Abstract
A 62-year-old male, who received immune suppressive therapy due to a lung transplantation several years ago, developed multiple painful abscesses in the right forearm. First misdiagnosed as staphylococcal abscesses, Mycobacterium tuberculosis was eventually cultured from the abscesses. In addition, the patient also suffered from pulmonary tuberculosis and respiratory specimens were also culture-positive for Mycobacterium tuberculosis. Cutaneous tuberculosis must be kept as a differential diagnosis in the case of abscess-like lesions on the skin, especially in immunocompromised patients. Mycobacteria specific tests (polymerase chain reaction in respiratory samples and wound smears) and antituberculotic combination therapy are necessary to treat Mycobacterium tuberculosis infection/reactivation adequately.
Introduction
Cutaneous tuberculosis is a rare event especially in a country with low incidence of tuberculosis. Here we discuss the management and pathogenesis of cutaneous tuberculosis of the right forearm in a patient on immune suppression following a double lung transplantation. In addition the patient turned out to have active pulmonary tuberculosis. Cutaneous tuberculosis should be included in the differential diagnosis of patients with skin lesions and immune suppression.
Case description
In June 2012, a 62-year-old Greek male (60 kg, 180 cm, body mass index 18.5 kg/m2) who immigrated to Germany 35 years ago, was referred to the Medical School of Hannover from another hospital with multiple painful abscesses located at the right forearm. Prior to referral, one abscess perforated spontaneously; the others were surgically opened, cleaned and two drainages were inserted (Figure 1). A culture from wound smears grew Staphylococcus hominis and the patient was started on clindamycin and oxacillin. A chest X-ray showed several infiltrates in the upper lobes of both lungs. The patient was on tacrolimus, prednisolone and mycophenolate because of a double lung transplantation in November 2005 due to bullous emphysema. The last presentation of the patient in the interdisciplinary transplant ambulance was in January 2012. At this time, the chest X-ray, bronchoscopy and pulmonary function tests showed no pathologic alteration. The blood level of tacrolimus was considerably elevated (18.7 ng/ml) and therefore a dose reduction was initiated.
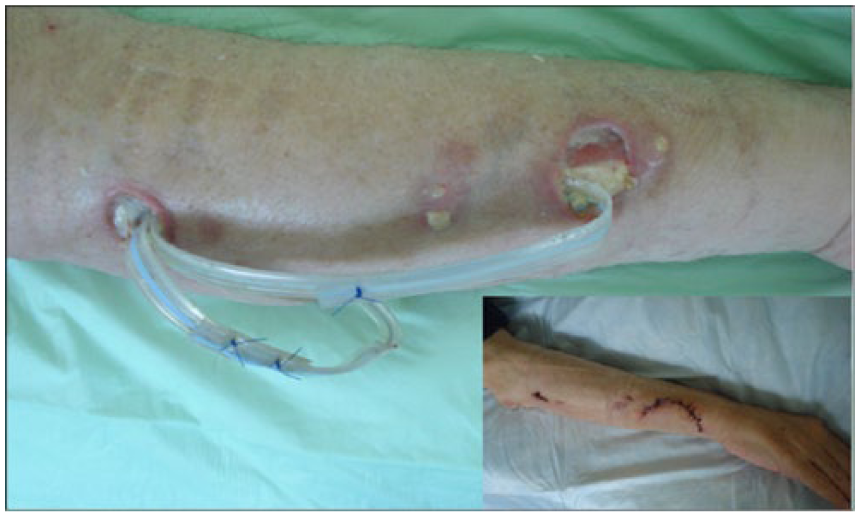
Multiple painful abscesses located at the right forearm of a lung transplant patient. One abscess perforated spontaneously. The others were surgically opened, cleaned and two drainages were inserted.
On admission at our hospital, the patient still complained about severe pain in his right arm. He had noticed a 15 kg loss of weight over the past 4 weeks. The physical examination showed multiple wounds on his right arm, but was otherwise unremarkable (temperature: 37.2°C, breath rate: 14/min, blood pressure: 110/70 mmHg, heart rate: 70 bpm). Blood tests revealed increased C-reactive protein (81 mg/l; reference value <3 mg/l), increased creatinine (311 μmol/l; reference value 59–104 μmol/l) and anemia (7.9 g/dl; reference value 13.5–17.5 g/dl). The other parameters were at normal range. A diagnostic bronchial lavage was performed. Smears were positive for acid fast bacilli and nucleic acid amplification-based techniques showed Mycobacterium tuberculosis complex. An interferon-γ release assay was negative (0.04 U/ml), with the mitogen control just above (0.63 U/ml), the cutoff (0.5 U/ml) indicating lowered immunity. The patient was started on rifampicin, isoniazid, ethambutol and pyrazinamide. Clindamycin and oxacillin were continued.
Over the next days the patient complained of persisting pain in his right arm, which showed progressive inflammation. Smears from the right arm revealed acid fast bacilli, which turned out to be M. tuberculosis complex by nucleic acid amplification-based techniques. Histology demonstrated granulomatous, necrotizing inflammation with epitheloid and giant cells. Clindamycin and oxacillin were stopped.
Under therapy with rifampicin, isoniazid, ethambutol and pyrazinamide the wounds healed. Later, culture of the initial respiratory and the wound specimens grew M. tuberculosis sensitive to rifampicin, ethambutol and pyrazinamide, but resistant to isoniazid and streptomycin. Isoniazid was stopped and the triple combination was continued for 9 months. After secondary wound closure (insert to Figure 1), the patient was discharged in good general condition.
Discussion
Although tuberculosis most often presents as pulmonary disease, in principle it may affect any organ. Tuberculosis of the skin mimics a wide range of differential diagnoses. In this case lesions were initially thought to be due to staphylococcal infection. However, progressive disease and the diagnosis of pulmonary tuberculosis led us to suspect M. tuberculosis as the causative pathogen of cutaneous lesions. Cutaneous tuberculosis may be secondary, either from hematogenous spread or from extension of an underlying focus such as an infected lymph node. Primary tuberculosis of the skin results from direct inoculation of bacilli into the skin [Pomeranz et al. 1995]. In the immunocompromised host, hematogenous spread of M. tuberculosis to the skin may cause a miliary form that consists of small erythematous papules or pustules [Stack et al. 1990]. Due to anergy, skin tests are typically negative [Pomeranz et al. 1995]. It also appears as metastatic tuberculosis abscesses (tuberculosis gummata). Single or multiple subcutaneous nodules develop anywhere on the limbs and trunk, and eventually form undermined ulcer, sinuses or both [Rietbroek et al. 1991]. Gummata are believed to be the product of latent foci of M. tuberculosis that reactivate under various immunosuppressive conditions [Bravo and Gotuzzo, 2007; Semaan et al. 2008]. The skin test is variable [Frankel et al. 2009]. This patient suffered from gumma of the skin on the right arm, which was most likely the result of reactivation of latent disease following immunosuppression after lung transplantation. Another possibility, although less likely, is a self-inoculation of M. tuberculosis by the patient’s own sputum when he covers his mouth with his arm at the moment of coughing. However, this route of infection requires an epithelial damage as M. tuberculosis is not capable of penetrating intact skin.
The cutaneous lesions caused the presenting symptoms of tuberculosis, which later turned out to involve the lung as well. Thus cutaneous tuberculosis should be considered in immunocompromised patients with atypical, nonhealing lesions and a suspected underlying infection.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
