Abstract
Background
Previous studies suggest that transient receptor potential melastatin 2 (TRPM2) plays a protective role in sepsis by enhancing bacterial clearance. This effect is mediated through the modulation of macrophage phenotypic changes, which strengthen the immune response against infection. However, the specific role and underlying mechanism of TRPM2 in macrophage polarization during sepsis remain unclear.
Method
Cecal ligation and puncture (CLP) was used to establish a mouse sepsis model, and bone marrow–derived macrophages (BMDMs) and peritoneal macrophages were prepared from C57BL/6 wild-type and TRPM2 knockout (trpm2−/−) mice. IPI549 was utilized as a specific inhibitor of PI3K. Macrophage polarization, bactericidal ability, and the PI3K/protein kinase B (AKT)/cyclic adenosine monophosphate response element-binding protein signaling pathway were assessed. In addition, survival rate, bacterial burden, lung wet/dry weight ratio, lung and liver injury scores, and cytokine levels were measured in CLP-induced septic mice.
Results
In lipopolysaccharide (LPS)-stimulated BMDMs, trpm2 deficiency increased the expression of characteristic markers associated with the M2b phenotype, reduced the bactericidal ability, and activated the PI3K/AKT/CREB signaling pathway. Consequently, both trpm2−/− BMDMs and trpm2−/− mice exhibited impaired bactericidal clearance during CLP-induced sepsis. Furthermore, IPI549 attenuated TRPM2 deletion-induced M2b polarization and restored the bactericidal function of BMDMs. Notably, IPI549 preconditioning reversed the increased susceptibility of the trpm2−/− mice to sepsis. The 7-day mortality rate was 92% in trpm2−/− mice, compared to 42% in IPI549-pretreated trpm2−/− mice. Moreover, IPI549-treated mice exhibited improved lung wet/dry ratios, reduced lung and liver injury scores, reversed M2b polarization and decreased bacterial load.
Conclusion
The PI3K/AKT/CREB pathway mediates the effect of TRPM2 by inhibiting M2b macrophage polarization and promoting bacterial clearance during sepsis.
Introduction
Sepsis is characterized by detrimental organ dysfunction resulting from an imbalanced host inflammatory response to infection, often leading to significant mortality and morbidity. 1 Septic shock represents a severe manifestation of sepsis, where metabolic and cellular changes substantially elevate the risk of a fatal outcome. 2 According to the World Health Organization, approximately 6 million people die from septic shock annually, with this number continuing to rise each year. 3 Furthermore, growing evidence highlights the critical role of immunosuppression in sepsis, which is associated with increased nosocomial infections and mortality rates.4,5
Macrophages play a pivotal role in the immune response to sepsis, particularly in infections caused by gram-negative bacteria, such as Escherichia coli. Lipopolysaccharide, a primary component of the outer membrane of Gram-negative bacteria, binds to toll-like receptor 4 (TLR-4) triggering an inflammatory response. 6 Classically activated macrophages (M1) release numerous pro-inflammatory mediators that support Th1 and Th17 immune responses, which are crucial for eradicating microbial threats. 7 In contrast, alternatively activated macrophages(M2s) are further categorized into several subtypes, including M2a, M2b, M2c, and M2d, each with distinct morphological and functional characteristics. 8 These macrophage subtypes participate in immune regulation, inflammation and tissue remodeling.9,10 The PI3Kγ/protein kinase B (AKT) signaling pathway plays a crucial role in promoting M2 polarization and immunosuppression in macrophages.11,12 Certain M2 subtypes, particularly M2b macrophages, have been demonstrated to reduce bacterial clearance and contribute to bacterial persistence in inflammatory diseases. 13 Activation of the PI3Kγ/AKT pathway has also been demonstrated to promote M2b polarization in alveolar macrophages during influenza viruse infection. 14
Transient Receptor Potential Melastatin 2 (TRPM2) is a nonselective, Ca2+-permeable channel that is abundantly expressed in macrophage membranes.15,16 Our previous studies have explored the role of TRPM2 in polymicrobial sepsis, demonstrating its regulation of Ca2+ influx and heme oxygenase-1 (HO-1) in TRPM2 knockout (KO) bone marrow-derived macrophages (BMDMs) and mouse model.17–19 These studies suggest that TRPM2 upregulation may play a protective role in polymicrobial sepsis by promoting bacterial clearance in macrophages. Meanwhile, TRPM2 has been shown to be crucial for survival and bacterial burden control during Listeria monocytogenes infection. 20 However, the precise role and underlying mechanism of TRPM2 in macrophage polarization and bacterial clearance during sepsis remain unclear.
In this study, we hypothesize that TRPM2 knockout induces M2b macrophage polarization by activating the PI3K/AKT/cyclic adenosine monophosphate response element-binding protein pathway, leading to increased bacterial burden and decreased survival in septic mice.
Materials and methods
Mice
TRPM2 knockout(trpm2−/−) mice were bred at the Animal Care Centre (Zhejiang University, Hangzhou, China), and age-matched male wild-type (C57BL/6J) mice were purchased from SLAC Biotechnology (Shanghai, China). All the animals were housed in specific pathogens free-facilities, with unrestricted access to food and water, and maintained on a 12-h light/dark cycle. The male mice were aged 7 to 9 weeks, weighted between 22 and 30 grams. This study adhered to the ARRIVE guidelines (https://arriveguidelines.org), and all the animal experiments were approved by the Ethics Committee of Zhejiang University, in compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (No. ZJU20210125).
Sepsis model
The cecal ligation and puncture (CLP) model was generated following a previously established protocol. 17 In brief, mice were anesthetized using intraperitoneal injection of 80 mg/kg pentobarbital, and a 1-cm midline laparotomy incision was made to expose the cecum. The cecum was then ligated and punctured with a 21-gauge needle at a point equidistant between the distal pole and the ileocecal junction, followed by gentle squeezing to expel a small quantity of fecal material. After repositioning the cecum, the abdominal cavity was closed. Mice in the sham CLP group underwent the same procedure, excluding the ligation and puncture steps. In the CLP + IPI-549 group, 1 mg/kg IPI-549 (Selleck, Houston, USA) was administered 2 h before the CLP operation. After the surgical procedure, each mouse received 1 ml of normal saline subcutaneously. The survival rate was monitored over a period of 7 days, and other in vivo experiments were performed 48 h after the procedure. At the end of experiment, euthanasia was performed by inhalation of carbon dioxide, followed by cervical dislocation and execution. To minimize bias, all further experiments were conducted with blinding to murine genotype and treatment.
Cell culture
Bone marrow-derived macrophages (BMDMs) were isolated from both the femurs and tibias of WT and trpm2−/− mice and then cultured in Dulbecco's modified Eagle medium (DMEM) (Cellgro, Corning, USA) supplemented with 20% heat-inactivated fetal bovine serum (FBS) (Cellgro, Corning, USA), 5% penicillin and streptomycin, and 20% granulocyte-macrophage colony-stimulating factor (GM-CSF) (acquired from the L929 culture medium). The cells were counted with a hemocytometer and seeded into a 10-cm dish at a density of 6.0 × 106 cells per dish. 6–7 ml old medium was discarded and replaced with fresh complete DMEM medium (20% FBS, 5% penicillin and streptomycin) with 20% GM-CSF on days 3 and 5. On day 7, adherent cells were harvested using Trypsin-ethylenediaminetetraacetic acid (EDTA) (0.25% trypsin and 0.02% EDTA, Keyi Co., Ltd, China) for a short duration (≤5 min at 37°C), then collected into 15 ml tubes and centrifuged at 1200×g for 5 min. The supernatant was removed, and the cells were resuspended and seeded into both 6-well and 24-well plates, then incubated overnight at 37°C in a 5% CO2 incubator. Then, the cells were treated with lipopolysaccharide (LPS) or Escherichia coli (E.coli )for the subsequent experiment.
Bacterial killing test
BMDMs were seeded into a 24-well plate at a density of 2 × 105 cells/well, and then incubated at 37°C for 2 h after adding E. coli (DH5ɑ) with multiplicity of infection(MOI) of 20. The cells were washed twice with phosphate buffered saline(PBS), and then gentamicin (100 μg/ml) was added to the medium to eliminate extracellular bacteria. After 6 h, the cells were re-washed with PBS and lysed with 500 μl of 0.1% Triton X-100 for 5 min. The cell lysates were serially diluted with 1× PBS, cultured on Luria broth agar plates, which were incubated for another 12 h at 37°C to determine the bacterial killing capacity by counting the colony-forming units (CFUs). For some experiments, the cells were pretreated with IPI-549 for 15 min. After the incubation periods, the supernatant was removed, and the cells were washed for subsequent experiments.
Peritoneal macrophage isolation and peritoneal lavage fluid (PLF) preparation
After 48 h of CLP, euthanasia was performed on the mice. A 25-gauge needle, attached with a vascular clamp, was inserted into the peritoneal cavity, and 5 ml cold 1× PBS was infused. The entire body was gently shaken for 10 s, and then the peritoneal lavage fluid (PLF) was collected. After centrifugation, the supernatant was discarded. The pellet was resuspended and plated in complete DMEM supplemented with 10% FBS (Cellgro, Corning, USA) and 5% penicillin/streptomycin. After 2 h of incubation in a 37°C, 5% CO2 incubator, the supernatant was discarded, and the adherent cells (considered to be macrophages) were washed twice with 1 × PBS. The macrophages were then harvested for Western blot experiments.
Determination of bacterial burden
The protocol for determining the bacterial burden was described in our previous study. 17 PLF was collected according to the above method, and 100 μl of PLF was plated on tryptic soy agar plates. The plates were incubated at 37°C for 12 h, after which CFUs were counted. The bacterial burden is presented as CFU per milliliter of PLF.
Hematoxylin-eosin (HE) staining and tissue histological analysis
Lung and liver tissues were fixed in 4% paraformaldehyde for 48 h and then embedded in paraffin after dehydration and clarification. Four μm thick slices were prepared and stained with H&E staining solution (H&E solution; Nanjingjiancheng, China). The slides were examined under a VS 200 digital slide scanner (Olympus, Tokyo, Japan). A scoring system was used to evaluate lung injury based on indicators such as capillary congestion, alveolar congestion, leukocyte infiltration, and alveolar wall thickness: 0 = normal, 1 = mild injury (<25%), 2 = moderate injury (25–50%), 3 = severe injury (50–75%), 4 = very severe injury (>75%). 21 Liver damage was assessed using a similar scale based on necrosis, characterized by loss of architecture, vacuolization, and karyolysis: 0 = normal, 1 = mild injury, 2 = moderate injury, 3 = severe injury, 4 = complete necrosis. 22
Western blot
BMDMs and peritoneal macrophages were lysed with radioimmunoprecipitation assay buffer (Boster, China) containing PhosSTOP (Roche), protease inhibitor cocktail (Roche) and phenylmethylsulfonyl fluoride (Sigma). Protein concentrations were determined, and 40 μg of protein was loaded onto 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The proteins were separated and transferred to nitrocellulose membranes (BioTrace) for 90 min at 300 mA using a Mini-protein II system (Bio-Rad). Later, the membranes were blocked with tris-buffered saline with 0.1% Tween20 detergent (TBST) containing 5% bovine serum albumin for 1 h at room temperature, followed by overnight incubation at 4°C with specific primary antibodies. Details of the primary antibodies are as follows:
Glyceraldehyde-3-phosphate dehydrogenase(GAPDH) (1:5000, db106, Digbio, China), p-AKT (Ser473) (1:1000, #4060, CST, USA), AKT (1:1000, ab1607, Abcam, USA), p-CREB (Ser133) (1:1000, AF3189, Affinity Biosciences, USA), CREB (1:1000, 12208-1-AP, Proteintech, China).
Afterwards, the membranes were washed six times with 1× TBST and incubated with the appropriate secondary antibodies, IRDye 800CW goat anti-rabbit or IRDye 680CW goat anti-mouse (1:5000, LI-COR Biosciences, USA), for 2 h. The protein bands were visualized using a LI-COR Odyssey infrared imaging system (LI-COR, USA).
Quantitative real-time polymerase chain reaction (qRT–PCR)
Total RNA was isolated from BMDMs and peritoneal macrophages using isoPlus (9109, Takara, Japan). The extracted RNA was reverse transcribed to first-strand cDNA. SYBR Green Master Mix reagent (Takara, Japan) was used for qRT–PCR analysis. GAPDH was used as a normalization control. The PCR mixture consisted of 4.5 μl of cDNA, 5 μL of 2× SYBR Green mix, 0.25 μ of forward primer and reverse primer. The qRT–PCR process was performed under the following thermal cycler program: 95°C for 10 min; 45 cycles of 95°C for 10 s, 60°C for 20 s, and 72°C for 30 s. All the primers used in this study were synthesized by Sangon Biotech (Shanghai, China). The sequence of the primers listed in Table 1.
Enzyme-linked immunosorbent assay (ELISA)
To determine the levels of cytokines and chemokines, the supernatants from the BMDMs and peritoneal macrophage culture medium were collected. The level of secreted cytokines were detected using ELISA kits. Specifically, tumor necrosis factor alpha (TNF-α), interleukin10 (IL-10), interleukin 6 (IL-6), interleukin12 (IL-12), and C-C motif chemokine ligand 1 (CCL1) were measured using kits from various manufacturers as follows: TNF-α (DY410), IL-10 (DY417) and IL-6 (DY406) from R&D Systems, Minneapolis, MN, USA; IL-12 (431604) from BioLegend, USA; and CCL1 (L210919515) from Cloud-Clone Corp., China. The measurements were done in accordance with the manufacturers instructions, and the optical density was measured at 450/570 nm. The optical density values were normalized to the blank well, and the results are expressed as pg/ml.
Statistical analysis
All the data are presented as the mean ± SD and represent more than three independent experiments. All the statistical analyses were performed using GraphPad Prism 9.0. Two-way ANOVA followed by Bonferroni's post hoc test was used for multiple comparisons. Student's t test was used for unpaired observations. Survival was analyzed by the Kaplan–Meier method and log-rank test. A value of P < 0.05 was considered to indicate statistical significance.
Results
TRPM2 deficiency promotes M2b polarization in BMDMs stimulated with LPS
Previous studies have shown that the biomarkers, such as CCL1 and IL-10, are more prominently in M2b macrophages compared to other macrophage subtypes.
23
Therefore, CCL1 plays a key role in maintaining the unique characteristics of M2b macrophages.9,24 In the current study, qRT-PCR analysis revealed that the expression of M1 markers, such as inducible nitric oxide synthase (iNOS), IL-12, was significantly lower in trpm2−/− BMDMs than in WT-BMDMs after LPS stimulation

Trpm2−/− BMDMs were prone to M2b-type macrophage polarization after LPS stimulation. BMDMs were isolated from wild-type (WT) and trpm2−/− mice and stimulated with 100 ng/ml LPS at different time points. (A) The mRNA expression levels of iNOS, IL-12, CCL1, IL-10, IL-6, and TNF-α in BMDMs were assessed via qRT–PCR and normalized to that of GAPDH. (B) The concentrations of CCL1, IL-10, IL-12, and TNF-α in the culture medium of BMDMs were measured using ELISA. The data are presented as the means ± SD (n = 3). Two-way ANOVA followed by Bonferroni’s post hoc test was used for multiple group comparisons. *p < 0.05, **p < 0.01, ***p < 0.001 vs WT mice.
TRPM2 deficiency activates the PI3K/AKT/CREB signaling pathway and M2b polarization in BMDMs
M2b macrophage polarization is regulated by several signaling pathways, including the PI3K/AKT pathway and the transcription factor CREB.11,12 To understand how TRPM2 impacts macrophage polarization, we examined the expression of AKT and CREB in BMDMs from both trpm2−/− and WT mice, which were stimulated with LPS. Notably, the ratios of p-CREB/CREB and p-AKT/AKT were higher in the trpm2-/- BMDMs compared to WT BMDMs following LPS stimulation. To investigate the relationship between PI3K pathway and TRPM2, we used the PI3K inhibitor IPI-549 to block PI3K/AKT signaling. Our results revealed that pretreatment with IPI-549 significantly reversed the p-AKT/AKT and p-CREB/CREB ratios (Figure 2). These findings indicate a potential correlation between TRPM2 and the PI3K/AKT/CREB pathway.
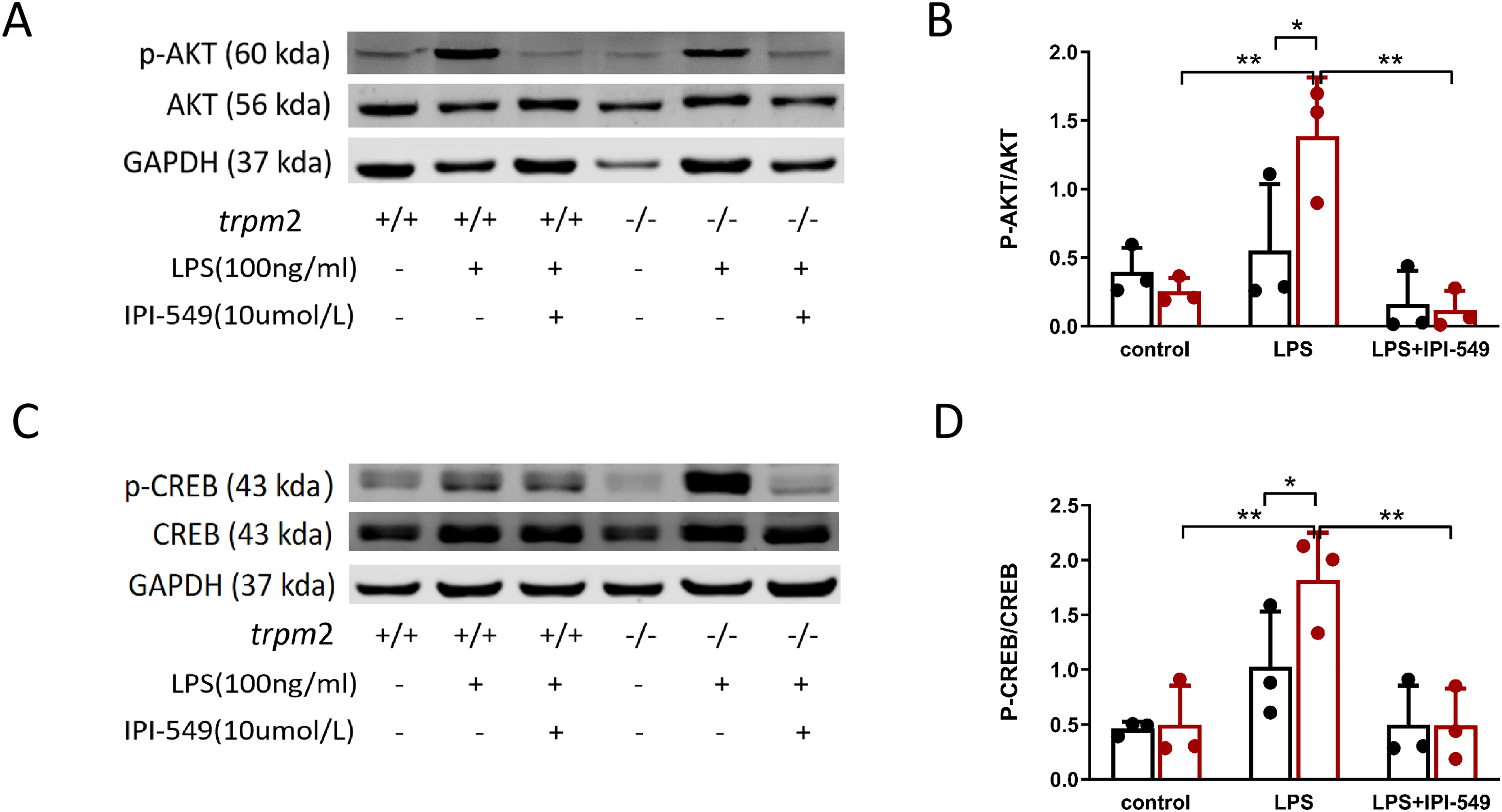
Inhibition of PI3K-γ reversed the change of the AKT/CREB pathway in trpm2−/− BMDMs. BMDMs from WT and trpm2−/− mice were pretreated with IPI-549 15 min before stimulation with 100 ng/ml LPS, and determined 30 min after LPS stimulation via western blotting. (A–B) The protein expression and densitometric quantification of AKT, p-AKT. (C–D) The protein expression and densitometric quantification of CREB, and p-CREB. The data are presented as the means ± SD (n = 3–4). Two-way ANOVA followed by Bonferroni’s post hoc test was used for multiple group comparisons. *p < 0.05, **p < 0.01, ***p < 0.001.
To further investigate the effects of TRPM2 and the PI3K/AKT/CREB pathway on M2b polarization, we assessed the mRNA expression of iNOS, IL-12, CCL1 and IL-10 (Figure 3A–D) and the secretory proteins IL-12, CCL1 and IL-10 (Figure 3E–G) by ELISA and qRT–PCR. We found that the expression of typical M2b markers, including CCL1 and IL-10, was dramatically downregulated, while the expression of the M1 markers iNOS and IL-12 was mildly upregulated in the trpm2−/− BMDMs compared with LPS-treated trpm2−/− BMDMs. These results suggest that the M2b markers CCL1 and IL-10 are partly attenuated through the inhibition of the AKT/CREB pathway in trpm2−/− BMDMs.
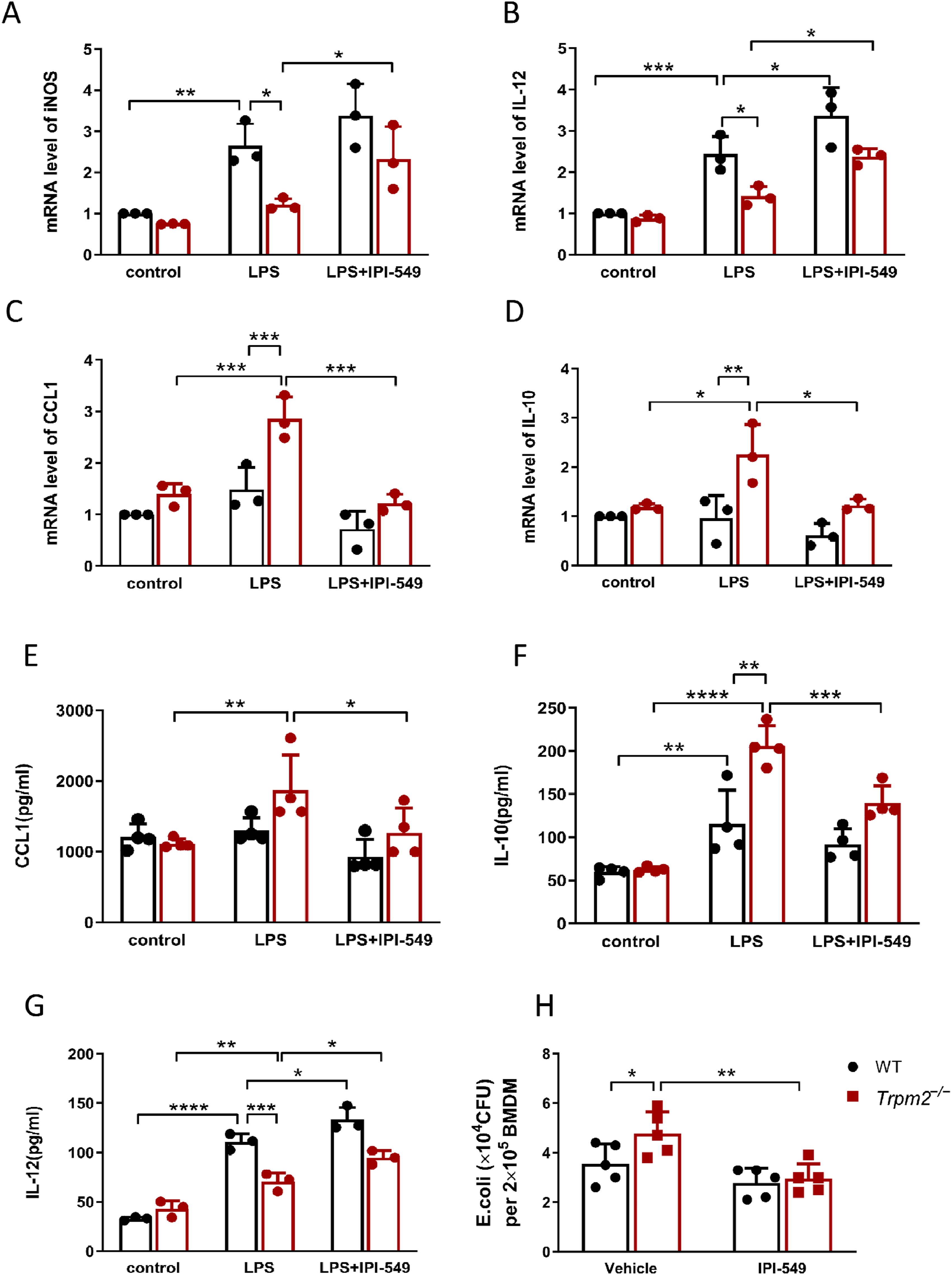
The PI3K inhibitor IPI-549 affect macrophage polarization and bacterial burdens in BMDMs from WT and trpm2−/− mice. (A–G) Cells were treated with DMSO or IPI-549 for 30 min, then stimulated with 100 ng/ml LPS, cells and supernatants were collected after 12 h. (A–C) mRNA expression of M1/M2b markers in the BMDMs. (D–G) Cytokine, CCL1, IL-10, and IL-12 levels in the supernatants. (H) Bacterial burdens of BMDMs from WT and trpm2−/− mice. Cells were treated with treated with DMSO or IPI-549 (10 μmol/ml) for 30 min, and subsequently challenged with Escherichia coli (DH5ɑ) for 1 h at 37°C (MOI = 20). Then, gentamicin (100 μg/ml) was added to the cells for 6 h, after which the cell lysates were inoculated on agar plates for 12 h, after which the colony-forming units (CFU) were counted. The data are presented as the means ± SD (n = 3–4). Two-way ANOVA followed by Bonferroni’s post hoc test was used for multiple group comparisons. *p < 0.05, **p < 0.01, ***p < 0.001.
PI3K inhibition enhances bacterial clearance in trpm2−/− BMDMs
Our previous study confirmed that there is no significant difference in the phagocytic capacity of peritoneal macrophages between trpm2+/+ and trpm2−/− mice,
24
and TRPM2 participates in intracellular bacterial elimination of peritoneal macrophages. To determine whether the TRPM2-regulated PI3K/AKT/CREB pathway influences the bactericidal activity of macrophages, we performed the intracellular bacterial killing assay. Consistent with our earlier findings, trpm2−/− BMDMs exhibited a higher bacterial load than WT BMDMs. Furthermore, treatment with the PI3K inhibitor IPI-549 reduced the bacterial burden in trpm2−/− BMDMs compared to untreated trpm2−/− BMDMs
PI3K inhibition reduces bacterial burden and mortality in trpm2−/− mice after polymicrobial sepsis
Our previous studies demonstrated that TRPM2 plays an essential role in controlling bacterial clearance and host defense during polymicrobial sepsis. 15 To further validate the involvement of the TRPM2-mediated bacterial clearance pathway via the PI3K/AKT/CREB pathway in the CLP model, we utilized the PI3K inhibitor IPI-549 to assess its impact on bacterial clearance in septic trpm2−/− mice. Remarkably, trpm2−/− mice treated with IPI-549 exhibited a significant decrease in bacterial load in the PLF at 48 h after CLP surgery (Figure 4A). Moreover, the survival rate was higher in trpm2−/− mice treated with IPI-549 compared to those treated with a vehicle control (Figure 4B).

The PI3K inhibitor IPI-549 improved survival by promoting bacterial clearance in trpm2−/− mice after CLP. (A) At 48 h after CLP, bacterial burdens were detected by plating serial dilutions of peritoneal lavage fluid (PLF) on agar plates. CFUs were counted 12 h after inoculation on agar plates. Each dot represents the CFU of one mouse. The horizontal bars represent the means (n = 6). *p < 0.05, Two-way ANOVA followed by Bonferroni’s post hoc test was used for multiple group comparisons. (B) Survival was monitored for 7 days (n = 12). *p < 0.05, Kaplan–Meier log-rank test.
PI3K inhibition partly mitigates tissue damage in trpm2−/− mice after polymicrobial sepsis
To investigate the pathological role of TRPM2 in the PI3K/AKT/CREB pathway during CLP-induced sepsis, we performed hematoxylin and eosin (H&E) staining to assess lung and liver injuries (Figure 5A). Notably, several indicators of organ injury, including lung injury score, total protein concentration in bronchoalveolar lavage fluid, wet-to-dry weight ratio of lung tissue, liver injury score, and serum alanine aminotransferase concentration (ALT) levels, were significantly decreased in trpm2−/− mice following treatment with the PI3K inhibitor. (Figure 5B and C). Taken together, these results suggest that the PI3K inhibitor improved pathological parameters and reduced mortality in septic trpm2−/− mice.

The PI3K inhibitor IPI-549 attenuates organ injury in trpm2−/− mice after CLP wt and trpm2−/− mice were intraperitoneally injected with 1 mg/kg IPI-549 or vehicle 30 min before CLP. Mice were sacrificed at 48 h after CLP. (A) Lungs and livers were collected for hematoxylin and eosin staining (original magnification, 20×; scale bar, 20 µm). Green arrow points out the neutrophil infitration in the lung . (B) The lung injury score (n = 5), total protein concentration in bronchoalveolar lavage fluid (n = 5), and lung wet-to-dry weight ratio (n = 4) represent the severity of lung injury. (C) The liver injury score (n = 5) and serum alanine aminotransferase concentration (ALT) (n = 4) represent the severity of liver injury. The data represent the means ± SD. Two-way ANOVA followed by Bonferroni’s post hoc test was used for multiple group comparisons. *p < 0.05, **p < 0.01, ***p < 0.001.
PI3K mediated TRPM2 deficiency induced M2b polarization in peritoneal macrophages from septic mice
To confirm these findings in vivo, we developed a CLP model to explore whether the PI3K/AKT/CREB pathway mediates the contribution of TRPM2 to macrophage polarization. At 48 h after CLP, the mRNA expression of M2b markers, including CCL1 and IL-10, was significantly higher in trpm2−/− mice compared to the WT group. Pretreatment with IPI-549 in trpm2−/− mice decreased the mRNA levels of M2b markers (Figure 6A–C). Similarly, the levels of CCL1, IL-10, and IL-12 in the supernatants followed the same trend (Figure 6D–F). Furthermore, protein expression of p-AKT and p-CREB in peritoneal macrophages 48 h after CLP was markedly upregulated in the trpm2−/− macrophages (Figure 7A and B). These findings suggest that TRPM2 deficiency may promote M2b-type macrophage polarization by enhanced phosphorylation of AKT and CREB.

M2b polarization was exhibited in peritoneal macrophages from trpm2−/− mice in the CLP model (A–C) mRNA expression of IL-12 (n = 3), Il-12, CCL1, and IL-10 (n = 5) in peritoneal macrophages 48 h after CLP and quantitative analysis via qRT–PCR. (D–F) Cytokine, CCL1, IL-10, and IL-12 levels in the supernatants were assessed via ELISA. The data represent the means ± SD. Two-way ANOVA followed by Bonferroni’s post hoc test was used for multiple group comparisons. *p < 0.05, **p < 0.01, ***p < 0.001.

PI3K/AKT/CREB pathway mediated M2b polarization in peritoneal macrophages from trpm2−/− mice in the CLP model, and the phenotypic shift from M2b to M1 was facilitated by the PI3Kγ inhibitor. (A) The pres The levels of AKT, phosphorylated AKT (n = 3) (B), CREB, and phosphorylated CREB (n = 4) (C)were detected by immunoblotting, and analysis. The data represent the means ± SD. Two-way ANOVA followed by a Bonferroni comparison test was used. *p < 0.05, **p < 0.01, ***p < 0.001.
Discussion
In the present study, we investigated the novel signaling pathway of TRPM2 in sepsis models both in vitro and in vivo. First, BMDMs derived from trpm2−/− mice expressed higher levels of M2b biomarkers upon LPS stimulation and activated the PI3K/AKT/CREB signaling pathway. Second, pretreatment with IPI-549 significantly mitigated M2b polarization and increased bactericidal ability in trpm2−/− BMDMs. Third, we revealed that M2b polarization mediated by the PI3K/AKT/CREB signaling pathway was elicited in peritoneal macrophages from CLP-induced septic mice. Finally, IPI-549 pretreatment enhanced the bacterial clearance capacity and survival in septic trpm2−/− mice. Collectively, our results suggested that TRPM2 deficiency impairs bacterial clearance during polymicrobial sepsis by promoting M2b macrophage polarization via the PI3K/AKT/CREB signaling pathway.
Macrophages are widely distributed innate immune cells that play an indispensable role in sepsis, and can be polarized into two main groups (M1 and M2), M2 are further subdivided into M2a, M2b, M2c, and M2d subtypes, in response to pathogen invasion. 25 Functionally, M1 macrophages contribute to immunity by eliminating foreign pathogens and tumor cells, but they also mediate tissue damage induced by reactive oxygen species, which can impede wound healing and tissue regeneration. 7 In contrast, M2 macrophages, particularly the M2b subtype, are associated with significantly reduced bacterial clearance, leading to increased mortality and nosocomial infections in patients with sepsis. 26 M2a macrophages express high levels of the mannose receptor and secrete pro-fibrotic factors, such as insulin-like growth factor (IGF) and fibronectin, which contribute to tissue repair during wound healing. 27 M2c subtype of macrophages exhibits potent anti-inflammatory activities by releasing large amounts of IL-10 and pro-fibrotic activity through secretion of transforming growth factor beta. 28 Additionally, M2c macrophages express high levels of Mer receptor tyrosine kinase (MerTK), which facilitates efficient phagocytosis of apoptotic cells. 9 M2d macrophages constitute the major inflammatory component in neoplastic tissue, where they contribute to angiogenesis and cancer metastasis as tumor-associated macrophages. 29
As an M2 subtype, M2b macrophages are characterized by heightened expression of CCL1 and secrete substantial quantities of the anti-inflammatory cytokine IL-10, alongside diminished secretion of IL-12, which contrasts with the functional characteristics observed in M1 macrophages.9,30 IL-10 serves as a specific marker of M2b cells, 9 involved in sepsis-induced immunosuppression. 31 Notably, phosphorylated CREB can bind to the regulatory domain of the IL-10 promoter, thereby activating IL-10 production. 32 In contrast, the bactericidal activity of M1 macrophages, the primary macrophage subtype responsible for killing bacteria, is impaired when the host harbors an abundance of M2b macrophages.33,34 Kobayashi et al. proposed that M2b macrophages from mesenteric lymph nodes displayed compromised antibacterial resistance in gamma-irradiated hosts, suggesting that targeting M2b macrophages could effectively regulate bacterial translocation and subsequent sepsis. 35 Furthermore, Ohama et al. found that elimination of M2b macrophages could enhance the resistance of mice with chronic alcohol consumption to opportunistic infections. 36 Our results demonstrate that BMDMs derived from trpm2−/− mice exhibit elevated expression levels of M2b biomarkers following LPS stimulation. Moreover, inhibition of macrophage polarization towards M2b phenotype enhances bacterial clearance and improves the survival rate of septic trpm2−/− mice.
Interestingly, Heather Knowles et al. reported that trpm2−/− mice exhibited uncontrolled bacterial growth and reduced numbers of iNOS-expressing monocytes following Listeria monocytogenes infection, which correlated with impaired innate immune system activation. 20 Consistently, our results indicated that TRPM2 protects mice from polymicrobial sepsis, partly by preventing immune paralysis and the recurrence of severe infection. In contrast, Beceiro et al. reported that deficiency of TRPM2 can induce polarization of M1 macrophages and exacerbate inflammation in the gastric mucosa. Importantly, chronically H. pylori-infected tpm2−/− mice exhibit augmented production of inflammatory cytokines and enhanced nicotinamide adenine dinucleotide phosphate oxidase activity. Compared with WT BMDMs, trpm2−/− macrophages showed significantly higher the expression of M1-associated markers, including IL-6, TNF-α and iNOS, after stimulation with LPS plus IFN-γ. In turn, reduced Helicobacter pylori gastric colonization and increased the inflammation of the gastric mucosa. 37 These discrepancies regarding the role of TRPM2 in bactericidal ability may stem from variations in the experimental models, the species and origins of bacteria, and other unidentified factors.
Our study utilized the CLP model, which closely resembles human sepsis by involving polymicrobial infection and a systemic inflammatory response.38,39 The CLP model shows a robust immune response, including immune cell activation, cytokine release, and multiorgan dysfunction, which are hallmarks of severe sepsis. 40 In contrast to LPS-induced models that primarily cause endotoxemia, the CLP model involves bacterial infection, allowing for a more comprehensive study of host immune responses to polymicrobial pathogens. 41 This approach provided a physiologically relevant setting to examine TRPM2 deficiency in macrophage polarization and immune dysfunction.
Activation of the PI3K/Akt pathway plays a critical role in modulating proinflammatory and anti-inflammatory responses in LPS-stimulated macrophages. 42 Peng Wan et al. suggested that PI3K/AKT signaling plays a crucial role in platelet activation, which accelerates endothelial cell damage in CLP-induced sepsis. 43 Recently, evidence has highlighted the essential role of the PI3K/Akt signaling pathway in regulating M2b macrophage polarization in sepsis. 44 Megan et al. reported that PI3Kγ controls the macrophage switch between immune stimulation and suppression. Blockade of PI3Kγ improved proinflammatory responses in macrophages, likely by decreasing the phosphorylation of AKT and CREB. 45 In our study, we found that the PI3K/AKT/CREB signaling pathway was markedly activated in trpm2−/− BMDMs compared with WT BMDMs. Consistent with these findings, similar results were found in the peritoneal macrophages of trpm2−/− and WT septic mice. These findings suggest that TRPM2 may play an important role in macrophage polarization following PI3K/AKT/CREB signaling.
In conclusion, TRPM2 deficiency promotes M2b macrophage polarization via the PI3K/AKT/CREB pathway, consistent with the established role of PI3K in driving M2 phenotypes. Elevated IL-10 and CCL1 levels reinforce this mechanism, but tissue-specific effects, such as those observed in gastric inflammation, underscore the complexity of TRPM2.
The primer sequences used for the qRT-PCR in the present study.

qRT–PCR: Quantitative real-time polymerase chain reaction.
Footnotes
Acknowledgment
We thank Li Liu and Guifeng Xiao in the core facility platform of Zhejiang University School of Medicine for their technical support. We thank Ping Liu in the Laboratory Animal Center of Zhejiang University for her technical assistance in the breeding of mice.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zhejiang Provincial Natural Science Foundation (grant number LY21H150004), and National Natural Science Foundation of China (grant number 82272183).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used and analyzed in the current study are available from the corresponding author upon reasonable request.
