Abstract
Innate lymphoid cells (ILCs) are the main resident lymphocytes that mostly reside in tissues owing to the lack of adaptive antigen receptors. These cells are involved in early anti-infective immunity, antitumour immunity, regulation of tissue inflammation, and maintenance of homeostasis in the internal environment of tissues and have been referred to as the “first armies stationed in the human body”. ILCs are widely distributed in the lungs, colon, lymph nodes, oral mucosa and even embryonic tissues. Due to the advantage of their distribution location, they are often among the first cells to come into contact with pathogens.Relevant studies have demonstrated that ILCs play an early role in the defence against a variety of pathogenic microorganisms, including bacteria, viruses, fungi and helminths, before they intervene in the adaptive immune system. ILCs can initiate a rapid, nonspecific response against pathogens prior to the initiation of an adaptive immune response and can generate a protective immune response against specific pathogens, secreting different effectors to play a role.There is growing evidence that ILCs play an important role in host control of infectious diseases. In this paper, we summarize and discuss the current known infectious diseases in which ILCs are involved and ILC contribution to the defence against infectious diseases. Further insights into the mechanisms of ILCs action in different infectious diseases will be useful in facilitating the development of therapeutic strategies for early control of infections.
Introduction
Infectious diseases are a large group of diseases caused by infection and include damage to various tissues and organs of the body caused by the attack by pathogenic microorganisms, such as bacteria, viruses, fungi, and helminths, which jeopardize the health of humans and can be life threatening in the case of serious infections. overreactive responses of the host immune system stimulated by pathogenic microorganisms could also contribute to the pathologies of infectious diseases. With the improvement of living standards and the advancement of medical technology, some infectious diseases, such as chronic hepatitis C, smallpox and leprosy, have been eliminated or gradually reduced. However, new or mutated microorganisms have continued to be produced and discovered, leading to the occurrence of new emerging infectious diseases, 1 such as coronavirus disease 2019 (COVID-19), Middle East respiratory syndrome (MERS) and mpox virus (MPXV) infection that have emerged in recent years. The emergence of superbugs that are stubbornly resistant to antibiotics, and an increase in the spread of previously known pathogens, such as Ebola virus and Zika virus. Research indicates that the number of deaths caused by infectious diseases is expected to reach 10 million in 2050. Therefore, the current state of prevention and control of infectious diseases requires serious attention.
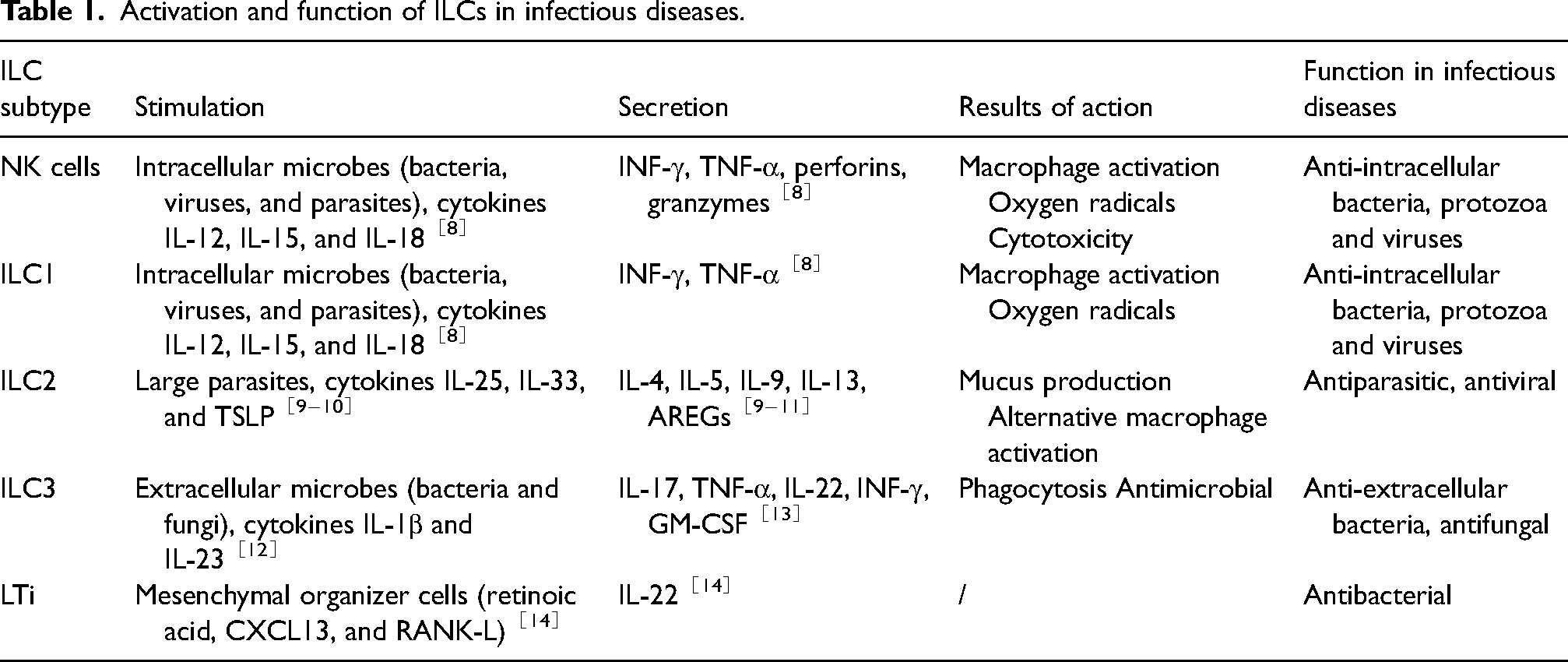
The control of infectious diseases by an organism primarily relies on the complementary actions of innate and adaptive immunity to suppress and eliminate the pathogen. Innate immunity serves as the initial line of defence, providing immediate and nonspecific attacks against pathogens. These responses are mediated by physical and chemical barriers, myeloid and innate lymphoid cells, and the complement system. Innate immune cells are alerted to danger when pathogen-or damage-associated molecular patterns (pathogen-associated molecular patterns(PAMPs)and damage associated molecular patterns(DAMPs), respectively) bind to surface or intracellular pattern recognition receptors (PRRs). This binding event triggers intracellular signalling cascades that activate classical innate immune responses, such as phagocytosis and cytokine production. 2 Innate lymphoid cells (ILCs), important effector cells of innate immunity, also modulate adaptive immune responses while playing a crucial role in early defence against infectious diseases due to their usual residence in tissues and organs as well as their ability to rapidly respond through intrinsic immunity in an antigen-independent manner. 3 Moreover, ILCs tend to expand on barrier surfaces, such as the gut, skin, and lungs. Unlike other immune cells, they lack recombination activating gene (RAG)-dependent antigen receptor rearrangement and do not express markers typically associated with other lymphoid and myeloid lineages. Instead, they express germline-encoded activating receptors that enable ILCs to sense signals produced by host myeloid and parenchymal cells following local injury, inflammation, or pathogen infection. This sensing mechanism leads to rapid production of both proinflammatory and regulatory cytokines. 4 The activation mechanism and functions of ILCs in infectious diseases are shown in Table 1. Currently, five distinct subpopulations of ILCs are defined based on their developmental trajectories: predominantly composed of cytotoxic natural killer (NK) cells, helper-like lymphocytes (The helper-like ILCs are classified into three subtypes based on their specific transcription factor and cytokine profiles: ILC1, ILC2, and ILC3, which approximately correspond to helper T1, T2, and T17 cells, respectively), and lymphoid tissue inducer (LTi) cells.4,5,6 Both NK cells and ILC1s can express the transcription factor T-bet (encoded by TBX21) and the natural cytotoxicity receptor NKp46 and can secrete interferon gamma (IFN-γ), while NK cells also express desmosomal eomesodermin. 7 NK cells and ILC1s are regulated primarily by cytokines, such as interleukin-12 (IL-12), IL-15, and IL-18. These cytokines possess stimulatory properties that induce the secretion of tumour necrosis factor alpha (TNF-α) and INF-γ by these cells. Moreover, they play a crucial role in conferring resistance against intracellular bacterial infections. Additionally, under the regulation by IL-12, NK cells and ILC1s actively participate in the immune response to early viral infections. 8 With their stable and persistent populations present in most adult tissues, ILC2 development and maintenance rely on the transcription factor GATA3, which induces the secretion of IL-4, IL-5, IL-9, and IL-13, as well as other type II cytokines and amphiregulin (AREG), in response to the stimulation by cytokines such as IL-33, IL-25, thymic stromal lymphopoietin (TSLP), and others. Secretions play a crucial role in tissue repair and parasite clearance and are also implicated in the mediation of airway hyperresponsiveness and participate in lung inflammation.9,10 The ILC2 population exhibits some degree of heterogeneity, with two distinct subpopulations identified in mouse models: the proinflammatory KLRG1+ ILC2 subset and the reparative inducible T-cell COStimulator(ICOS)+ ILC2 subset. Specifically, the KLRG1+ ILC2 subset is responsive to stimulation with IL-7 and IL-33; this leads to elevated production of IL-15 and IL-13, which are crucial for anti-infective responses. 11 ILC3s predominantly express the transcription factor RORγt (encoded by RORC) and can be categorized into two major subgroups, NCR- ILC3s and NCR + ILC3s based on their expression of the natural cytotoxicity receptor (NCR) NKp46 in mice and humans or NKp44 in humans. ILC3s can secrete IL-17A, IL-22, and granulocyte-macrophage colony-stimulating factor (GM-CSF) upon stimulation with interleukin-1β (IL-1β) and IL-23, which play crucial roles in mucosal immunity. Additionally, IL-22 promotes the expression of antimicrobial peptides by epithelial cells, providing protection against bacterial infections. Moreover, these cells exhibit plasticity in differentiating into other subtypes of ILCs for immune cell regulation and contribute to anti-infection processes. 12 The stimulation of ILC3s has been demonstrated to enhance the secretion of the cytokines IL-17 and IL-22, thereby promoting microbial clearance in both viral and bacterial pneumonia. 13 lTi cells are considered a distinct lineage of ILC3s that plays a crucial role in initiating secondary lymph node (LN) formation and promoting the development of Peyer's patches during embryogenesis. Elimination of LTi cells has been linked to the absence of LN formation. Primarily secreting IL-22, LTi cells may contribute to host defence against infections by activating ILC3s. 14 Infectious diseases caused by pathogenic microorganisms remain a significant risk factor for global mortality, with young children, elderly individuals, and immunocompromised individuals being particularly vulnerable to infections. Therefore, gaining a more comprehensive understanding of the response mechanisms of innate lymphoid cells during infectious disease development is crucial for early prevention and control. This article reviews the current research progress on ILCs in infectious diseases.
Activation and function of ILCs in infectious diseases.
ILCs and viral infections
Upon viral infection, the host immune system orchestrates a cascade of intricate immune response processes to combat the virus. Typically, the initial line of defence is initiated by the innate immune response, which aims to halt the primary infection, safeguard cells from further invasion, or eliminate infected cells. The activation of innate immune defences involves pattern recognition receptors (PRRs), such as Toll-like receptors (TLRs) and NOD-like receptors (NLRs), which recognize conserved features shared by viruses.
15
ILCs play a pivotal role as the primary responders to viral infections. The underlying mechanisms governing the antiviral functions of ILCs have been extensively investigated, with the following findings: (1) Immune cells should be targeted to the site of infection to kill the virus directly
15
16
When the virus infiltrates the body, peripheral blood NK cells can also be recruited to the infection site, where they exert direct cytotoxicity against virus-infected cells by releasing perforin and granzyme, thereby impeding viral replication. Furthermore, NK cells may augment host cell recognition susceptibility through infection-induced upregulation of self-encoded molecules and/or concurrent cellular stress responses; moreover, downregulation of MHC class I ligands, which inhibit receptor activity, further enhances target cell vulnerability.17,18 Furthermore, it has been demonstrated that NK cells exhibit an antibody-dependent, cell-mediated cytotoxic effect by engaging Fcγ receptor III/CD16 to bind virus-infected target cells coated with antibodies, subsequently leading to cellular lysis.
16
Indeed, NK cells can be effectively stimulated to eliminate infected cells through the recognition of viral particle fragments or through the engagement with viral ligands that activate specific NK cell receptors, such as KIRs and NKG2C.
19
(2) IFN-γ or TNF-α are produced to limit viral replication.
16
ILC1s and NK cells exhibit rapid responses during the early stages of viral infection, effectively limiting viral replication through prompt production of IFN-γ at the initial site of infection. In a murine model of
Mechanisms underlying the functional roles of ILCs in the clearance of viruses.

Influenza viruses
Influenza viruses belong to the Orthomyxoviridae family and are categorized into four types: A, B, C, and D. Types A, B, and C can infect humans. 27 These viruses mainly cause mild to moderate upper respiratory tract infections in patients, but a small percentage of patients progress to severe cases. Upon infection of respiratory epithelial cells by the influenza virus, pattern recognition receptors in the lung epithelium and innate immune cells in the tissue detect viral RNA, which leads to the production of proinflammatory cytokines and chemokines as well as the activation and recruitment of immune cells, including ILCs. 26 Activated ILC1s and NK cells secrete substantial quantities of IFN-γ, while NK cells also release perforin and granzyme to eliminate influenza virus-infected cells.17,28 Concurrently, the expression of the CD40 ligand by NK cells augments humoral immunity specific to the influenza virus. 26 The interaction between the NK cell surface receptor NKp46 and the influenza virus surface glycoprotein HA enhances the cytotoxicity of NK cells towards target cells and promotes cytokine production by activated NK cells. 28 Both subgroups of ILC2s c-kit + and c-kit2 ILC2s exhibit biphasic recruitment and progressive accumulation in the lungs during influenza virus infection, with c-kit2 ILC2s predominating in the acute phase and the number of c-kit + ILC2s dramatically increasing during the recovery phase. 29 ILC2s facilitate eosinophil accumulation by producing cytokines, including IL-5, which play a crucial role in the later stages of recovery from influenza virus infection. Additionally, NK cells can regulate IL-5 secretion during the recovery process by producing IL-33. 30 Monticelli et al. demonstrated that ILC2s played a crucial role in facilitating proper repair of the epithelial barrier and maintaining lung homeostasis through the secretion of AREG.31,32 In a murine model of influenza-induced injury, epithelial integrity was restored in the absence of ILCs through either pulmonary transfer of ILCs or administration of dual regulatory proteins. 33 It is evident that ILCs play a crucial role in the repair of influenza virus-induced damage to epithelial tissues. Specifically, pulmonary ILCs can contribute to early postinfectious airway hyperresponsiveness and compromise the sensitized respiratory epithelial barrier when exposed to high doses of H3N1 influenza virus through an IL-13-dependent mechanism, with ILC2s playing a predominant role. 29 While the influenza virus directly infects human NK cells and primarily induces apoptosis in infected NK cells through clathrin-mediated endocytosis, an alternative study revealed that the influenza virus could also impede NK cell cytotoxicity by reorganizing cell surface MHC class I proteins, thereby facilitating viral transmission.22,28,34 This implies that ILCs engage in interactions with influenza viruses during infection, playing a pivotal role in viral clearance, maintenance of pulmonary tissue homeostasis, and restoration of airway epithelial integrity postinfection.
SARS-CoV-2
SARS-CoV-2, a β-genus coronavirus, predominantly relies on the receptor-binding domain (RBD) located on its S protein to recognize and bind with high affinity to the angiotensin-converting enzyme 2 (ACE2) receptor present in the human respiratory tract for efficient host cell infection. 35 Numerous studies have consistently demonstrated a strong association between SARS-CoV-2 infection and the presence of ILCs. A study by Silverstein et al. revealed that the abundance of ILCs was significantly correlated with hospitalization requirements, the duration of the hospital stay, and the severity of inflammation in COVID-19 patients. 36 A study by García revealed a decrease in the overall population of CD127+ ILCs in moderately and severely ill patients, while the abundance of ILC1s remained relatively stable. However, there was an increase in both total ILC2 and CD117- ILC2 counts in the moderately ill group, whereas these counts decreased in severely ill COVID-19 patients. 37 Furthermore, a separate investigation revealed that the severity of the novel coronavirus was exclusively influenced by age-dependent disease tolerance mechanisms, with a significant association observed between lymphocytes, including ILCs, and the facilitation of disease tolerance. 38 Studies have also revealed a positive correlation between an advanced age and increased severity of COVID-19 in male patients exhibiting a lower ILC abundance.35,39 So how do ILCs work as antivirals in patients infected with SARS-CoV-2? ILC1s and NK cells primarily mediate direct antiviral effects by secreting type I cytokines such as IFN-γ and TNF-α, while ILC2s contribute to the antiviral response by secreting the type II cytokine IL-9. 40 Several studies have postulated that ILCs may potentiate antiviral effects by inducing a robust cytokine response in COVID-19 patients. SARS-CoV-2 may partially facilitate viral transmission by impeding type I interferon expression and inhibiting macrophage phagocytosis, thereby attenuating NK cell cytotoxicity to some extent. 41 These findings highlight the potential of enhancing immune system functionality and bolstering the body's antiviral capacity through the supplementation of expanded ILCs, thus offering a promising therapeutic avenue.
HIV
Human immunodeficiency virus (HIV) is a lentivirus that invades cells of the human immune system and belongs to the class of retroviruses. Upon infecting the human body, HIV aggressively targets CD4+ T cells, thereby compromising the functionality of the immune system. Kløverpris et al. reported an irreversible loss of circulating ILCs during early HIV infection and highlighted the potential of early antiretroviral therapy for preventing this loss; their findings may offer valuable insights for immunotherapy strategies in individuals with HIV infection. 42 The findings of another study corroborated these results, demonstrating rapid and persistent depletion of ILCs in untreated HIV/SIV infection in both human and nonhuman primates. 43 In addition to the decreased number of ILCs in the peripheral blood of HIV-infected patients, our study in an acute monkey HIV infection model revealed a significant reduction in the number of NKp44+ ILCs in all mucosal tissues as early as Day 6 postinfection. The decreases in both the number and function of ILCs were primarily attributed to the increased apoptosis-induced release of inflammatory cytokines from the intestinal environment and the enhanced inhibition of RORγt due to HIV infection. 44 Yetao Wang's research also show that inflammatory cytokines associated with HIV-1 infection irreversibly disrupt ILCs.Moreover, it was found that HIV-1 infection mainly caused an imbalance in ILC3 homeostasis and the expansion of TCF7-positive memory NK cells. 45 A comprehensive evaluation of the impact of acute SIV/SHIV infection on ILC3 and NK cell populations in infant rhesus monkeys demonstrated the indispensable contribution of these cells to antiviral defence during HIV infection. SIV/SHIV infection leads to a significant reduction in the number of ILC3s and an increase in that of NK cells, resulting in a functional transition from a homeostatic to a multifunctional proinflammatory state. In rhesus monkeys infected with the virus, there was a threefold decrease in the number of colonic ILC3s compared with that under normal conditions, while NK cell numbers were elevated in tonsils and oral lymph nodes. Additionally, heightened expression of CD107a, INF-γ, and TNF-α was observed, which enhanced antiviral effects through the increased NK cell activity and related cytokine production. 46 In conclusion, ILCs may play a pivotal role in the early immune response against HIV infection, and maintaining ILC homeostasis may be clinically important for the treatment of HIV-1 infection.
Cytomegalovirus (CMV)
CMV is a member of the herpesvirus family and is characterized by a double-stranded DNA genome. It is considered a low-virulence virus, which is widely distributed throughout the human body, and its pathogenicity remains limited in individuals with intact immune function. Using a murine model of cytomegalovirus infection, we discovered that hepatic ILC1s played a crucial role in the clearance of CMV. Depletion of zfp683-dependent hepatic ILC1s led to an increased viral load upon murine cytomegalovirus (MCMV) infection in mice with normal immune levels. 47 Furthermore, both peripheral blood NK cells and ILC1s can produce IFN-γ, which has been implicated in the antiviral response. 48 Moreover, the presence of a latent cytomegalovirus infection in the host organism significantly attenuates the motility response of both total and progenitor ILCs and impairs the exercise-induced mobilization of NK cells in individuals with a history of CMV infection. 49 In summary, ILCs play a pivotal role in both active and latent CMV infection.
Respiratory syncytial virus (RSV)
RSV, an RNA virus classified into the family Paramyxoviridae, is a significant causative agent of acute lower respiratory tract infection in children aged less than 5 years, elderly individuals, and immunocompromised individuals. ILCs play an important role in the initial response to RSV infection. Upon invasion of bronchial epithelial cells by RSV, the recognition of RSV transcripts and viral replication intermediates can trigger the release of TSLP, IL-33, HMGB1, and IL-25 from these cells. Subsequently, the release of IFN-γ by NK cells facilitates the activation of DCs and initiates T-lymphocyte responses while also exerting direct cytotoxicity against infected cells. 50 Activated receptors, such as NKG2D, expressed on lung NK cells can stimulate the production of substantial amounts of IFN-γ and facilitate the recruitment of other inflammatory cells .Toll-like receptors 4 (TLR-4) is an intrinsic immune receptor that is mainly expressed on cell membranes of macrophages, monocytes, and other cells, and its expression in human bronchial epithelial cells occurs only after infection with RSV. 51 Binding of RSV's F protein to TLR4 on host ciliated tracheal epithelial cells triggers the activation of P38 mitogen-activated protein kinase, which enhances the ability of RSV's ability to enter host cells.52,53 Is it possible to reduce RSV invasion by attenuating TLR-4 expression. However, Another study found that during RSV infection, TLR-4-deficient mice developed fewer NK cells trafficking than wild-type mice, resulting in reduced NK cytotoxic effects and delayed RSV virus clearance. 54 Therefore TLR4 may promote RSV virus clearance by affecting NK cell trafficking after host infection with the virus. Moreover, during early RSV infection, the involvement of NK cells may restrict the body's humoral immune response.50,54 In addition, epithelial-derived proteins stimulate the secretion of IL-4, IL-5, and IL-13 by ILC2s involved in antiviral responses. 55 Using an RSV mouse model, researchers further investigated the antiviral mechanism of ILCs and discovered that the STAT1 pathway played a crucial role in mediating their action and coordinating cytokine production. During viral infection, STAT1-deficient mice exhibit a decrease in the number of ILC1s and an increase in the production of ILC2s and ILC3s resulting in an imbalanced ratio of ILC subsets, which impairs both cytokine release by these cells and their antiviral capacity. 56 Therefore, maintaining the homeostasis of ILCs is crucial for bolstering the host antiviral defence.
Herpes simplex virus (HSV)
HSV is a neurotropic enveloped DNA virus that primarily infects the body through mucous membranes, the skin, and nerve tissues, leading to the development of associated lesions. It is classified into two main subtypes, namely HSV-1 and HSV-2. After herpes simplex virus infection, ILCs release various factors with antiviral properties. Notably, compared with ILC1s and ILC3s ILC2s exhibit elevated expression levels of granulocyte colony-stimulating factor (G-CSF), IL-1, IL-6, IL-9, a chemokine (RANTES), TNF-α, CXCL1, CXCL2, CXCL10, CCL3 and CCL4. 57 The release of these factors plays an important role in the clearance of HSV infection. By increasing ILCs may provide a new direction for immunotherapy of HSV.
Dengue virus
Dengue is a mosquito-borne disease that is mainly caused by 4 different antigenically related dengue virus serotypes (DENV-1, DENV-2, DENV-3 and DENV-4). 58 Similar to HSV infection, ILCs exert antiviral effects by releasing relevant cytokines following dengue virus infection. Notably, the production of inflammatory factors by ILCs is positively correlated with the severity of dengue disease, and more severe cases exhibit heightened secretion levels of factors such as INF-γ and IL-4. 59
Hepatitis
The immune composition of the liver differs from that of other organs and is characterized by a substantial population of resident natural immune cells, among which ILCs play a prominent role. Recent studies have increasingly indicated the crucial involvement of ILCs in antiviral defence against hepatitis viruses. Investigation of NK cells from individuals vaccinated against or infected with hepatitis B virus (HBV), compared with those unexposed to HBV, revealed variations in the cell phenotype and function, depending on the stage of chronic HBV infection. During early HBV infection, NK cells are activated through upregulation of activation markers such as NKp46 and exhibit an expansion of CD56+ NK cell subsets, enhanced cytotoxicity and IFN-γ production, thereby facilitating the clearance of HBV DNA. 60 In HBeAg-positive chronic hepatitis B patients treated with PEG-IFN-α-2a, we observed a significant correlation between the functional molecular expression by NK cells and HBsAg clearance. 61 Furthermore, our findings suggest that these cells may contribute to HBsAg clearance through relevant signalling pathways. In the liver tissues of patients with chronic hepatitis B, elevated numbers of ILC1s are significantly correlated with liver damage. Moreover, the expression of the differentiation-related transcription factor T-bet, the effector molecule IFN-γ, and the IL/receptor signalling molecules IL-12/IL-12R is significantly greater than that of factors related to ILC2s such as IL-33, ST2, GATA3, and IL-13, which are upregulated to a comparatively lesser extent. 62 After 52 weeks of antiviral treatment with tibivudine, the numbers of ILCs and the levels of cytokines released by the cells effectively normalized. 62 These findings suggest that ILCs, particularly ILC1s may play a significant role in antiviral therapy. Additionally, studies by Matsumoto and Liang demonstrated the protective potential of ILC2s and ILC3s through the release of cytokines in acute hepatitis.63,64 HBV infection is still a major global public health concern, and an in-depth understanding of the mechanism of action of ILC in HBV may provide an important direction for immunotherapy to achieve hepatitis B cure. In the context of hepatitis C, ILCs also play a pivotal role. Hepatic NKG2A + ILC1s can impede CD8+ T-cell-mediated antiviral immunity and facilitate HCV immune evasion within the liver. On the other hand, in patients infected with HCV, NKG2A expression is upregulated on the ILC1 surface, and blocking NKG2A on these cells enhances HCV clearance by suppressing the CXCL9 expression needed for IFN-γ+ NK cell recruitment. Moreover, inhibiting the NKG2A receptor on ILC1s promotes NK cell recruitment and IFN-γ release, thereby augmenting the host's ability to eliminate HCV.65,66This suggests that inducing a robust CD8+ T-cell response against persistent liver pathogens by inhibiting the NKG2A signalling pathway on ILC1s may be a novel therapeutic and preventive strategy against HCV.
ILCs and bacteria
Although the regulatory role of ILCs in immune responses against external pathogens has been acknowledged, their precise contributions and underlying mechanisms in protecting hosts against bacterial infections have not been fully elucidated.The most studied ligand-activated transcription factor, the aryl hydrocarbon receptor (AHR), senses environmental toxins and endogenous ligands, thereby inducing detoxification enzymes and regulating immune cell differentiation and responses and playing a key role in the innate defence against bacteria. Upon ligand binding, AHR exerts its antimicrobial effect by activating the degradation of virulence factors and modulating cytokine and chemokine production.67,68 Relevant studies have demonstrated that NK cells can engage with bacteria through the expression of AHR, thereby promoting INF-γ production and exerting antibacterial effects. 69 The expression of AHR is also significantly upregulated in ILC3s which facilitates their proliferation and enhances the secretion of IL-22.. 70 The observed reduction in the number of ILC3s in AHR-deficient mice is associated with an increased abundance of filamentous bacteria in the gastrointestinal microbiota, suggesting that heightened AHR expression in ILC3s may augment their antimicrobial efficacy. 71 In the dextran sulfate sodium (DSS) model of colitis, researchers observed a reduction in the proliferation of and IL-22 production by ILC3s lacking the SCFA receptor Ffar2, which resulted in a diminished synthesis of mucins and antimicrobial peptides such as Reg3α, β, and γ. Consequently, this exacerbated infection severity and disease progression. 72 Deficiency of the EP4 receptor may also impact antimicrobial efficacy. When bacteria are present in the body, their secreted lipopolysaccharide (LPS) stimulates the synthesis of prostaglandin E2 (PGE2), which interacts with ILC3s via the EP4 receptor to enhance ILC3 proliferation, the expression of ILC3 signature genes, and IL-22 production, thereby exerting an anti-infective effect. In mouse experiments, decreased expression of the EP4 receptor was shown to exacerbate infection owing to impaired intestinal barrier function, resulting in the spread of systemic inflammation associated with bacterial infection-related ectasia to the liver. 72 Additionally, ILCs can augment antimicrobial effects through the secretion of cytokines and antimicrobial peptides, among other factors. The secretion of GM-CSF by ILC3s promotes the maturation and polarization of intestinal macrophages in patients with inflammatory bowel disease, thereby enhancing the antibacterial capacity. 73 ILC3s can also induce the production of IL-17A, thereby promoting the synthesis of antimicrobial peptides (AMPs), including β-defensins, S100A8, liposome protein 2, and REG3A (the mouse homologue is regIIγ), in epithelial cells, which collectively contribute to an anti-infective effect. Furthermore, the produced IL-17F and IL-17A can synergistically cooperate with IL-22 to enhance the synthesis of antimicrobial peptides. 74 The subsequent sections delineate the involvement of ILCs in prevalent bacterial infections and elucidate their potential mechanisms of action (Figure 1).

Mechanisms underlying the functional roles of ILCs in the clearance of bacteria:  .
.
Salmonella Typhimurium and Listeria monocytogenes
The clearance mechanisms of ILCs against various bacteria vary. An abnormal function of ILCs in the body has an impact on the elimination of
Neisseria meningitidis
Tuberculosis
ILCs also play a crucial role in the prevention and control of
Klebsiella pneumoniae, Streptococcus pneumoniae and Pseudomonas aeruginosa
Citrobacter
When investigating the mechanism of
ILCs and fungi
The incidence of fungal infections has significantly increased in recent years, particularly among immunocompromised patients. In antibiotic-preexposed knockout mice (Rag2-/-Ilr2g-/-) lacking ILCs, susceptibility to invasive fungal infection was heightened, while transplantation of ILCs partially mitigated this susceptibility.
85
The pivotal role in antifungal defence is fulfilled by NK cells. First, a specific subset of NK cells exerts direct antifungal effects through the release of microbicidal molecules such as perforin. Additionally, NK cells exert indirect antifungal effects by releasing cytokines. For instance, the production of IFN-γ by NK cells plays a pivotal role in the eradication of
Additionally, Homeostatic imbalance of ILC3 subsets may heighten the susceptibility to fungal infections.
102
In one study focusing on intestinal homeostasis, researchers found a homeostatic imbalance of ILC3s in the intestines of old mice, with an increase in intestinal NKp46+ ILC3s and a decrease in the CCR6+ ILC3 subpopulation in old mice, especially a significant decrease in CCR6 + CD4+ ILC3s as well as a functional decline in ILC3s with these cells having a secretion of IL-22 and IL-17A diminished capacity, limiting host antifungal capacity in a mouse model of fungal infection.ILC3s also resist fungal infection by regulating the amount of Th17.In the context of
ILCs and helminths
Helminths are multicellular ancient organisms that parasitize on the mucosal surfaces of their hosts. Recent research has highlighted the importance of ILCs, particularly ILC2s and their type 2 immune responses in human defence against helminth invasion (Figure 2). ILC2s primarily mediate antihelminth immunity through the expression of the transcription factor GATA3 and the production of AREG, as well as the cytokines IL-4, IL-5, IL-9, and IL-13..104–107 The transcription factor RXRγ is strongly and selectively expressed in steady-state small intestinal ILC2s The suppression of RXRγ substantially amplifies the responsiveness of ILC2s to alarmin cytokines, thereby exacerbating alarmin-induced intestinal inflammation. Depletion of RXRγ in a murine model of parasitic infection further enhances parasite suppression, mediated by ILC2s within the homeostatic small intestine.
108
Helminth-induced intestinal tissue damage triggers the production of IL-33, IL-25, and TSLP by epithelial cells and IL-33 by stromal cells, as well as the release of IL-25 and the cysteine leukotriene LTC4 (an activator of IL-2 that promotes the release of IL-5 and IL-13) by cluster cells, thereby activating IL-2.104,109–111 After activation, ILC2s release the corresponding cytokines and AREG to participate in the helminth clearance process. The release of IL-4 primarily induces class switching in IgE-producing B cells, thus playing a crucial role in this process.
112
Additionally, IL-13 facilitates the expulsion of helminths by directly targeting intestinal epithelial and goblet cells, which leads to their proliferation, increased mucus production, and enhanced muscle contractility.
113
In the small intestine, chemosensing by epithelial tuft cells triggers the activation of ILC2s thereby driving an increase in the tuft cell frequency. This regulatory loop is commonly referred to as a feedforward loop. Notably, IL-13 and IL-4 play pivotal roles in promoting tuft cell differentiation and facilitating the establishment of feedforward circuits. Consequently, enhanced production of IL-25 further contributes to efficient worm clearance.
114
IL-13 and AREG can also exert anti-inflammatory effects on epithelial cells, promote wound healing, and facilitate tissue repair.
115
The crucial role of IL-9 in maintaining the response to ILC2s has been demonstrated, with studies showing that the administration of IL-9 antibodies affects the intestinal excretion of

Mechanisms underlying the functional roles of ILCs in the clearance of helminths:  .
.
During helminth infection, ILC2s can actively participate in the immune response against
The impaired amplification of ILC2s was observed to impede their expulsion by
NK cells and ILC1s also play crucial roles in the clearance of helminths, which are primarily associated with intracellular pathogen elimination. A study revealed that T-bet-dependent production of IFN-γ by ILC1s and NK cells contributed to cDC1-mediated host resistance against
ILCs also play a crucial role in the pathogenesis of cerebral malaria. Recently, in C57BL/6 mice infected with cerebral malaria, researchers have discovered that the activation of ILC2s through the release of IL-33 predominantly stimulates the production of IL-4 and IL-5, thereby attenuating the production of inflammatory mediators such as IFN-γ, IL-12, and TNF-α. Exogenous IL-33-driven ILC2s induce the production of M2 macrophages, with anti-inflammatory properties, thereby promoting the expansion of Foxp3 + regulatory T cells (Tregs) and subsequently preventing cerebral malaria. Gene regulation is also crucial for the antiparasitic mechanism of ILC2s. 131 Adenine N6 methylation (m6A), the most prevalent and abundant posttranscriptional modification of mRNAs in eukaryotes, plays a pivotal role in various aspects of RNA metabolism. This discovery opens up new avenues for parasite treatment. 132
Conclusions
ILCs are recognized as the primary responders to the lymphocyte lineage and are strategically located in diverse tissues, including the lung, liver, skin, and intestine. These cells exhibit rapid responsiveness to local environmental cues during early-stage pathogen invasion and play a crucial role in combating infections.While extensive research has been conducted on the role of ILCs against helminths and viruses, investigations regarding the role of ILCs against bacterial and fungal infections have been relatively limited, necessitating further exploration of the underlying mechanisms involved. ILCs are activated to produce effector molecules, which are key signaling factors for tissue defense against pathogens in the early stage of viral, bacterial, and fungal infections, and are involved in the promotion of tissue inflammation or repair. However, the specific mechanisms by which ILCs generate immune responses and their molecular pathways to promote inflammation or tissue repair after infection with the above pathogens have yet to be elucidated (especially in fungal infections). Furthermore, the mechanism of LTi's role in anti-infection has been poorly investigated. A comprehensive understanding of the intricate molecular and cellular communications of ILCs will provide valuable insights into their dynamics during infectious disease development, thereby paving new avenues for the treatment and prevention.
Footnotes
Author contributions
Ting Yuan: Writing, conceptualization, Figure making, proofreading.Qianhui Zhou and Yuqiu Tian:editing, data correction. Yangjing Ou and Yunzhu Long: Figure making, conceptualization. Yingzheng Tan: Supervision, Conceptualization, proofreading, data correction.
Data availability
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
