Abstract
Introduction:
Fatigue, which is also present in the healthy population, is a common but understudied symptom in chronic obstructive pulmonary disease (COPD). We hypothesized that clinically significant fatigue is also frequent in COPD and can be associated with an increased disease burden.
Methods:
An exploratory analysis derived from an ongoing cross-sectional study was carried out to evaluate levels of fatigue and impact on health-related quality of life/health status in patients with COPD (COPD group; n = 20) and healthy subjects (control group; n = 5). Health-related quality of life was measured using the Short Form Health Survey 36 (SF-36), health status with the Clinical COPD Questionnaire (CCQ), and airways obstruction with postbronchodilator forced expiratory volume in 1 s (FEV1 %predicted). Fatigue was measured with the vitality score of the SF-36, its clinical significance being defined by values of 50 or less. Fatigue was also measured using the Functional Assessment of Chronic Illness Therapy scale for fatigue (FACIT-F).
Results:
Vitality scores were significantly worse in the COPD group (45.60 versus 76.25; p = 0.004). FACIT-F scores were significantly lower in the COPD group versus the control group (74.5 versus 95.0; p = 0.03). Clinically significant fatigue was detected in 60% of the COPD group, and was associated with a worse FEV1 %predicted (47.71 versus 65.82%; p = 0.016), worse symptoms burden (CCQ symptoms score 3.75 versus 2.43; p = 0.019), and worse overall health status (CCQ total score 3.30 versus 2.11; p = 0.011). Its link with systemic inflammation remains to be clarified further.
Conclusions:
Clinically significant fatigue is common among patients with COPD and is associated with an increased disease burden. It should therefore be integrated as a measure of disease prognosis and control in patients with COPD.
Introduction
In patients with chronic obstructive pulmonary disease (COPD) fatigue is the most commonly detected ‘general’ symptom, and is also one of the most under-recognized and under-treated manifestations of this disease [Antoniu and Ungureanu, 2015]. Fatigue can impact significantly on the health status of such patients and can also negatively influence disease prognosis [Stridsman et al. 2015]. In COPD, fatigue is most commonly seen as a physical fatigue and is usually targeted with pulmonary rehabilitation. However, other dimensions of fatigue, such as mental fatigue or social fatigue, can also be relevant and should be evaluated in such patients. Fatigue is more frequently considered within the management approach to the underlying disease in patients with advanced airflow limitation and is often attributed to hypoxaemia [Amann et al. 2010; Casale et al. 2004; Zattara-Hartmann et al. 1995]. However, fatigue can also be due to other pathogenic mechanisms such as anaemia or systemic inflammation. Fatigue can also be diagnosed in patients with COPD and without hypoxaemia, but its relationship with the degree of airflow obstruction, exercise capacity, systemic inflammation or health status has only recently been addressed by a few studies, which however have not considered the level of fatigue in comparison with the healthy population. In this paper we hypothesized and demonstrated that in patients with COPD, fatigue is more severe than in healthy subjects and that when clinically significant it is associated with an increased disease burden in these patients.
Methods
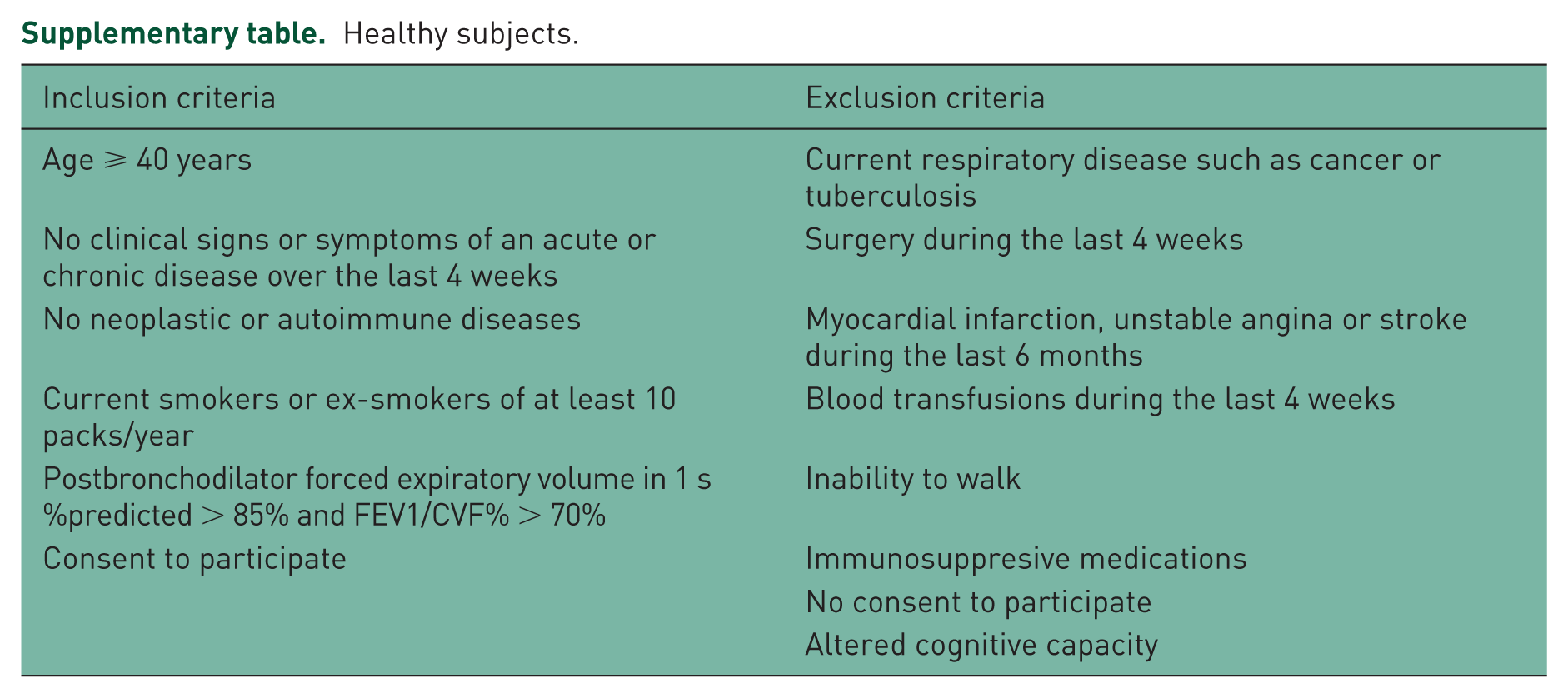
This was an exploratory (initial) analysis of a sample of patients with COPD (COPD group) and healthy, non-COPD subjects (control group), in whom the levels of fatigue and the related impact on health-related quality of life/health status were evaluated. The clinical study protocol received the approval of the Ethics Committee of the Grigore T. Popa University of Medicine and Pharmacy, Iasi, Romania. Participants were enrolled based on their signed informed consent. Among the inclusion criteria for COPD patients, the main ones were a COPD diagnosis known for at least 1 year, the patient was a current or ex-smoker with a history of smoking of at least 10 packs/year, the patient had received no systemic corticosteroid therapy during the last 4 weeks, and had no signs of exacerbations over the last 4 weeks. The exclusion criteria for both COPD patients and healthy subjects as well as inclusion criteria for healthy subjects are listed in the supplementary table.
Patients with chronic obstructive pulmonary disease.

Healthy subjects.
Airway obstruction was measured by postbronchodilator forced expiratory volume in 1 s (FEV1 %predicted) according to the American Thoracic Society criteria [American Thoracic Society, 1995].
Exercise capacity was measured with the 6 min walking test (6MWT) based on the standardized procedure [ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories, 2002].
The level of dyspnoea was evaluated before and at the completion of the exercise testing using the Borg scale [Grant et al. 1999].
Health-related quality of life was measured in both patients with COPD and healthy subjects using the Short Form Health Survey 36 (SF-36) [Ware and Sherbourne, 1992]. This is a generic, 36-item questionnaire, which has been widely used in patients with COPD, and generates several domain scores including vitality, physical functioning, social functioning and two general scores, mental component score (SF-MCS) and physical component score (SF-PCS). The scores can range from 0 to 100 and a lower score corresponds to a more altered aspect of health-related quality of life [Casaburi et al. 2002]. Vitality domain score was used to define the clinical significance of fatigue. Clinically significant fatigue was defined with a SF-36 vitality subdomain score of 50 or less [Cho et al. 2013]. It represents a validated method to evaluate this symptom in various inflammatory debilitating diseases such as multiple sclerosis or rheumatoid arthritis in which a supplementary threshold of 35 or less has been defined for severe fatigue [Fernández et al. 2011; Boosman et al. 2010; Druce et al. 2015].
Fatigue was also measured using the Functional Assessment of Chronic Illness Therapy scale for fatigue (FACIT-F). This is 26-item scale with four domain scores, physical fatigue (FACIT-PWB), emotional fatigue (FACIT-EWB), social fatigue (FACIT-SWB) and functional fatigue (FACIT-FWB), and a total score (FACIT-T). The 13-item shortened version of this scale was previously used in other studies involving patients with COPD [Al-Shair et al. 2012; Stridsman et al. 2013]. We used this scale to back up the vitality score and to further characterize this symptom in the populations studied.
Health status was evaluated with the Clinical COPD Questionnaire (CCQ) in patients with COPD. This is a 10-item disease-specific questionnaire generating three domain scores for symptoms (CCQ-S), functional status (CCQ-F) and mental status (CCQ-M), and a total score (CCQ-T) [van der Molen et al. 2003]. A higher score indicates a worse health status.
Serum C-reactive protein (CRP) was measured by the latex-agglutination method (Spectrum Laboratories, Rancho Dominguez, CA, USA), serum tumour necrosis factor-alpha (TNF-α) was measured by the chemiluminescence method (Siemens), and a monocyte blood count was performed with a Sysmex XS 1000I® (Sysmex Corporation, Kobe, Japan) as a part of a complete blood count test.
SPSS Statistics for Windows version 17.0 software (SPSS Inc. 2008, Chicago, IL, USA) was used for statistical analyses. To compare two sets of data, the Student’s t-test was used when the variables analysed had a normal distribution, and the nonparametric test Mann–Whitney U test was used for variables without a normal distribution. For the first test, mean values were compared, whereas for the second one median values were compared (when the nonnormal distribution shapes were the same). Goodness-of-fit with normal distribution was assessed by the Shapiro–Wilk test (suitable for small samples). Also the limits for skewness and kurtosis were considered appropriate if they were between -2 and 2. Significance was considered for a p value of less than or equal to 0.05 (standard value). We defined pT respectively and pMW for student’s t-test and the Mann–Whitney U test significance. Spearman’s correlation was used to evaluate relationships between certain variables. The confidence interval for the correlation was computed with the bootstrap method (lower limit [LL], upper limit [UL]). The statistically significant differences of certain variables analysed comparatively between the predefined subjects sets were represented by box and whiskers plots (where the middle line is the median or the second quartile). Normally distributed variables were described by mean ± standard deviation, while nonnormal variables were described by the median and interquartile range (which contains 50% of data).
Results
There were 20 subjects in the COPD group and five in the control group. The main descriptive data for both groups are presented in Table 1. Most notably the groups were comparable in terms of age (mean value 66.3 years in the COPD group versus 61.0 years in the control group; p = 0.16), and this ensured that the impact of age-related fatigue was minimized.
Main descriptive data for the chronic obstructive pulmonary disease group and the control group.

pM, Mann–Whitney U test; pt, Student’s t-test; SD, standard deviation; SpO2, amount of oxygenated haemoglobin in the blood.
Fatigue: symptom severity in the COPD group compared with the control group
Mean vitality SF-36 score was found to differ significantly between groups being 45.6 ± 19.98 for patients with COPD and 76.25 ± 15.56 for healthy subjects (pT = 0.004) (Figure 1). The SF-PCS scores also differed significantly between groups (mean ± standard deviation scores were 41.50 ± 7.15 versus 50.46 ± 3.08; pT = 0.013) (Figure 2), whereas the SF-MCS score was also significantly lower in the COPD group (41.47 ± 10.15 versus 52.91 ± 7.51; pT = 0.028).

Fatigue severity in patients with chronic obstructive pulmonary disease versus healthy subjects (controls).
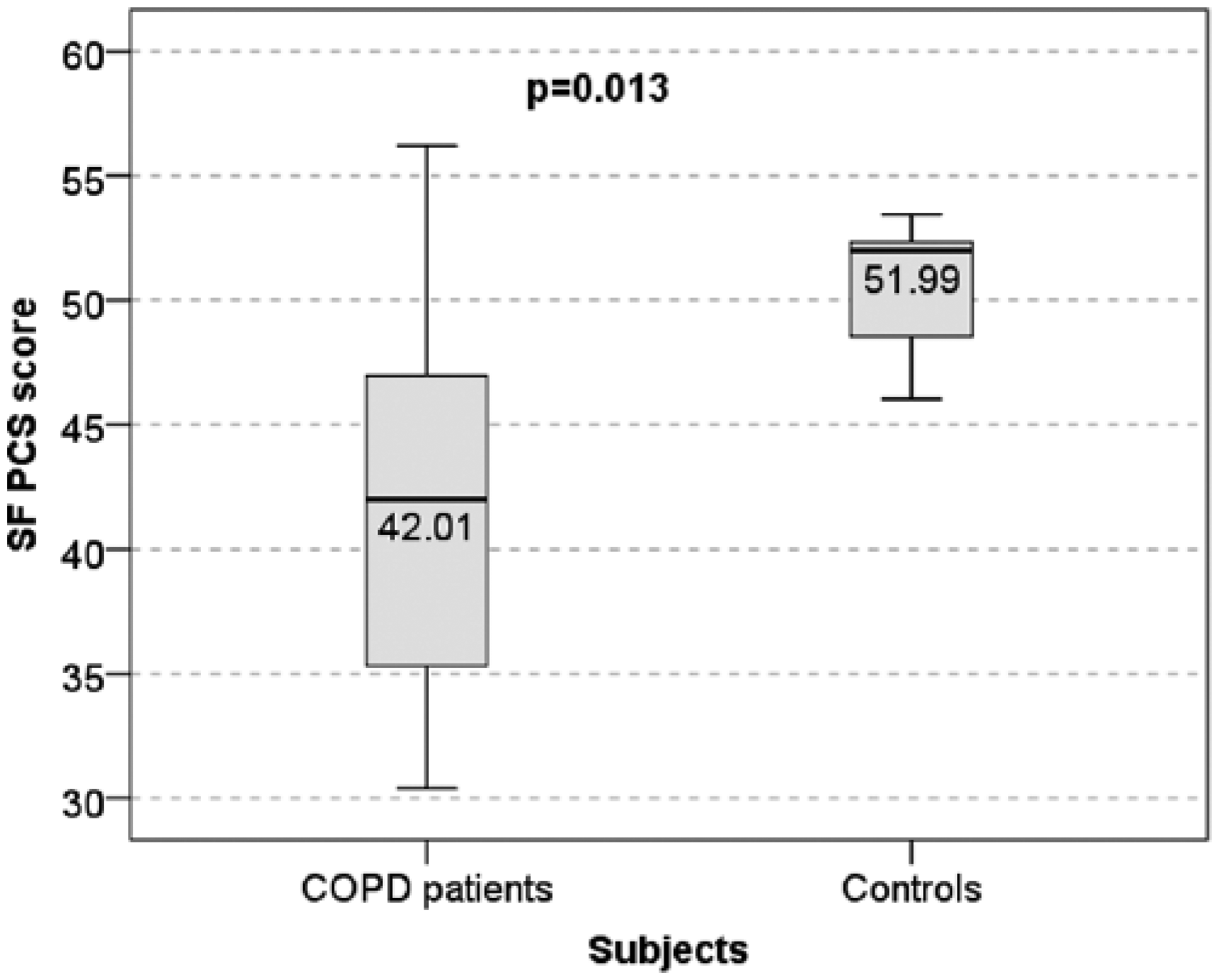
Health-related quality of life (Short Form Health Survey 36 physical component scores) in patients with chronic obstructive pulmonary disease versus healthy subjects (controls).
The FACIT-T score was also found to be significantly lower in the COPD group with a median and interquartile range 74.5 (12.7) versus 95 (24.08); pMW = 0.03. The FACIT-PWB was found to be marginally significant between the COPD group and the healthy subjects (20 ± 4.23 versus 24 ± 2.64; pT = 0.057), the FACIT-FWB was the score with the most significant differences between the groups with median 23.00 (7) in healthy subjects versus 20 (8) in patients with COPD; pMW = 0.026. The other two domain scores FACIT-EWB and FACIT-SWB did not differ significantly between the groups.
Clinically significant fatigue: impact on underlying disease severity
In the COPD group, vitality SF-36 score was found to correlate significantly with the CCQ-T, r = -0.587, 95% confidence interval was between LL = -0.864 and UL = -0.089 (bootstrap method); p = 0.006. In the COPD group 12 (60%) of patients had clinically significant fatigue. Among these 12 patients, seven were ex-smokers and most of them were men (11 subjects). Age or smoking status were not found to differ significantly in the COPD subset with significant fatigue versus the set without.
In the COPD group the presence of clinically significant fatigue was associated with a worse degree of airway obstruction, mean FEV1 %predicted was 47.71% ± 14.92% in patients with clinically significant fatigue versus 65.82% ± 13.99% in the COPD subset without clinically significant fatigue (pT = 0.016) (Figure 3). However, exercise capacity was not found to be significantly worse in the COPD subset with clinically significant fatigue with median 340 m (162 m) versus 389 m (52 m); pMW = 0.147. Physical activity was also evaluated on the SF-PCS scale and significantly worse scores were found in the COPD subset with clinically significant fatigue when compared with the COPD subset without significant symptoms (39.67 ± 17.82 versus 67.5 ± 15.11; pT = 0.002). Significant differences in the SF-36 social functioning scores were also found (48.75 ± 23.87 versus 79.68 ± 17.59; pT = 0.006).
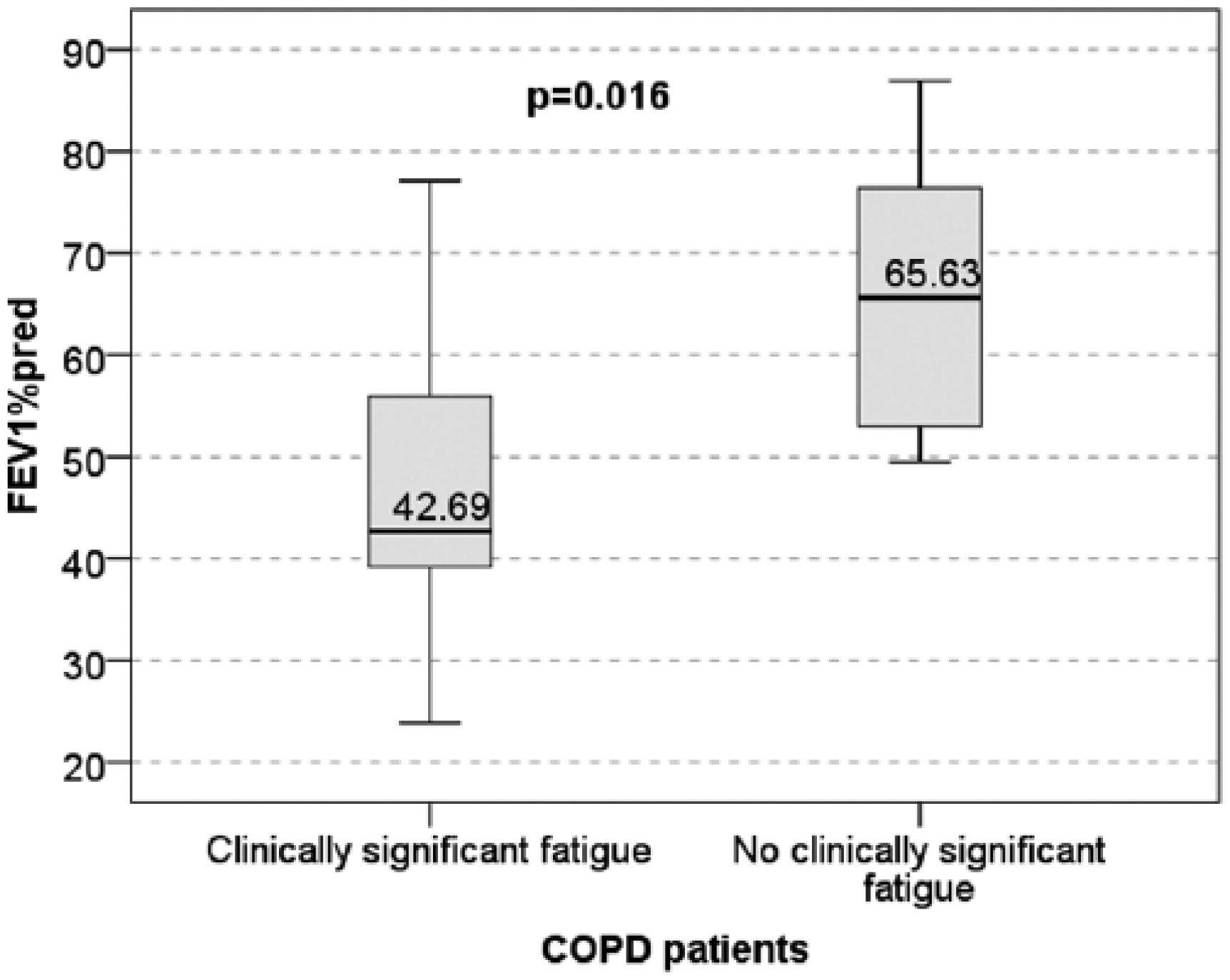
Severity of airways obstruction in relationship to the presence or absence of clinically significant fatigue in patients with chronic obstructive pulmonary disease.
The respiratory symptoms burden assessed with the CCQ-S was also demonstrated to be higher in the COPD subset with clinically significant fatigue 3.75 ± 1.16 versus 2.43 ± 1.05 (pT = 0.019) compared with the COPD subset without clinically significant fatigue.
Surprisingly the score of the CCQ-F was not found to be significantly worse in the COPD subset with clinically significant fatigue and similar results were obtained for CCQ-M scores.
The CCQ-T also differed significantly between the two COPD subsets with median 3.30 (0.60) in the COPD subset with clinically significant fatigue versus 2.11 (0.93) in the COPD subset without clinically significant fatigue (pMW = 0.011) (Figure 4).
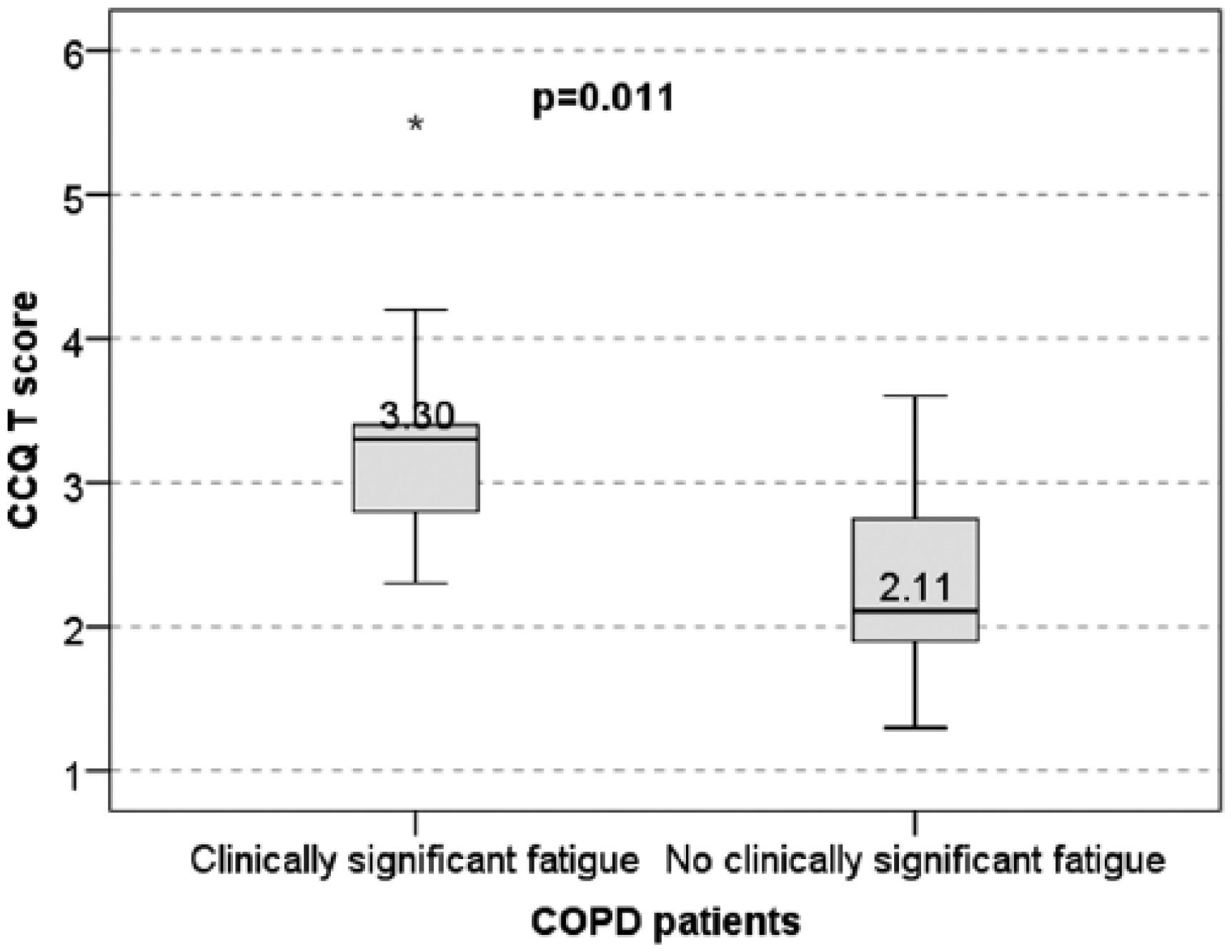
Health status worsening in relationship to the presence or absence of clinically significant fatigue in patients with chronic obstructive pulmonary disease.
Although the severity of effort dyspnoea as measured with the Borg scale at the end of the 6MWT was alsohigher in this category of patients, there was no statistically significant difference when compared with the COPD subset without clinically significant fatigue (median 3 interquartile range 3.3 versus median 3.0 interquartile range 2.8 with pMW = 0.39).
Clinically significant fatigue in COPD patients: systemic inflammation a possible pathogenic explanation?
Finally we wanted to test if systemic inflammation might be a plausible potential explanation for clinically significant fatigue and we compared serum CRP, serum TNF-α and monocytosis in the COPD subsets with and without clinically significant fatigue. Although clinically significant fatigue was associated with higher serum levels of inflammation mediators (CRP and TNF-α) and of monocytosis, the differences between the two COPD subsets were not statistically significant (Table 2).
Variables of systemic inflammation (mean values) in patients with chronic obstructive pulmonary disease and healthy subjects.

SD, standard deviation.
Discussion
In this analysis we demonstrated that patients with COPD with less advanced disease experience fatigue and that this symptom is significantly worse than in the general population. Furthermore, we detected that the presence of clinically significant fatigue was commonly detected in patients with COPD. We also demonstrated that the presence of clinically significant fatigue is associated with a worse health status, a worse level of physical and social functioning and worse chronic respiratory symptoms, and that a potential pathogenic mechanism of this symptom might be related to the presence and expression of systemic inflammation, although this latter finding still requires further validation.
Other studies have tried to evaluate fatigue in COPD in various settings and in relationship to depression, biomarkers of systemic inflammation or the body mass index, airflow obstruction, dyspnoea and exercise capacity (BODE) index. In a study performed on a sample of patients with COPD with a lung impairment comparable to that in the present analysis, fatigue was measured with a ‘disease (COPD)-specific’ questionnaire and serum CRP, TNF-α, receptors 1 and 2, and interleukin-6 were also measured: in a sample of 120 patients fatigue scores were not found to be significantly correlated with the serum levels of these biomarkers. Fatigue levels assessed with the Borg scale before and after exercise testing were found to be weakly correlated to the serum level of this mediator (TNF-α). On the other hand, depression, which was also measured, was found to be predicted by serum TNF-α levels [Al-Shair et al. 2011]. However, this study did not assess the COPD-related health status and did not compare the severity of fatigue with that of the general population as did our exploratory analysis. Our study considered depression analysis in both the COPD and healthy groups but the related scores were comparable and demonstrated no clinically relevant depression so it was decided that this aspect was not worth being discussed in relationship to fatigue. In our study, the level of exertional dyspnoea, although more severe in the COPD subset with significant fatigue, was not significantly different from that of the COPD subset without clinically significant fatigue.
Another more recent study performed by the Evaluation of COPD Longitudinally to Identify Predictive Surrogate Endpoints (ECLIPSE) study team evaluated the appropriateness of a shortened version of the FACIT-F scale for documenting fatigue and its severity in a very large (ECLIPSE) cohort of patients with COPD [Al-Shair et al. 2012]. In this study health status was evaluated with the disease-specific Saint George Respiratory Questionnaire and its scores correlated well with those of fatigue. Fatigue was found to correlate significantly with health status and dyspnoea severity and was worse in patients with poorer exercise capacity and more depressive symptoms [Janssen et al. 2014]. In our study we also found similar correlations for fatigue using the CCQ and also demonstrated that in the COPD subset with clinically significant fatigue, health status (overall or particularly due to symptoms) was significantly worse than in the COPD subset without clinically significant fatigue.
Among the existing studies regarding COPD patients the OLIN study used the SF-36 questionnaire and FACIT-F scale to study fatigue and its impact on health-related quality of life in a large cohort, which included patients with and without COPD. In this study clinically relevant fatigue was defined using the FACIT-F scale, was detected in both COPD and non-COPD patients, was found to worsen the quality of life in both categories of patients, and was also demonstrated to predict mortality in patients with COPD [Stridsman et al. 2015]. In our exploratory analysis clinically significant fatigue was defined using predefined SF-36 vitality-score thresholds and was documented in most of the enrolled patients with COPD. It was associated with a moderate degree of airways obstruction, with worse health status and was not detected in the sample of healthy subjects.
A subsequent analysis performed in the same OLIN sample examined the relationship between fatigue and physical activity, the latter being assessed by the International Physical Activity Questionnaire [Andersson et al. 2015]. A lower level of physical activity was found to be associated with more significant fatigue. In our study physical activity was analysed indirectly with exercise capacity and the physical functioning domain of the SF-36 scale, and was found to be impaired in patients with worse fatigue.
As mentioned above, none of these studies relied on the vitality domain of the SF-36 scale to detect clinically significant fatigue in patients with COPD, and none of the existing studies used healthy subjects as a control population to evaluate the severity and burden of this symptom. This approach can therefore be considered as the main original feature of this analysis and consequently can be used in other, larger cohort analyses.
Fatigue is generally seen as a feature of more advanced COPD and as a potential consequence of hypoxaemia [Antoniu and Ungureanu, 2015]. The aim of our study was to draw attention to the fact that fatigue can develop earlier and in the absence of hypoxaemia, and that its presence or better, its clinically significant severity, might represent a measure of poor disease control. Another interesting issue is the fact that fatigue in COPD can no longer be seen as a simple physical fatigue and once detected should be better documented, from an emotional and social point of view as well, in order to document better its determinants and con-sequently to intervene on therapeutic grounds. Such an approach also needs a repeated evaluation of fatigue at predefined periods of time, especially in order to see which of its domains best parallels the progression of the underlying respiratory disease.
One of the main limitations of our study was the fact that it was not powered to follow up for the dynamics of fatigue or its impact on disease prognosis, but this would indeed be useful in clinical practice: if COPD is viewed as a systemic disease then a general symptom should also be integrated with the existing prognostic indexes in order to characterize better the disease. Prognostic indexes such as the BODE index currently provide a useful tool to foresee the progression of the disease, mostly from a respiratory point of view, and the addition of fatigue might possibly allow a more accurate tool for this purpose.
Based on the results of this preliminary analysis the authors hope to draw attention to the need to study further fatigue and its pathogenic determinants in COPD and the importance of this symptom in the management of this disease, not only when it reaches the most advanced stages.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
