Abstract
The application of biological therapy and glucocorticoids in Auto-immune diseases (AID) patients will cause immunocompromised host (ICH) prone to infection. And monocytes play a key role in both innate and adaptive immune responses. We aimed to investigate the changes of circulating monocyte subsets in AID or AID-ICH patients with pulmonary infection. The subgroups and PD-L1 expression of monocytes were measured by flow cytometry in healthy individuals (HC), new-onset AID patients (AID cohort) and AID-ICH patients with pulmonary opportunistic infection (AID-ICH cohort). Flow cytometry analysis was used to determine the distribution of monocyte subsets, including classical monocytes (CL, CD14++CD16−), intermediate monocytes (ITM, CD14++CD16+) and non-classical monocytes (NC, CD14+/−CD16++), as well as the dynamic change of PD-L1 + cluster among monocyte subsets. Among total monocyte, AID-ICH displayed decreased CL subset along with increased NC subset compared to HCs and AID. Regarding PD-L1 + monocytes, although CL subset constituted the majority of that in HCs, AIDs and AID-ICHs, imbalance of NC/CL within PD-L1 + monocytes was only noticed in AID-ICHs (
Introduction
Auto-immune diseases (AID) are a group of chronic inflammatory diseases, which can be characterized by the damage of auto-immune tolerance mechanisms and an immune response to antigenic components of the host itself.1,2 Various overactivated immune cells and diverse cytokines regulate each other to form a complicated imbalanced immune network. 3 Biological therapy and Glucocorticoids are increasingly employed for AID patients, which have dramatically improved patient outcomes and transformed treatment paradigms. Despite their outstanding efficacy, patients under biological therapy are considered as immunocompromised host (ICH), who are at considerable risk for a variety of infections.4,5
Monocytes play a key role in both innate and adaptive immune responses for the reason that they can produce large amounts of soluble cytokines. According to immunological studies, monocytes are a heterogeneous population and can be divided into three different subsets relied on the presence and strength of CD14 and CD16 expression levels6,7: classical monocytes (CD14++CD16−), intermediate monocytes (CD14++CD16+) and non-classical monocytes (CD14+/−CD16++). 8 There are many studies have suggested that the percentages of monocyte subsets may vary with the presence of disease or inflammation.9–11 For example, the imbalance of monocyte counts have been observed in Coronavirus disease (COVID-19) as well as Gram-negative sepsis and have proven to be associated with unfavorable outcome.11,12
Moreover, PD-L1 is expressed in a variety of innate immune cells, including monocytes. The PD-1/PD-L1 pathway exerts inhibitory effects by regulating T-cell activation, tolerance, and immunopathology. Previous studies have demonstrated that the increase of PD-L1 expression in monocytes subsets was related to worse outcome in HIV/TB patients. 10
Currently, little is known about monocyte subsets in patients with AID or AID-ICH. In this study, we aimed to examine the proportions of the three monocyte subsets and their correlations with the opportunistic infection in AID-ICHs. Additionally, the PD-L1 expression was measured in the entire monocytes as well as NC/CL subsets to evaluate the dynamic change of PD-L1+ cells and their contribution to opportunistic infection process in these patients.
Methods and material
Patients
The study group consisted of 40 AID patients and 18 health controls (HC) from August 2019 to November 2023 in the department of respiratory and critical medicine of the First Affiliated Hospital of Soochow University. AID patients who had comorbidities of malignant tumor and blood system diseases were excluded from this study. The study was approved by the Ethics Committee of the Institute of the First Affiliated Hospital of Soochow University. All blood samples were collected after the patient gave their informed consent.
Moreover, the subjects with AID were divided into two different cohorts according to the exist of ICH and pulmonary opportunistic infections, including new-onset AID subjects diagnosed for the first time (AID cohort) and AID subjects with pulmonary opportunistic infections under immunocompromised condition (AID-ICH cohort).
Data collection
Clinical characteristics, including age, gender, and underlying disease, clinic status, and therapeutic response all collected from an electronic medical record.
Clinical definition
AIDs can be defined as a group of diseases with the main pathogenesis of tissue, organ damage and corresponding dysfunction caused by autoimmune response, including connective tissue diseases (such as systemic lupus erythematosus and rheumatoid arthritis), neuromuscular diseases (such as multiple sclerosis and myasthenia gravis) and the endocrine system diseases.
AID patients such as various rheumatologic, dermatologic, neurologic, and gastrointestinal diseases take biologic therapies or targeted disease modifying antirheumatic drugs for immunosuppression.13,14
The occurrence of opportunistic pulmonary infections is predominantly observed in patients under immunocompromised condition, wherein the primary causative agents encompass cytomegalovirus, pneumocystis jirovecii and aspergillus. Diagnosis of pulmonary opportunistic infection was defined as the combination of compatible symptoms of infection, imaging findings and proven pathogen. 15
Clinical outcome
Our study mainly explored the response to active comprehensive treatment after 7 days of flow cytometry analysis. All patients in AID-ICH cohort have received standardized clinical interventions upon admission, with respiratory support therapy, anti-opportunistic infection therapy, and low-dose glucocorticoids administered based on individual condition requirements.
Based on the clinical response, AID-ICH cohort could be further subdivided into two subgroups: progressive group and well-controlled group. Progressive group was defined as the presence of radiographic progression, escalation of respiratory support, development of septic shock or the occurrence of severe complications within 7 days of testing. 16
Flow cytometry analysis
Approximately 5 mL of peripheral blood sample from each subject was collected in the anti-coagulant tube. The samples were transported to the laboratory at 4°C within 2 h. Antibodies were purchased from Biolegend (USA). According to the manufacturer's recommendations monoclonal antibodies were used: per 50 μl of peripheral blood mononuclear cells strained with PECF594-labeled-anti-CD56 (1 μl, clone 5.1H11), PE-labeled-anti-CD14 (1 μl, clone 63D3), FITC-labeled-anti-CD16 (2 μl, clone 3G8), PC7-labeled-anti-PD-L1 (1 μl, clone 29E_2A3) were incubated for 30 min at room temperature in the dark. Samples were run on a Gallios™ Flow Cytometer (Beckman Coulter, Inc.) and analyzed using Flowjo 10 software.
In detailed, monocytes were first gated by forward scatter (FSC) and side scatter (SSC); The enriched monocyte fraction was then labeled with anti-CD14, anti-CD16, and anti-CD56 for fluorescence activated cell sortin into different subsets. CD14−CD56+ NK cells were excluded, as they also express CD16. The remaining cells were gated into CL (CD14+/CD16−) and NC (CD14−/CD16+) subsets. 17 Moreover, the baseline for measuring PD-L1 expression was determined using a negative-control gate consisting solely of CD56\CD14\CD16. Additionally, PD-L1 molecules were introduced into the experimental tube to facilitate the assay. A fluorescence activated cell sorter Calibur flow cytometer was used for analyzing the samples.
Statistical analysis
All analysis were performed using Graphpad Prism 6. The continuous analytical data were not all normally distributed, as assessed with the Shapiro-Wilk test. Therefore, continuous variables are expressed as medians (25th-75th percentiles) for non-normal distributions. The Kruskal-Wallis test was applied to compare multiple groups of data. And multiple testing was corrected using the Dunn's multiple comparison test. The Mann-Whitney test was used for comparisons between two groups. Statistical significance was set at
Results
Patient characteristics
The recruited subjects were composed of three population cohorts (HC, AID and AID-ICH), and the overall characteristics of these cohorts are described in Table 1. AID cohort consisted of 20 individuals with new-onset AID, and AID-ICH cohort was composed of 20 AID patients who suffered from pulmonary infection under immunocompromised condition.
Demographic and clinical characteristics of all subjects.

Data presented as median (25th percentile, 75th percentile) or n (%)
As shown in Table 1, no statistically significant difference between AID and AID-ICH cohorts was observed in age (61.5 years [IQR 47.5,72.8] vs. 67.0 years [58.0, 72.0],
In terms of underlying diseases, 55.0% of patients in AID cohort and 70.0% of patients in AID-ICH cohort had a history of underlying diseases, respectively. But the difference was not statistically significant (
Identification of monocytes subsets
The proportions of monocyte subsets may fluctuate with the presence of disease or inflammation.11,18 As shown in Figure 1A, NC and CL subsets were marked with different expression of CD14 and CD16: the CL subset with high expression of the CD14 cell surface receptor and no CD16 expression (CD14++CD16−) and the NC subset otherwise with a low/negative level of CD14 expression and co-expression of the CD16 receptor (CD14−/+CD16++).

Imbalance of NC/CL in AID when suffering from pulmonary opportunistic infection (AID-ICH)
Following, we explored the distribution of NC/CL monocyte subsets in new-onset AID patients and AID patients with pulmonary opportunistic infection due to immunocompromised condition. Firstly, circulating monocytes subsets among healthy controls, AID patients and AID-ICH patients were compared in Figure 1B. In detail, the percentage of three monocyte subsets did not show significant difference between healthy controls and AID patients (NC 21.9% [16.43, 28.95] vs. 15.70% [4.29, 33.06]; CL 71.9% [66.23, 76.63] vs. 77.90% [62.45, 88.13],
However, a significantly lower median proportion of CL subset in AID-ICH patients compared to healthy controls (53.20% [44.78, 69.65] vs. 71.9% [66.23, 76.63],
Shift from CL to NC among PD-L1+ monocytes noticed in AID-ICH patients
The presence of the programmed death ligand-1 (PD-L1) molecule on the surfaces of monocytes can indicate their role in suppressing the immune response and have association with a poor prognosis.
19
We explored the ratios of CL and NC among the PD-L1+ monocytes, to evaluate the distribution of dominant monocytes subsets in this context. The gating strategy of PD-L1+ monocyte subsets in flow cytometry was shown in Figure 2A. Interestingly, we observed that CL subset still constituted consistently the majority of PD-L1+ monocytes in no matter which cohorts, (HC: 85.50% [76.75, 87.25]; AID: 85.00% [70.75, 93.00]; ICH: 67.50% [46.25, 75.25]), while the ratio of CL subset in PD-L1+ monocytes decreased in AID-ICH cohort (P both <0.01). Moreover, it was noticed the ratio of NC subset in PD-L1+ monocytes increase obviously in AID-ICH cohort than that in HC and AID cohort (30.50% [23.23, 48.00] vs. 10.50% [7.75, 18.25] vs. 7.50% [2.00, 23.75], P both <0.01). Correspondingly, the ratio of PD-L1+ NC/CL subset in the AID-ICH cohort (0.445 [0.320, 1.028]) obviously increased than HC cohort (0.115 [0.090, 0.240],

PD-L1+ cluster in NC and CL subsets
To be more specific, we further analyzed the proportion of PD-L1+ cells in NC/CL subsets. As shown in Figure 3B, when compared to healthy controls (22.90% [18.38, 27.00],), PD-L1+ cluster in NC subset in AID (42.35% [29.53, 58.93],
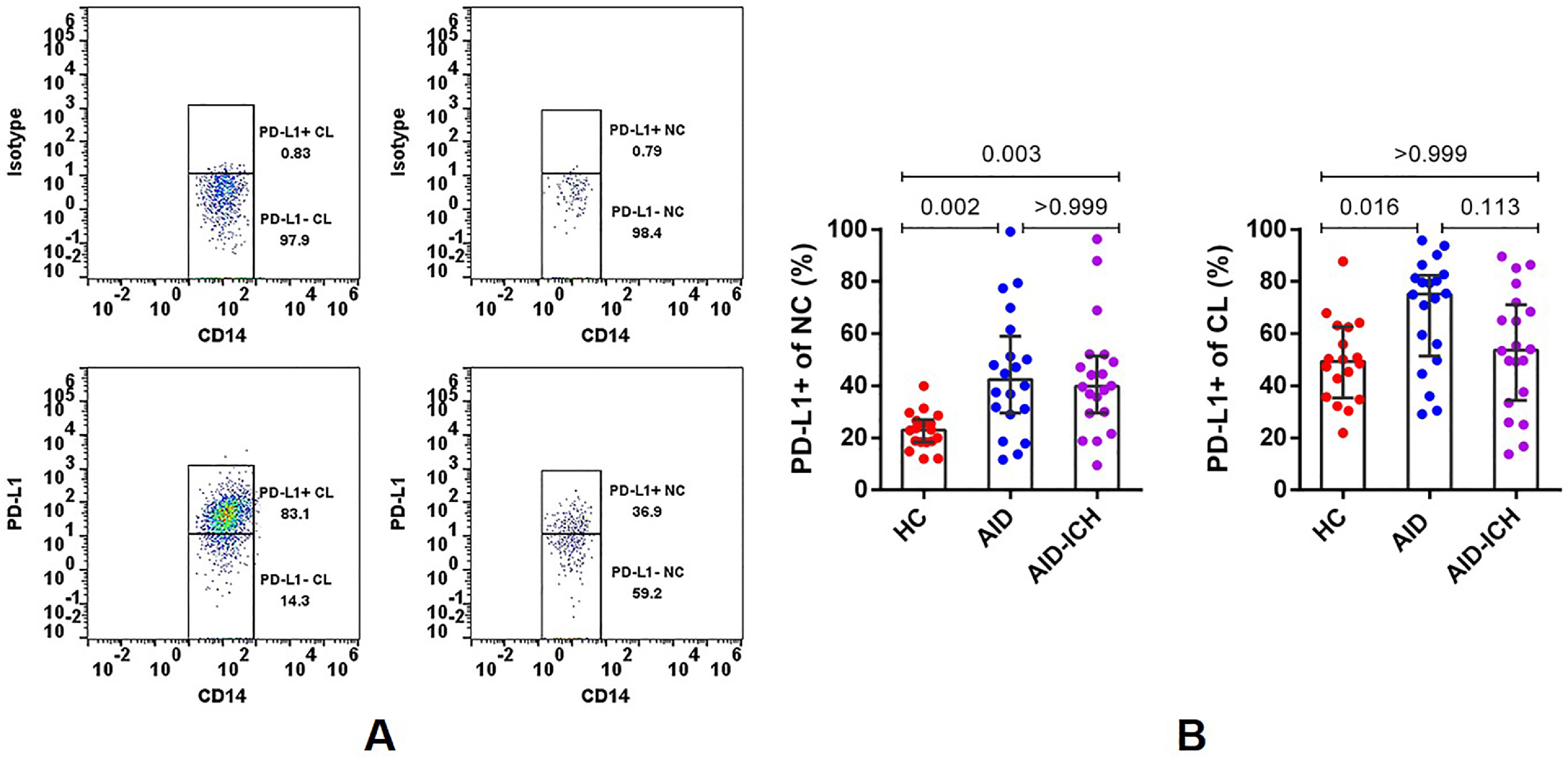
Circulating PD-L1 expressing-monocytes subsets and 7-day disease condition
Finally, AID-ICH patients were divided into progressive (

Discussion
The purpose of our study was to investigate the changes of circulating monocyte subsets in AID or AID-ICH patients with pulmonary infection, and explore the association of monocytes subsets with the response to active comprehensive treatment after 7 days of flow cytometry analysis, as well as to assess the PD-L1 expression on CL and NC subsets. The key findings are as follows: 1) AID-ICH patients showed distinct redistributions of monocyte subsets in the peripheral blood; 2) The shift of PD-L1+ CL subset to PD-L1+ NC subset was selectively found in AID-ICH patients and also reflected poor response to initial therapy, which served as a potential biomarker in clinical practice; 3) The upregulation of PD-L1+ cluster in NC subset was associated with autoimmune responses and the down-regulation of PD-L1+ cluster in CL subsets was consistent with disease progression in AID-ICH patients.
The AIDs as well as AID-ICHs with pulmonary opportunistic infections are associated with a large number of immunological abnormalities, which includes phenotypic and functional alterations in monocytes. 20 The proportion of classical monocytes exhibited a significant decrease in AID-ICHs compared with AIDs and HCs in our study, whereas the proportion of nonclassical monocytes demonstrated an increase. It is known that classical monocytes are critical for the initial inflammatory response, which can engage in proinflammatory response and differentiate into macrophages in tissue, while non-classical monocytes have been widely viewed as anti-inflammatory cells and they are a first line of defense in the recognition and clearance of pathogens. 21 Therefore, this finding suggested that pulmonary opportunistic infections in AID-ICH patients were more likely to promote intermediate monocytes translate to nonclassical monocytes in order to recognized and clear the pathogens.
The shift of CL cells into NC cells is suspected to be associated with the administration of immunosuppressive drugs. Research has demonstrated that the use of glucocorticoids can induce an upregulation in CD16 expression within monocytes, facilitating their transition towards NC cells through an intermediate state. 22 Hence the effect of immunosuppressive drugs such as glucocorticoids may rely not only on their direct action on T cells, but also on their ability to shift the balance between different monocyte subsets. Overall, there is a paucity of research dedicated to the mechanism of CL-to-NC shift.
Immune checkpoint molecules such as PD-1 and its ligand PD-L1 play an important role in regulating the immune response. Danielle Rosa Beserra et al. underlined the role of PD-1/PD-L1 modulation in infection and pointed out an increased PD-1 expression on CD14+ monocytes in severe/critical patients in association with the time of infection. 23 Another research found an increase of PD-L1 expression on intermediate monocytes in COVID-19 patients compared to the convalescent patients. 11 Therefore, further explored the ratios of CL and NC among the PD-L1+ monocytes to better define functional implications of PD-L1 expression on monocyte subsets. According to our study, CL subset still played the dominant role in PD-L1+ monocytes in HCs, AIDs and AID-ICHs. But deviating from prior studies, we observed the ratio of NC subset increased and CL subset decreased in PD-L1+ monocytes obviously in AID-ICH cohort than that in HC and AID cohort. And another research revealed that increased percentages of nonclassical monocytes significantly correlated with increased levels of monocytic PD-L1 expression in patients with human papilloma virus (HPV)-related oropharyngeal cancer. 24 All these data suggested that a higher level of immune suppression and greater susceptibility to opportunistic infections was associated with the increased abundances of NC subset in PD-L1+ monocytes.
Furthermore, our study classified AID-ICH cohort into progressive subgroup and well-controlled subgroup according to the response to active comprehensive treatment after 7 days of flow cytometry analysis. And in clinical practice, although the proportion of NC/CL subsets did not show any difference between progressive and well-controlled groups, the ratio of NC subset in PD-L1+ monocytes in progressive group also significantly increased compared with well-controlled group, whereas the opposite tendency was observed in CL subset. Moreover, it has been shown that PD-L1 is upregulated in neutrophils and correlates with sepsis-induced immunosuppression. 25 And another study has suggested increased PD-L1 expression on human neutrophils delays cellular apoptosis by triggering specific pathway to drive lung injury and increase mortality. 26 Based on our findings, we postulated that the predominant presence of NC cells among PD-L1 positive cells may delay NC cell apoptosis through specific signaling pathways, thereby exacerbating the inflammatory response within the body. Therefore, the shift of PD-L1+ CL subset to PD-L1+ NC subset could serve as a potential biomarker for AID-ICH patients in clinical practice.
As for the PD-L1 + cluster in these monocyte subsets, we have observed that the upregulation of PD-L1+ cluster in NC subset was associated with autoimmune responses. Despite the absence of statistical significance, the down-regulation of the PD-L1+ cluster in CL subsets should not be overlooked, and this trend warrants verification through experiments with larger sample sizes. Notably, a more pronounced downregulation of PD-L1+ cluster in CL subset in progressive AID-ICH patients has been discovered, which has demonstrated that monitoring the changes of PD-L1+ cluster in CL subset throughout the course of the disease remains meaningful.
Limitations of this study should be noted. First, the number of AID patients, AID-ICH patients and healthy controls was limited. Second, dynamic changes in the levels of monocyte subsets and functions of PD-L1 expressed in monocyte subsets remain targets for future studies. Further experiments could be performed to enhance the understanding of monocyte subsets. Moreover, our study solely focused on investigating the expression of PD-L1 molecules within the monocyte population, overlooking the involvement of other relevant biomarkers. This has prompted us to incorporate additional biomarkers in subsequent studies for further exploration.
Footnotes
Acknowledgements
We thank the patients, the nurses and clinical staff who are providing care for the patient, and staff at the local and state health departments.
Author contributions
QQX and CC conceived the idea and designed and supervised the study. ZDH, JYJ, JYF and SY performed the experiment and drafted the manuscript. ZDH and JYJ collected data, analyzed data and performed statistical analysis.
Consent for publication
Not applicable
Data availability
The datasets analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This study was approved by the Institutional Review Boards of The First Affiliated Hospital of Soochow University (2023-089).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was mainly supported by the National Natural Science Foundation of China (NSFC) grant 81672280 (to CC); Natural Science Foundation of Suzhou City grant SYS2021042 (to CC); The Project of Invigorating Health Care through Science, Technology and Education grant ZDXM2024002 (to CC) and Ncommunicable Chronic Diseases-National Science and Technology Major Project grant 2024ZD0522702 (to CC)..
