). Additionally the percentage of patients with air trapping and uncontrolled asthma declined significantly from 83% to 14%. Future studies using different indicators for air trapping are needed to further confirm our findings.
The main aim of this study was to evaluate, in a real-life setting, the clinical outcomes of patients previously treated with standard therapy for asthma and eventually switched to extrafine therapy with BDP/F. The results show significant improvements in terms of lung function, asthma control, and symptoms compared to baseline and standard therapy received prior to study start. This evidence suggests that the observed effects are induced by the treatment received during the study. Patients participating in our study were previously treated with drugs formulated in larger particles (except seven patients who received extrafine ciclesonide), thus suggesting that the positive outcomes may be linked to the extrafine formulation of the administrated drug. One limitation of our study is the absence of a parallel control group and therefore it is not possible to rule out that the results observed can be attributed, at least in part, to a spontaneous relief or a placebo effect. However, the results are in line with previous real-life studies comparing extrafine BDP/F with larger-particle treatments and reporting significant benefits in terms of better asthma control, symptoms and quality of life [Terzano et al. 2012; Allegra et al. 2012; Muller et al. 2011]. Although all patients were followed in a clinical-practice setting, we cannot rule out that the improvement in lung function and asthma control could be due to improved medication compliance or adherence from study participation (i.e. regular contact with investigators during their assessments). In addition, about one out of six patients enrolled in the study had actually received BDP/F as an add-on to ongoing treatment and not as a single therapy: this may potentially represent a confounding factor when evaluating the study results. Another possible limitation is that, despite the large number of patients enrolled in the overall population, subgroup analysis necessarily evaluated smaller populations. Therefore, the findings of this study may benefit from further confirmation in larger cohorts of patients. Lastly, data on asthma control at 4–8 weeks and information on the proportion of patients with asthma COPD overlap syndrome (ACOS) were not available.
Conclusion
This real-life observational study showed that patients receiving extrafine BDP/F fixed combination for the first time experienced significant improvements of asthma control and symptoms compared to treatments received previously. Positive clinical outcomes were observed in the overall population as well as in asthma patients with small airway phenotypes that proved to be less responsive to treatment with larger particle formulations.
Footnotes
Acknowledgements
Editorial assistance for the preparation of this manuscript was provided by Luca Giacomelli, PhD, Simona Barlera, PhD, Siobhan Ward, PhD, and Silvana Pileggi, PhD, on behalf of Content Ed Net; this assistance was funded by Chiesi Farmaceutici. The authors would like to thank all contributing physicians for their active participation in the trial, Claudia Höller (Chiesi, Austria) for her contribution and scientific assistance with the manuscript, Ursula Sturm (Chiesi Austria) for her contribution in study design and initiation, and Dr Wolfgang Rejzlik (Marketmind GmbH) for statistical support.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Chiesi Pharmaceutical GmbH, Vienna Austria.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Wolfgang Pohl has given presentations at symposia and /or served on scientific advisory boards sponsored by Almirall, Astra Zeneca, Boehringer Ingelheim, Chiesi, Mundipharma, Meda, Novartis, Teva.
Katharina Marth has no conflict of interest. Milos Petrovic has no conflict of interest.
