Abstract
A previous study found that repeated intrauterine infusions of lipopolysaccharide (LPS) followed by an LPS infusion into the mammary glands attenuated the mammary inflammatory response. This suggests that repeated LPS infusion into the uterus promotes endotoxin tolerance (ET) in the mammary gland. However, the specific changes in mammary glands under ET conditions remain unclear. We hypothesized that ET affects leukocyte function in milk. This study aimed to investigate leukocyte function in milk under ET conditions induced through repeated LPS infusions into the uterus for three days followed by LPS infusion into the mammary glands of goats. Goats in the IU group (n = 17) received an infusion of 100 μg LPS in 5 ml saline into the uterus for three consecutive days (d −3, −2, and −1), whereas the goats in the control group did not receive this infusion (n = 19). On d 0, 1 μg LPS in 5 ml saline was infused into the mammary glands of both groups. Milk was collected 0, 4, 8, 12, 24, 48, 72, and 120 h after LPS intramammary infusion. The IU group decreased cytokine production (interleukin (IL)-1β, tumor necrosis factor (TNF)-α, and IL-1Ra) in milk following intramammary LPS infusion. Moreover, leukocyte activation, measured by phagocytic activity and CD11b expression, was higher in the IU group than in the control group. These findings suggest that goats exhibit enhanced leukocyte function in mammary glands under ET conditions, induced by repeated intrauterine infusion of LPS.
Introduction
The mammary gland plays a vital role in supporting the growth of offspring through the production of nutrient-rich milk and enhancing the immune function of newborns. 1 However, inflammation of the mammary gland or mastitis is the most prevalent disease in dairy animals. This condition exerts a multitude of detrimental effects, such as lower milk production and quality as well as higher treatment costs.2–4 Mammary gland provides an immune function to avoid this mastitis.5,6 After the bacteria invade the teat canal and enter the cistern, the interactions between these pathogens and milk leukocytes or epithelial cells critically establish inflammation in the mammary gland. 7
Leukocytes, which are part of the innate immune system of the mammary gland, actively participate in pathogen recognition and elimination. Polymorphonuclear leukocytes (PMNs) are the predominant leukocyte type in the uninfected mammary glands of cows and goats.8,9 These PMNs detect pathogens or pathogen-associated molecular patterns (PAMPs) through their recognition receptors (PRRs) such as toll-like receptors (TLRs). The PRR-PAMP interaction triggers intracellular signaling cascades, involving the activation of transcription factors such as nuclear factor (NK)-κB, subsequently regulating the expression of various soluble mediators that initiate inflammatory responses. 10
Lipopolysaccharide (LPS) is a component of the gram-negative bacterial cell wall. Recognition of LPS by leukocytes via the TLR4 pathway is a crucial step in the initiation of the immune response. This pathway upregulates the expression of CD11b, a cell surface protein and marker of PMNs activation.11–13 Zhou et al. (2005) demonstrated that PMNs from TLR4-deficient mice failed to induce CD11b expression following LPS challenge, whereas wild-type PMNs exhibited significantly increased CD11b expression within 3 h. 14 CD11b-positive PMNs can phagocytose bacteria, leading to a significant elevation in neutrophil number, along with the production of cytokines, including interleukin (IL)-1, IL-6, IL-8, IL-10, and tumor necrosis factor (TNF)-α within mammary glands.15,16 These cytokines play a pivotal role in initiating an inflammatory response in mammary glands.
Our previous study found that repeated intrauterine infusion of LPS for 7 days caused inflammation in the mammary glands. 17 However, the inflammatory response recovered after a few days, even though the infusion was still ongoing. The subsequent infusion of LPS into the mammary gland after repeated intrauterine infusion of LPS attenuated mammary inflammatory responses. 18 This indicates that repeated intrauterine infusions of LPS can induce inflammation, followed by tolerance to endotoxins in the mammary gland. Preconditioning human peripheral blood monocytes (PBMCs) with LPS markedly reduced the expression of pro-inflammatory cytokines, including TNF-α and IL-1β, upon subsequent LPS exposure. 19 This phenomenon, where repeated exposure to the same pathogen results in diminished inflammatory responses, is known as endotoxin tolerance (ET).20,21 According to Petzl et al. (2012), pre-treatment of cow udder with LPS inhibited the upregulation of gene expression of both inflammatory (TNF-α and IL-6) and anti-inflammatory (IL-10 and IRAK-M) cytokines in mammary tissue at 24 h after subsequent intramammary infusion with Escherichia coli. 22 Moreover, cows that were previously intramuscularly injected with LPS showed suppressed responses in terms of cortisol, cytokine (IL-6), and chemokines (CCL2, CCL3, and CCL4) upon subsequent intramuscular re-challenge with LPS. 23 ET serves as a crucial negative feedback mechanism that prevents excessive inflammation. 24 Regulation of the TLR4 signaling cascade plays a vital role in the establishment of ET. Decreased expression of TLR4 weakens the cell's response to LPS by preventing TLR4 from interacting with signaling molecules such as TLR4-MyD88 dependent signaling, leading to a weak immune response.25–27 This phenomenon is a complex physiological process involving several cell signaling pathways, changes in receptors, and immunological molecules. However, precise changes in mammary leukocyte function under ET conditions in ruminants are not yet fully understood. Therefore, the current study aimed to investigate the function of leukocytes in goat milk under ET conditions using repeated infusion of LPS into the uterus for 3 days followed by infusion of LPS into the mammary gland.
Materials and methods
Animals
This study included a total of 36 udder from 20 healthy Tokara goats, with body weights ranging from 20 to 25 kg, ages between 1 and 11 years. The goats were randomly assigned to two groups: the IU group (n = 17) and the CON group (n = 19). Both groups were in the mid-lactation stage, with the IU group had a day in milk (DIM) of 123.67 ± 94.5 (mean ± standard deviation), while the CON group had a mean DIM of 137.09 ± 75.45. No goats used in this study became pregnant during the experiment period. The parity in the IU group was 1 ± 0.5 (median ± interquartile range), while the CON group had a parity of 2 ± 2. Additionally, milk production before the mammary LPS infusion was 67.55 ± 42.15 ml (mean ± SD) in the IU group and 102.73 ± 57.94 ml in the CON group.
The goats were housed at Hiroshima University farm and provided 0.3 kg of hay and 0.1 kg of barley twice daily, along with free access to a trace mineral salt block and water. Their diet followed the Japanese feeding standard for sheep, as reported previously. 28 Only goats with a somatic cell count (SCC) below 1 × 106 cells/mL and no signs of udder inflammation were included at the start of the experiment or before reuse in subsequent trials. This study was conducted with approval from the Hiroshima University Animal Research Committee and adhered to their ethical guidelines (No. C 19–4).
LPS infusion
The IU group was induced ET condition by intrauterine pretreatment with 100 μg of LPS (E. coli O111:B4; Wako Pure Chemical Industries, Ltd, Osaka, Japan) dissolved in 5 ml of saline for three consecutive days, while the CON group served as a control and did not receive LPS pretreatment. The goats were inserted a speculum through the vulva, followed by the insertion of a blunt-ended stainless-steel needle through the cervix into the uterus for the intrauterine infusion. Prior to intrauterine infusion, sedation was administered using Xylazine (Bayer Healthcare, Osaka, Japan). After establishing ET in the IU group, both groups received an intramammary infusion of 1 μg of LPS dissolved in 5 ml of saline. Five goats from each group were used with intervals ranging from 2 weeks to 6 months.
Milk sample collection
Milk samples were collected by hand from the udder at 0, 4, 8, and 12 h after intramammary LPS infusion and then daily for 5 days. The milk samples were then centrifuged at 2300× g for 5 min at 4°C to remove fat. Skim milk samples were then stored at −20°C for subsequent enzyme immunoassays. The precipitated somatic cells (SCs) were used to measure SCC, differential leukocyte count (DLC), leukocyte activation, phagocytic activity (PA), and real-time polymerase chain reaction (RT-PCR) analysis. Leukocyte activation was assessed using CD11b as a marker, which was measured through both immunocytochemistry (ICC) and RT-PCR. PA was evaluated using a method involving fluorescent beads and viable bacteria culture.
Analysis of somatic cells (SCs)
The remaining SCs were resuspended in PBS to measure the SCC using a Countess Automated Cell Counter (Thermo Fisher Scientific, Waltham, MA, USA), as reported by Purba et al. (2019). 29 ICC was used to detect CD11b expression in milk SCs. The adjusted SCs solution (1 × 107 cells/mL) was fixed using 10% formalin for 10 min at 20°C, then incubated with 0.2% Triton X-100 for 30 min at 20°C for membrane solubilization. After centrifugation, the precipitate was incubated with anti-mouse CD11b IgG (Santa Cruz Biotechnology, Texas, USA) in BSA/PBS overnight at 4°C. SCs were incubated with anti-mouse IgG labeled with FITC (Proteintech, Tokyo, Japan) for 1 h at 37°C, and fluorescence was measured using a Countess Automated Cell Counter (Thermo Fisher Scientific).
Additionally, a part of the SCs solution was smeared onto a glass slide and stained with Giemsa to examine the DLC. Leukocytes were divided into lymphocytes, neutrophils, macrophages, and eosinophils and counted manually for 100 cells under a microscope.
Real-time polymerase chain reaction analysis
The protocol was previously described by Terada et al. (2020). 30 Breifly, total RNA was extracted from SCs using Sepasol RNA I Super (Nacalai Tesque Inc., Kyoto, Japan) following the manufacturer's instructions. The extracted total RNA was then resuspended in TE buffer (10 mM Tris-HCl, pH 8.0, with 1 mM EDTA) and subsequently stored at −80°C until PCR analysis. The concentration of total RNA in each sample was determined using NanoDrop Lite (Thermo Fisher Scientific). Subsequently, RNA samples were reverse-transcribed using ReverTra Ace qPCR RT Master Mix with gDNA Remover (Toyobo, Osaka, Japan) in a PTC-100 programmable thermal controller (MJ Research, Waltham, MA, USA). Real-time PCR was conducted using an AriaMX real-time PCR system (Agilent Technologies, Santa Clara, CA, USA) with the Brilliant III Ultra-Fast SYBR Green QPCR Master Mix (Agilent Technologies). Integrin subunit alpha M (ITGAM; CD11b) primers were used for PCR (Table 1), with the amplification cycle parameters set to a denaturation phase at 95°C for 5 s and an annealing phase at 56°C for 10 s; these were repeated for 60 cycles. The cycle parameters for the melting step were as follows: 95°C for 30 s, 65°C for 30 s, and 95°C for 30 s. Real-time PCR data were analyzed using the 2−ΔΔCT method and the expression levels were normalized using the ribosomal protein S18 (RPS18) housekeeping gene. 31
Primers for the study of gene expression in milk leukocytes.
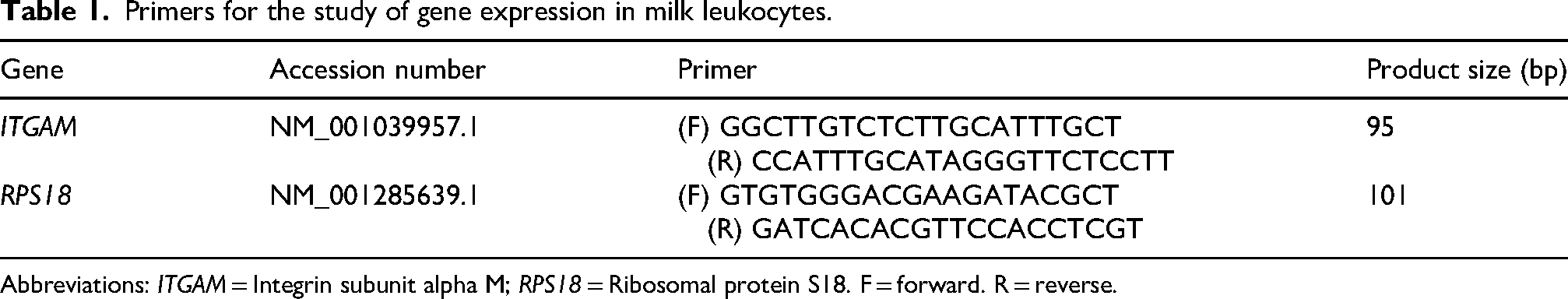
Abbreviations: ITGAM = Integrin subunit alpha M; RPS18 = Ribosomal protein S18. F = forward. R = reverse.
Phagocytosis activity (PA) of SCs
Two methods were used to examine the PA of the SCs: the fluorescent bead method and viable bacterial culture. For fluorescent bead method, SCs were resuspended in PBS and adjusted to 1 × 107 cells/mL, then incubated with Fluoresbrite® Carboxylate YG Microspheres (Polysciences, Pennsylvania, USA) for 30 min at 37°C; then, PA was determined by counting the population of cells that had phagocytosed microspheres using a Countess Automated Cell Counter (Thermo Fisher Scientific).
Viable bacteria culture method, E. coli strain was isolated from cows with clinical mastitis. The isolates have been previously described by Suzuki et al. (2022). 32 Briefly, isolated E. coli were identified using 16 s rRNA particle gene sequencing with primers 10F and 800R (Applied Biosystem 3500 xl Genetic Analyzers; Thermo Fisher Scientific). The BLAST algorithm from the National Center for Biotechnology Information (NCBI) was used for identification. Then, the isolated colonies were inoculated in nutrient agar (Pearlcore Nutrient Agar; Eiken Chemical, Tochigi, Japan) and cultured for 24 h at 37°C. Bacterial density was determined turbidometrically (575–660 nm) using a photoabsorbance sensor (PICOEXPLORER; Yamato Scientific, Tokyo, Japan) and adjusted to 1 × 108 CFU/ml. The prepared bacterial suspension was diluted to approximately 100 CFU/ml and incubated with milk SCs (1 × 107 cells/mL) resuspended in PBS. The contents were gently mixed and 100 µl was plated on deoxycholate-hydrogen sulfide-lactose agar (DHL; AccuDia DHL Agar; Shimadzu Diagnostics, Tokyo, Japan) at the initial time point (0 h) and after incubation at 21°C for 1 h. The plates were then incubated at 37°C for 24 h, and the number of colonies was recorded. The relative number of colonies was compared between 0 and 1 h.
Enzyme immunoassays
Concentrations of IL-1β, TNF-α, and IL-1Ra in milk were determined using a competitive enzyme immunoassay. Antibodies against IL-1β, TNF-α, and IL-1Ra were produced from peptides (CGEERDNKIPVA, CAEAKPWYEPIYQ, CITDLNQNREQDKR, respectively). Immunization was performed in rabbits (SCRUM, Tokyo, Japan), and the antibodies were purified using a HiTrapTM Protein G high-performance affinity column (Altiva, Uppsala, Sweden). The target peptides were conjugated with horseradish peroxidase (HRP) using a Peroxidase Labeling Kit-SH (Dojindo Laboratories, Kumamoto, Japan). The protocol for measuring IL-1β and TNF-α concentrations has been described previously by Tsugami et al. (2022) with 200 sample dilutions. 33 Additionally, IL-1Ra levels were measured with a 96-well microtiter plat, which was coated with 10 µg/ml goat anti-rabbit IgG, followed by blocking in borate buffer containing BSA. Rabbit anti IL-1Ra IgG (1 µg/ml), standard, and milk samples were added to each ELISA plate in triplicate. The milk samples were diluted 200 times. HRP-labeled IL-1Ra was added to the wells, followed by incubation. After washing, the optical density was measured using a microplate reader (Multiskan FC Microplate Type 357; Thermo Fisher Scientific).
Statistical analysis
All statistical analyses were performed using SAS software (JMP® Pro 18.1.0 version; SAS Institute, Inc., Cary, NC, USA) and the distributions of the means and residuals were examined to verify model assumptions of normality using the UNIVARIATE procedure. The effects of treatment, time, and interaction between treatment and time were assessed using mixed-model analysis. The data with normal distribution were analyzed using one-way analysis of varience (ANOVA) followed by Each Pair Student's t Test and Dunnett's for comparing with 0 h. For non-normal distribution, data were performed using nonparametric test by Wilcoxon Each Pair and Steel's test for comparing with 0 h. Differences were considered significant if the P-value was less than 0.05.
Results
Following intrauterine infusion
Following intrauterine LPS infusion, the vaginal wall swelled and redness was observed. Additionally, after the intrauterine infusion, the IU group exhibited a loss of appetite, which persisted and further declined after the subsequent intramammary LPS infusion. Both the CON and IU groups showed reduced appetite, with food consumption resuming at 24 and 48 h, respectively, following the intramammary LPS infusion.
After infusion of LPS into the uterus, goats in the IU group exhibited a significant decrease in milk yield, along with a marked increase in SCC levels (Supplementary file). Remarkably, these changes returned to the pre-infusion levels by d 0 (the day of intramammary infusion). Additionally, only IL-1Ra expression in milk showed a significant increase following the 2nd infusion of LPS into the uterus, then returned to the pre-infusion level.
Following intramammary infusion
Milk yield and SCC
In response to LPS intramammary infusion, the control groups showed a significantly decreased milk production at 4 until 24 h compared to that at 0 h (P < 0.05), while the IU had a significantly decreased milk yield at 8 and 12 h compared to those at 0 h (Figure 1A; P < 0.05). However, there was no significant difference in milk production between the IU and the CON group. In contrast to milk production, SCC levels showed a significant increase in both the IU and the CON group at 4 until 48 h compared to those before LPS intramammary challenge (Figure 1B; P < 0.05). However, the CON group exhibited a significantly lower SCC at 72 h (P < 0.05) than those in the IU group.

Effect of lipopolysaccharide (LPS) intramammary infusion on milk yield (A) and somatic cell count (SCC; B) in goats with (IU; n = 11) and without (CON; n = 11) pre-intrauterine infusion of LPS for 3 consecutive days. Values with different letters show significant differences between groups (a and b; P < 0.05). Asterisks show significant differences as compared with d 0 (*; P < 0.05). Trt = treatment.
CD11b expression
The immunocytochemistry results showed a significantly higher percentage of CD11b-positive leukocytes in the IU group at 0 h than in the control group (Figure 2A; P < 0.05). However, the IU group showed a significant decrease in the percentage of CD11b-positive cells at 72 and 120 h compared with that at 0 h (P < 0.05).
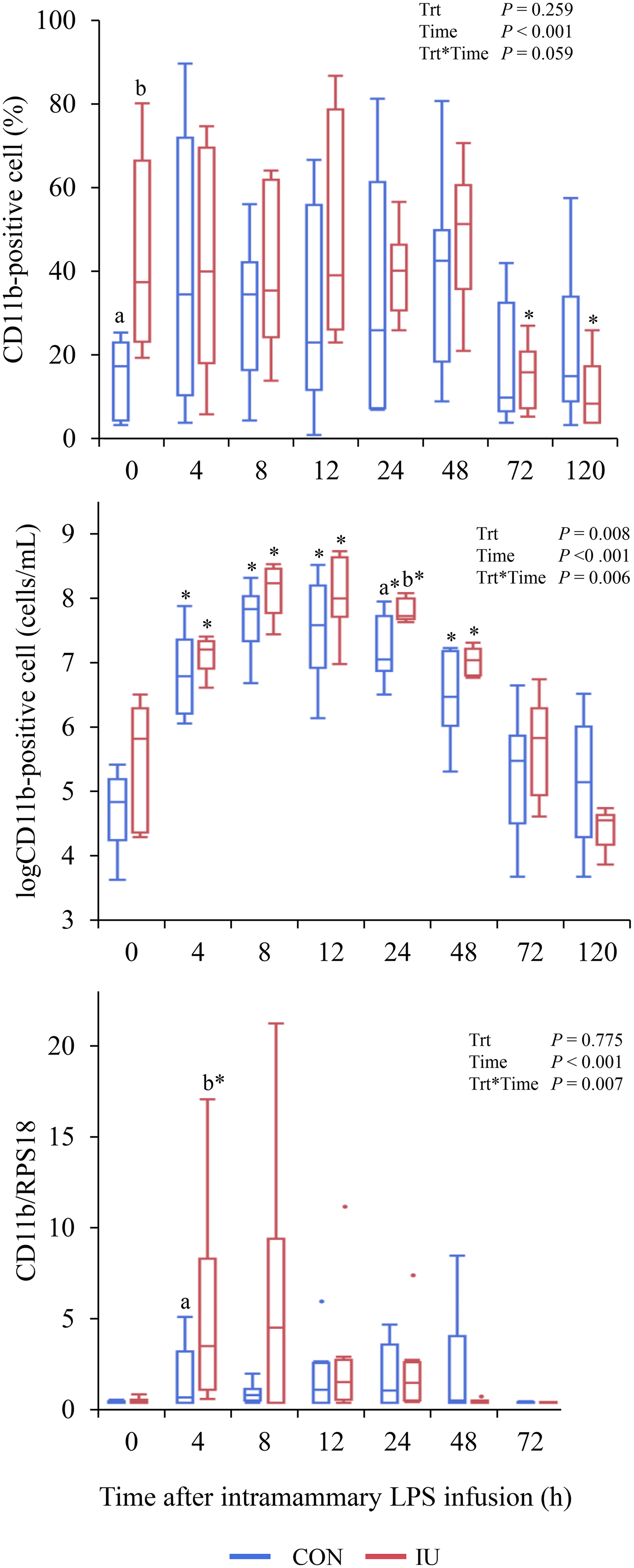
Effect of lipopolysaccharide (LPS) intramammary infusion on CD11b expression of milk leukocytes. Result of CD11b expression in terms of percentage (A), total number (B), and gene expression (C). Total number of CD11b-positive cells was calculated by multiplying the percentage of CD11b-positive cells by the somatic cell count (SCC). Goats were separated into two groups: IU (n = 8), which received and CON (n = 9), which did not receive the intrauterine infusion of LPS for three consecutive days. Gene expression (IU; n = 9 and CON; n = 9). Values with different letters indicate significant differences between groups (a and b; P < 0.05). Asterisks indicate significant differences compared with d 0 (*; P < 0.05).
Additionally, the total number of CD11b-positive cells was calculated by multiplying the percentage of CD11b-positive cells by the number of SCC. The results showed a significant increase in CD11b-positive cells in both the IU and control groups at 4 to 48 h compared to that at 0 h (Figure 2B; P < 0.05). However, the control group exhibited a significantly lower number of CD11b-positive cells than the IU group at 24 h (P < 0.05).
Furthermore, CD11b gene expression in milk leukocytes showed that the IU group had significantly higher CD11b expression at 4 h than the control group (Figure 2C; P < 0.05).
Differential leukocytes counts
Following LPS infusion into the mammary gland, the control group showed a decrease in lymphocytes and an increase in neutrophils at 24 h compared with those at 0 h (Table 2; P < 0.05). The IU group showed an increase in the percentage of neutrophils at 12 h compared with that at 0 h (P < 0.05). Additionally, the IU group had significantly higher lymphocyte percentages than the control group at 24, 48, and 72 h (P < 0.05). This contrasts with the lower neutrophil percentages in the IU group than in the control group at the same time points (P < 0.05). The percentage of macrophages in the IU group also significantly decreased at 8 h compared to that at 0 h (P < 0.05). Moreover, the IU group showed a significantly higher percentage of macrophages than the control group at 48, 72, and 120 h (P < 0.05). However, the control group had higher eosinophil percentages at 8 and 12 h than the IU group (P < 0.05).
Percentage of differential milk leukocyte count 1 after LPS infusion into the mammary gland.

Abbreviations: LPS = Lipopolysaccharide; h = hour.
Results are expressed as median ± interquartile range (IQR).
Leukocytes type
CON = control group (n = 6)
IU = goats with preinfusion of LPS into the uterus for 3 consecutive days (n = 6)
Values within a row with different superscripts differ significantly at P < 0.05.
Values within a row with asterisks differ significantly at P < 0.05 compared with 0 h.
Phagocytosis activity of SCs
PA was measured using fluorescent beads. Cells that engulfed these beads were considered to be positive for phagocytosis (Figure 3A). The control group showed a significant increase in the percentage of PA-positive cells at 8 h compared to that at 0 h (P < 0.05). However, a significantly higher percentage of PA-positive cells were present in the IU group at 12 h than in the control group (P < 0.05).

Phagocytosis activity (PA) of milk leukocytes following lipopolysaccharide (LPS) intramammary infusion in goats with (IU; n = 8) and without (CON; n = 8) the pre-intrauterine infusion of LPS for three consecutive days. The results of the beads method as a percentage (A) and the total number of PA-positive cells (B). The total number of positive cells was calculated by multiplying the percentage of positive cells by the somatic cell count (SCC). PA of viable bacterial culture methods were compared for colonies between 0 and 1 h (C; IU; n = 9 and CON; n = 8). Values with different letters indicate significant differences between groups (a,b; P < 0.05). Asterisks indicate significant differences compared with d 0 (*; P < 0.05). Trt = treatment.
The total number of phagocytosing cells (PA-positive cells) was calculated by multiplying the percentage of PA-positive cells with the number of SCC (Figure 3B). Both the IU and CON groups showed a significant increase in PA-positive cells at 4 to 24 h, whereas the control group had an elevated number of PA-positive cells until 48 h than those at 0 h (P < 0.05). However, IU group exhibited a significantly higher PA-positive cells than the CON group at 8 until 72 h (P < 0.05).
When leukocytes were incubated with viable bacteria for 1 h, the control group showed a decrease in bacterial numbers from 4, 8, 24, and 48 h compared with those at 0 h (Figure 3C; P < 0.05). In contrast, the IU group only showed a significantly lower bacterial count at 4, 8, and 24 h than at 0 h (P < 0.05). However, the CON group had lower bacterial counts at 12 and 48 h than the control group (P < 0.05).
Cytokines level
In response to intramammary LPS infusion, the IU group showed a significant decrease in TNF-α levels at 12 and 24 h compared with that at 0 h (Figure 4A; P < 0.05). Similarly, the control group also showed a significant decrease in TNF-α levels at 12 and 120 h compared with that at 0 h (P < 0.05). However, at 24 and 120 h, the control group had significantly different TNF-α levels in milk compared to the IU group (P < 0.05).

Effect of lipopolysaccharide (LPS) intramammary infusion on milk TNF-α (A), IL-1β (B), and IL-1RA (C) level. Goats with (IU; n = 11) and without (CON; n = 11) intrauterine infusion of LPS for three consecutive days. Values with different letters indicate significant differences between groups (a and b; P < 0.05). Asterisks indicate significant differences compared with d 0 (*; P < 0.05). Trt = treatment.
Milk IL-1β levels also showed a significant decrease at 24, 48, and 120 h compared with 0 h in the IU group (Figure 4B; P < 0.05). However, the control group showed significantly decrease IL-1β expression at 120 h (P < 0.05). The IU group exhibited a significantly higher milk IL-1β expression than the CON group at 0, 4, 8, 12, 48, 72, and 120 h (P < 0.05).
Furthermore, the IU group showed a significantly higher in milk IL-1Ra expression than the CON group at 0, 4, 8, and 12 h (Figure 4C; P < 0.05). Moreover, IU group exhibited a significant decrease at 120 h after LPS intramammary challenge (P < 0.05).
Discussion
Following LPS infusion into the mammary gland, Jaisue et al. (2023) 18 reported that goats pre-treatment with repeated intrauterine LPS infusion exhibited higher milk production and lower SCC levels compared to those without pre-treatment, suggesting reduced inflammation in pre-treated goats. This finding aligns with the concept of ET. However, the exact changes that occur in the mammary gland under ET conditions remain unclear. We hypothesize that ET may affect leukocyte function in milk. Therefore, this study focused on the leukocyte function in milk under ET conditions following LPS infusion into the mammary gland.
Leukocytes play a crucial role in mammary defense mechanisms by recognizing and eliminating invading microorganisms to protect against infection. LPS or endotoxin is a potent outer membrane component of Gram-negative bacteria. During inflammation, leukocyte migration is tightly regulated by the interactions between adhesion molecules on leukocytes and the vascular endothelium.34,35 L-selectin (CD62L) on endothelial cells binds to Mac-1 (CD11b/CD18) receptors on leukocytes, facilitating extravasation and migration to infection sites. 13 Diez-Fraile et al. (2003) demonstrated that LPS intramammary infusion in cows increased CD11b expression in circulating leukocytes, which correlated with an increase in SCC. 13 These findings suggest that CD11b is a potential marker of leukocyte trafficking and activation during inflammation. To estimate the total number of CD11b-positive and phagocytic cells in the mammary gland, we multiplied the percentage of these cells by the SCC count. At 0 h, the percentage of CD11b-positive cells was significantly higher in the IU group than that in the control group. Additionally, the CD11b gene expression in milk leukocytes was higher in the IU group at 4 h. These results suggest that repeated intrauterine LPS infusions enhance leukocyte activation in the milk.
In addition, leukocytes are crucial for combating bacterial infections during inflammation through phagocytosis and microbial elimination.36–38 To confirm PA, leukocytes were incubated with fluorescent beads and viable bacteria. The results showed that the IU group had a higher percentage of phagocytic-positive cells than the control group at 12 h after LPS intramammary infusion. Additionally, the IU group exhibited a greater PA-positive cells from 8 to 72 h compared to the control group. Moreover, both the IU and CON group demonstrated reduced bacterial clearance at 4, 8, and 24 h compared to 0 h, these indicating enhanced phagocytic ability. Zhong et al. (2021) demonstrated that macrophage can phagocytose LPS after the incubation with LPS for 24 h. 39 The increased in leukocyte activation and phagocytic ability in the IU group may improve bacterial component clearance following LPS infusion.
Our study showed that the IU group decreased in milk TNF-α and IL-1β expression significantly at 24 h after intramammary LPS infusion. This finding supports the concept of ET, in which a diminished inflammatory response occurs following repeated endotoxin exposure. When LPS binds to TLR4, accessory molecules, such as LPS-binding protein (LBP), MD2, and CD14 are required for activation. This triggers the MyD88-dependent and TRIF (TIR-domain-containing adapter-inducing interferon-β) pathways, leading to the upregulation of both pro-inflammatory (IL-1β and TNF-α) and anti-inflammatory (IL-1Ra) genes via NF-κB activation.20,40,41 Restimulation of tolerized human monocytes showed reduced TNF-α and IL-1β mRNA expression, indicating that these cytokines are markers of ET. 19 IL-1Ra is a potent competitive antagonist of the IL-1 family and binds to the same receptor as IL-1β.42,43 Therefore, a balance between IL-1 and IL-1Ra levels is crucial for regulating inflammation.42,44,45 In addition, Chen et al. (2025) observed a positive correlation between IL-1Ra and IL-1β expression in whole blood after in vitro LPS stimulation. 46 This explains why their expression levels are simultaneously observed in this study. However, the increasing IL-1Ra expression in the IU group after ET induction may play a crucial role in the regulation and progression of the inflammation. This sustained cytokine expression may have contributed to limiting the inflammatory response following subsequent intramammary LPS infusion, particularly when compared by ratio levels. Further research is necessary to determine whether the rise in IL-1Ra levels may actually reduce inflammation or not.
While alterations in signaling pathways are considered crucial in inducing ET, it is likely that other factors also contribute to this process. The ET mechanism may include decreased production of LBP, which hinders the interaction of LPS with immune cells. However, the blunted response observed in our study might be attributed to higher LPS clearance in the IU group, which subsequently led to a lack of LPS recognition by TLRs. Although we did not investigate LBP in this study, our previous study showed that repeated intrauterine LPS infusion for 7 days decreased plasma LBP levels on d 3, along with a lower SCC level and milk TNF-α level on d 4 compared to the control group. 17
In addition, leukocyte composition can be altered by bacterial recognition and the development of infection. 47 Therefore, a distinct leukocyte population in milk was compared between groups in the present study. Lymphocytes were the predominant cell population in the IU group, while neutrophils were dominant in the control group. However, the control group exhibited a stronger inflammatory response than the IU group, suggesting that neutrophils trigger a shift toward early inflammation.5,6,48 As the first cell population recruited to the site of infection, neutrophils encounter invading pathogens, engulf and eliminate them through phagocytosis. 8 Similar to our study, Draisma et al. (2009) reported a neutrophil population on d 1 after LPS intravenous bolus injection into humans. 49 However, whether neutrophils or macrophages were primarily responsible for the phagocytic activity in this study, remains unclear. Further research is needed to determine the cell type that exhibits phagocytic ability.
Understanding the effects of ET on leukocyte function could lead to the development of therapeutic strategies that can reduce inflammation, decrease antibiotic use, and potentially reduce the risk of antibiotic resistance. Further research is needed to explore the application of ET in mitigating mastitis-related damage from bacterial infections in dairy cows, although the present study was conducted using a goat model. Additional studies are essential to implement ET in bovines. Ultimately, clarifying the protective and regulatory mechanisms of ET is the key to understanding the role of the immune system in maintaining animal health.
In conclusion, these findings suggest that goats under ET conditions induced by repeated LPS infusion into the uterus exhibit enhanced leukocyte function in the mammary gland, resulting in improved bacterial clearance and moderate inflammatory responses to subsequent LPS intramammary infusion.
Supplemental Material
sj-pptx-1-ini-10.1177_17534259251341659 - Supplemental material for Mammary leukocytes function of endotoxin tolerant goat induced by intrauterine infusion of lipopolysaccharide
Supplemental material, sj-pptx-1-ini-10.1177_17534259251341659 for Mammary leukocytes function of endotoxin tolerant goat induced by intrauterine infusion of lipopolysaccharide by Jirapat Jaisue, Naoki Suzuki, Takahiro Nii and Naoki Isobe in Innate Immunity
Footnotes
Ethical considerations
This study was approved and conducted in accordance with the guidelines of the Hiroshima University Animal Research Committee (No. C 19–4).
Author contributions
Funding
This research received grant from Ministry of Education, Culture, Sports, Science and Technology, Japan Society for the Promotion of Science (21K05893).
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
