Abstract
Keywords
Introduction
Preterm birth is a major worldwide cause of neonatal mortality, morbidity, and long-term complications.1,2 Sonographic short cervix (SCX; cervical length ≤25 mm) in the midtrimester is an early risk factor and powerful predictor of spontaneous preterm delivery (SPTD).3–6 A SCX has been identified in approximately 2–8% of all pregnant women in the midtrimester, with 7–40% of these women delivering at < 32+0 weeks of gestation.4,6–9 Several interventions (i.e., progesterone, cerclage, antibiotics, indomethacin, and pessary) for treating SCX have been suggested but with variable therapeutic success in the clinical setting.3,5,10–12 This therapeutic variability is most likely due to the multiple etiology of this cervical disorder or its consequences.13,14 Thus, a better understanding of the SCX-underlying mechanisms (or of SPTD pathogenesis following cervical shortening) and identification of associated specific biomarkers are paramount to guiding therapy options and developing novel treatment strategies for women with SCX.
Evidences suggest that subclinical inflammation and infection in the amniotic cavity likely contribute to the etiology, pathophysiology, or consequence of SCX.7,12–16 In particular, the presence of intraamniotic inflammation (IAI), regardless of the microbial status of the amniotic cavity (MIAC), was reported to pose an additional risk for SPTD development in women with SCX.7,16 However, the inflammatory response does not simply manifest as present or absent, but occurs as a continuum of severity. In fact, in the setting of acute cervical insufficiency, preterm labor (PTL), and preterm premature rupture of membranes (PPROM), several studies have shown that the risk severity for SPTD correlates with the degree of intraamniotic inflammatory responses, which manifest on a continuum from “mild” to “moderate” to “severe”.17–20 Nevertheless, to date, no rigorous studies have directly evaluated these reported correlations in the SCX setting.
Most previous studies on amniotic fluid (AF) from women with midtrimester SCX have primarily focused on the inflammatory cytokines/chemokines, with elevated levels being robust predictors of subsequent SPTD.15,21,22 However, complex disorders with multiple causes, such as SPTD (particularly following the midtrimester identification of a SCX), may be typically caused by the interaction of multiple molecules that can mediate different biological mechanisms, rather than the malfunctioning of a single causal molecule.22–24 Thus, exploring the expression profile of specific proteins involved in other than the currently known inflammatory pathogenic pathways (i.e., cytokine-chemokine cascade) is critical for allowing the identification of novel biomarkers for early risk prediction and therapeutic targets for SPTD associated with SCX.
Acute-phase response (APR)- and extracellular matrix (ECM)-related signaling pathways potentially contribute to the underlying pathogenic mechanisms of SPTD, membrane rupture, and IAI.25–30 Furthermore, resistin, as well as the aforementioned signaling pathways-related molecules, represent useful AF biomarkers for SPTD in the context of cervical insufficiency, PTL, and PPROM.27,31–33 However, little or no information is available on the role of various APR- and ECM-related proteins and resistin in the AF in association with SPTD risk following the midtrimester identification of SCX. Thus, in the present study, we aimed to determine whether altered levels of various APR-, inflammation-, and ECM-related proteins in the AF are independently associated with SPTD risk in asymptomatic women with midtrimester SCX. In addition, we also aimed to assess whether the severity of SPTD risk is linked to the expression profile of those AF proteins. Lastly, we aimed to develop a better combined prediction model for SPTD risk based on the herein identified significant AF biomolecules.
Materials and methods
Study cohort and design
This was a single center, retrospective, observational cohort study of asymptomatic pregnant women who were diagnosed with a sonographic SCX at 17+0–25+6 weeks of gestation and who were admitted to the Department of Obstetrics and Gynecology at the Seoul National University Bundang Hospital (Seongnamsi, Korea) between August 2004 and March 2022. AF samples were selected from a comprehensive database of all consecutive patients at high risk of SPTD if they met the following inclusion criteria: (i) singleton gestation; (ii) a live fetus; (iii) asymptomatic sonographic cervical length (CL) < 25 mm; (iv) transabdominal amniocentesis performed for assessing in utero inflammation/infection, according to the prevalence of IAI and MIAC in patients with SCX (approximately 22% and 10%, respectively)7,34,35; (v) invisible amniotic membrane, as confirmed by sterile speculum examination; and (vi) available pregnancy outcomes data. Patients with the following conditions were excluded: multiple-gestation pregnancy, PPROM and PTL before AF collection, visible or prolapsed membranes beyond the external cervical os upon speculum examination, placenta previa, prophylactic cerclage earlier in the pregnancy, clinical signs of chorioamnionitis at presentation, missing (or consumed) AF sample, and major fetal congenital anomalies. SCX was defined as CL <25 mm, measured by transvaginal ultrasonography, and concurrently presenting without visible amniotic membrane based on the visual examination. Gestational age (GA) was calculated based on the last menstrual period and adjusted, if necessary, by the crown-rump length in the first trimester of pregnancy. The primary endpoint of the study was SPTD at <34 weeks of gestation, and the secondary endpoints were SPTD at <32 weeks and interval from sampling to delivery. The demographic and clinical data (but not the laboratory data measured in the AF samples) of several patients contained in this manuscript have been previously reported in papers published in Arch Gynecol Obstet (n = 47), J Korean Med Sci (n = 27), and PLoS One (n = 12).35–37 This study received approval from the Institutional Review Board of the Ethics Committee of Seoul National University Bundang Hospital (approval no. B-1311/228-010). All participating women provided written informed consent for the amniocentesis and for the use of their biological specimens, and clinical and ultrasound data for research purposes.
Collection and processing of AF samples
The procedures for collection, preparation, and storage of AF samples, and the detection of microorganisms have been previously reported in detail. 38 Briefly, AF samples were obtained at the time of admission by ultrasound-guided transabdominal amniocentesis. These AF samples were sent to the clinical laboratory for conventional culture of aerobic/anaerobic bacteria and fungi and isolation of genital mycoplasmas (Ureaplasma urealyticum and Mycoplasma hominis) using a commercial kit (Mycoplasma IST-3 kit; BioMerieux, Marcy l’Etoile, France). The clinical laboratory also determined the AF white blood cell (WBC) count. The rest of the AF samples was centrifuged at 1500 ×g at 4°C for 10 min, and the supernatant was aliquoted and stored at −70°C until assayed. The managing physicians gained access to the results of the AF culture and WBC counts. The primary indication for amniocentesis in asymptomatic women with midtrimester SCX was to rule out subclinical MIAC and/or IAI.7,12,34,35 This is because antibiotic treatment in women with SCX and MIAC eradicates intraamniotic infection, thereby achieving term delivery. 12
Analysis of AF proteins
The levels of hepcidin, kallistatin, mannose-binding lectin (MBL), pentraxin-2, retinol binding protein 4 (RBP4), and serpin A1 as APR molecules; interleukin (IL)-6, IL-8, and resistin as inflammatory molecules; lumican, matrix metalloproteinase (MMP)-8, transforming growth factor beta-induced (TGFBI), and urokinase-type plasminogen activator (uPA) as ECM-related molecules were determined in the stored AF samples by enzyme-linked immunosorbent assay (ELISA) (DuoSet ELISA kits; R&D Systems, Minneapolis, MN, USA) according to the manufacturer's instructions. The AF dilutions used and the assay's working range for each protein are provided in detail in Supplementary Materials. The intra-assay coefficient of variation was <10% for all analyzed proteins, except for IL-8 (10.5%). The inter-assay coefficient of variation was not calculated because all samples for each mediator were run on the same ELISA plate. Good assay performance of the ELISA kits for the aforementioned proteins was confirmed in the AF samples using spike-and-recovery and linearity-of-dilution experiments before the primary ELISA assays were performed. These protein molecules were chosen for the present study because: i) they were demonstrated to be important mediators of biological signals (such as acute-phase-, ECM-, and inflammation/immune-related homeostasis [https://www.uniprot.org/]) that are potentially involved in the pathogenesis of SPTD and in utero inflammation/infection25–27,39 and ii) they are considered to be valuable as predictive AF biomarkers for SPTD or in utero inflammation/infection of women with PTL or PPROM27,30,40,41; however, whether their altered expression in the AF is associated with SPTD in the setting of midtrimester SCX remains unclear. The aforementioned proteins in the AF were measured for research purposes only and their results were unavailable to the managing clinicians.
Management of asymptomatic SCX and clinical definitions
CL measurements using transvaginal sonography and obstetric management of asymptomatic SCX was previously described in detail.4,35,42 Briefly, CL was measured at least three times between the internal and external cervical os, and the shortest CL was used for analysis. Clinical decisions regarding amniocentesis, placement of cervical cerclage, use of progesterone, antibiotics, tocolytic agents, and bed rest were made primarily based on the discretion of the attending clinician due to the lack of uniformity concerning these issues among attending obstericians, as well as because study participnats included patients treated during a long-term study period (18 years). In general, before 2012–2014, women with SCX were offered the option to have cerclage, but progesterone was not offered for preterm birth prevention when a SXC was first detected. Since 2015, vaginal progesterone was recommended to most woman with SCX, and cerclage was also offered to most patients with SCX and a prior preterm birth in our hospital, in accordance with the then published American College of Obstetricians and Gynecologist guidelines on “Prediction and Prevention of Preterm Birth” and “Cerclage for the Management of Cervical Insufficiency”.43,44 Both cerclage and vaginal progesterone were recommended to any patients with SCX according to the preferences of the clinicians and patients. In our hospital, 200 mg micronized vaginal progesterone capsules (Utrogestan; Besins International, Belgium) were only used daily until 34–36 weeks of gestation if it was prescribed for SCX. For patients who underwent ultrasound-indicated cerclage for SCX, the McDonald cervical cerclage was performed by attending Maternal-Fetal Medicine physicians under spinal anesthesia, as previously reported. 18 Amniocentesis was not routinely performed to rule out subclinical intraamniotic inflammation/infection in women with SCX. Antenatal corticosteroids were administered to patients at risk of PTB between 23 and 34 weeks of gestation.
MIAC was defined as a positive AF culture for microorganisms. IAI was characterized by elevated AF IL-6 levels (≥2.6 ng/mL), based on previous reports.7,22 Acute histologic chorioamnionitis was diagnosed based on the presence of neutrophils in the chorionic plate, fetal membranes, or umbilical cord, using previously described criteria.45,46 Clinical chorioamnionitis was diagnosed based on the criteria proposed by Gibbs et al. 47 SPTD was characterized as preterm delivery caused by PPROM, PTL, or the development of clinical evidence of chorioamnionitis. Early SPTD was defined as SPTD at <32 weeks of gestation.
Statistical analysis
Mann–Whitney U test was used for comparison of continuous data, and the χ2-test or Fisher's exact test was used for comparison of categorical data between groups. AF proteins levels showing a significant association or trend for significance (P < 0.1) in the univariate analysis were further analyzed in a multivariable Firth logistic regression model to assess whether these were independent predictive factors for SPTD. The model was adjusted for the effect of baseline clinical variables which had a P-value <0.1 in univariate analyses. Additionally, to develop the best combined models (AF-based multimarker panels) for SPTD prediction, multivariable logistic regression analyses with forward selection were performed based on the identified AF markers (P < 0.1, in the univariate analysis). The linearity of significant continuous predictors was assessed prior to modeling using the Box-Tidwell test, which demonstrated that all continuous factors were linearly related to the logit of the outcome variables. Moreover, all models were checked for multicollinearity among the independent variables using the variance inflation factor (VIF), which did not reveal collinearity problems in all regressions (all VIFs <2). Receiver operating characteristic (ROC) and area under the curve (AUC) analyses were used to determine the optimal cutoff and diagnostic performance of each of the identified significant proteins and combined models that best predicted SPTDs at <34 and <32 weeks. The AUCs for each protein and combined model were compared pairwise using a previously described method. 48 Spearman rank correlation was used to determine which, if any, AF proteins correlated with CL. The sampling-to-delivery interval was assessed according to the low or high levels of each significant protein using Kaplan-Meier curves, with censoring at the time to medically indicated delivery or last follow up; and the difference between the two curves was determined by log-rank test. For variables that were statistically significant from the log-rank test (P < 0.05), multivariable Cox proportional hazards models were further constructed to evaluate the independent effect of each protein on the sampling-to-delivery interval, while controlling for other confounders. The proportional hazards (PH) assumption was assessed using the global Schoenfeld residual test, 49 which showed that the PH assumption is violated for AF serpinA1 (≥ 304.25 μg/mL) (P = 0.028), but not for IL-6 (≥ 0.98 ng/mL) (P = 0.028) and kallistatin (≥ 0.83 μg/mL) (P = 0.849) in the AF. To address the violation of the PH assumption by AF serpinA1, the follow-up period was divided into two periods: “early” (the first 60 days of follow-up) and “late” (subsequent days); Cox model analyses were conducted separately for each period.50,51 Finally, to evaluate the correlation between the expression levels of significant inflammatory mediators in the AF and the risk severity for subsequent SPTD following SCX diagnosis, the data corresponding to inflammatory mediators were further analyzed according to subgroups stratified by the quartiles of each inflammatory mediator. Firth logistic regression analyses were used to estimate the crude and adjusted odds ratios (OR) with 95% confidence intervals (CI) for the primary outcome (i.e., SPTD at <34 weeks), comparing each quartile to the lowest quartile (as the reference group) for each inflammatory mediator. The distribution of the primary outcome stratified by quartiles of each mediator concentration were compared using trend analysis. All probability values were two-sided and the level of type 1 error was set to P < 0.05. Statistical analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA) and R 4.1.1 software (R Foundation for Statistical Computing, Vienna, Austria).
Results
During the study period, 195 consecutive women with SCX between 17+0 and 25+6 weeks of gestation who were admitted to our hospital were identified. Of these 195 women, 83 did not undergo an amniocentesis owing to unwillingness to undergo the procedure or participate in the study. One woman had a history-indicated cerclage during early index pregnancy, and 18 were diagnosed with a SCX but presented with a visible amniotic membrane (cervical dilatation of 10 mm [n = 16] or 5 mm [n = 2]). Thus, these 102 women were excluded from the study. Of the remaining 93 women who underwent amniocentesis, 12 had no AF available for analysis, owing to limited remaining sample volume or inability of sample collection at admission, and 11 were lost to follow-up after being discharged without giving birth. These 23 women were also excluded from the study, leaving 70 women suitable for evaluation (Figure 1).
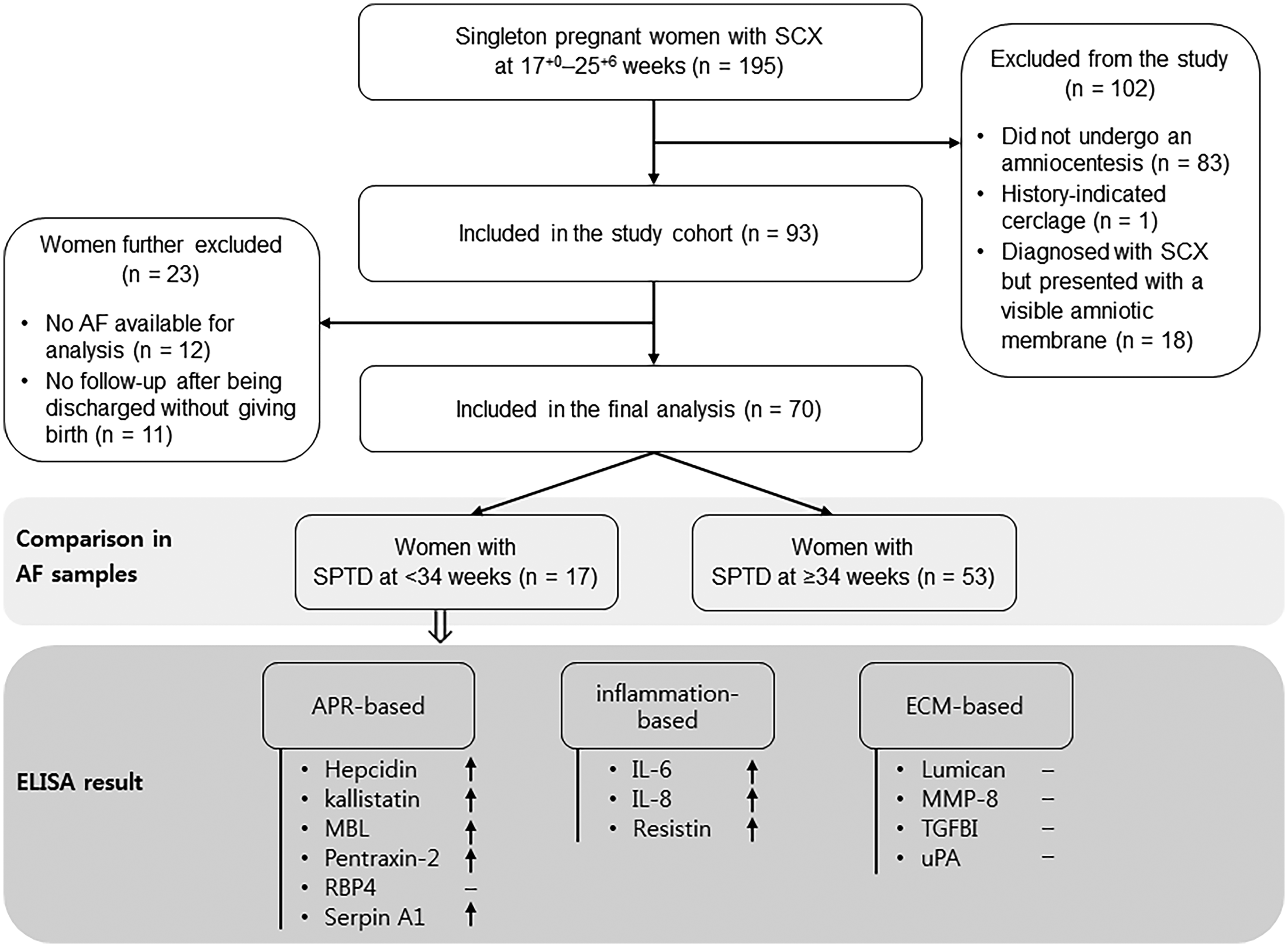
Study flow chart showing the participant recruitment process. SCX, short cervix; AF, amniotic fluid; SPTD, spontaneous preterm delivery; APR, acute-phase response; MBL, mannose-binding lectin; RBP4, retinol binding protein 4; serpin A1, alpha-1-antitrypsin; IL, interleukin; ECM, extracellular matrix; MMP, matrix metalloproteinases; TGFBI, transforming growth factor beta-induced; uPA, urokinase-type plasminogen activator.
Characteristics of the study participnats
Overall, 70 asymptomatic women with SCX were enrolled in the study. The mean GA (± standard deviation [SD]) of the study cohort was 21.9 ± 2.1 weeks (median, 21.8; range, 17.5–25.6 weeks) at sampling and 35.2 ± 5.5 weeks (median, 37.3; range, 18.0–40.0 weeks) at delivery. SPTDs at <34 and <32 weeks of gestation were observed in 24.2% (17/70) and 17.1% (12/70) of the women, respectively. MIAC and/or IAI, MIAC, and IAI were identified in 20.0% (14/70), 10.0% (7/70), and 17.1% (12/70) of the patients, respectively. The presence of concomitant MIAC and IAI was observed in 7.1% (5/70) of the women. The microorganisms isolated from the AF of asymptomatic women with MIAC were U. urealyticum (n = 7) and M. hominis (n = 3), among whom 42.8% (3/7) had polymicrobial infection.
Baseline demographic and clinical characteristics were similar between women with SPTDs at <34 vs. ≥ 34, as well as between those with SPTD at <32 vs. ≥ 32 weeks, including age, parity, CL, medications, cerclage placement, and rates of positive AF culture (Table 1). The exceptions were as follows: (1) patients who delivered at <34 weeks had significantly lower GA at sampling, had a higher rate of corticosteroid administration, and tended to be less likely to receive progesterone therapy and had more positive AF cultures (P = 0.057 and P = 0.054, respectively) compared with patients who delivered at ≥34 weeks; and (2) patients who delivered at <32 weeks had significantly lower GA at sampling and tended to be more likely to receive antibiotic therapy, and be less likely to receive cerclage placement (P = 0.057 and P = 0.088, respectively) compared with patients who delivered at ≥32 weeks (Table 1).
Baseline demographic and clinical characteristics of the study population stratified according to spontaneous preterm delivery status in women with a short cervix.

CRP, C-reactive protein; AF, amniotic fluid; WBC, white blood cell.
Values are given as the median (interquartile range) or % (n/N).
Defined as AF IL-6 ≥ 2.6 ng/mL.
Associations between AF proteins and SPTD at <34 and <32 weeks of gestation
The median AF levels of hepcidin, IL-6, IL-8, kallistatin, MBL, pentraxin-2, resistin, and serpin A1 were significantly higher in women with STPB at <34 weeks than in those who delivered at ≥34 weeks (Figure 2). Univariate analysis of SPTD at <32 vs. ≥ 32 weeks also showed that the median AF levels of hepcidin, kallistatin, MBL, pentraxin-2, and serpin A1 were significantly higher in women who delivered before 32 weeks than in those delivering at or after 32 weeks (Figure 3). Moreover, AF RBP4, resistin, and TGFBI levels tended to be higher in women with STPB at <32 weeks; however, these differences were not statistically significant (P = 0.059, P = 0.082, and P = 0.066, respectively; Figure 3). The rates of IAI were significantly higher in women with SPTDs at <34 and <32 weeks of gestation than in those who delivered later on (Table 1). However, in univariate analyses, no differences in the AF levels of lumican, MMP-8, and uPA were found to be associated with SPTD at <34 or <32 weeks (Figures 2 and 3).
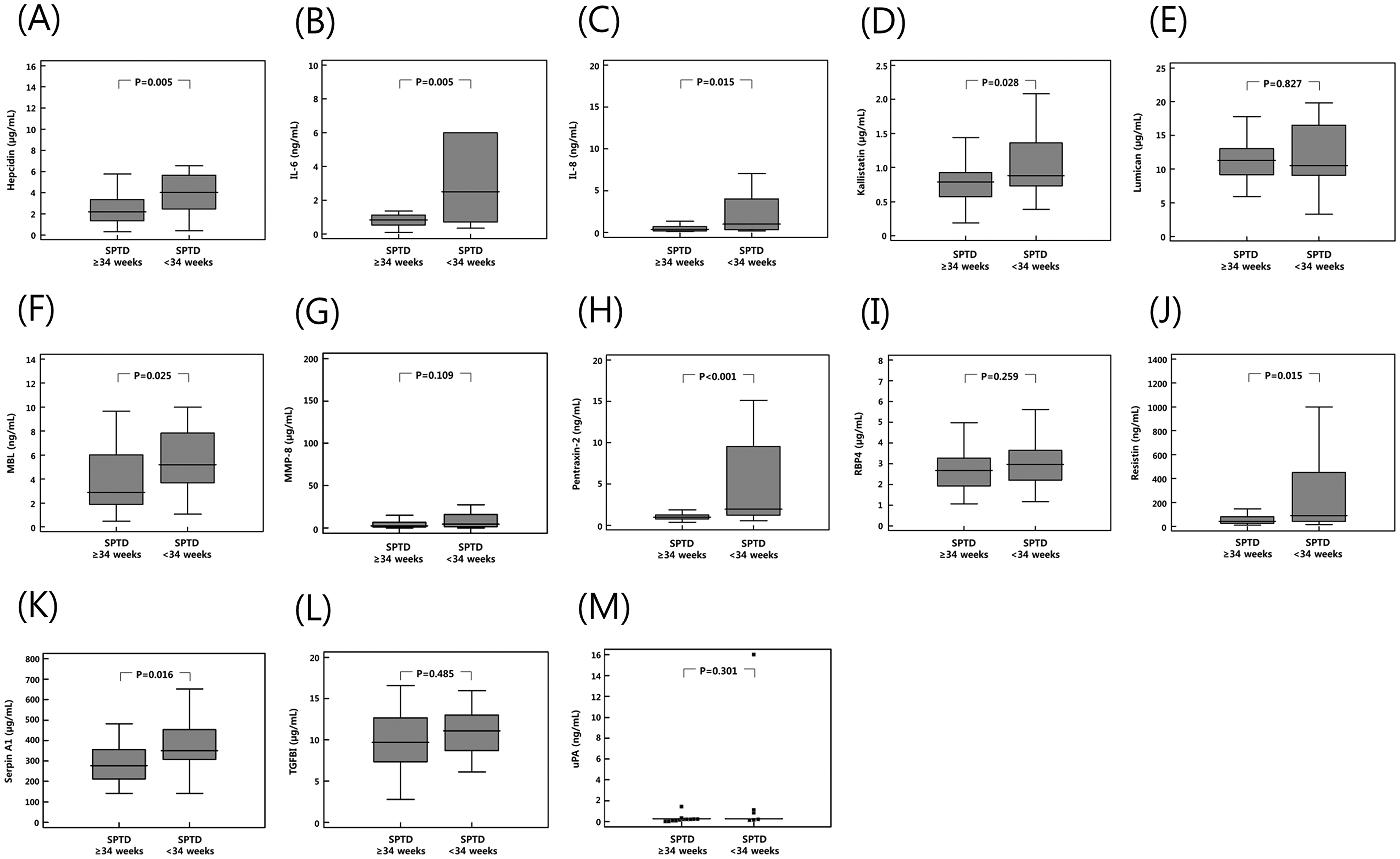
Box plots displaying median values (horizontal lines), interquartile range (25th percentile to 75th percentile; boxes), and extreme values (vertical lines) of (A) hepcidin, (B) IL-6, (C) IL-8, (D) kallistatin, (E) lumican, (F) MBL, (G) MMP-8, (H) pentraxin-2, (I) RBP4, (J) resistin, (K) serpin A1, (L) TGFBI, and (M) uPA in the amniotic fluid from women with spontaneous preterm delivery at <34 vs. ≥ 34 weeks and a short cervix. IL, interleukin; MBL, mannose-binding lectin; MMP, matrix metalloproteinases; RBP4, retinol binding protein 4; serpin A1, alpha-1-antitrypsin; TGFBI, transforming growth factor beta-induced; uPA, urokinase-type plasminogen activator.

Box plots displaying median values (horizontal lines), interquartile range (25th percentile to 75th percentile; boxes), and extreme values (vertical lines) of (A) hepcidin, (B) IL-6, (C) IL-8, (D) kallistatin, (E) lumican, (F) MBL, (G) MMP-8, (H) pentraxin-2, (I) RBP4, (J) resistin, (K) serpin A1, (L) TGFBI, and (M) uPA in the amniotic fluid from women with spontaneous preterm delivery at <32 vs. ≥ 32 weeks and a short cervix. IL, interleukin; MBL, mannose-binding lectin; MMP, matrix metalloproteinases; RBP4, retinol binding protein 4; serpin A1, alpha-1-antitrypsin; TGFBI, transforming growth factor beta-induced; uPA, urokinase-type plasminogen activator.
We further assessed the independent association of various AF protein levels with SPTD at <34 and <32 weeks (Table 2). Elevated AF levels of IL-6, IL-8, kallistatin, pentraxin-2, resistin, and serpin A1 (but not of hepcidin and MBL), as well as the presence of IAI, were found to be significantly associated with SPTD at <34 weeks after adjusting for GA at sampling and progesterone and corticosteroid use. Similarly, using SPTD at <32 weeks as an endpoint in the multivariable Firth logistic regression model, elevated AF levels of kallistatin, MBL, and serpin A1 (but not hepcidin, pentraxin-2, RBP4, resistin, TGFBI, nor the presence of IAI) were found to be significantly associated with SPTD at <32 weeks when adjusted for GA at sampling, cerclage placement, and antibiotics administration.
Multivariable firth logistic regression model showing the association between the various amniotic fluid proteins and spontaneous preterm delivery in women with a short cervix.

SPTD, spontaneous preterm delivery; AF, amniotic fluid; IL, interleukin; MBL, mannose-binding lectin; RBP4, retinol binding protein 4; serpin A1, alpha-1-antitrypsin; TGFBI, transforming growth factor beta-induced.
Adjustment for gestational age at sampling and use of vaginal progesterone and corticosteroid.
For the adjusted odds ratio.
Adjusted for gestational age at sampling, cerclage placement, and use of antibiotics.
Defined as AF IL-6 ≥ 2.6 ng/mL.
For the prediction of SPTD at <34 weeks, the AUC values of AF IL-6, IL-8, kallistatin, pentraxin-2, resistin, and serpin A1 ranged from 0.678 to 0.792, with no significant differences from each other (all variables: P = 0.079–0.989) (Table 3 and Figure 4A and B). Similarly, the AUCs for the prediction of SPTD at <32 weeks were of 0.723 for AF kallistatin, 0.772 for AF MBL, and 0.760 for AF serpin A1, which were not significantly different from each other (all variables: P = 0.574–0.910) (Table 3 and Figure 5). None of the the measured AF proteins was correlated with CL (all variables, r = −0.061–0.063, P > 0.5), except for hepcidin levels (r = 0.246, P = 0.04) (Table S1).

Receiver operating characteristic curves for predicting spontaneous preterm delivery at <34 weeks (A) by IL-6, IL-8, and resistin in AF (AF IL-6: AUC 0.725, SE 0.080, P = 0.006; AF IL-8: AUC 0.696, SE 0.077, P = 0.015; AF resistin: AUC 0.698, SE 0.086, P = 0.015) and (B) by kallistatin, pentraxin-2, and serpin A1 in AF (AF kallistatin: AUC 0.678, SE 0.077, P = 0.028; AF pentraxin-2: AUC 0.792, SE 0.070, P < 0.001; AF serpin A1: AUC 0.695, SE 0.076, P = 0.016). IL, interleukin; AF, amniotic fluid; AUC, area under the curve; SE, standard error; serpin A1, alpha-1-antitrypsin.

Receiver-operating characteristic curves for predicting spontaneous preterm delivery at <32 weeks by kallistatin, MBL, and serpin A1 in AF (AF kallistatin: AUC 0.723, SE 0.096, P = 0.016; AF MBL: AUC 0.772, SE 0.064, P = 0.003; AF serpin A1: AUC 0.760, SE 0.084, P = 0.005). MBL, mannose-binding lectin; serpin A1, alpha-1-antitrypsin; AF, amniotic fluid; AUC, area under the curve; SE, standard error.
Diagnostic indices of various amniotic fluid proteins to predict spontaneous preterm delivery.

SE, standard error; ROC, receiver operating characteristics; CI, confidence interval; PPV, positive predictive value; NPV, negative predictive value; SPTD, spontaneous preterm delivery; AF, amniotic fluid; IL, interleukin; serpin A1, alpha-1-antitrypsin; MBL, mannose-binding lectin.
Cut-off values corresponding to the highest sum of sensitivity and specificity or the point on the ROC curve closest to (0, 1).
Values are presented as % (95% CI).
Combined model A consists of AF IL-6 and pentraxin-2 levels.
P < 0.05 compared with combined model A by the method of DeLong et al.
P < 0.05 compared AF pentraxin-2 by the method of DeLong et al.
Relationship between AF proteins and sampling-to-delivery interval
Kaplan-Meier survival analyses revealed that women with higher AF levels of kallistatin (≥0.83 μg/mL) (log-rank test, P = 0.037) or serpin A1 (≥304.25 µg/mL) (log-rank test, P = 0.049) exhibited significantly shorter sampling-to-delivery intervals (Figure 6). However, Kaplan-Meier and log-rank test analyses of the sampling-to-delivery intervals for a IL-6 of ≥0.98 or <0.98 ng/mL, IL-8 of ≥0.37 or <0.37 ng/mL, pentraxin-2 of ≥1.26 or <1.26 ng/mL, or resistin of ≥71.32 or <71.32 ng/mL showed no statistically significant difference (log-rank test, P = 0.053, 0.120, 0.791, and 0.172, respectively) (Figure S1). In Cox proportional hazard analyses, high AF levels of kallistatin (hazard ratio: 1.25; 95% CI, 0.69–2.28, P = 0.457) were not significantly associated with the sampling-to-delivery interval after adjusting for GA at sampling, corticosteroid use, and cerclage placement (Table S2). As stated in the statistical analyses section, multivariable Cox model analyses for AF serpinA1 were performed separately for the first 60 days of follow-up and for subsequent days. Cox proportional modeling, adjusted for GA at sampling, corticosteroid use, and cerclage placement, indicated that high AF levels of serpinA1 were not significantly associated with longer sampling-to-delivery interval in the first 60 days (hazard ratio: 4.69; 95% CI, 0.57–38.41, P = 0.149) and in the subsequent period (hazard ratio: 1.41; 95% CI, 0.80–2.50, P = 0.230) (Table S3).
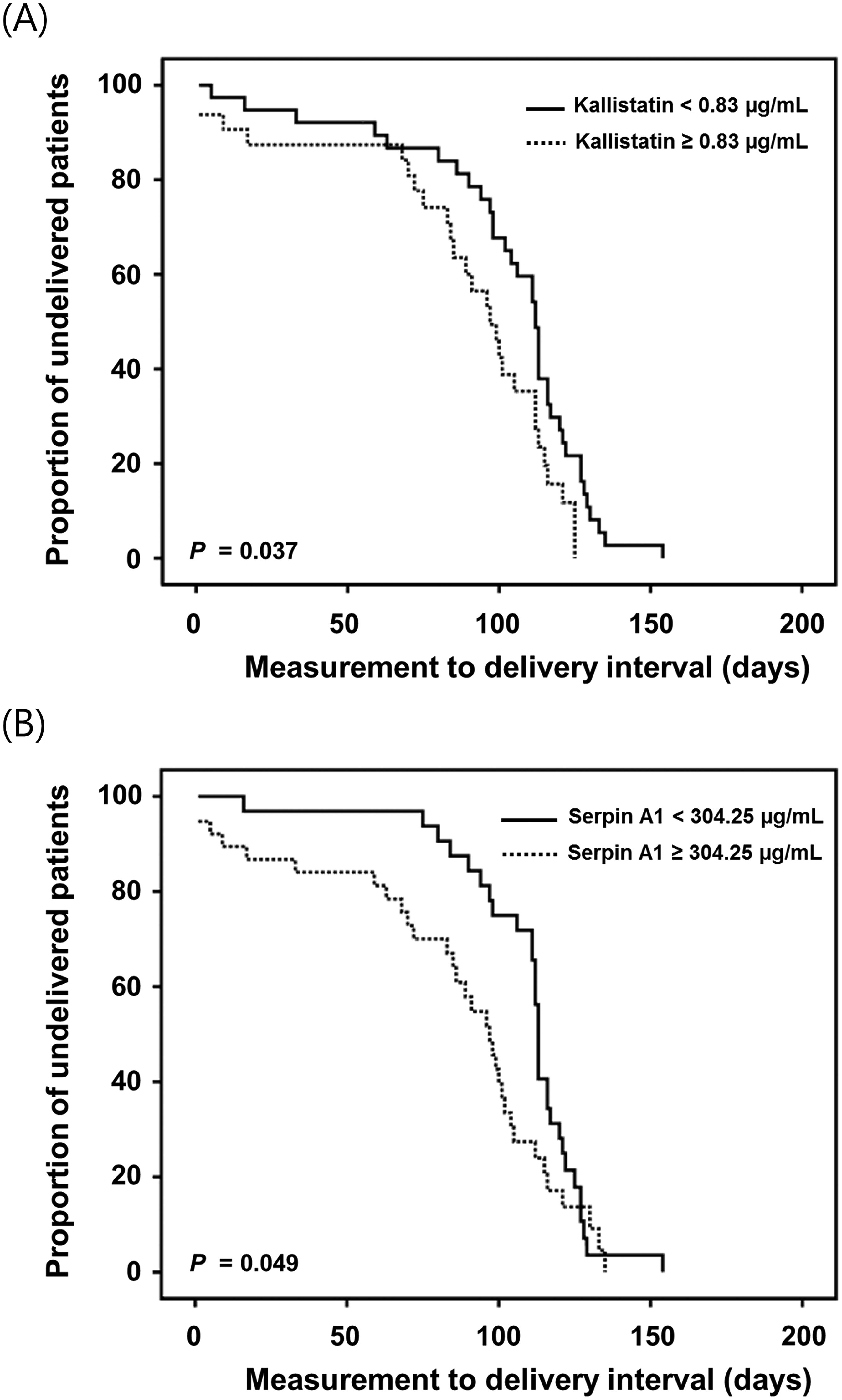
Kaplan-Meier survival estimates of the sampling-to-delivery intervals for (A) AF kallistatin of ≥0.83 or <0.83 μg/mL (median, 97.00 days [95% CI, 85.42–108.57] vs. 112.00 days [95% CI, 106.79–117.20]; P = 0.037) and (B) AF serpin A1 of ≥304.25 or <304.25 µg/mL (median, 97.00 days [95% CI, 86.00–107.99] vs. 113.00 days [95% CI, 111.91–114.08]; P = 0.049). Kaplan Meier curves were generated based on the optimal cut-off values derived from the ROC curves to predict SPTD at <34 weeks. AF, amniotic fluid; CI, confidence interval; serpin A1, alpha-1-antitrypsin; ROC, receiver operating characteristic; SPTD, spontaneous preterm delivery.
Severity of SPTD risk based on the interquartile range of significant inflammation-related proteins
The odds of SPTD at <34 weeks increased significantly with each increasing quartile of baseline AF concentrations for IL-6/8, pentraxin-2, and serpin A1 (except for kallistatin or resistin) (P for trend <0.05; Table 4 and Figure S2). In particular, this relationship remained significant for IL-6/8, pentraxin-2, and resistin, when adjusting for relevant baseline confounders, such as GA at sampling and use of vaginal progesterone and corticosteroid (P for trend <0.05; Table 4 and Figure S2). For example, women with SCX who had the highest levels of pentraxin-2 (highest quartile) showed 6.92-fold increased odds of SPTD at <34 weeks as compared with those with the lowest levels of AF pentraxin-2. Furthermore, the adjusted OR for the highest AF pentraxin-2 levels (4th quartile) was 15.76 (95% CI: 1.72–322.42) compared with the lowest quartile, after adjusting for potential confounders.
Unadjusted and adjusted odds ratios of spontaneous preterm delivery at <34 weeks according to the expression quartiles of inflammation-related proteins in the amniotic fluid.
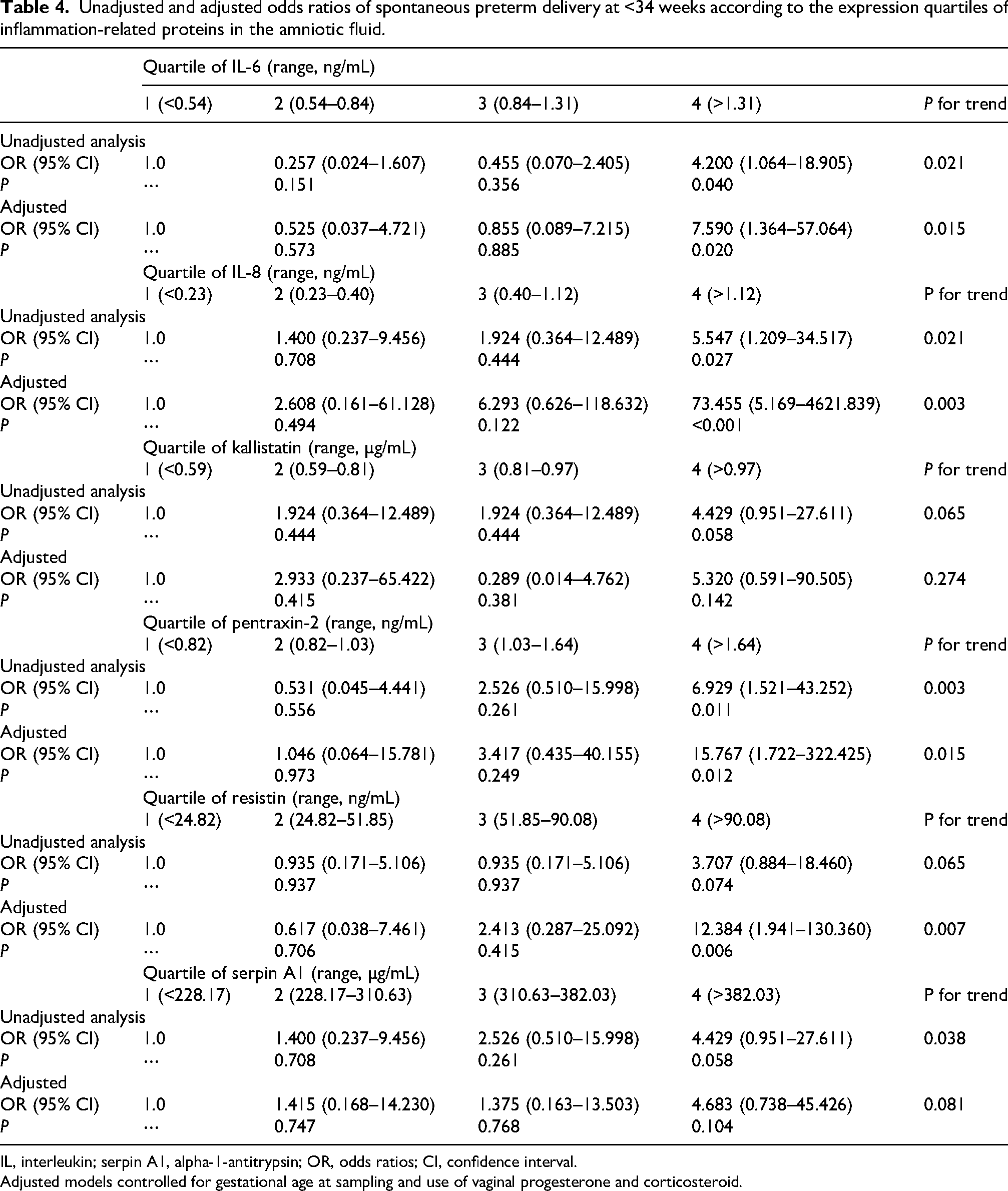
IL, interleukin; serpin A1, alpha-1-antitrypsin; OR, odds ratios; CI, confidence interval.
Adjusted models controlled for gestational age at sampling and use of vaginal progesterone and corticosteroid.
Development of combined prediction models
To develop a combined prediction model for SPTD at <34 weeks, the newly identified eight AF biomolecules (hepcidin, IL-6, IL-8, kallistatin, MBL, pentraxin-2, resistin, and serpin A1) were included in a stepwise logistic regression model (Table S4). Only AF pentraxin-2 and IL-6 were finally selected for the best combination, with an AUC value of 0.853 (95% CI: 0.753–0.954; Hosmer–Lemeshow test, P = 0.822) (Table S4; Figure S3A). The AUC value of this combined model was significantly greater than that of AF IL-6 alone (P < 0.048), but not of AF pentraxin-2 (P = 0.383) (Table 3).
Similarly, a combined prediction model was also developed for SPTD at <32 weeks using eight AF biomolecules, namely AF hepcidin, kallistatin, MBL, pentraxin-2, RBP4, resistin, serpin A1, and TGFBI. Of note, only AF pentraxin-2 was selected for the model in the forward stepwise regression analysis, with an AUC value of 0.930 (95% CI: 0.868–0.991; Hosmer–Lemeshow test, P = 0.452). The AUC value of AF pentraxin-2 was significantly greater than the AUC values of AF hepcidin, kallistatin, MBL, pentraxin-2, RBP4, resistin, and TGFBI (P < 0.05 for each), but was not significantly different from the AUC value of AF serpin A1 (P = 0.0678) (Table 3; Figure S3B).
Relationship between AF proteins and IAI
We further analyzed the data to determine associations between the altered levels of AF proteins and IAI. The median AF levels of hepcidin, pentraxin-2, serpin A1, IL-6, IL-8, resistin, lumican, and MMP-8 were significantly higher in women with IAI than in women without this condition (Table S5). AF MBL levels tended to be higher in women with IAI; however, this difference did not reach statistical significance (P = 0.056).
Discussion
The present study demonstrates that, in asymptomatic women with midtrimester SCX, (i) elevated AF concentrations of kallistatin, MBL, pentraxin-2, and serpin A1 (APR molecules), as well as IL-6/8 and resistin (inflammatoty molecules) are significantly associated with an increased risk of SPTD; (ii) SPTD risk increases in a dose dependent manner of each of these inflammation-based mediators; and (iii) notably, combination of AF IL-6 and pentraxin-2 can effectively help predict the risks of SPTD at <34 weeks (AUC=0.853), whereas AF pentraxin-2 alone showed the best predictive potential for SPTD at <32 weeks (AUC=0.930). However, (iv) altered AF levels of lumican, MMP-8, TGFBI, and uPA (ECM-related molecules) are not associated with SPTD risk. Taken together, these observations highlight the importance of inflammatory mechanisms occurring in the amniotic cavity, especially those contributing for the severity of the local inflammatory response activated in the AF in addition to the presence or absence of IAI, for developing SPTD in the context of a midtrimester SCX.
IAI presenting in women with midtrimester SCX has been consistently reported as an important risk factor for SPTD.7,15,16,21,22,35 Herein, we extend these finding and show for the first time that the adverse pregnancy outcomes of IAI associated with SCX cannot be characterized by an “all-or-nothing” process, but rather have grades of severity that correlate with the expression levels of AF inflammatory mediators, as previously described in several other obstetric conditions (i.e., PTL, PPROM, and cervical insufficiency).17–20 These findings have important clinical implications for the treatment of SCX, since the severity of IAI may be regulated (reduced or eliminated) to improve pregnancy outcomes for women with SCX, with early treatments to reduce IAI severity, prevent its progression to a more severe condition of IAI, or resolve it. In fact, previous studies on women with SCX or cervical insufficiency reported that intraamniotic infection or inflammation can be eradicated or ameliorated with the use of antibiotics (particularly clarithromycin/azithromycin) in a subset of patients, many of whom can reach term or near term pregnancy.12,52,53 Similarly, our data also may support the current use of progesterone supplementation, which has antiinflammatory protperties, to prevent SPTD in women with SCX. Indeed, Kiefer et al. previously showed that only patients with IAI-associated SCX could benefit from progesterone or cerclage treatment. 54 There is, therefore, a critical need to develop a noninvasive test that can accurately detect the presence of in utero inflammation/infection in women with SCX.
The present study demonstrates that the earlier the midtrimester SCX is diagnosed, the higher the risks of SPTD at < 32 and < 34 weeks, which is in agreement with previous studies.22,55 Our finding of 10% rate of MIAC also agree with Hassan et al., which reported a rate of MIAC of 9% in asymptomatic women with CL < 2.5 cm and without cervical dilatation. 12 However, in contrast with previous studies showing that proinflammatory cytokines levels in the AF are inversely correlated with CL in women with midtrimester SCX,16,22,56 we did not find any associations between AF inflammatory and APR proteins levels and CL. This discrepancy may be related to one of the enrollment criteria of this study, namely, “invisible amniotic membrane” based on the visual examination vs. SCX with cervical dilatation ≤20 mm or CL of “0” mm, which condition may present with a visible amniotic membrane employed in other studies.16,22,56 “Invisible amniotic membrane” was chosen as the enrollment criterion of this study to exclude patients with premature cervical dilatation. It should be noted that visible amniotic membrane could potentially affect AF inflammatory proteins levels and SPTD risk, which are generally found to be higher than in women without this condition.35,37 Indeed, Kiefer et al. showed the lack of inverse correlations between CL and AF cytokine levels when the CL was between 6–25 mm, and that significant inverse correlations between these parameters observed in all women with CL between 0–25 mm were driven largely by high cytokine levels at very short CL measurtements (≤5 mm). 56
In the present study, we firstly showed that increases in kallistatin, MBL, pentraxin-2, and serpin A1 levels in the AF (all APR proteins) were associated with SPTD following the midtrimester SCX diagnosis. These associations indicated that APR signaling pathway of SPTD may take place over several weeks before the symptoms of PTL or PPROM manifest. Thus, dysregulated APR signaling may be involved in the early activation of the parturition process as a common pathway of SPTD. 57 Kallistatin (also known as Serpin A4) is an endogenous protein that exerts pleiotropic biological functions, including antiinflammatory, antioxidative, antiapoptotic, and antiangiogenic properties, and is expressed in various body fluids, tissues, and organs.58,59 In our previous studies, kallistatin levels were found to be elevated in the AF of women with PTL demonstrating SPTD within 7 days and MIAC/IAI, whereas significantly decreased kallistatin levels (as a negative APR protein) were observed in the plasma obtained from women with PTL/PPROM complicated by in utero inflammation/infection.27,60–62 These findings are in agreement with the herein reported kallistatin data in the context of asymptomatic women with midtrimester SCX.
Pentraxin-2 (also known as serum amyloid P), which belongs to a pentraxin family as a short pentraxin (similar to CRP), is a pattern recognition molecule that plays a pleiotropic role in the innate immune system and inflammation. 63 This protein also functions as a factor that decreases neutrophils adhesion and spreading, regulates macrophage activation, and enhances phagocytosis of cell debris. 63 Pentraxin-2 is mainly produced by the liver and is constitutively present in circulation in the blood as a naturally circulating soluble pattern recognition receptor.63,64 In humans, this protein can also be expressed by macrophage and smooth muscle cells at the inflammatory site via the stimulation of proinflammatory cytokines, microbial components, and toll-like receptor agonists. 64 To our knowledge, only one study on AF samples from women with PPROM explored the role of pentraxin-2 in the obstetric field, showing that its levels are significantly increased in the AF of women with IAI or microbial-associated IAI (but not MIAC). 65 Overall, the known inflammatory properties (particularly for neutrophil) of pentraxin-2 and the findings from the aforementioned study by Musilova et al. 65 are consistent with and support the herein described relationship between high AF pentraxin-2 levels and SPTD risk, given the reported associations between IAI, elevated AF WBC counts (mainly neutrophils), and SPTD risk.33,66 Finally, it should be noted that AF pentraxin-2 yielded the highest predictive potential (AUC=0.792 and 0.930, respectively) for SPTDs at <34 and <32 weeks as a single marker, which may be used as an important indicator of adverse pregnancy outcomes in women with a SCX.
MBL, belonging to the collectins family, is an APR protein that activates the lectin pathway of the complement system and subsequent inflammatory mechanisms; and is mainly synthesized in the liver and secreted into the bloodstream, but also by human placenta, decidua, and vaginal epithelial cells.67–69 This protein acts as a pattern recognition molecule of the innate immune system that interacts with invading microorganisms to promote their removal by opsonophagocytosis, and thus plays an important role in the innate immune response. 67 In line with its role at the first-line immune defense, MBL polymorphisms (which result in lower MBL serum concentrations) were reported to be associated with increased risks of preterm birth in several ethnic groups, histologic chorioamnionitis, and PPROM.70–74 In the setting of asymptomatic women (at 16–23 weeks), a previous study revealed that MBL levels are not significantly different in the plasma of women with preterm births and controls with term delivery. 75 A recent study on cervicovaginal fluid of asymptomatic pregnant women (in the early and late second trimester) showed that MBL levels are significantly increased in the cervicovaginal fluid of preterm delivery patients compared with the term delivery group, 76 which is in agreement with and supports the findings reported herein for AF MBL. Although few studies have explored the role of MBL in the AF in relation to parturition, IAI, or MIAC, our data strongly support its important role in the AF for the regulation of preterm parturition.
Serpin A1, which is also called α1-antitrypsin, is an APR protein and natural inhibitor of neutrophil elastase, plays a significant role in regulating neutrophil activation and neutrophil-initiated processes.77,78 Serpin A1 is mainly produced in the liver and secreted into the circulation, with immune-modulatory and antiinflammatory functions, but is also locally synthesized in the amniotic tissues, monocytes, macrophages, and trophoblasts through the stimulation of proinflammatory cytokines (IL-6 or tumor necrosis factor-α).79–81 Traditionally, serpin A1 has been extensively investigated for its ability to prevent membrane collagen breakdown in the context of PPROM and was shown to be decreased in the AF and amnions of patients with PPPOM as compared with those with a normal pregnancy.79,82 Moreover, deficiencies of this protein activity may be considered to be a cause of preterm birth. 83 In the context of asymptomatic pregnant women, Li et al. reported that serpin A1 expressions are significantly upregulated in the AF of women who delivered at <34 weeks as compared with those who delivered at ≥34 weeks, 41 which agree with the results of the present study. Similarly, a previous study on serum samples of asymptomatic pregnant women of 1st and 2nd trimesters showed a significant higher level of serum serpin A1 in a preterm delivery group compared with that in a term delivery group. 84 Taken together, these findings suggest that serpin A1 levels may be reflected in circulation and in the local in utero environments in various manners (high or low level) according to pathologic factors associated with SPTD. However, further in vitro and in vivo studies are required to elucidate its roles in the regulation of preterm parturition.
Elevated AF levels of resistin were reported to be one of the strongest predictors of SPTD or MIAC/IAI in pregnancies complicated by cervical insufficiency and PTL/PPROM, with a diagnostic potential similar to those of AF IL-6/8.31,32,85 We confirm these findings in the setting of asymptomatic midtrimester SCX. Human resistin is a 12.5-kDa cysteine-rich protein that is mainly secreted by macrophages and monocytes in inflammatory lesions, and is significantly implicated in innate immunological and inflammatory responses. 86 Resistin also acts as a proinflammatory cytokine capable of stimulating the production and release of other cytokines/chemokines, including IL-1, IL-6, IL-12, and tumor necrosis factor-α. 86 Given its biological functions and production site, the observed significant increase in AF resistin expression in women with SPTD following midtrimester SCX diagnosis may reflect a response to the microorganisms or inflammation in the amniotic cavity (Table S5), leading to the terminal cascade of events that result in preterm birth.
In contrast to our previous studies conducted in women with PTL, where SPTD was associated with high expression of ECM-related proteins in the AF (such as TGFBI, MMP-2/8/9, uPA, and osteonectin),20,27 we found that altered AF levels of ECM-related molecules (namely lumican, MMP-8, TGFBI, and uPA) are not associated with SPTD risk in asymptomatic women with SCX. This discrepancy concerning the role of ECM-related proteins in the regulation of SPTD between SCX and PTL is probably related to the secondary production of ECM-related proteins and prostaglandins triggered by proinflammatory cytokines during local in utero inflammation. 87 IAI may be identified in asymptomatic women with SCX at very early inflammation stages using AF samples obtained at the time of amniocentesis; hence, the activation of ECM-related proteins and prostaglandins production are not yet initiated by proinflammatory cytokines. In contrast, IAI in symptomatic women with PTL may represent a more advanced stage of inflammation in the amniotic cavity to the extent that it may eventually cause uterine contractions via locally produced prostaglandins; thus, ECM-related proteins are already upregulated in the AF, along with proinflammatory proteins.88,89 Alternatively, this discrepancy may be explained by the differences in sample size between the studies.
Major strengths of the study are that it is, to our knowledge, the first to investigate (i) the potential role of several APR-, ECM-, and other inflammation-related proteins in the AF with respect to SPTD risk in asymptomatic women presenting with midtrimester SCX; and (ii) whether risk severity for SPTD is associated with the degree of intraamniotic inflammatory response, as evidenced by AF inflammation-related proteins levels. Another strength of this study is the strict inclusion criteria employed for SCX, such as SCX without visible amniotic membrane, which excluded the possible effects of premature cervical dilatation associated with cervical insufficiency on AF proteins levels and SPTD risk (increase).35,37 Despite these strengths, the present study had also some limitations. First, the clinical application of AF-based biomarkers is limited in contemporary clinical practice for asymptomatic women with SCX as the AF sampling requires an invasive procedure that women with SCX (and even clinicains) may decline. Second, the clinical management was not uniform among all cases with SCX, which could result in selection bias, due to (i) the long-term study period; (ii) the number of attending obstetricians cared for women with SCX during this period; and (iii) establishment of a global standard management only since 2015 (still ongoing). 43 Consequently, not fully controlling for this factor could potentially introduce bias into the study. Third, we used AF samples stored for up to 18 years at −70°C to measure the analytes, which may lead to alterations in their levels associated with protein degradation. 90 Fourth, this study (i) had a retrospective design, which may be subject to inherent selection bias; (ii) included a relatively small number of patients from a single center, which may lead to type II error (underpowered analysis); (iii) established only associations, rather than causal relationships, between identified AF proteins and SPTD outcomes owing to its observational nature, and (iv) the cutoff values used for the identified AF markers were not validated in an independent cohort, all of which may limit the generalizability of our findings. Therefore, replication of the study findings in other cohort samples is warranted. Fifth, multiple comparison corrections, such as Bonferroni adjustments and P < 0.0038 (0.05 divided by 13), were not applied in this study because of its exploratory nature and the limited sample size, which is not sufficiently powered to withstand corrections. Although Bonferroni adjustments are effective at reducing the risk of Type I errors (false positives), they could increase the risk of Type II errors (false negatives) when assessing various biomarkers and lead to an even smaller number of significant results. In fact, when applying multiple comparison corrections using the Mann-Whitey U test with Bonferroni adjustment (P < 0.0038) in this study, only one protein with a P value <0.001 (pentraxin-2 for SPTD at <34 weeks) was confirmed as a significant predictor of the outcomes. Sixth, not every woman with SCX who was admitted to our hospital routinely underwent amniocentesis owing to the lack of uniformity among attending obstetricians concerning its benefits and associated risks. This may have potentially led to a selection bias in the current study. Seventh, the data for AF sludge was not available for this study, despite the reported associations of its presence with SPTD and in utero inflammation and infection in asymptomatic women with SCX or at high risk for SPTD.91,92 Eighth, the analyses that were performed in this study did not include polymerase chain reaction for the identification of MIAC and other important mediators levels (i.e., monocyte chemoattractant protein-1 or macrophage inflammatory protein-1), known to be highly predictive of SPTD in the setting of SCX.21,22 Ninth, the statistical power of the biomarker analyses performed ranged between 6.2–98.6% for SPTD at <34 weeks and 9.0–99.9% for SPTD at <32 weeks (Table S7). This may explain the lack of associations between the AF lumican, MMP-8, RBP4, TGFBI, and uPA levels and the SPTD outcomes. Thus, our biomarker results should be interpreted with caution, requiring the validation of these findings in larger independent cohorts.
Conclusions
In conclusion, our data demonstrate that several APR (kallistatin, MBL, pentraxin-2, and serpin A1)- and inflammation (resistin and IL-6/8)-related proteins in the AF are involved in the subsequent development of SPTD in asymptomatic women with midtrimester SCX. In particular, this SPTD risk (especially risk severity) is closely associated with the degree of the inflammatory response in the AF, as categorized by the aforementioned APR- and inflammation-related proteins levels, as well as IAI presence or absence. These findings may provide valuable insights for developing novel therapeutic strategies to reduce residual risk of SPTD in women with midtrimester SCX and for supporting the current use of progesterone supplementation (antiinflammatory effect) in these patients who had IAI. Additional research is necessary to evaluate whether the levels of the aforementioned significant proteins measured in less-invasively obtained samples (e.g., plasma, cervicovaginal fluid, or saliva) are also effective for predicting SPTD and MIAC/IAI risks in women with midtrimester SCX.
Supplemental Material
sj-docx-1-ini-10.1177_17534259251372138 - Supplemental material for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix
Supplemental material, sj-docx-1-ini-10.1177_17534259251372138 for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix by Kyo Hoon Park, Bo Young Choi, Kyong-No Lee, Eunji Oh, Min Jung Lee, Hee Young Cho and Da Eun Jeong in Innate Immunity
Supplemental Material
sj-docx-2-ini-10.1177_17534259251372138 - Supplemental material for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix
Supplemental material, sj-docx-2-ini-10.1177_17534259251372138 for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix by Kyo Hoon Park, Bo Young Choi, Kyong-No Lee, Eunji Oh, Min Jung Lee, Hee Young Cho and Da Eun Jeong in Innate Immunity
Supplemental Material
sj-tif-3-ini-10.1177_17534259251372138 - Supplemental material for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix
Supplemental material, sj-tif-3-ini-10.1177_17534259251372138 for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix by Kyo Hoon Park, Bo Young Choi, Kyong-No Lee, Eunji Oh, Min Jung Lee, Hee Young Cho and Da Eun Jeong in Innate Immunity
Supplemental Material
sj-tif-4-ini-10.1177_17534259251372138 - Supplemental material for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix
Supplemental material, sj-tif-4-ini-10.1177_17534259251372138 for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix by Kyo Hoon Park, Bo Young Choi, Kyong-No Lee, Eunji Oh, Min Jung Lee, Hee Young Cho and Da Eun Jeong in Innate Immunity
Supplemental Material
sj-tif-5-ini-10.1177_17534259251372138 - Supplemental material for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix
Supplemental material, sj-tif-5-ini-10.1177_17534259251372138 for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix by Kyo Hoon Park, Bo Young Choi, Kyong-No Lee, Eunji Oh, Min Jung Lee, Hee Young Cho and Da Eun Jeong in Innate Immunity
Supplemental Material
sj-sav-6-ini-10.1177_17534259251372138 - Supplemental material for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix
Supplemental material, sj-sav-6-ini-10.1177_17534259251372138 for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix by Kyo Hoon Park, Bo Young Choi, Kyong-No Lee, Eunji Oh, Min Jung Lee, Hee Young Cho and Da Eun Jeong in Innate Immunity
Supplemental Material
sj-docx-7-ini-10.1177_17534259251372138 - Supplemental material for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix
Supplemental material, sj-docx-7-ini-10.1177_17534259251372138 for Acute-phase response- and inflammation-, but not extracellular matrix-related proteins in the amniotic fluid are associated with spontaneous preterm delivery in asymptomatic women with midtrimester short cervix by Kyo Hoon Park, Bo Young Choi, Kyong-No Lee, Eunji Oh, Min Jung Lee, Hee Young Cho and Da Eun Jeong in Innate Immunity
Footnotes
Acknowledgments
The authors thank the Division of Statistics in the Medical Research Collaborating Center at Seoul National University Bundang Hospital for assistance in statistical analysis.
Ethics approval and consent to participate
This study received approval from the Institutional Review Board of the Ethics Committee of Seoul National University Bundang Hospital (approval no. B-1311/228-010). All participating women provided written informed consent for the amniocentesis and for the use of their biological specimens, and clinical and ultrasound data for research purposes.
Authors’ contributions
KH Park: conceptualization, protocol/project development, supervision, funding acquisition, data analysis, manuscript writing/editing; BY Choi: data collection or management, data analysis, manuscript writing/editing; KN Lee: data collection or management, data analysis.
E Oh: data collection or management, data analysis; MJ Lee: data collection or management, data analysis; HY Cho: data collection or management, data analysis, manuscript writing/editing; DE Jeong: data collection or management, data analysis. All authors approved the final version of the manuscript before submission
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Seoul National University Bundang Hospital Research Fund, (grant number 13-2024-0013).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and analyzed in the current study are available in the Supplementary File.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
