Abstract
The morphology of the choriocarcinomatous variant of cervical squamous cell carcinoma (SCC) suggests an undifferentiated aggressive biological behaviour and a poor outcome, for which standard treatment has not been established. In addition, cases are rarely reported, with only five cases of patients with cervical carcinoma with choriocarcinoma reported previously in the literature. This current case report describes in detail a patient who was diagnosed with cervical SCC mixed with choriocarcinomatous differentiation. The case report includes details of the diagnosis, pathology, short tandem repeat genotyping, treatment and follow-up of this patient. As there is no standard treatment for this variant, the patient underwent surgery followed by radiotherapy. Unfortunately, 4 months after therapy discontinuation, radiological evaluation and laboratory tests documented a recurrence of the disease and the patient died. This report also systematically reviews the literature on cervical cancer associated with choriocarcinomatous differentiation and the five previous cases. It provides the most up-to-date summary of this disease, including its clinical manifestations, histopathology, diagnosis, treatment and prognosis.
Keywords
Introduction
Cervical squamous cell carcinoma (SCC) combined with a choriocarcinomatous component is rarely encountered and diagnosed, but it is associated with an aggressive clinical course and an overall worse prognosis. To the best of our knowledge, there have only been five cases reported previously that showed the coexistence of cervical carcinoma with choriocarcinoma, of which the majority died within 4 months of therapy.1–5 Choriocarcinoma is a highly aggressive tumour, predominantly made up of syncytiotrophoblasts and cytotrophoblasts. Based on its origin, choriocarcinoma can be either gestational or non-gestational. Non-gestational choriocarcinoma can be of germ cell origin, or it can be seen in association with a somatic malignancy differentiation. 6 Given that the difficulty with differentiating gestational from non-gestational choriocarcinoma according to clinical presentation and histopathology, short tandem repeat (STR) genotyping is widely applied.6,7 A recent review reported that if the STR patterns of the tumour are matched to the genetic pool of the patient, then the diagnosis of non-gestational choriocarcinoma is confirmed. 8 This current case report presents a rare case in which the cervical SCC was mixed with choriocarcinoma dedifferentiated from SCC according to the histopathological features and STR genotyping. This report also systematically reviews the literature on cervical cancer associated with choriocarcinomatous differentiation and provides the most up-to-date summary of this disease, including its clinical manifestations, histopathology, diagnosis, treatment and prognosis.
Case report
In July 2022, a female in her late 70s presented to the Department of Oncology, Zhejiang University School of Medicine Women’s Hospital, Hangzhou, Zhejiang Province, China with erratic vaginal bleeding. She had a history of hypertension and type 2 diabetes mellitus. Her last pregnancy was 50 years ago. She had no history of molar pregnancy. Pelvic examination demonstrated a 4-cm protruding tumour mass occupying the uterine cervix with bleeding and necrotic tissue, without vaginal wall or parametria involvement. Results of the blood analysis demonstrated an increased level of serum β-human chorionic gonadotrophin (β-hCG) (2065.0 IU/l; normal, <5.3 IU/l) and no abnormalities of a range of tumour biomarkers that included cancer antigen (CA) 125, squamous cell carcinoma antigen, CA 19-9 and CA 15-3. A pap smear suggested a squamous cell carcinoma. The pelvic magnetic resonance imaging scan revealed a 1.9*4.9*2.5 cm mass in the endocervix that was invading the right uterine side and uterine isthmus wall, without spreading outside the cervix or showing any evidence of lymph node enlargement. Cervical biopsy indicated cervical malignancy, choriocarcinoma or squamous cell carcinoma, but it was not very clear. After extensive discussion, she underwent radical hysterectomy with bilateral salpingo-oophorectomy as well as bilateral pelvic lymphadenectomy.
The excised uterus contained a 3*2.5*1.5 cm necrotic cervical tumour, with a cauliflower-like appearance, invading the upper 2/3 of the vaginal canal. The parametria and bilateral adnexa were ordinary (Figure 1(a)). Five right pelvic and five left pelvic lymph nodes were identified with no enlargement. The microscopic findings of the surgical specimen illustrated an endocervical tumour with two distinct but intermingled components (Figure 1(b)). The major portion of the tumour consisted of a choriocarcinoma (approximately 80%) with multinucleate syncytiotrophoblast-like cells and mononuclear cells (Figure 1(c)); with the minor portion consisting of a squamous cell carcinoma with nonkeratinizing squamous cells, which showed irregular nests of cells and an infiltrating growth pattern (Figure 1(d)). Vascular space invasion was seen. There was no lymph node or parametrium involved. There was no evidence of germ cell tumour components.

Excised uterus from a female in her late 70s who presented with erratic vaginal bleeding and subsequently underwent a radical hysterectomy with bilateral salpingo-oophorectomy and bilateral pelvic lymphadenectomy. The excised uterus contained a 3*2.5*1.5 cm necrotic cervical tumour (a). There were two components present within the tumour: a squamous cell carcinoma (left) and choriocarcinoma (right) (b). The choriocarcinomatous component contained multinucleate syncytiotrophoblast-like cells and mononuclear cells (c). The squamous cell carcinoma contained nonkeratinizing squamous cells and had an infiltrating growth pattern (d). The colour version of this figure is available at: http://imr.sagepub.com. Haematoxylin and eosin, scale bar 50 µm.
Immunohistochemically, the choriocarcinoma component was positive for hCG and focally positive for GATA3 (Figure 2). The squamous cell carcinoma component showed positive staining for P40 and cytokeratin 5/6 (CK5/6), while negative staining for Sal-like protein 4 (SALL4) and P16 (Figure 3). According to the aforementioned histological results, a squamous cell carcinoma of the cervix mixed with a choriocarcinomatous carcinoma, IIA1 by The International Federation of Gynecology and Obstetrics classification, was the final diagnosis.

The excised uterus from a female in her late 70s who presented with erratic vaginal bleeding contained a 3*2.5*1.5 cm necrotic cervical tumour. The choriocarcinomatous component of that tumour stained positive for human chorionic gonadotrophin (hCG) and GATA3. The colour version of this figure is available at: http://imr.sagepub.com. Scale bar 50 µm.

The excised uterus from a female in her late 70s who presented with erratic vaginal bleeding contained a 3*2.5*1.5 cm necrotic cervical tumour. The squamous cell carcinoma component is positive staining for P40 and cytokeratin 5/6 (CK5/6), while negative staining for Sal-like protein 4 (SALL4) and P16. The colour version of this figure is available at: http://imr.sagepub.com. Scale bar 50 µm.
An RNAscope assay was then performed on a paraffin block, which was negative for human papillomavirus (HPV). For STR analysis, an endometrial section was selected as normal tissue. Designated areas of the choriocarcinoma and squamous cell carcinoma were microdissected from a 10-μm formalin-fixed, paraffin-embedded tissue block. Then the three types of tissue were placed into new microfuge tubes, followed by DNA extraction according to manufacturer’s instructions. STR genotyping was performed using 15 tetranucleotide repeat loci amplification with the AmpFLSTR® Identifiler® Plus PCR Amplification Kit (Invitrogen, Carlsbad, CA, USA) according to the protocol. The data showed that the DNA from the choriocarcinoma, squamous cell carcinoma and the normal tissue of the patient demonstrated the same genetic content at all loci, confirming a non-gestational choriocarcinoma (Figure 4(a)–(c)).

The excised uterus from a female in her late 70s who presented with erratic vaginal bleeding contained a 3*2.5*1.5 cm necrotic cervical tumour with squamous cell carcinoma and choriocarcinoma components. Genotyping of the two components of the tumour demonstrated that both the choriocarcinoma (a) and squamous cell carcinoma (b) elements matched the DNA from the patient (c). The colour version of this figure is available at: http://imr.sagepub.com.
The patient then received radiotherapy and follow-up. One month after the operation at the initial follow-up, the hCG had decreased to 395.0 IU/l. However, the HCG increased to 50347 IU/l at 4 months post-radiotherapy, indicating recurrence. At the same time, a computed tomography scan demonstrated multiple lung metastases and mediastinal lymphadenectasis. Unfortunately, the patient died 1 month later.
The reporting of this study conforms to CARE guidelines. 9 Written informed consent was obtained from the patient for publication of this case report and any accompanying images. The patient’s personal details have been de-identified for this case report. The patient provided written informed consent for treatment before the study. The report was approved by the ethics committee of Zhejiang University (Approval no: IRB-20230154-R).
Discussion
Choriocarcinoma is an aggressive, malignant trophoblastic neoplasm, which can be classified as either gestational or non-gestational. Gestational choriocarcinomas secondary to normal or abnormal pregnancy, are mostly well treated. Non-gestational choriocarcinoma is not gestational in origin and has been described at multiple sites, including the jejunum, 10 hypophysis, 11 ovary 12 and uterine cervix. 13 Notably, non-gestational choriocarcinoma of the uterine cervix is very rare and difficult to diagnose. The distinction between gestational and non-gestational choriocarcinomas is extremely important because the latter has a poor prognosis and needs more aggressive treatment. However, in most cases the distinction may not be clear based on clinical information and histological examination. To help resolve this problem, STR genotyping is widely used, 6 as non-gestational choriocarcinoma will have the same STR alleles as the patient. 14 In this current case, STR genotyping analysis demonstrated that the choriocarcinoma element matched the patient’s DNA at all loci, supporting a diagnosis of non-gestational choriocarcinoma.
Non-gestational choriocarcinomas fall into two types in origin. The first category arises from a germ cell tumour. 2 The second category of choriocarcinoma can arise from a metaplastic transformation of epithelial cells, which is associated with an aggressive biological behaviour and a poor outcome. 5 This dedifferentiation hypothesis has been proposed by many authors and described in many organs, including the stomach, 15 renal pelvis 16 ovary 17 and cervix. 3 In this current case, the neoplasm did not demonstrate the presence of other germ cell tumour components, such as yolk sac tumour, germinoma or teratoma, precluding the origin being from a germ cell tumour. Based on these considerations, the choriocarcinomatous element was speculated to have been derived from the squamous cell carcinoma, although there was no definite evidence of this transformation, which was a limitation of this investigation. Maybe the precise diagnostic criteria on which the diagnosis was based can be further explored and improved. Squamous cell carcinoma of the uterine cervix is the most common histological variant of cervical cancer. 18 The World Health Organization (WHO) Classification of gynaecological tumours describes several histomorphological variants of cervical SCC, including squamotransitional and lymphoepithelioma-like. 19 However, the WHO Classification does not describe the rare choriocarcinomatous variant with SCC. To the best of our knowledge, only one previous report has demonstrated a case of cervical SCC with a small elevation of hCG (8 IU/l); 3 and the authors also found that the tumour was immunohistochemically positive for hCG, but clear histopathological features of choriocarcinoma were not described. The report also lacked the detection of HPV and molecular genotyping. 3 This current case report describes an unusual type of uterine cervical SCC with choriocarcinomatous variant supported by genetic studies, histological evidence, immunohistochemical staining, serum beta-hCG and an inter-institutional pathological panel consensus.
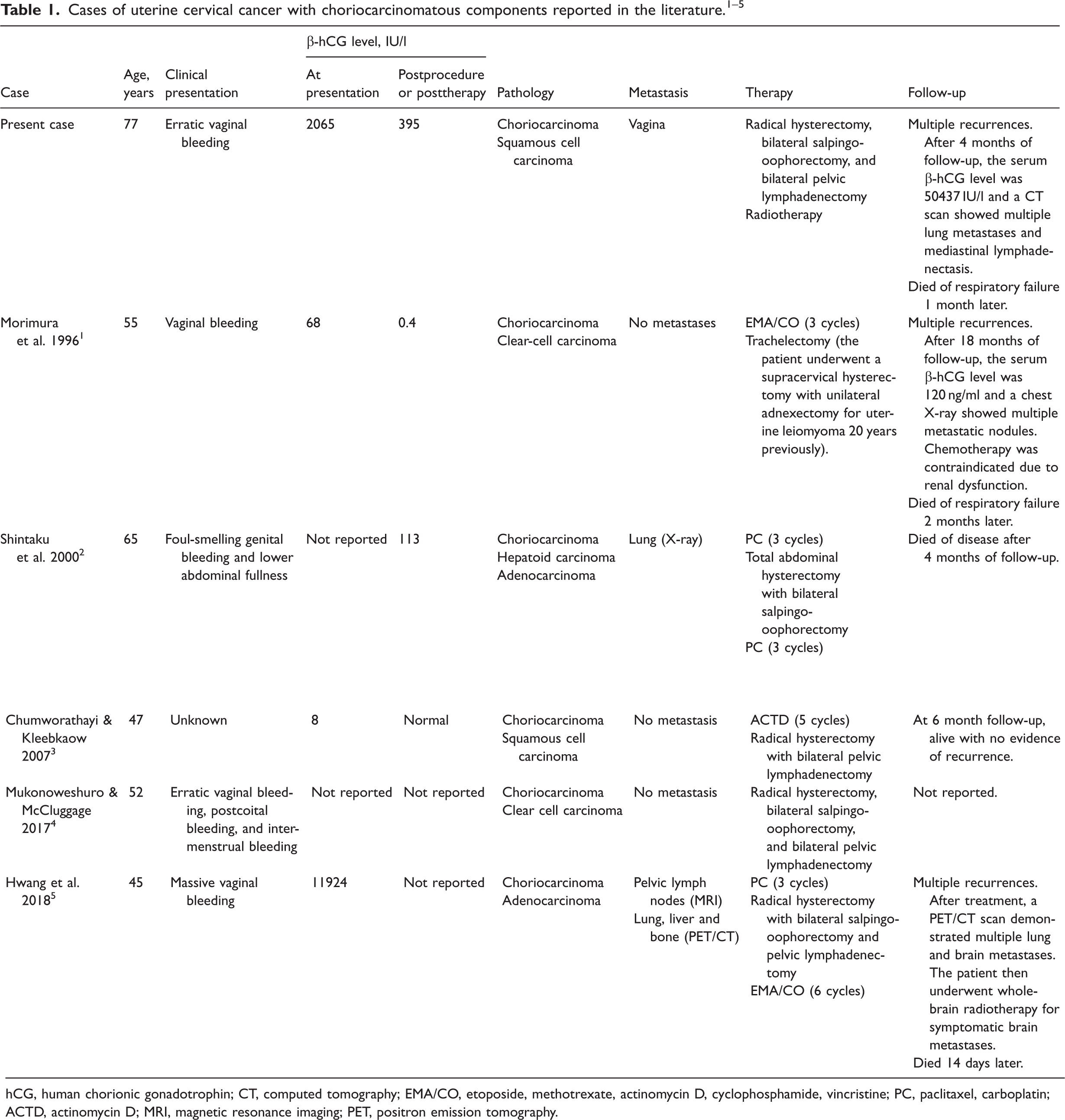
The morphology of the choriocarcinomatous variant of cervical SCC suggests an undifferentiated aggressive biological behaviour and a poor outcome, for which standard treatment has not been established. Although it is difficult to draw conclusions about the best therapy for cervical SCC with choriocarcinomatous variant, surgery is generally acknowledged as an important option because of the potential size of the tumour and its poor response to chemotherapy, as shown by previous reports.3,4 There are a number of unanswered questions. For example, do these patients need subsequent radiotherapy or chemotherapy? Which is the preferred treatment approach? If chemotherapy is used, how should the agents be chosen? These issues remain controversial. To have a more thorough understanding, the previous five cases were reviewed in detail and their treatment is summarized in Table 1.1–5 Except for one patient that underwent surgery and was subsequently lost to follow-up, all of the other four patients received chemotherapy with different regimens, including etoposide/methotrexate/actinomycin D/cyclophosphamide/ vincristine (EMA/CO), paclitaxel/carboplatin (PC) and actinomycin D (ACTD). Of note, most of them died within 4 months, except a 47-year-old woman who presented with a non-gestational choriocarcinoma of the cervix associated with SCC, who underwent five cycles ACTD chemotherapy and radical hysterectomy with bilateral pelvic lymphadenectomy. 3 She was alive with no evidence of recurrence at the 6-month follow-up. 3 In the current patient, the cervical biopsy indicated a tumour and a pap smear suggested an SSC, so all of the current authors agreed to undertake surgery first. However, after receiving the results of the pathology, there was an intense discussion. Some of the authors suggested chemotherapy with ACTD because of the findings in the previous patient with choriocarcinoma and SCC of the cervix that had no evidence of recurrence after 5 cycles of ACTD. 3 However, most of the authors had the opposite opinion, because there is no evidence that ACTD is helpful for SCC, especially as the choriocarcinomatous element was regarded as an aberrant dedifferentiation of the SCC. The authors agreed to treat the tumour as cervical cancer. To this end, the current authors agreed with a previous report that treated a similar case as a cervical cancer; 2 however, radiotherapy was used rather than the PC regimen as a first line treatment of cervical cancer. Unfortunately, 4 months after therapy discontinuation, radiological evaluation and laboratory tests documented a recurrence of the disease. These current findings suggest that it might not be enough to treat SCC with choriocarcinoma according to cervical cancer standard therapy. The identification of optimal treatment regimens requires further treatment and reports of patients with these rare malignancies.
hCG, human chorionic gonadotrophin; CT, computed tomography; EMA/CO, etoposide, methotrexate, actinomycin D, cyclophosphamide, vincristine; PC, paclitaxel, carboplatin; ACTD, actinomycin D; MRI, magnetic resonance imaging; PET, positron emission tomography.
Footnotes
Acknowledgement
The authors thank the patient for participating in the study.
Author contributions
F.W., S.X. and Y.C. drafted the manuscript. Y.Q. and X.Z. revised the manuscript content. Y.C. approved the final version of the manuscript. All authors have read and approved the final version of the manuscript.
Data availability statement
All data are available upon reasonable request from the corresponding author.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research was supported by the Zhejiang Natural Science Foundation (no. LY21H160032).