Abstract
Background/objective:
Recurrent hemoptysis is a debilitating complication of cystic fibrosis (CF) and likely results from mucosal erosions into abnormal bronchial blood vessels due to chronic respiratory infection. We hypothesize that the use of beta-blockade will decrease mean arterial pressure resulting in lower bronchial artery blood flow and, subsequently, decrease the frequency and severity of hemoptysis, rate of hospitalizations, and usage of intravenous antibiotics.
Methods:
Retrospective chart review was performed on 12 CF patients with recurrent hemoptysis, aged 13–40 years old, along with a follow-up telephone survey to assess the effectiveness of beta-blockade for hemoptysis, tolerance of inhaled respiratory medications, activity tolerance, and potential adverse effects. A beta-blocker, specifically atenolol, was initiated in all subjects within 24 hours after experiencing recurrent hemoptysis episodes.
Results:
A majority of patients (72.7%) had complete cessation of hemoptysis. There were significant decreases in the frequency of hemoptysis (p = 0.02) and the amount of hemoptysis (p = 0.004). The rate of hospitalizations significantly decreased from 1.33 to 0.67 (p = 0.05) after initiation of atenolol. There was a trend toward statistical significance in the reduction of intravenous antibiotics use (p = 0.08). No statistical difference was found when comparing the pre- and post-treatment means of forced expiratory volume in 1-second (p = 0.59). Very minimal adverse effects were observed with only one patient reporting intermittent facial flushing.
Conclusion:
Beta-blockade, particularly with atenolol, appears to successfully treat, if not resolve, recurrent hemoptysis refractory to conservative therapy in CF. Beta-blocker therapy appears to maintain an effective safety profile in CF.
Introduction
Cystic fibrosis (CF) is an autosomal recessive disease with abnormal CF transmembrane conductance regulator (CFTR). The resulting mucosal airway disease is characterized by dehydration of the airway surface liquid and impaired mucociliary clearance. Complications include airway obstruction, exaggerated inflammatory response, and persistent infections, and long-term complications include hemoptysis, pneumothorax, hyperglycemia, and steady decline in pulmonary function testing, particularly forced expiratory volume in 1-second (FEV1) [Flume et al. 2010].
Hemoptysis is a common complication in the CF disease course. Specific hemodynamic pathophysiology of hemoptysis in CF has not been succinctly delineated. Chronic respiratory infections with bronchiectasis likely result in mucosal erosions into abnormal bronchial blood vessels with resultant hemoptysis. Scant hemoptysis is quantified as less than 5 ml, mild as 5 ml, moderate as 5 ml to 240 ml, and massive hemoptysis as greater than 240 ml per day or significant recurrent hemoptysis interfering with a patient’s lifestyle [Barben et al. 2002; Flume et al. 2005]. One 5-year study demonstrated 9.1% of CF patients developed hemoptysis with approximately half experiencing moderate to massive hemoptysis [Efrati et al. 2008]. A recent study by Flume and associates [Flume et al. 2005] showed that approximately 4.1% of CF patients will suffer massive hemoptysis during their lifetime with an average annual incidence of 0.87%.
Scant to moderate hemoptysis is typically managed with bed rest, intravenous antibiotics for acute pulmonary exacerbations, withholding airway clearance therapies, withholding aerosolized medications, and supplemental vitamin K therapy [Barben et al. 2002; Flume et al. 2010]. The majority of patients show resolution of symptoms after several days of the aforementioned conservative therapy. However, a subset of CF patients may require further intervention including selective bronchial artery embolization (BAE) and, as a last resort, surgical lung resection. The Cystic Fibrosis Pulmonary Guidelines in 2010 advocate BAE in CF patients with massive hemoptysis who are clinically unstable [Flume et al. 2010]. However, no consensus is reported on the management of massive and submassive hemoptysis in clinically stable patients. Recurrent hemoptysis is a debilitating complication of CF which disrupts essential respiratory treatment regimen, affect daily activities, and worsen overall disease state with decline in FEV1 [Antonelli et al. 2002].
Bronchial artery circulation is the source of hemoptysis in CF patients. The bronchopulmonary circulation is a high-pressure system receiving its blood flow from either the aorta, intercostals arteries, or rarely, internal mammary and coronary arteries. The bronchial artery blood supply to the bronchial mucosa and visceral pleura extend through a plexus of communicating vessels, which ultimately terminates in the pulmonary vein [Nicolls and Zamora, 2010]. Refer to Figure 1 which details the complexity and intricacies of the bronchopulmonary circulation [Deffebach et al. 1987]. The bronchial artery blood flow normally accounts for 1–2% of the total pulmonary blood supply. Bronchiectasis, which is very common in CF, is associated with angiogenesis of the bronchial vasculature leading to a significant increase in bronchial blood flow up to 35% of the total cardiac output [Charan et al. 1997]. We speculated that use of chronotropic inhibition and afterload reduction with beta-blockade would decrease mean arterial pressure (MAP) resulting in lower bronchial artery blood flow. We further surmised that a decrease in bronchial artery flow and pressure would result in decreased frequency, if not cessation of hemoptysis. In this study, we report the results of the first documented interventional study of beta-blockade as a therapeutic modality in CF hemoptysis.

Bronchopulmonary circulation. Bronchial artery supply blood to the bronchial mucosa and visceral pleura through a plexus of communicating vessels, which ultimately terminates in the pulmonary vein.
Methods
Our pediatric and adult accredited CF center at Miller Children’s Hospital / Long Beach Memorial located in Long Beach, CA, USA consists of over 210 pediatric and 115 adult patients. A retrospective chart review along with follow-up telephone survey was performed on 12 CF patients, aged 13–40 years old, with recurrent hemoptysis refractory to conservative management. All study subjects were on vitamin K supplementation, either 5 mg twice per day or 10 mg daily, and had normal prothrombin time, international normalized ratio (less than 1.3), and platelet count. The study was approved by the Memorial Health Systems IRB (number 704–11) and informed consents were obtained. One CF patient was unable to be contacted for the follow-up survey and excluded from that portion of the study. Atenolol, a selective β1 receptor antagonist, was initiated at a starting dose of 12.5 mg once daily in all subjects within 24 hours after experiencing recurrent hemoptysis episodes. Recurrent hemoptysis was defined as having at least two separate episodes of hemoptysis within a 12-month period. The dose was subsequently titrated up until cessation of hemoptysis was accomplished. Atenolol was continued daily on the final effective dose. Outcome variables include rate of hospitalizations, use of intravenous antibiotics, and frequency and severity of hemoptysis for 3 months before and after initiation of atenolol. Pulmonary function testing, specifically FEV1, and systolic blood pressure (SBP) were regularly followed based upon clinical follow up (minimum monthly).
A follow-up survey was administered to assess effectiveness of beta-blockade for hemoptysis. Subjects were asked to rate 10 statements regarding hemoptysis, tolerance of inhaled respiratory medications, activity tolerance, and potential adverse effects associated with beta-blocker agents (see Table 1). Ranking of statements 1–5 and 9 as high values and statements 6–8 and 10 as low values suggests positive effects of beta-blocker, and vice versa.
Follow-up survey.

Statistical analysis
Owing to the small sample size, the mean differences of variables between the beta-blocker pre- and post-treatment were unable to meet normative distribution assumptions. Therefore, the nonparametric Wilcoxon signed rank sum test was utilized. Statistical tests were two-sided, and significance was determined at the 0.05 level or less. Analyses were performed using SAS (SAS Institute, Inc., Cary, NC, USA).
Results
Clinical characteristics of the 12 CF study subjects who were treated with beta-blocking agents are shown in Table 2. The average age was 21.2 years old. Seven patients were female (58.3%). Seven patients have homozygous delta F508 mutations (58.3%). Average FEV1 was 53.9% of predicted with a range of 23% to 82%. Hemoptysis events prior to the initiation of a beta-blocker agent were quantified as scant in three patients (25%), mild in six patients (50%), moderate in one patient (8.3%), and massive in two patients (16.7%). After beta-blocker treatment, eight patients (72.7%) had complete resolution of hemoptysis, one patient (9.1%) had scant hemoptysis, and two patients (18.2%) had mild hemoptysis.
Characteristics of 12 cystic fibrosis (CF) subjects with recurrent hemoptysis on beta-blockers.

The majority of subjects (58.3%) did not have recurrent hemoptysis on atenolol 12.5 mg daily. Patients older than 17 years appeared to require slightly higher doses. The highest required dosage of atenolol was 25 mg three times per day in a patient with concomitant systemic hypertension. Overall, average time to resolution of hemoptysis events was 3 days with a range of 1–7 days. One subject with a history of chronic scant to mild hemoptysis and hypertension, concurrently on metoprolol, did not achieve complete resolution of the hemoptysis but had a decrease in both frequency and severity of the episodes. Another patient with chronic hemoptysis despite multiple previous BAEs achieved significant improvement in symptoms after atenolol was deployed.
A significant decrease in hospitalization rates was noted with a mean for the pre-treatment period of 1.33 compared with the post-treatment period of 0.67 with a p-value of 0.05 (see Table 3). A trend toward statistical significance was evident in reduced incidence of intravenous antibiotics use (p-value of 0.08). Although no statistical difference was found comparing the pre- and post-treatment means of FEV1 (p-value of 0.59) and SBP (p-value of 0.39), its clinical significance will be discussed later as this CF patient population is expected to demonstrate marked decline in FEV1 after development of CF hemoptysis, and the lack of such decline represents a net gain [Flume et al. 2005].
Average rate of hospitalizations, intravenous antibiotic usage, FEV1, and SBP.

FEV1, forced expiratory volume in 1 second; IV, intravenous; SBP, systolic blood pressure.
In our study group, there were significant decreases in the frequency of hemoptysis from about twice per week to less than twice per month (p-value of 0.02) and in the amount of hemoptysis from mild hemoptysis to none-scant (p-value 0.004) after initiation of the beta-blocker agent (see Table 4).
Frequency and severity of hemoptysis.
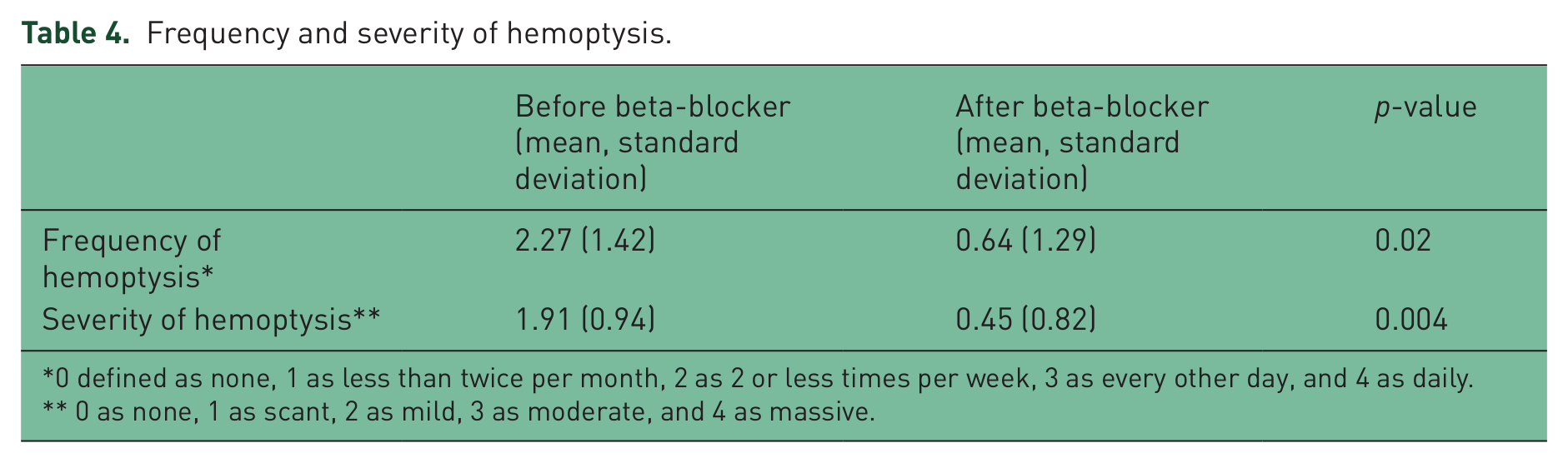
0 defined as none, 1 as less than twice per month, 2 as 2 or less times per week, 3 as every other day, and 4 as daily.
0 as none, 1 as scant, 2 as mild, 3 as moderate, and 4 as massive.
Responses revealed that use of beta-blockade appeared to have a positive effect by significantly diminishing the number of episodes of hemoptysis, decreased amount volume of hemoptysis, and improved tolerance of inhaled respiratory medications. Beta-blockers did not affect degree of coughing during inhaled respiratory medications or the ability to perform daily activities. Subjects ranked statements 6–8 and 10 between 1.55 to 2.18 suggesting that use of beta-blocker did not negatively impact perceived breathlessness, activity of scheduled beta-2 agonist use, or increase frequency of orthostatic symptoms (see Figure 2).
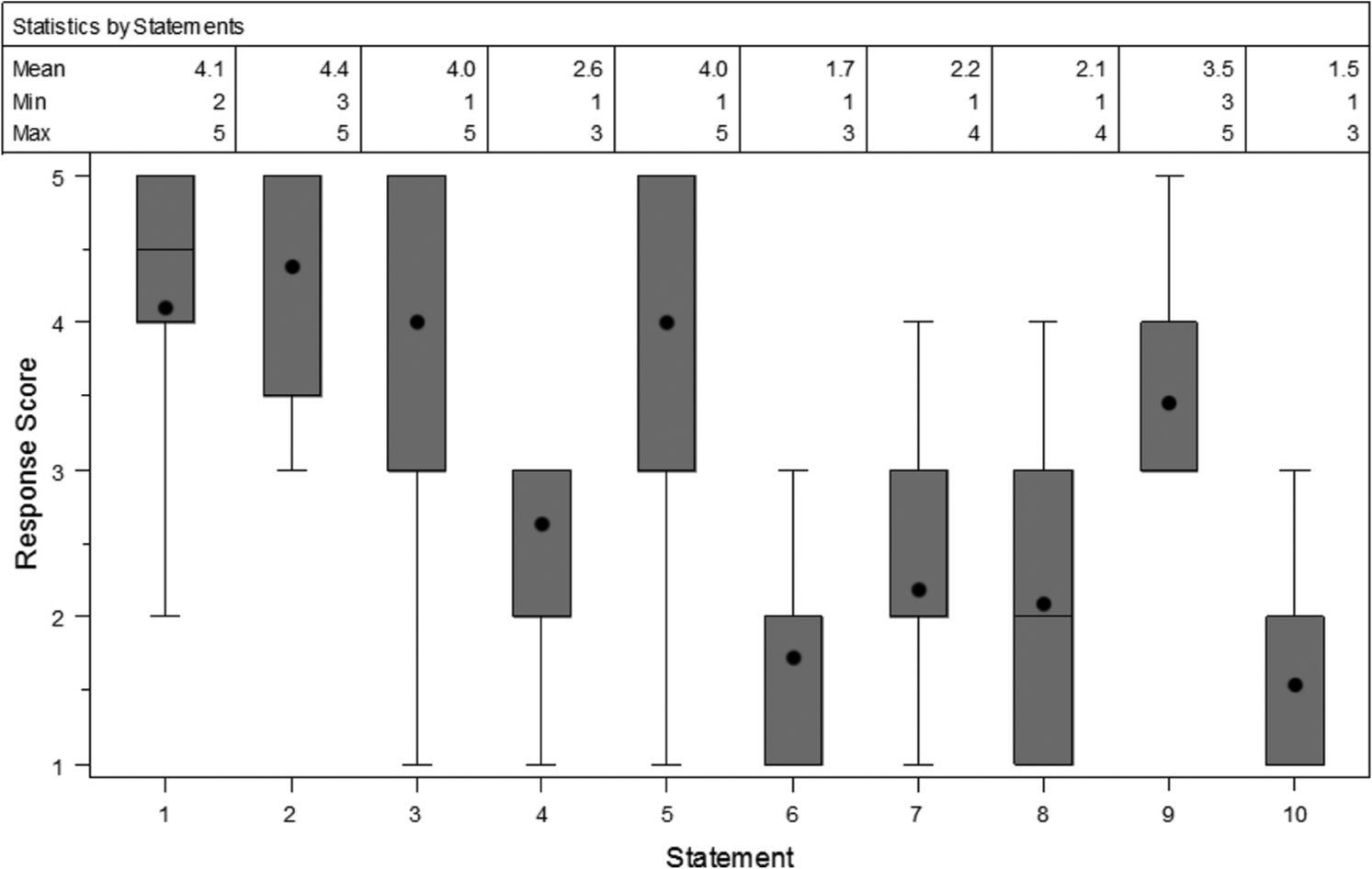
Boxplot of statement responses. Boxes represent the 25th and 75th percentile of statement responses. Mean responses are denoted by a dot within the boxplot, and median by a line through the box plots.
Adverse effects
Very minimal adverse effects were observed with only one patient reporting intermittent facial flushing since starting atenolol. There were no reported episodes of bradycardia, hypotension, syncope, dizziness, bronchospasm, or other potential beta-blocker side effects.
Discussion
Our study demonstrated that beta-blocking agents, specifically atenolol, significantly reduced hospitalization rates and decreased the frequency and severity of refractory hemoptysis in CF patients with chronic hemoptysis. Therapeutic success in this pilot study spanned age, gender, bacterial colonization, and severity of lung function. Atenolol was well tolerated in our study group with very minimal adverse effects. Indeed, our study subjects reported better tolerance of inhaled respiratory medications after starting beta-blockade.
Approximately 90% of massive or refractory hemoptysis originates from the bronchial circulation, due in large part to erosion into the bronchial vessels [Yoon et al. 2002]. Several maintenance daily aerosol medications such as dornase alpha, hypertonic saline, and acetylcysteine aggravate hemoptysis. Therapeutic options for chronic hemoptysis in CF remain limited with BAE as the current mainstay of therapy for significant hemoptysis. The majority of patients (95–97%) who undergo BAE have resolution of hemoptysis within 24 hours [Barben et al. 2002; Vidal et al. 2006]. However, long-term results of BAE reveal a low success rate with 55% of patients undergoing a second BAE at a median time of 4 months with its associated severe complications [Barben et al. 2002]. Sidhu and colleagues reported 75% of such recurrent hemoptysis cases may initiate bleeding from nonbronchial systemic collaterals instead of the commonly suspected bronchial arteries [Sidhu et al. 2008]. The bronchial artery blood flow is determined by the MAP with influences by vasodilatory mediators, vascular remodeling, and vascular proliferation [Charan et al. 2007]. Our study reflects a likely unique aspect of CF hemoptysis whereby bronchial bleeding may not resolve from isolated occlusion of localized bronchial artery circulation. Perhaps a consistent reduction of MAP achieved by beta-blockade in our study overrides irregular bronchial artery dynamics and mimics the localized effect seen early in BAE.
Our study suggests beta-blocker agents in CF specifically decrease MAP resulting in diminished flow and pressure to the bronchial arteries and bronchopulmonary collaterals. Although our data does not reveal significant differences between pre- and post-beta-blocker SBP, specific patients required maintenance of low MAP to maintain cessation of hemoptysis. In addition, a decrease in bronchial artery pressure may precede a measurable decrease in MAP. Thus far, to the best of the authors’ knowledge, no animal or human studies have been published objectively measuring the effect of beta-blockade on bronchial artery flow or pressure.
Beta-blockade may be effective in hemoptysis via other mechanisms. Propranolol, a nonselective beta-blocker agent, has been demonstrated to cause significant reduction in size of infantile hemangiomas within 7 days of initiating therapy. Storch and Hoeger proposed that propranolol works through mechanisms via vasoconstriction, inhibition of angiogenesis, and induction of apoptosis [Storch and Hoeger, 2010]. Vasoconstriction results from propranolol antagonistically binding to beta-2 adrenergic receptors. Subsequent decrease in endothelial nitric oxide synthase function inhibits nitric oxide release. Nonselective beta-blocker also inhibits angiogenesis through downregulation of proangiogenic proteins such as vascular endothelial growth factor and hypoxia-inducible factor-1α. Both selective beta-1 adrenergic and nonselective adrenergic antagonists have been shown to induce apoptosis in cultured endothelial cells, 55% and 67%, respectively [Sommers Smith and Smith, 2002]. Given the nature of immune activation, lymphoproliferation, and bronchiectasis associated angiogenesis in CF, the mechanism of action for beta-blocker therapy is likely multifactorial and remains unclear.
Beta-2 agonists are regularly utilized in maintenance CF therapy. Hence, the utilization of beta-blockade in this population is counterintuitive. Previously, beta-blockers were contraindicated in obstructive lung disease and in diseases with significant airway hyperreactivity component. Beta-blockers were reported to increase airway hyperresponsiveness and may decrease response to beta-receptor agonists [Benson et al. 1978]. Bronchial hyperresponsiveness occurs in 40% of CF patients between ages 4 and 7 years old and 77% between 8 and 18 years old [Sanchez et al. 1993]. Atenolol was specifically utilized in this study since it is a selective beta-1 adrenergic receptor antagonist, thereby minimizing potential effects on the beta-2 adrenergic receptors of the respiratory system. Beta-blockers have been deemed safe in large longitudinal cohort studies in both chronic obstructive pulmonary disease and asthma [Morales et al. 2011; Short et al. 2011]. In our study, no statistical change in FEV1 was observed after initiation of beta-blocker therapy. CF patients with hemoptysis typically show a significant decline in lung function in general and FEV1 in particular. Study subjects did not experience increased dyspnea or feel a reduced effect of beta-2 agonists while on beta-blockade, which is consistent with current literature [Salpeter et al. 2005]. Hence, we surmise atenolol may have attenuated the expected decline of FEV1 by allowing the continued and uninterrupted maintenance of therapeutic CF airway clearance regimens associated with the absence of hemoptysis. Therefore, lack of decline of FEV1 in this CF population actually represents a net gain on relative improvement in FEV1. Although negative impact on the respiratory system was not observed in our study, CF patients who are started on beta-blockers should be monitored closely with pulmonary function testing and serum electrolyte studies due to potential adverse effects.
There are multiple limitations to our study. The small study size limits clinical extrapolation despite achieved statistical significance in primary and secondary outcome variables. The utilization of surveys despite several rounds of questioning inherently allows for recall and interviewer bias. The absence of research on the pharmacodynamics of beta-blockers, specifically on the bronchopulmonary vasculature, and the lack of cardiac catheterization studies along with measurements of vascular endothelial growth factor and hypoxia-inducible factor-1α in our study leaves absent a clear mechanism of action of beta-blocker therapy in CF.
Conclusion
Beta-blockade, particularly with atenolol, appears to successfully treat, if not resolve, recurrent hemoptysis refractory to conservative therapy in CF. Beta-blocker therapy is effective in improving symptoms and appears to maintain an effective safety profile in CF. Beta-blocker therapy may be considered a viable option for CF patients with refractory hemoptysis in lieu of or in conjunction with BAE or surgical lung resection. Molecular, pharmacodynamic, and vascular physiology research is needed to further elucidate the therapeutic mechanism of action described. Although our study findings are promising, larger longitudinal, controlled clinical studies are required to confirm our findings.
Implications
Beta-blocking agents deployed in CF patients with refractory hemoptysis may potentially reduce the need for selective angiography with bronchial artery embolization.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
IRB approval
IRB approval was obtained from Memorial Health Systems IRB with approval number 704-11.
Conflict of interest statement
None declared.
