Abstract
Community-acquired pneumonia (CAP) is a leading cause of morbidity and mortality worldwide, affecting approximately 5.6 million patients annually in the USA, where the annual cost exceeds US$12 billion. Optimal management should be based on knowledge of the most likely etiologic pathogens for each patient, based on an assessment of specific risk factors. It is also essential to assess severity of illness, to determine the appropriate site of care, and to order appropriate diagnostic testing. New developments in CAP management have focused on recognizing newly identified pathogens, such as methicillin-resistant Staphylococcus aureus and novel H1N1 influenza, understanding when to utilize new microbiological diagnostic techniques, and how to use biomarkers to direct the appropriate utilization of antibiotics and to define the duration of therapy. This paper reviews recent advances in our knowledge about the diagnosis and optimal management of CAP.
Keywords
Epidemiology
Community-acquired pneumonia (CAP) is a serious clinical problem with associated high mortality and morbidity. It can be potentially life threatening in the elderly and in patients with other comorbid diseases. Its clinical spectrum ranges from rapid resolution of symptoms to severe medical complications and death.
CAP remains a leading cause of death from infectious diseases. Together influenza and pneumonia are the eighth leading cause of death in the USA [Heron et al. 2009]. Approximately 5.6 million cases of pneumonia occur annually in USA [Niederman et al. 1998]. In 2006, 1.2 million people in the USA were hospitalized with pneumonia and 55,477 people died from the disease [Heron et al. 2009]. Various epidemiological studies worldwide have reported the annual incidence of CAP as 5–11 per 1000 population with the incidence rising in elderly patients. In a nationwide study carried out in Germany, the incidence of hospitalized CAP was 2.96 per 1000 population with a high incidence 7.65 per 1000 population in patients over 60 years of age [Ewig et al. 2009]. The estimated yearly cost associated with CAP is more than US$12 billion [Colice et al. 2004]. In a cohort of 1339 CAP patients, 13.5% of patients who received care in the intensive care unit (ICU) accounted for 42.9% of the total hospital costs [Angus et al. 2002].
The major issues in optimal management of CAP are accurately diagnosing if a lung infiltrate is due to pneumonia, identifying the responsible pathogen, and deciding the appropriate treatment. A number of respiratory and infectious disease societies around the world have established guidelines for managing CAP, taking into account local therapeutic factors.
CAP is an alveolar infection that develops in the outpatient setting or within 48 h of admission to a hospital. However, current pneumonia guidelines recommend different treatment for pneumonia patients who are at risk for specific antibiotic-resistant organisms, such as nursing home patients and patients with frequent contact with healthcare (e.g. dialysis, home infusion therapy or recent hospitalization), called healthcare-associated pneumonia (HCAP). They should be treated with broad-spectrum antibiotics similar to hospital-acquired pneumonia and ventilator-associated pneumonia.
There has been an emergence of antibiotic resistance in common etiologic bacteria, the development of new microbiological diagnostic techniques, new understandings in the area of severity assessment, and the development of new antibiotics. This paper reviews recent advances in the diagnosis and management of CAP.
Etiology
Specific pathogens based on epidemiological data.

Certain risk factors make patients susceptible to uncommon organisms. Methicillin-resistant Staphylococcus aureus (MRSA) is recognized both as a nosocomial and, more recently, a community-acquired pathogen, particularly after influenza. Though not common, patients with chronic lung disease, such as bronchiectasis, cystic fibrosis, and chronic obstructive pulmonary disease (COPD), can get infections with Gram-negative organisms, such as Pseudomonas aeruginosa. Others risk factors associated with Pseudomonas infection are corticosteroid therapy ≥10 mg prednisone/day, broad-spectrum antibiotic therapy for ≥7 days in the past month, and malnutrition [Mandell et al. 2007]. Underlying malignancy, cardiovascular disease, and smoking are risk factors for Gram-negative pneumonia [Kang et al. 2008]. In a cohort of 3272 hospitalized CAP patients, history of COPD, current use of corticosteroids, prior antibiotic therapy, tachypnea ≥30/min, and septic shock on admission were associated with Gram-negative bacilli causing pneumonia, and these patients had a significantly longer hospital stay and increased mortality (36% versus 7%; p < 0.001) than those patients without these organisms [Falguera et al. 2009a].
Mucociliary clearance and cough are important in preventing bacteria from reaching the lower respiratory tract. Certain diseases, such as cerebrovascular accidents, esophageal and swallowing disorders, drug overdoses, and neuromuscular disorders, increase the risk for CAP due to aspiration. Gram-negative organisms are the main pathogenic organisms causing aspiration pneumonia in the elderly. Aspiration pneumonia can also be polymicrobial involving Klebsiella spp., Peptostreptococcus, Bacteroides spp., Fusobacterium spp., and Prevotella spp. Alcoholism (daily alcohol intake ≥80 g/day) is an important independent risk factor for severe CAP [Almirall et al. 2008; Ruiz et al. 1999]. Various other risk factors associated with CAP are poor dental health, and active and passive smoking [Almirall et al. 2008].
Viral causes of pneumonia include respiratory syncytial virus (RSV), influenza virus, parainfluenza virus and epidemic viruses, such as coronavirus (which caused the severe acute respiratory syndrome [SARS]), and avian influenza. In a study carried out in Chile, respiratory viruses were the second most common pathogens (32%) isolated in CAP patients [Diaz et al. 2007]. Recently, the use of nucleic acid amplification tests has increased yield to isolate viruses in clinical samples compared with conventional methods such as serology, viral culture, and immunofluorescence. In a study of CAP patients, respiratory viruses were detected with real-time polymerase chain reaction (PCR) assay in 56.2% compared with 14.2% when using conventional methods [Templeton et al. 2005]. Viral pneumonia (SARS and influenza) can lead to respiratory failure. In April 2009, an outbreak of H1N1 influenza infected approximately 61 million people with the death rate as high as 13,000 approximately. H1N1 influenza, in contrast to seasonal flu, affected younger people more than the elderly. The Centers for Disease Control and Prevention estimated that 90% of estimated hospitalizations and 87% of estimated deaths occurred in people younger than 65 years, whereas with seasonal influenza about 60% of flu-related hospitalizations and 90% of flu-related deaths occur in people 65 years and older [CDC, 2010]. Occasionally tuberculosis or fungi can result in severe pneumonia including coccidiomycosis and histoplasmosis mainly in endemic regions.
Clinical presentation: history and physical examination
Clinical presentations of CAP can vary from mild to fatal in severity. In the past, the clinical presentation was classified as being either ‘typical’ or ‘atypical’, but data have shown that this approach is not clinically useful [Lim et al. 2009; Mandell et al. 2007]. CAP can present with various symptoms most commonly fever, chills, cough, purulent sputum, dyspnea, and pleuritic chest pain. Cough is the most common symptom. However, elderly patients may not have classical symptoms and they may present with only confusion, deterioration in functional capacity, and other nonrespiratory findings [Metlay et al. 1997]. One study showed that tachypnea could be the initial presenting sign of pneumonia in elderly patients emphasizing the need to count carefully the respiratory rate in this population [McFadden et al. 1982]. Severely ill patients with CAP may present with hypotension, organ failure, and extrapulmonary infections such as empyema and meningitis.
Pertinent history can sometimes point to the etiology of pneumonia. Travel to endemic areas, sick contacts, exposure to specific pathogens, and risk factors may help point to an etiology (see Table 1). However, the clinical features and history cannot reliably tell the specific etiology of CAP, or separate viral from bacterial infection.
Physical signs of pneumonia are predominantly tachypnea, fever and crackles, rhonchi, or bronchial breath sounds, which can be heard on auscultation. If consolidation is present egophony and dullness to percussion may be heard. Specific signs, such as skin lesions (e.g. chickenpox, measles, blastomycosis), bullous myringitis (Mycoplasma pneumoniae), peripheral manifestations of endocarditis, evidence of congestive heart failure, and phlebitis, can be used to point to specific organisms or to differentiate pneumonia from other diseases.
Recommended diagnostic testing
Radiographic imaging
Inexpensive and readily available plain chest radiograph is recommended to confirm a clinical diagnosis of pneumonia [Mandell et al. 2007]. The presence of a new infiltrate on a chest radiograph along with clinical features is considered the ‘gold standard’ for identifying patients for clinical trials. A chest radiograph can also give more information regarding location, extent of pneumonia, and complications such as cavitation, pleural effusion, multilobar disease, and acute respiratory distress syndrome. However, chest radiographs are of limited value in predicting the causative pathogen [Boersma et al. 2006]. In most cases the plain film findings may be diagnostic of pneumonia and no additional radiographic imaging is required.
Chest radiograph with decubitus views is performed if there is a suspicion of loculated pleural effusion, but ultrasonography has largely replaced the use of decubitus films. Chest ultrasound can be used to evaluate the size of effusion, the free-flowing or loculated nature of the effusion, and to mark the appropriate site for sampling the parapneumonic effusion. Though not widely used for this purpose, ultrasound can also assess the presence of consolidation. Lichtenstein and colleagues compared the diagnosis of alveolar consolidation by computed tomography (CT) scan and ultrasound and found that alveolar consolidation was diagnosed using ultrasound with a sensitivity of 90% and a specificity of 98% [Lichtenstein et al. 2004]. Out of 65 cases of consolidation seen on CT scan, ultrasound was positive in 59. In addition, ultrasound can detect micro-abscesses.
CT scan is rarely used to diagnose CAP but is more sensitive than a chest radiograph. In a study of 47 patients with suspected CAP, high-resolution CT scan was found to be more accurate than chest radiograph in diagnosing CAP [Syrjälä et al. 1998]. A CT scan can give detailed information about nonresolving pneumonia or might help in severely ill patients. It can help diagnose a tumor or foreign body in a patient with suspected postobstructive pneumonia. A CT scan can also give detailed information about cavitation and effusion, but it is not used as a primary modality given its cost and the effects of radiation exposure.
Microbiological testing
Radiography and routine blood tests are neither sensitive nor specific enough to aid decisions regarding therapy in an individual patient. However, a white blood cell count (WBC) may be helpful if it is very low, suggesting neutropenia, which may be a sign of severe pneumonia, or may raise the suspicion of specific pathogens if the patient is known to be immune suppressed.
Microbiological tests are not completely reliable in identifying the etiology of pneumonia, and in 40–70% of pneumonia cases, the etiology is never determined [Restrepo et al. 2010; Dambrava et al. 2008; File et al. 1998]. Some authors have even suggested that because empiric antibiotics work well, isolating a bacterial pathogen in sputum culture is not helpful nor does it exclude the presence of atypical pathogen co-infection. In one study, the authors concluded that investigations for CAP etiology are not cost effective [Theerthakarai et al. 2001]. Mostly, results of microbiological tests are available after therapeutic decisions have been made. The impact of diagnostic testing on CAP is uncertain. Several studies have suggested that establishing etiological diagnosis may not affect the outcome. In one study, the management of CAP with empiric therapy was compared with a pathogen-directed approach. In that study, even with extensive testing, nearly 40% of 262 patients had no etiology established. Although pathogen-directed therapy had no overall impact on mortality or length of stay, it did lead to less adverse events than empiric therapy, and also was accompanied by a lower mortality for patients admitted to the ICU. In patients with severe CAP, diagnostic testing may be valuable for guiding modifications of antibiotic therapy, rather than impacting the choice of initial therapy [Van der Eerden et al. 2005].
Gram's stain of sputum is widely available but its value is controversial. It may, in conjunction with sputum culture, help to identify certain pathogens (e.g. S. pneumoniae, S. aureus, and Gram-negative bacteria) by their characteristic appearance. To be adequate for culture, a sputum sample must have >25 neutrophils and <10 squamous epithelial cells per low-power field. The accuracy of Gram stain depends heavily upon the quality of sample and observer. Previous antibiotics also decrease the yield of Gram stain and culture.
A meta-analysis of several published studies (1966–1993) comparing Gram stain with culture results showed that the sensitivity of Gram stain in pneumococcal pneumonia ranges from 15% to 100%, and its specificity ranges from 11 to 100% [Reed et al. 1996]. However, the sensitivity and specificity was high when infectious disease or pulmonary specialists interpreted the test and a definition of greater than 10 organisms per oil-immersion field was used. Several other studies evaluated the usefulness of Gram stain for detecting S. pneumoniae and reported a sensitivity of 57–70% and a specificity of 79–100% [Musher et al. 2004; Rosón et al. 2000; Lehtomaki et al. 1988].
In the study by Roson and colleagues, in 533 patients hospitalized with CAP, the sensitivity of the Gram stain for the diagnosis of bacteremic pneumococcal pneumonia was 34% and the specificity was 100%, but the sputum Gram stain finding of Gram-negative coccobacilli was highly specific (99%) for H. influenzae pneumonia [Rosón et al. 2000]. However, one third of all patients were unable to give a sputum sample, whereas another third had a poor quality sample. Sputum has also been considered as a low yield diagnostic tool, because of the failure to obtain a sample from most patients, delays in processing of samples in primary care settings, low diagnostic yield, and minimal impact on therapeutic decisions [Ewig et al. 2002].
The Infectious Diseases Society of America/American Thoracic Society (IDSA/ATS) guidelines suggest that more aggressive diagnostic testing should only be performed on the subset of patients with more serious illness (e.g. admitted to the ICU, in whom the causative organism diagnosis is highly uncertain, and outcome may depend on optimal antimicrobial therapy), patients with structural lung disease or pleural effusion [Mandell et al. 2007]. Routine diagnostic testing in outpatients is not recommended but can be helpful either for epidemiologic implications or where treatment will be modified such as in an influenza outbreak (the Gram stain and culture finding of Gram-positive cocci in clusters and MRSA, respectively), in disease caused by agents of bioterrorism, with Legionella spp. infection, community-acquired MRSA infection, M. tuberculosis infection, or endemic fungal infection.
Blood culture and other cultures
Pneumococcal bacteremia is associated with increased mortality [Garcia-Vidal et al. 2008], but most studies have shown that the rate for false-positive blood culture is high and false-positive blood culture results can increase costs, prolong hospital stays, and increase the use of broad-spectrum antibiotics [Corbo et al. 2004; Chalasani et al. 1995]. There is no proven benefit of the finding of positive blood cultures. In one prospective study of 760 patients with CAP, a change of antibiotic therapy based on blood cultures may have improved clinical outcome in only three cases (0.4%) [Campbell et al. 2003]. Owing to the low yield and lesser clinical benefit, the IDSA/ATS guidelines recommend blood cultures only in severe CAP. Certain high-risk patients, including those with neutropenia secondary to pneumonia, asplenia, complement deficiencies, or chronic liver disease, should have blood cultures drawn.
In one study Falguera and colleagues found that the presence of chronic liver disease, pleuritic pain, tachycardia, tachypnea, and systolic hypotension, and the absence of prior antibiotic therapy were predictors of bacteremia in a multivariate analysis [Falguera et al. 2009b]. They concluded that blood cultures should be drawn in patients with the coincidence of two or more of these predictive factors, because they have a substantial risk of bacteremia. These findings confirm the older data of Metersky and colleagues, which showed that bacteremia was more common in patients with no recent antibiotic treatment, but with liver disease, and when vital-sign (i.e. systolic blood pressure, temperature, heart rate) and laboratory (serum sodium, blood urea nitrogen [BUN], and WBC) abnormalities were present [Metersky et al. 2004].
Diagnostic testing to determine etiology has limited value, and should never delay the administration of antimicrobial therapy because delay in treatment increases mortality [Meehan et al. 1997]. In patients at risk for P. aeruginosa and other Gram-negative pathogens, Gram stain and culture of sputum may help exclude the need for empiric coverage.
Diagnostic thoracentesis should be performed for all parapneumonic effusions and fluid sent for cell count, differential, pH, Gram’s stain, and culture. Even though the yield is low, Gram’s stain and culture of aspirates of parapneumonic effusions can impact both antibiotic choice and management decisions [Mandell et al. 2007].
Bronchoscopy to obtain a sample has not proven to be better in detecting common pathogens, however, it may be valuable in patients with severe CAP and in those with suspicion of Pneumocystis jiroveci infections and in patients with Mycobacterium tuberculosis infection.
Laboratory tests to detect M. pneumoniae infection include culture, serology, and PCR. Serology with IgM and IgG antibody becomes elevated in many cases, but the response is often delayed, so the usefulness of these tests for early detection is limited, and their specificity may be low.
Urinary antigen detection assays
Two tests to detect pneumococcal and certain Legionella spp. antigens in urine are available and can detect antigen even after the initiation of appropriate antibiotic therapy. Legionella urinary antigen is very specific and sensitive for culture-confirmed cases of L. pneumophila serogroup 1 disease. In a study carried out by Helbig and colleagues, urinary antigen had a sensitivity of more than 80% in patient culture-positive Legionella infection [Helbig et al. 2003]. However, the urinary antigen assay only detects L. pneumophila (serogroup 1), but this organism accounts for about 80% of sporadic cases of Legionnaires’ disease. The more expensive Legionella PCR detects all serotypes. The use of Legionella urinary antigen to guide therapy choices in CAP may lead to a more complicated course of illness. Falguera and colleagues randomized 177 pneumonia severity index (PSI) class IV and V CAP patients to receive either empiric treatment or targeted treatment based on results of Legionella and pneumococcal urinary antigen tests [Falguera et al. 2010]. The patients who received targeted treatment had higher clinical relapse rate (12% versus 3%, p < 0.04) with no clinical outcome or economical benefit.
The pneumococcal urine antigen test is also quite sensitive and specific (80% and >90%). In a prospective study of 269 patients with CAP and no identifiable pathogen, S. pneumoniae urinary antigen was detected in 69 (27.5%) of those patients [Gutiérrez et al. 2003].
Direct enzyme immunoassay antigen detection performed on respiratory secretions is widely available for diagnoses of influenza A and RSV and is very useful both therapeutically and for isolation procedures.
Prognostic scoring
Severe pneumonia has a high mortality, so a key step in the management of pneumonia is assessing the severity of the illness and deciding where to treat the patient. There are several clinical prediction rules used to determine the mortality risk [Lim et al. 2003; Fine et al. 1997], and those with a high risk of death are admitted to ICU, whereas patients with moderate risk can be managed on a medical floor. Studies have shown that delay in treatment or delay in admission to the ICU leads to high mortality [Restrepo et al. 2010].
Validated clinical prediction tools can help acute care physicians better define the site of care but due to their complicated nature they are underused in clinical practice [Aujesky et al. 2009]. Multiple prediction rules are the IDSA/ATS criteria for severe CAP, and the PSI, the British CURB-65, the Australian SMART-COP, and the Spanish CURXO-80 are also available. These prediction rules vary widely in their ease of use, resource requirements, accuracy, sensitivity, specificity, and positive and negative predictive value.
None of the scoring systems are ideal but the two major tools of severity assessment are the PSI and the CURB score and its modification (CURB-65). In 1997, Fine and colleagues developed the PSI from a database of 14,199 CAP patients hospitalized in 78 hospitals, and then used a separate Medicare database of 38,039 patients to validate the PSI [Fine et al. 1997]. It was also validated in the pneumonia PORT study cohort of 2287 adults. Its goal was to identify low-risk patients who could be discharged home safely and receive outpatient treatment.
The PSI is based on 20 factors that are evaluated at the time of clinical presentation and include three demographic characteristics (i.e. age, sex, and nursing home residence), five coexisting illnesses (i.e. active neoplastic disease, congestive heart failure, cerebrovascular disease, renal disease, and liver disease), five physical examination findings (i.e. pulse rate, respiratory rate, systolic blood pressure, temperature, and mental status), six laboratory measurements (i.e. BUN, glucose, hematocrit, and sodium levels; partial pressure of arterial oxygen; and arterial pH), and one radiographic finding (i.e. pleural effusion). In the original study, patients were grouped into five risk classes for 30-day mortality based on the PSI scores: three with a low risk of 30-day mortality (class I = 0.1–0.4%; class II = 0.6–0.7%, and class III = 0.9–2.8%), a fourth with an increased risk (4–10%), and a fifth with a high risk (27%). Fine and colleagues recommended that all class I patients and many class II and III patients were candidates for outpatient therapy, which could lead to significant cost savings while class IV and V patients, associated with high mortality, should be managed in hospital [Fine et al. 1997]. The PSI score is limited, due to the impact of age on the score, and the possibility of underestimating the severity of illness in younger populations while overestimating the severity in an elderly population and patients with comorbidities [Ewig et al. 2004]. In addition, the PSI is a measure of mortality risk, not of pneumonia severity.
Another score, which is a modified form of the British Thoracic Society rule, the CURB-65, has the benefit of being easy to calculate and simple to use. The CURB-65 score was developed in a study of 1068 prospectively studied patients with CAP from three countries, UK, New Zealand, and the Netherlands. CURB-65 is an acronym based on a six-point score (range 0–5) that gives one point each for: confusion; urea >7 mmol/l; respiratory rate ≥30/min; low blood pressure (systolic blood pressure <90 mmHg or diastolic blood pressure ≤60 mmHg); and age 65 years or more. In the derivation and validation cohorts, the 30-day mortality among patients with 0, 1, or 2 factors was 0.7%, 2.1%, and 9.2% compared with 14.5%, 40%, and 57%, in patients with 3, 4, or 5 factors [Lim et al. 2003]. While CURB-65 is easy to calculate, it lacks formal assessment of vital signs and oxygen level, a major drawback in light of the importance of assessing oxygenation immediately on arrival in the emergency department (ED) [Blot et al. 2007]. Another drawback of any pneumonia severity score is that it only measures severity at the time of hospital admission, and usually serial measurements of severity of illness are necessary to make decisions.
A simpler version of CURB-65, omitting blood urea, so that a decision can be made only with clinical data without the need for laboratory data, showed that CRB-65 was equally effective as CURB-65 and PSI for predicting 30-day mortality [Capelastegui et al. 2006]. However, another study showed that CRB-65 underestimated mortality risk [Bauer et al. 2006]. Chalmers and colleagues provided data from a large population showing that the CRB-65 score can be simplified even more by omitting diastolic blood pressure [Chalmers et al. 2008a].
Various studies have compared the PSI to CURB-65. Capelastegui and colleagues found, in a large single-center Spanish cohort, an equivalence of the predictions made by the PSI, CURB-65, and CRB-65 [Capelastegui et al. 2006] score. However, this study showed that the CURB-65 score had a slightly lower performance in predicting ICU admission and death, probably because it may have given too much weight to the impact of age. In another study, Aujesky and colleagues compared PSI, CURB-65, and CURB for predicting 30-day mortality in 3181 CAP patients and concluded that PSI had higher sensitivity and higher negative predictive value for mortality than CURB and CURB-65 [Aujesky et al. 2005]. The area under the receiver operating characteristic curve was higher for the PSI (0.81) than for either the CURB (0.73) or CURB-65 (0.76) scores (p < 0.001) [Aujesky et al. 2005].
A similar approach has been developed by the Japanese Respiratory Society, the A-DROP scoring system, that assesses: age (male ≥70 years, female ≥75 years); dehydration (BUN ≥210 mg/l); respiratory failure (SaO2 ≤90% or PaO2 ≤60 mmHg); orientation disturbance (confusion); and low blood pressure (systolic blood pressure <90 mmHg) [Miyashita et al. 2006]. Shindo and colleagues compared A-DROP and CURB-65 in CAP patients and found that the sensitivity, specificity, and 30-day mortality predictive value of the A-DROP scoring tool were equivalent to the CURB-65 [Shindo et al. 2008].
Prognostic scores.

BP, blood pressure; BUN, blood urea nitrogen; ICU, intensive care unit.
Rello and colleagues developed the CAP-PIRO score based on a tool used to predict the risk for sepsis. This score evaluates variables related to the PIRO score that include: predisposition, infection, response, and organ dysfunction (Table 2). Considering the observed mortality from each PIRO score, the patients were stratified into four levels of risk: (a) low, 0–2 points; (b) mild, 3 points; (c) high, 4 points; and (d) very high, 5–8 points. This score was able to predict consistently the ICU mortality and healthcare use in a cohort of 529 patients admitted to the ICU with CAP [Rello et al. 2009].
España and colleagues developed a scoring system for recognizing patients requiring the ICU (the SCAP score) [España et al. 2006]. The need for ICU admission could be defined by the presence of one of two major criteria (arterial pH <7.39 or a systolic blood pressure below 90 mmHg), or the presence of two out of six minor criteria, which included confusion, BUN >1.7 mmol/l, respiratory rate >30/min, PaO2/FiO2 ration <250, multilobar infiltrates, and age >80 years. España and colleagues compared PSI, CURB-65, and their SCAP score as predictors of 30-day mortality and showed that the PSI and CURB-65 had higher sensitivities than their score, while the SCAP score had higher specificity [España et al. 2010]. Patients identified as low risk based on the SCAP score had lower ICU admission rates than patients identified as low risk based on CURB-65 and PSI in both cohorts. When severity criteria were met, the tool was 92% sensitive for identifying those with severe CAP and was more accurate than the PSI or CURB-65.
Another prospective study compared SCAP, PSI, and CURB-65 in Spain for the assessment of adverse outcomes, such as ICU admission, development of severe sepsis, need for mechanical ventilation, and therapeutic failure. The authors concluded that the SCAP score had a higher sensitivity, specificity, and area under curve (AUC) than the PSI and CURB-65. The SCAP score was shown to be slightly more accurate than the PSI and CURB-65 [España et al. 2009].
The IDSA/ATS 2007 CAP guidelines recommend ICU admission for patients with severe CAP, defined as the presence of one of two major criteria (i.e. the need for invasive mechanical ventilation or vasopressors) or three out of nine minor criteria.
The IDSA/ATS criteria for severe community-acquired pneumonia:
Minor criteria
Respiratory rate ≥30 breaths/min PaO2/FiO2 ratio ≤250 Multilobar infiltrates Confusion/disorientation Uremia (BUN level ≥1.1 mmol/l) Leukopenia (WBC <4000 cells/mm3) Thrombocytopenia (platelet count <100,000 cells/mm3
Hypothermia (core temperature <36°C) Hypotension requiring aggressive fluid resuscitation
Major criteria
Invasive mechanical ventilation Septic shock with the need for vasopressors
Several studies have validated these and prior versions of the ATS criteria for severe CAP [Phua et al. 2009; Valencia et al. 2007; Ewig et al. 2004]. A study carried out in 457 PSI class V patients showed that the modified ATS severity rule was more specific than the CURB score in predicting ICU admission. The ATS rule had the best results in predicting ICU admission and a very high negative predictive value (91%), and the CURB score had the best sensitivity for mortality [Valencia et al. 2007]. The ATS criteria have been shown to have the best accuracy in predicting the need for ICU admission. However, the need for ICU care for patients meeting only the minor criteria is uncertain.
Phua and colleagues carried out a validation study in Singapore and showed that the ATS minor criteria for severity of CAP had greater discriminatory power in the prediction of severity, ICU admission, and mortality than the PSI and CURB [Phua et al. 2009]. In another validation study, the ATS criteria had a sensitivity of 71% and specificity of 88% for predicting ICU admission and for patients meeting only minority severity criteria, there was no reduced mortality from ICU admission [Liapikou et al. 2009]. In a retrospective cohort, the ATS criteria performed significantly better than CURB-65, SMART-COP, and CURXO-80, with an AUC of 0.88 (95% confidence interval [CI] 0.85–0.90). By using four rather than three minor criteria, the positive predictive value of ATS criteria increased from 54 to 81% [Brown et al. 2009]
Biomarkers
In addition to the above severity prediction tools, several biomarkers have been recognized as markers of severe inflammation in CAP such as procalcitonin (PCT), C-reactive protein (CRP), and pro-adrenomedullin, and all are being evaluated as tools for predicting host response and prognosis in severe CAP. Elevated levels of pro-adrenomedullin, copeptin, natriuretic peptides, cortisol, pro-atrial natriuretic peptide and coagulation markers are significantly related to mortality in CAP. The two main biomarkers currently available are the CRP and PCT. Both are acute phase reactants with low circulating levels normally, rising with inflammatory disease, particularly bacterial.
PCT is a peptide precursor of the calcitonin hormone released in response to bacterial toxins, and its levels are elevated in patients with bacterial infections whereas the levels are low in patients with viral infections [Christ-Crain et al. 2004; Assicot et al. 1993]. Several studies have shown that biomarkers have a high negative predictive value for ruling out bacterial infections. Hence, PCT can be used to assess the necessity of antibiotic therapy. Several studies have shown that a PCT-guided strategy to treat bacterial infections can help reduce antibiotic exposure with no apparent adverse outcomes [Bouadma et al. 2010; Christ-Crain et al. 2004]. Using the highly sensitive Kryptor assay, a PCT level of <0.1 µg/l suggests the absence of bacterial infection, 0.1–0.25 µg/l indicates that a bacterial infection is unlikely, 0.26–0.5 µg/l indicates a possible bacterial infection, and >0.5 g/l strongly suggests the presence of bacterial infection [Christ-Crain and Müller, 2007]. In another study, carried out in 925 patients in Switzerland among patients with a PCT value <0.1 µg/l and 0.1–0.25 µg/l, only 0.9% (1 out of 117) and 0.9% (2 out of 224) had positive blood cultures, while 16.8% (61 out of 364) had positive results with PCT levels >1.0 µg/l. The authors concluded that PCT level in the ED accurately predicted bacteremia in CAP patients [Müller et al. 2010].
Other studies have shown that PCT levels correlate with the severity of pneumonia. Patients with a higher PSI score or with complications or death had significantly higher PCT levels than those with an uncomplicated clinical course [Masia et al. 2005]. In the CAPNETZ study, Krüger and colleagues reported that PCT had comparable predictive potential as CRB-65 [Krüger et al. 2008]. Nonsurvivors had significantly higher median PCT levels than survivors (1.2 versus 0.3 mmol/l; p = 0.0001). Low PCT accurately predicted patients at very low risk of death even in patients with high CRB-65. Given its high negative predictive potential (98.9% with PCT level of <0.6 mmol/l), patients with low PCT might be safely treated as outpatients [Krüger et al. 2008]. Huang and colleagues found that 23.1% (126 out of 546) of high-risk patients defined by PSI had low PCT and this subgroup had very low mortality similar to low-risk patients [Huang et al. 2008]. Serial measurements of PCT have also been used to define prognosis in severe CAP patients. A recent study published by the German CAPNETZ group showed that levels of precursor fragments of atrial natriuretic peptide (ANP) and atrial vasopressin, midregional pro-atrial natriuretic peptide (MR-proANP), and C-terminal pro-atrial vasopressin (CT-proAVP; copeptin) increased with increasing severity of CAP. This study also showed that CRP, WBC and PCT levels were associated with increasing pneumonia severity, however, the AUCs for short-term mortality of CT-proAVP (0.84, 95% CI 0.82–0.86) and MR-proANP (0.81, 95% CI 0.79–0.83) were significantly higher than those of PCT (0.76, 95% CI 0.73–0.78; p < 0.02), CRB-65 (0.74, 95% CI 0.71–0.76, p < 0.02), CRP (0.67, 95% CI 0.65–0.69, p < 0.001) and WBC (0.60, 95% CI 0.57–0.62, p < 0.001). The authors concluded that the MR-proANP and CT-proAVP were significantly better predictors for 28-day and 180-day mortality due to their high negative predictive value [Krüger et al. 2010].
CRP is an acute-phase reactant, and CRP measurements are frequently used to aid in the diagnosis of bacterial infections. CRP is synthesized by the liver, mainly in response to interleukin-6, which is produced during infection and inflammation. Almirall and colleagues showed that high plasma levels of CRP are more common when the pathogens involved are S. pneumoniae and L. pneumophila or when the illness is more severe [Almirall et al. 2004]. Thus higher levels can be used as a surrogate for severity and hence aid in deciding whether hospitalization is necessary. The initial concentration of CRP does not correlate closely with the severity of disease, but it can be used as an indicator of response to treatment. A persistent high or rising CRP level suggests treatment failure or complications [Smith et al. 1995].
A larger study of 570 patients with CAP, enrolled over 2 years, showed that admission CRP <100 mg/l has a high negative predictive value. CRP levels <100 mg/l indicated reduced risk, whereas failure of CRP to fall by 50% or more at day 4, was associated with increased risk for 30-day mortality, need for mechanical ventilation, and/or inotropic support, and complicated pneumonia [Chalmers et al. 2008b].
A recent meta-analysis of 110 studies concluded that PCT was more accurate than CRP in the distinction between viral and bacterial infections. CRP was only 75% sensitive compared with PCT levels with a sensitivity of 88%, and CRP was also less specific (67%) than PCT (81%) in differentiating bacterial from noninfective causes of inflammation [Simon et al. 2004]. Christ-Crain and colleagues in their study of cortisol levels reported that CRP could not differentiate between different severities of CAP, as defined by the PSI and was not a significant independent predictor of outcome [Christ-Crain et al. 2007].
High cortisol levels have also been investigated as a marker of severity of CAP. In a single-center study of 72 patients, significantly higher total baseline cortisol levels were observed in nonsurvivors than survivors (745 nmol/l versus 489 nmol/l; p = 0.003) [Salluh et al. 2008]. In another study of 278 patients, free and total cortisol levels correlated with severity of illness, as reflected by the PSI score, with a total cortisol level above 960 nmol/l having a sensitivity of 75% and a specificity of 71.7% for predicting mortality [Christ-Crain et al. 2007]. However, in the presence of sepsis, cortisol levels may be unreliable especially in patients with relative adrenal insufficiency due to sepsis. Also cortisol levels can have variable results during the day.
In conclusion, biomarkers can never be diagnostic on their own and can only be interpreted along with other clinical and pathologic data. Given the difficulties in choosing between viral and bacterial pathogens based on clinical assessment, biomarkers together with clinical parameters can aid clinicians in assessing the severity of the illness and the need for the use of antibiotics. Biomarkers can help to differentiate patients with pneumonia from heart failure and COPD exacerbation, with the latter not requiring antibiotics. Another advantage of biomarkers is that serial measurements can be used to assess the treatment response.
In the future, biological markers may become a part of routine diagnostic testing for CAP. It has been suggested that in future CAP trials, patients with low PCT levels should be excluded as these patients could dilute any beneficial effect that occurred in other patient subsets as patients with low PCT are unlikely to benefit from antibiotic therapy [Niederman, 2008].
Using available data to define the site of care
Despite the use of modern-day antibiotics and management strategies, mortality from pneumonia remains significantly high. A key decision for the physician after diagnosis of pneumonia is whether to treat the patient as an outpatient, in the hospital or ICU.
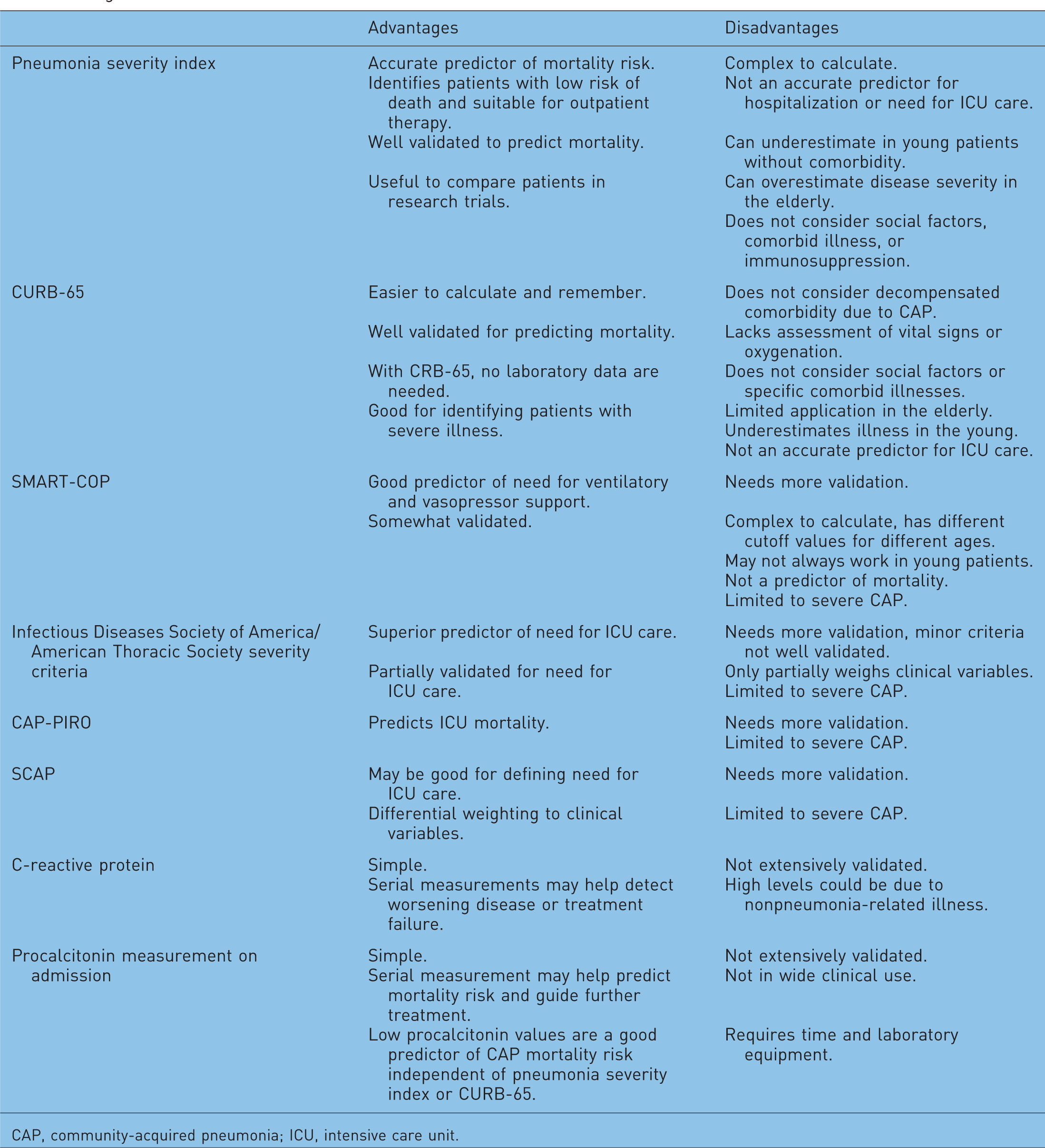
In the USA, 80% of patients with CAP are treated as outpatients, and 20% are treated as inpatients [Colice et al. 2004]. The majority of inpatients are handled in a hospital ward, and 10% of those who are admitted to the hospital are treated in the ICU. Accurate prognostication will allow physicians to determine the most appropriate site of treatment (home versus hospital), the intensity of hospital management (medical floor versus ICU), and the intensity of diagnostic testing and antibiotic therapy. Clinical judgment alone can be misleading in estimating disease severity, thereby leading to under or overestimation of the severity of CAP. A simple algorithm can aid in deciding the site of care (see Figure 1). Prognostic scores help clinicians to predict severity and manage patients effectively and avoid unnecessary risks and costs. Advantages and disadvantages of various scoring systems and biomarkers are listed in Table 3.
Algorithm for decision on site of care. Prognostic score. CAP, community-acquired pneumonia; ICU, intensive care unit.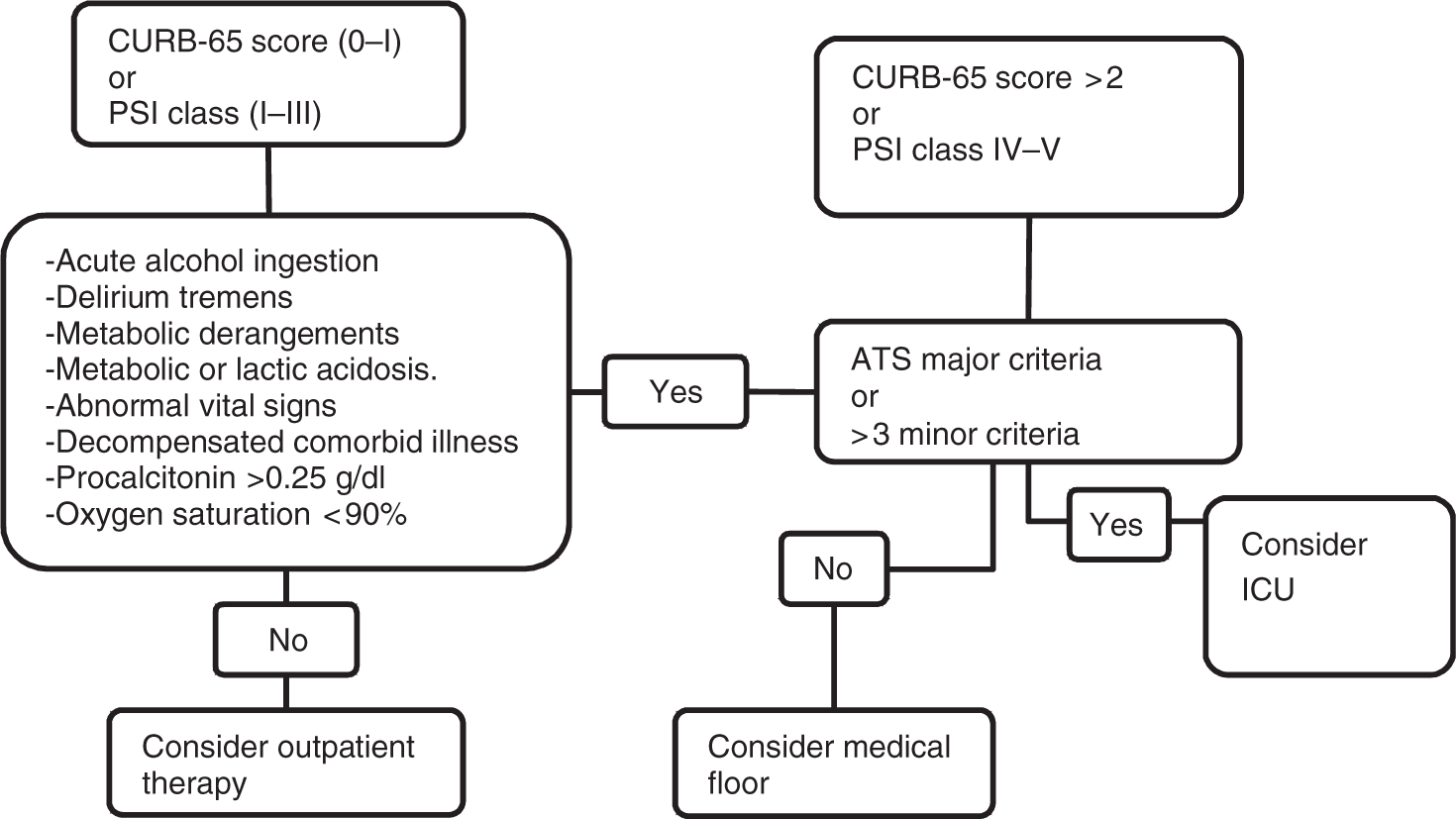
There are large cost differences associated with the site of treatment, and thus increasing outpatient management of low-risk patients is one approach to improve the cost efficiency of care for this illness [Niederman et al. 1998]. In the ICU, nonsurvivors had higher daily costs of (US$2168 versus US$1343) than survivors; p < 0.001. Total hospital costs were US$35,346 for ICU nonsurvivors and US$20,347 for ICU survivors [Angus et al. 2002]. Current guidelines recommend the hospitalization of patients with an increased risk for adverse medical outcome, and to discharge patients as soon as clinical stabilization is documented. In USA the total annual inpatient treatment cost for CAP is US$10.3 billion compared with the total outpatient treatment cost for CAP of US$1.9 billion [Colice et al. 2004].
Patients with low PSI scores (class I, II, or III) have lower 30-day mortality and they can often be safely treated as outpatients, whereas class IV or V have higher mortality and usually require hospitalization [Restrepo et al. 2008; Fine et al. 1997]. Carratalla and colleagues randomly assigned 224 adults with CAP in the low-risk PSI classes II or III to outpatient treatment or to inpatient care [Carratalla et al. 2005]. An overall successful outcome was observed in 83.6% of outpatients and 80.7% of hospitalized patients.
Most prognostic tools only assess 30-day mortality, however CAP is associated with increased long-term mortality. Delays in recognizing severe forms of CAP can increase mortality. Restrepo and colleagues showed that delay in admission to ICU is associated with high mortality [Restrepo et al. 2010]. In this study of 161 patients, 19 patients who were admitted to the ICU after 24 h of admission had a 30-day mortality of 47% versus 23% of patients admitted to the ICU within 24 h (p = 0.02). In another study, after severity adjustment, delayed ICU transfer was associated with a higher 28-day mortality rate (23.4% versus 11.7%; p < 0.02), and longer median hospital length of stay (13 days versus 7 days; p < 0.001) [Renaud et al. 2009]. In a study of 1339 patients, the principal reasons for ICU admission were respiratory failure (57%), hemodynamic monitoring (32%), shock (16%), and 27% of the ICU admissions were in PSI classes I–III [Angus et al. 2002].
Some low-risk patients may have medical and psychosocial contraindications to outpatient therapy. Patient preference, ability to maintain oral intake, history of substance abuse, cognitive impairment, patient functional status, and complications of pneumonia are other factors that may alter decision making. Aujesky and colleagues showed that ED physicians admitted 258 (37.4%) of 689 low-risk patients and treated 20 (3.2%) of 617 higher risk patients as outpatients [Aujesky et al. 2009]. The low-risk patients were admitted because of the presence of a comorbid illness (71.5%) laboratory value, vital sign, or symptom that precluded ED discharge (29.3%); or a decision of a primary care or consulting physician (19.3%).
Treatment
Timing of antibiotics
Delayed antibiotic therapy has been associated with increased risk of death. Therefore, a correct and rapid diagnosis is mandatory. Two major studies have suggested that early diagnosis and early antibiotic administration are associated with improved survival and decreased length of stay in CAP [Houck et al. 2004; Meehan et al. 1997]. The US Centers for Medicare and Medicaid Services use the timing of first administration of antibiotics to patients admitted for pneumonia as a quality core measure for public reporting. In 2004, the Centers for Medicare and Medicaid Services adopted a 4-h antibiotic rule as a quality measure. The 4-h cutoff time for initial antibiotic administration as a quality core measure was questioned in some studies [Welker et al. 2008; Kanwar et al. 2007]. Kanwar and colleagues compared hospitalized CAP patients before and after the 4-h rule and found that endorsement of the 4-h rule led to increased use of antibiotics [Kanwar et al. 2007]. After the 4-h rule, more patients were diagnosed as CAP despite normal radiographs (28.5% versus 20.6%; p = 0.04), and the proportion of patients from the cohort with a final diagnosis of CAP decreased from 76% to 59% (p < 0.001). Ferrer and colleagues in a prospective observational study showed that early administration of broad-spectrum antibiotics in severe sepsis is associated with lower hospital mortality [Ferrer et al. 2009]. Pneumonia was most common infection in this study (36.9 %). The most recent IDSA/ATS CAP treatment guidelines state that the first dose of therapy should be given in the ED (preferably within 6–8 h of arrival to the ED), but did not designate a specific time threshold [Mandell et al. 2007]. Giving the first dose in the ED rather than on arrival at the hospital ward is associated with more rapid time to first dose of antibiotic and shorter length of hospital stay. Data from a study concluded that ICU patients compared with ward patients were more likely to receive antibiotic therapy within 4 h of ICU admission (40% versus 25%; p < 0.001), and to undergo appropriate blood culture collection (83% versus 74%; p = 0.03), but were less likely to receive guideline-concordant antibiotic therapy (67% versus 82%; p < 0.001) [Restrepo et al. 2008].
Antibiotic treatment
Due to the lack of an ideal etiological test that is rapid, sensitive and specific, physicians must start empiric antibiotic therapy. An empiric therapy is typically based on the likelihood that a group of pathogens is most likely responsible. The selection of antimicrobial regimens for empiric therapy is based on a number of factors, including the risk factors for antimicrobial resistance and medical comorbidities, which may influence the likelihood of a specific pathogen and may be a risk factor for treatment failure. Discussion of empiric therapy can be found in guidelines [Lim et al. 2009; Mandell et al. 2007]. Information continues to evolve about the optimal therapy of CAP. One population that is being extensively studied is that of HCAP. This group of patients, who have contact with the healthcare environment prior to hospital admission, is a heterogeneous group, and some require CAP-type therapy, while others require therapy directed against nosocomial, drug-resistant organisms. A proposed algorithmic approach to antibiotic therapy in these patients has been published [Brito and Niederman, 2009].
Duration of therapy
Current recommendations are to treat outpatients with mild-to-moderate CAP for 7 days or fewer if they have no fever for 48–72 h, and no sign of extrapulmonary infection. In a meta-analysis of 15 randomized controlled trials, a short course antibiotic regimen was as effective as an extended course. There was no difference in the risk of clinical failure, risk of mortality and bacteriologic eradication between a short course antibiotic regimen (≤7 days) versus an extended antibiotic regimen [Li et al. 2007]. For inpatients, antibiotics can be switched from intravenous to oral antibiotics once the patient is afebrile and symptoms improve, and the patient is able to take medications orally. In bacteremic patients antibiotics can be switched from intravenous to oral antibiotics when patients are clinically stable, although in bacteremic patients, the time to reach clinical stability is significantly longer than in nonbacteremic patients [Ramirez and Bordon, 2001].
Biomarkers could potentially be used to decide the duration of treatment and avoid the risks of undertreating some patients while substantially reducing overall antibiotic use. In a cohort of 302 CAP patients, PCT guidance significantly reduced the duration of antibiotic therapy (5.8 versus 12.9 days; p < 0.001), with no significant difference in outcome in two groups [Christ-Crain et al. 2006].
Treatment failure
Treatment failure is a lack of response or clinical deterioration despite antibiotic therapy, and occurs in up to 15% of all patients. The 2007 IDSA/ATS guidelines distinguish early treatment failure from later nonresponse to therapy. Early failure is defined as progressive pneumonia or clinical deterioration, usually with respiratory failure or septic shock, occurring in the first 72 h of therapy. This is usually due to an uncovered or resistant pathogen, severe illness on presentation, metastatic infection, or an inaccurate diagnosis. Later failure or nonresponse is defined as the absence or delay in achieving clinical stability (change in fever, and resolution of other vital sign abnormalities), and is usually due to a nosocomial infection, a noninfectious complication, or diagnosis (e.g. pulmonary embolism, inflammatory lung disease, etc.) [Mandell et al. 2007].
In a cohort of 2457 immunocompetent hospitalized patients in Spain, patients with early deaths (57 patients) had altered mental status, renal failure, tachycardia, increased respiratory rate, high fever, multilobar infiltrates, respiratory failure, and shock [Garcia-Vidal et al. 2008]. The most common causes of treatment failure are delayed response by the host despite appropriate antibiotics, or infection with an organism that is not covered by the initial antibiotic regimen. One study showed that discordant initial empiric therapy was due to lack of coverage of P. aeruginosa leading to increased early deaths in that cohort [Garcia-Vidal et al. 2008]. Other causes of treatment failure include the presence of a resistant pathogen, an unusual pathogen, empyema, abscess, or obstruction. Further investigation with arterial blood gas determination, CT scan, bronchoscopy, or lung biopsy may be required.
The chest radiograph usually clears within 4 weeks in patients younger than 50 years without underlying pulmonary disease. In contrast, resolution may be delayed for 12 weeks or longer in older individuals and those with underlying lung disease and the bacteremia delays radiographic resolution. A chest radiograph should be repeated in 6 weeks to see resolution of infiltrate in patients at higher risk of underlying malignancy.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
None declared.
