Abstract
Acute exacerbations of chronic obstructive pulmonary disease (AECOPD) are major contributors to the morbidity and mortality associated with this disease. Current approaches that likely reduce chronic obstructive pulmonary disease (COPD) exacerbations include smoking cessation, influenza and pneumococcal vaccinations, long-acting bronchodilator and inhaled corticosteroid therapy, pulmonary rehabilitation, and mucolytic drugs. However, with optimal treatment using all of these modalities, we are only able to reduce exacerbations by about 40%. A significant proportion of COPD exacerbations are bacterial, therefore long-term antimicrobial therapy could have a role in preventing exacerbations. Long-term antibiotic treatment in COPD regimens that are being evaluated include low-dose macrolide therapy, pulsed fluoroquinolone administration and the use of inhaled antibiotics. Although initial results have been promising with significant reductions in exacerbations with these regimens, additional studies are required to identify the appropriate patient and regimen and elucidate the risk–benefit as well as cost effectiveness of long-term antibiotics in COPD.
Introduction
It is now well recognized that acute exacerbations of chronic obstructive pulmonary disease (AECOPD) are major contributors to its morbidity and mortality. Up to 69% of patients with chronic obstructive pulmonary disease (COPD) will have at least one exacerbation per year. The approximate combined annual cost of COPD in the US is estimated to approach US$30 billion in 2010 [National Heart, Lung and Blood Institute, 2007]. About 50% of the cost is exacerbation related. Apart from the financial implications of COPD exacerbations to the healthcare system, patients with frequent exacerbations have faster lung function decline, prolonged time to recovery of previous health status (especially if the initial exacerbation is followed by another one within 6 months) as well as increased incidence of depression and anxiety [Quint et al. 2008; Spencer and Jones, 2003; Donaldson et al. 2002; Kanner et al. 2001]. Patients with three or more AECOPD per year requiring hospitalization have significantly reduced 5-year survival [Soler-Cataluna et al. 2005].
Current approaches to the prevention of COPD exacerbations include smoking cessation, influenza and pneumococcal vaccinations, long-acting bronchodilator and inhaled corticosteroid therapy, pulmonary rehabilitation, and, potentially, mucolytic drugs [Tashkin, 2010]. However, at this point, with optimal treatment using all of these modalities, we are only able to reduce the number of exacerbations by about 40% [Aaron et al. 2007]. Therefore, development of further AECOPD reduction strategies would be beneficial from both patient quality of life and healthcare cost point of view. In this paper, we review the current evidence for a potential role of long-term antimicrobial treatment in the reduction of exacerbations and for other benefits in the management of COPD.
Rationale for long-term antimicrobial use in COPD
Bacteria cause up to 50% of COPD exacerbations, either as the initial pathogen or as a superinfection after a viral illness [Sethi and Murphy, 2008; Papi et al. 2006]. Compared with both healthy controls and patients with stable COPD, significantly increased bacterial populations are found in bronchoscopic specimens from patients in AECOPD [Rosell et al. 2005]. However, the primary underlying mechanism of bacterial exacerbation is not simply an increase in bacterial concentration in the airway, but is the acquisition of a new strain of bacterial respiratory pathogen from the environment (Figure 1(a)) [Sethi and Murphy, 2008]. Such acquisition is clearly associated with increased symptoms of an exacerbation, a mucosal and systemic inflammatory response and a strain-specific host immune response to the infecting bacterial strain. The leading bacterial pathogen in AECOPD is nontypeable (a) Cycles of infection in COPD. The arrow depicts current administration of short-term antibiotic use for exacerbations. (b) Conceptual benefits of long-term antibiotic use in COPD. The arrows depict the sites of action of long-term antibiotics.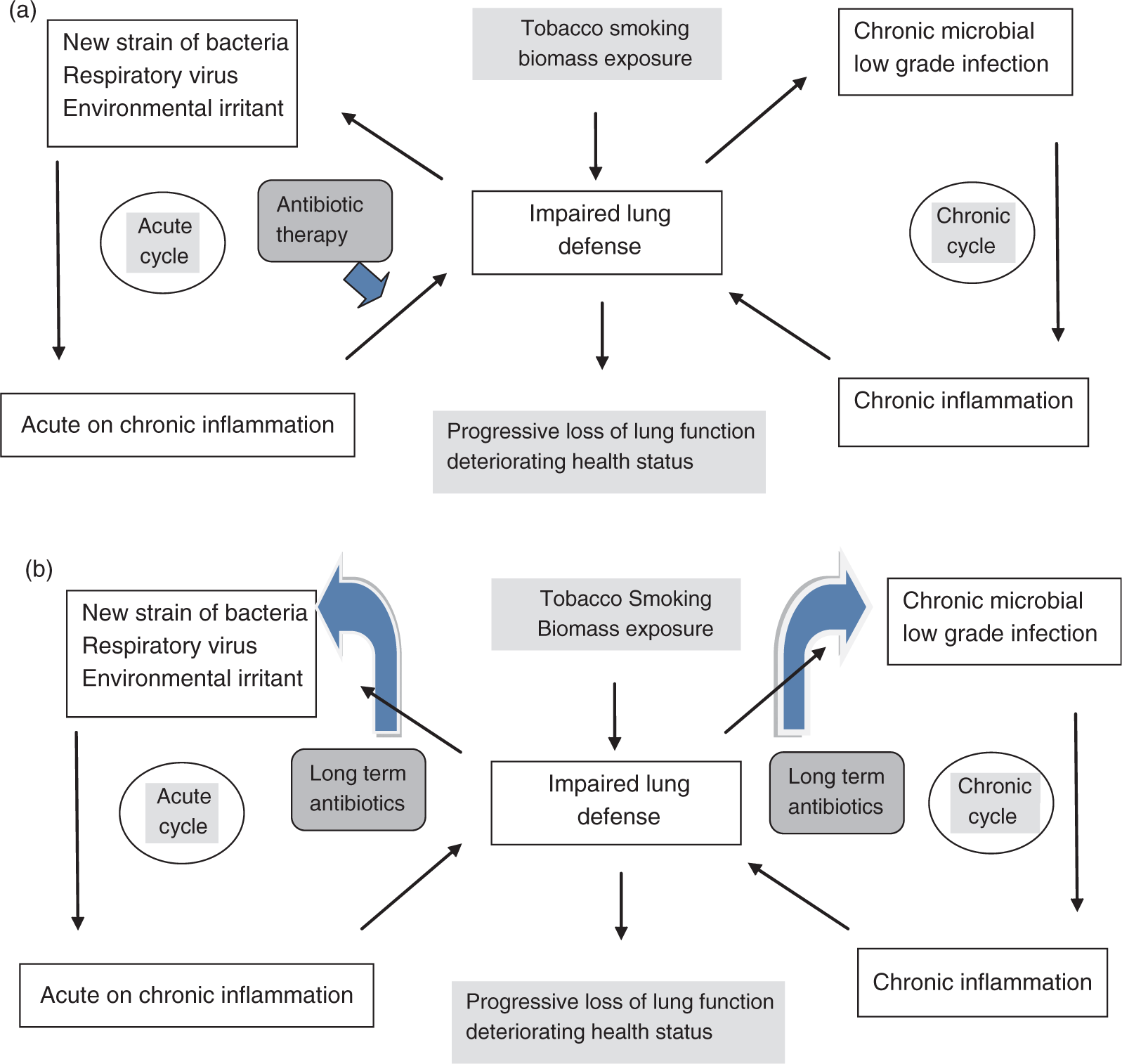
The healthy human lung, because of very effective and redundant innate defense mechanisms, is able to maintain a sterile airway. Disruption of these mechanisms by inhaled insults such as tobacco smoke permits bacterial pathogens to persist in the tracheobronchial tree of patients with stable COPD. Long regarded as innocuous, this abnormal bacterial presence in the lower airway is likely detrimental to the host. Several recent studies of bronchial inflammation in sputum or bronchoalveolar lavage in COPD have shown that it is enhanced with bacterial colonization [Sethi et al. 2008; Banerjee et al. 2004b]. Chronic airway inflammation underlies disease progression in COPD [Hogg et al. 2004]. Therefore, bacterial colonization-associated inflammation could contribute to disease progression in COPD. Conversely, an inflammatory milieu in the COPD airway could further compromise the lung defense mechanisms, making it more hospitable to infection, thereby setting up a vicious cycle of infection and inflammation (Figure 1(a)) [Sethi and Murphy, 2008]. Measures to abort this cycle could be directed at reducing infection or reducing the inflammation.
A significant proportion of COPD exacerbations are bacterial, therefore long-term antimicrobial therapy could have a role in preventing exacerbations (Figure 1(b)). As exacerbations are driven by the acquisition of new strains, prevention would require continuous exposure to antibiotics, so that these new strains cannot establish in the lower airway. However, intermittent antibiotics could also be useful in preventing exacerbations by decreasing the effect of bacterial colonization driven inflammation and making the airway less hospitable to new bacterial strains, by persistence of antibiotics intracellularly in cells such as lung macrophages or because of the postantibiotic effect, which is a period of time after complete removal of an antibiotic during which there is no growth of the target organism.
Cystic fibrosis, diffuse panbronchiolitis and bronchiectasis are conditions similar to COPD whose course and pathogenesis involves chronic airway inflammation, as well as colonization and exacerbations with bacterial pathogens. In these conditions, long-term antimicrobials have been shown to be of benefit, and in the case of the first two, are widely used in practice [Wolter et al. 2002; Tsang et al. 1999; Kudoh et al. 1998; Koh et al. 1997].
Older studies of long-term antibiotics in COPD
Prior to 1970, the British Hypothesis of COPD causation was prevalent, which emphasized the importance of chronic mucus hypersecretion and recurrent infection in COPD. During this time, there were several small trials conducted with long-term antibiotics to prevent COPD exacerbations. Nine of these trials were summarized in a Cochrane systematic review by Black and colleagues [Black et al. 2003]. The total number of subjects in these trials was 1055. Their baseline lung disease was usually not well described, with six of the nine trials including working men with chronic bronchitis. Antibiotics such as oleandomycin, penicillin and oxytetracycline were employed. Antibiotics were used daily for more than 3 months, usually during winter. A small reduction of exacerbations per patient per year was observed with an odds ratio of 0.91 (0.84–0.99) with treatment. There was also a statistically significant reduction in number of disability days per patient per month treated. Days of disability per exacerbation were reduced as well. A small increase in adverse effects with antibiotic use was observed. These studies are now mainly of historical interest, as antibiotics, bacterial pathogens and their susceptibility have evolved in the past 30 years.
New approaches to long term antibiotics in COPD
Owing to studies that did not support the British Hypothesis, and with the identification of tobacco smoking as a major cause of COPD, there was a waning of interest in bacterial infection in COPD and in the use of long-term antibiotics in COPD. Therefore, no studies were done with such treatment for nearly 3 decades. In the last decade, there has been a re-establishment of the important role of bacteria in COPD with studies using new investigative techniques of molecular epidemiology and modern immunology [Sethi and Murphy, 2008]. This has led to renewed interest in long-term antimicrobials in COPD.
The approach of using a penicillin or tetracycline continuously during the winter months for long-term antimicrobial treatment of COPD, as was done in the studies discussed above, is now regarded as a suboptimal approach. Bacterial pathogens implicated in COPD have evolved. For example,
These considerations have prompted new approaches to long-term antibiotic treatment in COPD and other lung diseases that include low-dose macrolide therapy, pulsed fluoroquinolone administration and the use of inhaled antibiotics, each of which is discussed in the following.
Long-term macrolide therapy in COPD
Summary of studies of macrolide antibiotics for prevention of exacerbations of chronic obstructive pulmonary disease.

In 2005 a prospective, double-blind, randomized placebo-controlled study of 67 patients with moderately severe COPD with 3 months of clarithromycin 500 mg daily was reported by Banerjee and colleagues [Banerjee et al. 2005]. These investigators failed to demonstrate benefit in terms of a decrease in sputum bacterial colony counts, exacerbation rate or health status (assessed by St George’s Respiratory Questionnaire [SGRQ]). However, there was a statistically significant improvement in symptom domain of the SGRQ in the clarithromycin arm. Major limitations of this study were the short duration of treatment and a small number of subjects.
Recently, Seemungal and colleagues conducted a single-center, randomized, double-blind, placebo-controlled study of twice-daily erythromycin 250 mg, in patients with moderate to severe COPD for 12 months. Fifty-three patients received the active drug, while 56 patients were randomized to placebo [Seemungal et al. 2008]. Outcome measures were exacerbation frequency and changes in airway and systemic inflammation, assessed using sputum IL-6, IL-8 and myeloperoxidase, and serum C-reactive protein (CRP) and IL-6. Changes in bacterial flora were also studied. There was a significant reduction in amount of moderate to severe exacerbations (requiring antibiotic and/or systemic corticosteroid treatment) with treatment as compared with placebo. Median time to first exacerbation also significantly increased (Figure 2). However, in contrast to the clinical benefits, macrolide treatment was not associated with reduction in any of the inflammatory markers or in decline of lung function by spirometry over the course of the study. The erythromycin treatment was well tolerated with no significant adverse effects. Macrolide resistance was rarely observed with one resistant Kaplan–Meier curves showing the proportion of patients without an exacerbation 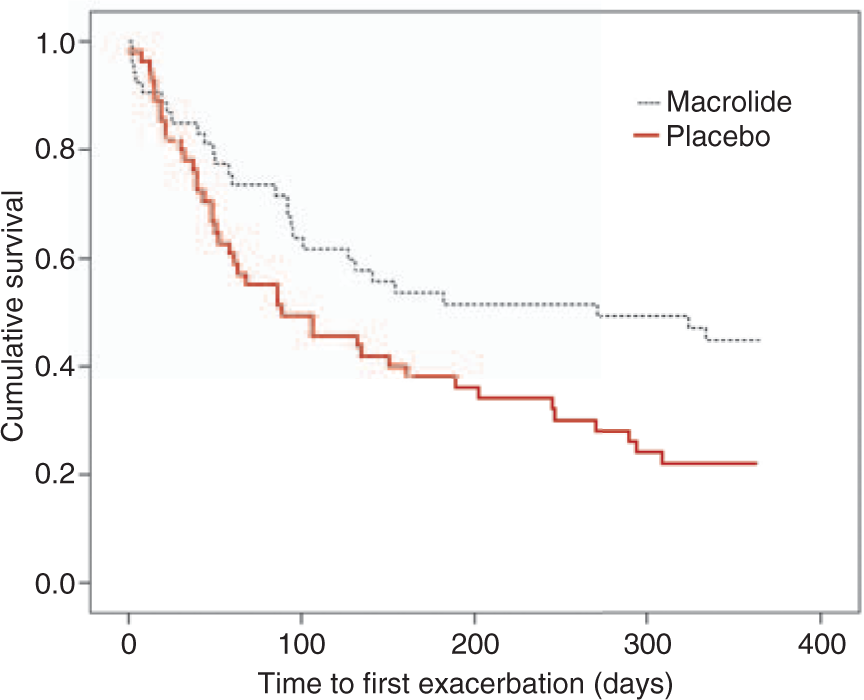
Although these studies are generally supportive of long-term macrolide use in COPD to prevent exacerbations, none of them are large enough or are multicenter studies. Therefore, a definitive study is required. The COPD Clinical Research network in the US is conducting a large, multicenter, randomized, placebo-controlled, double-blind study of azithromycin 250 mg daily for 12 months, with the primary endpoint of time to first exacerbation [ClinicalTrials.gov identifier: NCT00325897]. The results of this study will provide us with the kind of clinical evidence required to consider long-term macrolide use in COPD.
The exact mechanism by which macrolides achieved the beneficial results in COPD discussed above is not clear. At the low doses of erythromycin used in these studies, anti-inflammatory or immunomodulatory effects may be more important than its antimicrobial action. However, there was no decrease in inflammatory markers in these studies [Seemungal et al. 2008; Banerjee et al. 2004a; Suzuki et al. 2001]. Although direct antimicrobial effects are unlikely at low doses, indirect pathways, including altered biofilm synthesis or enhanced phagocytosis and bacterial clearance by alveolar macrophages could provide an antimicrobial action with low-dose macrolides [Peckham, 2002]. The daily dose of azithromycin currently being tested will provide direct antimicrobial activity, and lead to intracellular drug accumulation.
Fluoroquinolone therapy
Fluoroquinolones are extremely effective drugs in treating exacerbations of COPD. They achieve therapeutic concentrations in both the intracellular and extracellular compartments of the lung; achieve high rates of bacterial eradication for most of the major bacterial pathogens that cause exacerbations. In two studies, the GLOBE and the MOSAIC study, a longer time to next exacerbation was seen with a fluoroquinolone used to treat a exacerbation [Wilson et al. 2004, 2002]. In spite of widespread use over the last decade, there has been very little emergence of resistance to this class of drugs among the pathogens implicated in exacerbations of COPD, with the exception of
Based on these concepts, the PULSE study was designed. This was a randomized, double-blind, placebo-controlled, multicenter, international study that evaluated intermittent use of moxifloxacin 400 mg once a day for 5 consecutive days every 8 weeks for total of 48 weeks in the prevention of exacerbations of COPD [Sethi et al. 2010]. Patients included in this study had moderate to severe COPD with chronic bronchitis and at least two exacerbations requiring treatment in 12 months preceding the study, and had to be not colonized with Clinical efficacy of moxifloxacin 
Intensive microbiological monitoring of repeated sputum samples in all patients and rectal swab in a subgroup was conducted in the PULSE study. Only one strain of
The PULSE study demonstrates that intermittent short courses of a potent respiratory fluoroquinolone represent a safe and effective therapeutic option to decrease COPD exacerbations. However, the magnitude of the benefit was not as large as was desired, except in the subgroup with nonmucoid sputum at baseline. One possible reason could be the lower than expected overall frequency of exacerbations in the study. Another potential explanation has emerged in a recent study by Miravitlles and colleagues, in which after a single 5-day course of moxifloxacin in patients with stable COPD, there was a 75% eradication of potentially pathogenic microorganisms at 2 weeks but this effect was lost at 8 weeks [Miravitlles et al. 2009]. This suggests that the 8-week interval chosen in the PULSE study was too long an interval and shorter intervals between antibiotic courses may have been more effective. Although additional studies with different regimens of an oral fluoroquinolone would be desirable, with the evolution of inhaled drug delivery, the emphasis is shifting to the development of inhaled fluoroquinolones as discussed in the following.
Inhaled antibiotics
The inhaled route is the preferred route for all current medications used in COPD, with the exception of theophylline. Inhaled antibiotics have been used successfully in cystic fibrosis, with the available armamentarium continuing to expand [McCoy et al. 2008; Ramsey et al. 1999]. Attractive qualities of inhaled antibiotics include decreased systemic antibiotic exposure and the ability to achieve very high concentrations in the lung. Theoretically, this combination should result in more effective bacterial killing, less resistance emergence in and outside the lung and minimal systemic side effects. There are potential disadvantages to the inhaled route, including bronchospasm, lung tissue damage from local overdose and uneven distribution of the drug.
Summary of studies of inhaled antibiotics in patients with bronchiectasis or chronic obstructive pulmonary disease.

Drobnic and colleagues reported a double-blind, placebo-controlled crossover trial of inhaled tobramycin 300 mg twice daily for 6 months, with a 1-month washout and subsequent placebo for 6 months in 30 patients with radiographically proven bronchiectasis [Drobnic et al. 2005]. In contrast to the previous study, they were not able to demonstrate improvement in number of exacerbations, spirometry or health status assessed with the SGRQ. There was however a statistically significant decrease in number of hospitalizations, as well as decrease in sputum
The only published study in COPD with inhaled antibiotics is with inhaled tobramycin 300 mg twice daily in 13 patients with severe COPD for 14 weeks with a subsequent follow up of 6 months by Dal Negro and colleagues [Dal Negro et al. 2008]. These patients were colonized with multi drug resistant
Several other trials of inhaled fluoroquinolones in COPD are currently underway [ClinicalTrials.gov identifiers: NCT00739648 and NCT01052298]. Results of these trials will dictate our future use of this modality in the treatment and prevention of exacerbations of COPD.
Conclusions
Numerous questions need to be answered prior to routine treatment of patients with COPD with long-term antibiotics. Duration of ‘long term’, route of administration, pulsed
The risks of long-term antibiotic use need to be weighed against the benefits. The major concern is the potential for emergence of multiresistant organisms, both in the lungs and at extrapulmonary locations, for example in the gastrointestinal tract. Studies to date in COPD have shown a reassuringly low rate of resistance development. However, historical precedence is for some extent of resistance emergence with prolonged use and especially with continuous low-dose use, as the high rate of macrolide insusceptibility in cystic fibrosis [Phaff et al. 2006; Kasahara et al. 2005]. However, the clinical relevance of the development of such resistance is unclear. Furthermore, intermittent use and the inhaled route of delivery may result in negligible resistance emergence even with prolonged use. Ultimately, resistance emergence will have to be weighed against the clinical benefit of the treatment in a risk–benefit analysis.
Which subset of patients with COPD is most likely to benefit from long-term antibiotics? The emerging patient profile is a patient with at least moderate COPD who has mucopurulent/purulent sputum production in the stable state, and has frequent (>1) exacerbations per year and poor quality of life, in spite of optimal therapy with inhaled steroids and long-acting bronchodilators. Depending on the results of ongoing and planned studies, it is likely that long-term antibiotic therapy will have a future role in routine clinical practice in subsets of patients with COPD.
Footnotes
Conflict of interest statement
None declared.
