Abstract
The study of cellular immune responses in animal disease models demands detailed knowledge of development, function, and regulation of immune cells, including natural killer (NK) cells. Listeria monocytogenes (LM) bacterium has been explored in a large area of research fields, including the host pathogen interaction. Although the importance role of NK cells in controlling the first phase of LM burden has been investigated, the interaction between NK cells and infected cells in details are far from being comprehended. From in vivo and in vitro experiments, we can drive several important pieces of knowledge that hopefully contribute to illuminating the intercommunication between LM-infected cells and NK cells. Experimental studies performed in rats revealed that certain NK cell ligands are influenced in LM-infected cells. These ligands include both classical- and non-classical MHC class I molecules and C-type lectin related (Clr) molecules that are ligands for Ly49- and NKR-P1 receptors respectively. Interaction between these receptors:ligands during LM infection, demonstrated stimulation of rat NK cells. Hence, these studies provided additional knowledge to the mechanisms NK cells utilise to recognise and respond to LM infection outlined in the current review.
Listeria monocytogenes bacterium
The Gram-positive bacterium Listeria monocytogenes (LM) is a rod-shaped facultative intracellular foodborne pathogen responsible for a disease called listeriosis. LM is widespread in nature and thrives in various environments such as water, soil, in plants and in animals. 1 Varied and complex mechanisms of regulation and diverse responses to stress permit LM to survive in highly distinct environmental conditions and to change from saprophytism to virulence. 2 LM infection is relatively uncommon and usually eliminated by the immune system in the gut without causing systemic infection in healthy individuals. However, outbreaks due to contaminated products occurs with the high overall fatality rate of systemic infection (30%) compared to other pathogens such as Salmonella (< 1%). 3 LM may cause sepsis and spread to the nervous system causing cranial nerve palsies, encephalitis, abscesses, or meningitis. At-risk people including immunocompromised individuals and pregnant women are in particular vulnerable to LM infection. 2 LM is often used to study the mammalian immune response to infection because it is easy to culture, is relatively safe to handle and causes a robust infection in laboratory mice. LM has evolved several mechanisms to evade host immunity. The LM pathogen model has yielded comprehensive pictures of the ‘battle’ between host and microorganism.
Natural killer (NK) cells
Natural killer (NK) cells are capable of recognizing and destroying a wide variety of target cells including transplanted, antibody-coated, stressed, transformed cells and MHC-I-disparate normal hematopoietic cells. 4 NK cells constitute the third major population of lymphocytes, and are speculated to be relatively short-lived although more long-lived subpopulations of NK cells have been identified in lymph nodes (LNs) and thymus. 5 In addition to killing NK cells also produce cytokines, of which IFN-γ is critically important both in the innate and adaptive immune responses. 6 In humans, NK cells have been phenotypically defined as CD3−CD56+ lymphocytes that may be further subdivided into CD56dimCD16bright (90% of all NK) and CD56brightCD16− cells 7 in which differ in cytotoxic capacity and cytokine profiles. 8 In mice NK cells are defined as CD3−NK1.1+CD122+, 5 and in rats as CD3−NKR-P1A+ (Figure 1) 9 or NKp46+ cells. 10

The role of NK cells to Listeria monocytogenes infection
The role of NK cells in the protection against LM has been subjected to controversies, as both beneficial9,11–14 and detrimental effects of NK cells in rodents have been reported.15–20 These contrariety reports might be explained by the route of infection, 20 amount of LM used to infect the animals, causing subsequent excessive NK cell-response which might contribute to listerial-mediated immunopathology. There are unique aspects of rat NK cells (discussed in this review) that could account for protective roles in rats versus detrimental roles in mice. A line of independent reports support the protective role of NK cells to early granulomatous stage in listeriosis through a diverse repertoire of antibacterial effector mechanisms by secreting several TH1-type cytokines – IFN-γ and TNF-α, which are known to play a crucial role in activation of MØs and granuloma formation,1,6 apoptosis of infected cells by secretion of perforins and granzymes contained within the cytotoxic granula and by engagement of death receptors on the target cell,1,6 and cytotoxic activity they regulate immune responses through the elimination of suppressor cell populations, such as Tregs. 21 Decidual NK cells directly cytotoxic to LM through secreting granulysin, have also been reported. 22
The mechanisms NK cells specifically utilise to recognize LM-infected cells are largely unknown. A line of evidence points to indirect activation of NK cells through accessory cells. For instance, LM-infected MØ produce cytokines such as IL-12, which, in turn, activate NK cells to produce and release IFN-γ. 23 IFN-γ synergizes with other activation stimuli to activate MØ which efficiently kill ingested LM bacteria through different mechanisms, including reactive NO radicals, but also because LM bacteria no longer are able to escape into the cytoplasm and are killed by lysosomal enzymes. 24 Direct recognition LM-infected cells has been proposed by pattern recognition receptors, such as TLRs expressed on NK cells, 25 or ligands for activating NK cell receptors, such as NKp46 26 and NKG2D. 27 There are still other surface NK-ligands such as MHC-I molecules that can be modulated on LM-infected cells. The role of these ligands has been poorly investigated.
The current review aims to give the readers a summary of hitherto findings and conclusion of our experimental investigation in rats. These studies focused on two rat receptor families (Ly49- and NKR-P1 receptor families) that are described below.
NK Gene Complex (NKC) and Leukocyte Receptor Complex (LRC)
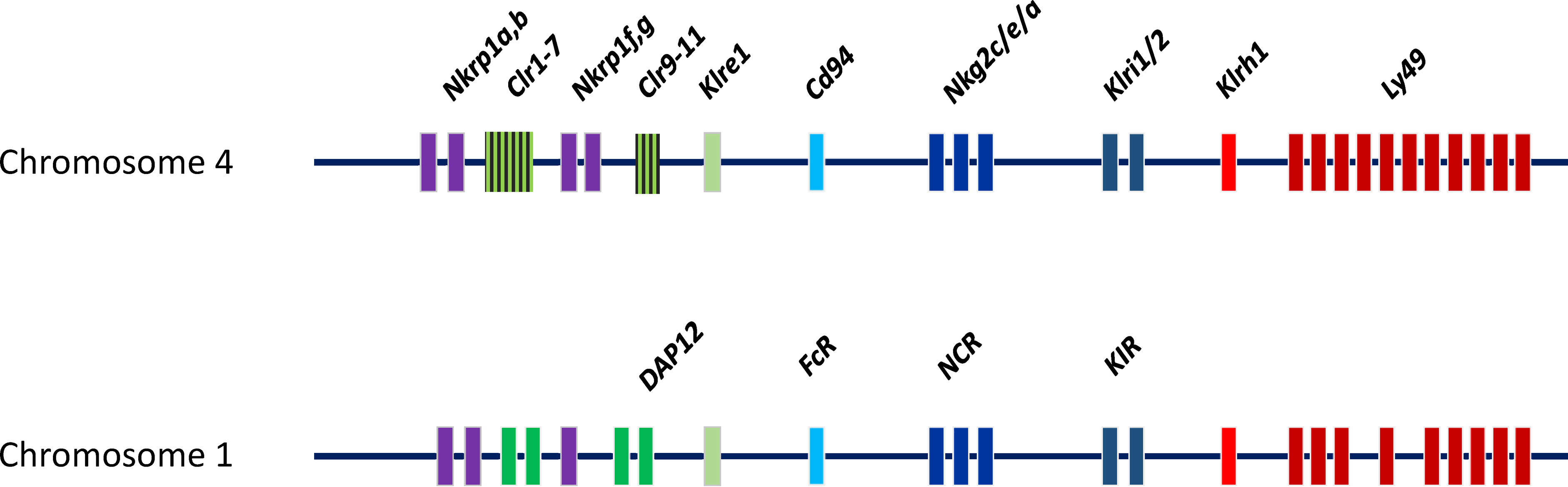
The NKC and LRC encode most of NK cell receptors in rats. Rats have 21 pairs of chromosomes. The NKC is located on chromosome 4 and encodes lectin-like molecules that can be classified into families of highly homologous members28,29 (Figure 2). These receptors are encoded by clusters of closely linked genes that usually are separated from the other gene families. One exception is the intertwined Nkrp1, and C-type lectin related (Clr). 8 Between families, the receptors share some general structural features but display limited homology. 30 This region spans from the Nkrp1a to the Ly49i8 gene. 31 The located rat contains about 67 killer cell lectin-like receptors (KLR) genes in the NKC. The number of Ly49 genes differs widely between NKC haplotypes. So far investigated, the rat NKC seems to contain more Ly49 genes than the mouse where many of these seem to have activating function.31,32 LRC is located on chromosome 1 and encodes NKp46 and ‘unfunctional’ rat human killer cell Ig-like receptor (KIR).33,34 (Figure 2). Other receptors genes are located on different chromosomes such as 2B4 on chromosome 13, DNAM-1 on chromosome 18 35 NKp30 on chromosome 20 (close to RT1 region). 36
Schematic
Rat NK cell receptors
Rat NK cell receptors can be structurally divided in two groups: the killer cell lectin-like receptors (KLR) and immunoglobulin (Ig) superfamily receptors. The former includes the Ly49 (KLRA), NKR-P1 (KLRB), CD94/NKG2 (KLRC/D), KLRE, KLRI, KLRH and NKG2D (KLRK) receptor families.5,30 These families have varying number of members in which the Ly49 family is the one with most members. The latter includes the natural cytotoxicity receptors (NCR) NKp30 and NKp46. KLR are type II C-type-lectin-like transmembrane molecules expressed as disulphide-linked homodimers or noncovalently heterodimers. These receptors differ from classical C-type lectins in that they recognize proteins instead of carbohydrates. Although termed ‘NK cell receptors’, many of these are not restricted to NK cells and can also be found on other cells. 4 A comprehensive review of all rat NK cell receptors is beyond the scope of this paper. In the following, the Ly49- and the NKR-P1 families are briefly described. A list of identified or characterized rat NK cell receptors with their identified ligand(s) and the respective available antibodies are summarized in Table 1.
Identified or characterized rat NK cell receptors and their cognate ligand(s).
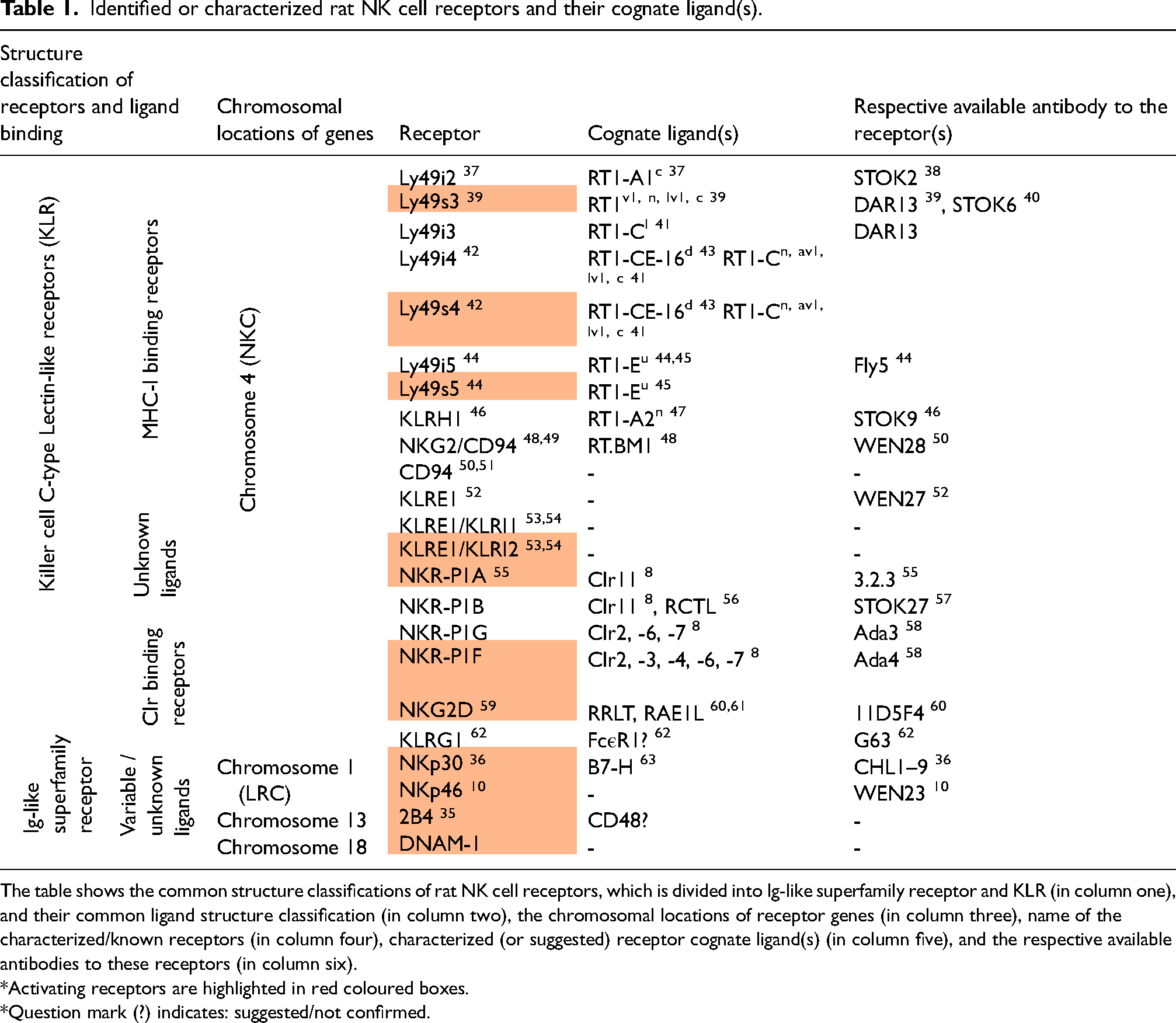
The table shows the common structure classifications of rat NK cell receptors, which is divided into Ig-like superfamily receptor and KLR (in column one), and their common ligand structure classification (in column two), the chromosomal locations of receptor genes (in column three), name of the characterized/known receptors (in column four), characterized (or suggested) receptor cognate ligand(s) (in column five), and the respective available antibodies to these receptors (in column six).
*Activating receptors are highlighted in red coloured boxes.
*Question mark (?) indicates: suggested/not confirmed.

A
The Rat MHC (RT1) Complex
The RT1 complex has been central to NK cell research since it encodes ligands for some of NK cell receptor families. This region is located on chromosome 20 and contains both classical (Ia) and nonclassical (Ib) genes. The classical molecules are encoded from the RT1-A region, while the nonclassical ones are encoded from the RT1-CE, -N, -M1, -M4, -M3A, -M3B, and -M2 regions. 95 The centromeric class Ia region and the first telomeric class Ib gene cluster, the RT1-CE region, are separated by the class II and class III regions (Figure 2). The RT1-CE region contains 16 nonclassical Ib genes in the RT1n haplotype, in which most of them are probably expressed. 96 They are less polymorphic than classical class I molecules, and often have a restricted tissue distribution and low surface expression. Some RT1-CE molecules may present peptides. Ly49 receptors have been shown to recognize MHC-I molecules encoded both from the RT1-A and -CE regions. 97 The second telomeric class I gene cluster, RT1-N, also contains 16 class Ib genes in the BN rat. The most telomeric part of the MHC contains the M-like class I gene clusters; M1, M4, M3A, M3B, and M2. M1 and M4 match the positions of human class I gene regions encoding HLA-92 and HLA-A/G/F. There are close similarities between the mouse and rat M-region class I genes. H2-M3 present N-formylated peptides to CD8+ T cells,98,99 however the role of these molecules from this region in rats is undetermined.
Role of NK cells in Listeria monocytogenes infection, studies in rats
Throughout the experiments we used Listeria monocytogenes strain L 242/73 type 4 b, a kind gift from Arja De Klerk, National Institute of Public Health, and the Environment, Bilthoven, The Netherlands. The infection by this strain was successful and caused a robust infection both in laboratory rats and cell lines or primary cells that have previously been described and demonstrated.42,45 Representative cytospins from these experiments are shown in Figure 4.
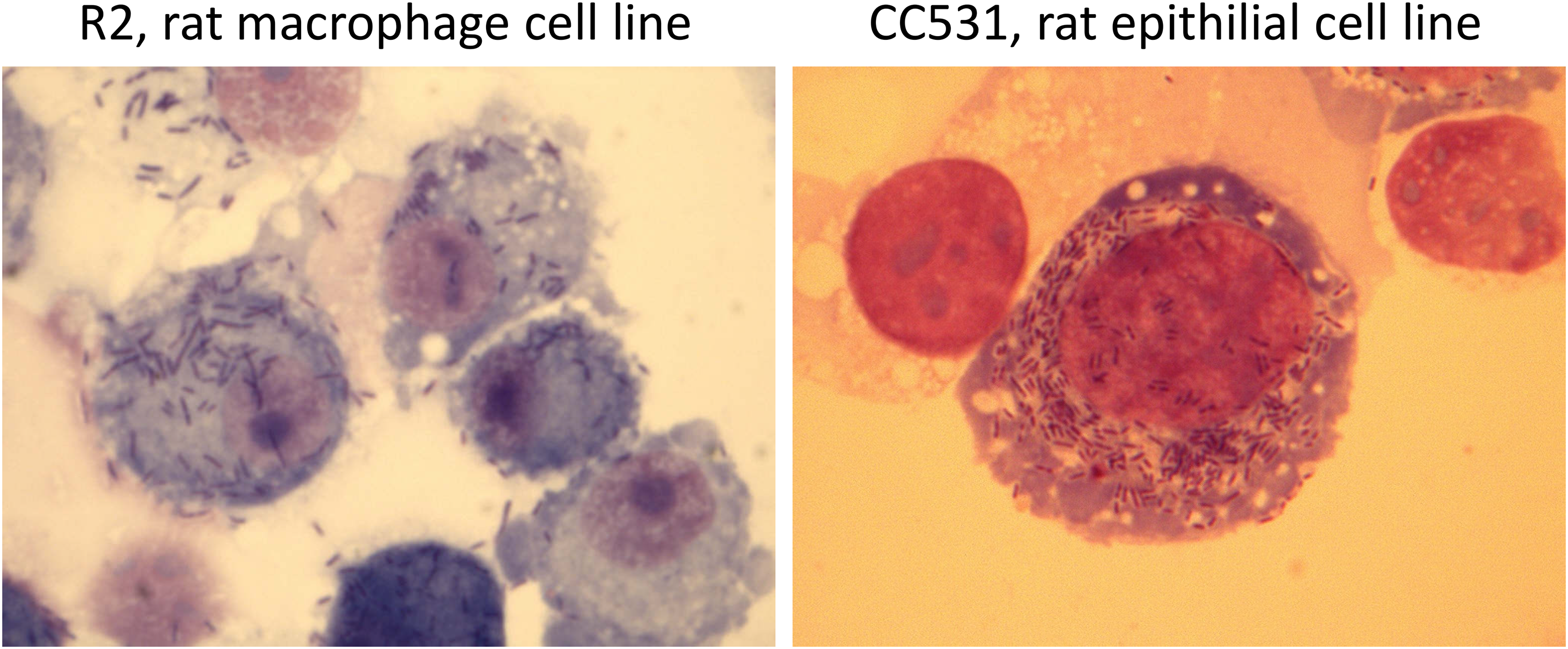
Giemsa-stained cytospin of Listeria monocytogenes-infected rat cell lines after 48 h.
In vivo experiments show NK cells have a protecting roll against Listeria monocytogenes infection
For more in-depth details of the roll and engagement of NK cell receptors and their cognate ligands we employed first an in vivo rat model. Rats, bred in our animal house or purchased from Harlan UK (Bicester, United Kingdom), were injected intravenously with LM, and sacrificed 1- or 3-days postinfection which is consistent with the putative effect of the innate immune system. As a readout, we counted the total number of colony-forming units (CFU) from tissues mainly spleen, blood, bone marrow and lymph nodes.
We chose to work with three rat strains: PVG [RT1c or c; i.e., RT1-Ac -B ⁄Dc -CE ⁄N⁄Mc (class Ia-class II-class Ib), abbreviated c-c-c], AO RT1u (u-u-u) and DA (a-a-av1) has differ in their expression of NK cell receptors. PVG and AO rats have a broad repertoire of Ly49 receptors reacting with MHC-I molecules28,68 while DA rats that lack most of the Ly49 receptors. 28 This dissimilarity in NK receptor repertoire was not reflected in the resistance to LM infection as LM colonies were detected in PVG and DA rats at approximately the same frequencies. The number of colonies after 24 h was higher in AO rats than in PVG and DA rats, insinuating that AO rats were somewhat more affected to LM-burden.
Comparison of athymic nude rats (rnu/rnu, RT1c) which lacks normal T cells while containing a robust NK cell repertoire 69 was a great the possibility to investigate how they handle LM infection compared to normal PVG rats (RT1c). Intriguingly, athymic nude rats were more resistant to bacterial replication than normal littermate rats since they eliminated the infection within 3 days. These results contrasted with severe combined immunodeficiency (SCID) mice studies. Although, SCID mice control the early Listeria infection equally well as normal animals, they become chronic carriers of the bacterium.70–72 Since the innate immune system in these mice does not induce complete clearance of the bacteria, suggesting Ly49 receptors repertoire differences between these species. Another evident observation was the number of NK cells enhancement in athymic nude rats compared with normal animals pointing again to NK cells importance in early controlling of LM infection. An in vitro conformation to the importance of NK cells against LM infection comes from coincubation of LM-infected CC531 cells with isolated splenic NK cells that clearly inhibited LM growth and increased LM-infected CC531 survival (unpublished observations).
Ly49-subset expansion
Another valuable observation in these experiments was the influence of LM on NK-subsets. The first evident observation was the expansion of splenic Ly49+ subset in LM-infected PVG rats. This subset increased from 32% up to 47% (∼45%), while the Ly49− subset decreased from 68% down to 56% (∼18%), described in the first report of our experimental investigation. 9 This may mean that, after engagement of the cognate ligand, these receptors contribute to the specific activation and ‘preferential’ population expansion of defined NK-subsets, which partially show some features of adaptive lymphocytes. The selective in vivo expansion of Ly49+ subset indicated that the NK cells recognizing MHC-I antigens may undergo ligand-induced expansion during the responses to LM.9,39 The frequency increscent of Ly49+ subset during LM infection may result from either a local expansion or direct recruitment from other tissues. The latter was supported by increased frequency of Ly49+ NK cells in the spleen paralleled by a concomitant decrease in the bone marrow during the early phase of LM infection. 73 Re-localization and migration of NK cells is likely controlled by chemokines, which are differentially expressed in tissues at steady-state and during infection (discussed in the in vitro section). 74 Whether the increscent Ly49 subset reconstitute at later time point or remains higher compared to non-infected littermates remains to be determined.
In addition to NK-subset alteration due to LM infection we also observed a unique change in STOK-27 population in the blood of infected rats. This subset is divided into two subsets: STOK-27dim and STOK-27bright subsets in uninfected rats. However, the blood (but not other tissues) STOK-27bright population was reduced/diminished in the infected rats which might be suggested that they have recruited toward other infected sites such as spleen (unpublished observation).
One further fact deduced from the results of these experiments was the expression levels of MHC-I molecules on MØ isolated from different tissues in infected rat. MHC-I molecules were highly up-regulated on OX41+ MØ from spleen, blood, and in the BM, but not cervical LN 24 and 48 h postinfection. 73 The later bias observation would be worth to investigate further. The same was found for splenic NK cells. The MHC-I upregulation was concomitant with a decreased surface expression of Ly49 receptors (known as receptor calibration). This is another indication of a plastic process that adapts NK cells to environmental changes.
Depletion experiments
Expansion Ly49-subset in response to LM infection and indication of the NK cells involvement in athymic rats urged us the depletion strategy of total NK cells and the Ly49-subset. 9 Monoclonal antibody (mAb) 3.2.3 towards NKR-P1A receptor expressed on most NK cells and subset of NKT cells, and mAb DAR13 towards the subset of NK cells encompassing the majority of Ly49 receptors were used. However, depletion experiments were most efficient with AO rats for studies of the Ly49 repertoire. These experiments documented increased bacterial loads in the spleen after systemic administration of the bacterium which indicated the engagement of NK cells and perhaps specifically the Ly49+ subset in the resistance to LM. Interestingly, DA rats that possess very few Ly49 expressing NK cells 28 also cleared LM as efficiently as did PVG rats, insinuating that alternative mechanisms for LM resistance exist.
IFN-γ production by NK-subsets
The indirect killing of infected cells by NK cells are preferred since direct killing may cause organ failure. IFN-γ production by NK cells is therefore thought to be a key factor controlling LM infection. 6 In our experiments we determined the peak IFN-γ responses by NK cells upon infection to be 24 h postinfection, with a decline 48 h postinfection in line with other researchers. We found further IFN-γ production by Ly49+ NK cells were equivalent levels of IFN-γ were produced by these subsets (Ly49− subset), suggesting that IFN-γ production is not unique to a particular subset of NK cells.9,73
Modulation of MHC-I molecules in response to Listeria monocytogenes infection
Changes in MHC-I expression can be a sign of infection. The selective in vivo expansion of Ly49+ subset indicated that the NK cells recognizing MHC-I antigens may undergo ligand-induced expansion during the responses to LM.9,39 Since Ly49+ NK cells kill target cells restricted by class Ib -encoded structures, we speculate that activating receptors on NK cells might recognize nonclassical class Ib ligands. We focused particularly on MØ and epithelial cells since these cells are natural targets for LM infection in vivo and yet important for the defence against LM. In addition, these cells originate from rats expressing either MHC ligands for the activating Ly49s4 or the Ly49s5 receptor. These experiments gave us the several important additional insights, as delineated below. Since normal enterocytes are both difficult to isolate and keep in culture, and normal MØ are quickly destroyed even after exposure to small amounts of LM, we selected three cell lines: R2 MØ (RT1d haplotype) from Wistar rats, 42 rat colon carcinoma cell line (CC531, RT1u haplotype) 45 and the myeloid cell line RmW originated from PVG (RT1c haplotype) rat spleen as a side-product during the generation of an in vitro-passageable variant of the in vivo-restricted Roser leukaemia (a rat T cell leukemic cell line)75,76 to investigate the impact of LM on the surface expression of MHC-Ia and –Ib molecules.
We have reported that LM-infected R2 and CC531 cells, similar to IFN-γ treatment of uninfected cells, displayed increased expression of classical and nonclassical MHC-I molecules, as determined by FACS staining with specific mAbs43–45,73 (summarized in table 1). The enhancement starts after 24 h post infection and reach its maximum level at 72 h that, to our knowledge, was the first clear description of this phenomenon. The most interesting observation was the upregulation of low expressed non classical molecules on LM-infected cells. MHC-I alteration, such as virus-infected cells that display mitigated MHC-I expression, may permit discrimination of normal healthy cells from diseased ones by NK cells. 4 From our experiments, much evidence suggests that the MHC-I alteration may act as ‘danger’ signal for NK cells while the potential role of LM-peptides loaded on RT1-CE molecules recognized by NK cell receptors may be another scenario for the recognition. “Danger signalling” (as we have postulated earlier 77 ) by upregulation of low expressing nonclassical MHC-I on LM-infected cells is unique to the rats. The explanation might come from the fact that recombinant inbred rat strains were devised much more recently compared to mice. Hence, studies in the rats may more likely reflect functions important for survival in the wild. And yet, this could be due to the higher number of activating Ly49 receptors in rat NKC compared to Ly49 genes in mice.31,32
Rat Ly49 receptors recognize both classical and nonclassical MHC-I molecules
Increased expression of both MHC-Ia and MHC-Ib molecules in LM-infected cells contrasted with infection with other intracellular pathogens like CMV, which either downregulate MHC-I or instructs the infected cell to express “decoy” MHC-I like molecules that are not recognized by CTL and inhibit NK cells. This MHC-Ib upregulation was beneficial to our studies as Ly49:MHC-Ib interactions are difficult to investigate due to low levels of MHC-Ib expression in uninfected cells.
In vitro experiments with reporter cell lines, expressing Ly49s5, Ly49s4 or Ly49i4 receptors strongly indicated that increased expression of RT1-CE molecules on infected cells may be detected by Ly49 receptors. 43 –45 However, since each NK cell expresses several stimulatory and inhibitory NK cell receptors, the overall result of NK cell recognition of a target cell will depend on several NK-receptor MHC-I interactions. 78 An indication to support this assumption comes from comparison of IFN-γ production of NK cells against LM-infected RmW and R2-MØ cells. LM-infected RmW cells upregulated their MHC-I molecules weak or not at all and consequently NK cells produced less IFN-γ in response as it was compared with LM-infected R2-MØ. It is worth mentioning that these rat cell lines do not themselves produce significant amounts of IFN-γ, and therefore LM upregulates their MHC-I molecules independently of IFN-γ which is not like the in vivo situation, where bystander cells may produce enough IFN-γ for upregulation to occur.
The Ly49s4 and Ly49i4 receptors recognize the same ligand(s). 42 Often signals generated by inhibitory NK cell receptors dominate over their stimulatory counterparts. However, if the activating receptors for a given MHC ligand are expressed by different NK cell subsets than their inhibitory counterparts, 72 the chance that activation is overridden by inhibitory receptors is minimized. This is the case for NK cells expressing Ly49s5 and Ly49i5 being present on different subpopulations of NK cells. 72 Whether Ly49s4 and Ly49i4 expressed by different subsets of NK cells remains to be determined.
Notably, rodent Ly49 receptors in contrast to KIRs, which bind at the top of MHC class I molecules, they bind laterally (also called ‘site 2’), making contact with amino acid residues in the α2 and 3 domains of the heavy chain as well as residues in the associated β2-microglobulin molecule. 79 Always Ly49 receptors usually do not show peptide specificity, but peptides may stabilize the MHC-I molecule which is important for sufficient surface expression. However, there are some reports that indicate Ly49 receptors to be peptide selective, such as the Ly49G2 receptor.80,81 Ly49 receptors may also recognize virally encoded MHC-I like molecules, such as m157 encoded by MCMV recognized by Ly49H. 82
Adaptive-like NK cell response
The studies with Rag-deficient mice have suggested that NK cells may have the ability to recognize virally infected cells in an antigen-specific manner, allowing for subsequent NK cell-mediated recall responses.83–88 O'Leary and co were among the first investigators who proposed memory NK cells and the hypothesis has become more and more accepted by independent researchers. Some of our following experimental evidence may support this hypothesis, summarized in table 2.
Comparison of different rat cell types and transfected mouse RAW cells expressing RT1-Ib with Listeria monocytogenes or IFN-γ and subsequent responses.

The table shows three rat cell types (R2, CC531 and RmW cells) and one mouse cell line (RAW cell line) transfected with a gene from the RT1-CE region of haplotype d (termed RAW-CE16d) infected either with LM or stimulated with IFN-γ. The first row shows the modulation of MHC-I molecules on these cells after LM infection. Row two after stimulation with IFN-γ. Rows three and four show how reporter cell line stably transfected with Ly49 receptors responses against LM-infected or IFN-γ stimulated cells respectively. Arrows indicate ↑ increscent, ↓ reduction and ↔ unchanged response or regulation.
By extending our experiments to a single nonclassical MHC-I molecule expression on mouse cell line (mouse RAW cells) we found some interesting results that may support adaptive behaviour. After successful RT1-CE16 d surface upregulated expression on LM-infected we observed stronger response by Ly49 receptor compared to IFN-γ upregulated cells which deserve a follow-up-analysis. 43 Theoretically speaking, listerial peptide loaded MHC-I molecules may influence the interaction by conformational changes in MHC-I molecules in which Ly49 recognition enhances. Although NK alloreactivity in mice is regulated primarily by inhibitory Ly49 receptors, activating Ly49 receptors are equally important in rats. 44 NK triggering by activating Ly49 receptors need to meet minimal thresholds to establish the triggering cascade and are likely more amenable to pathogen-mediated changes in ligands or in peptide mediated altered avidity. Indeed, peptide specificity of Ly49 has been proven to be true in the case of mouse Ly49P, which binds MCMV-derived peptide in the context of self MHC. 89 This has led to an apparently novel “danger system” in rats using Ly49's directed against normally low expressing MHC-I molecules that are performed by other receptor system such as NKG2D in other species. Here the lines between innate and adaptive immune receptors are further blurred. What makes the mechanism of this interaction particularly difficult to solve is this conformational change. Determination of detailed structure of Ly49:MHC-I by X-ray crystallography should shed light on the interaction.
Modulation of Clr molecules in response to Listeria monocytogenes infection
Functional interactions of NKR-P1:Clr is believed to be a MHC-independent ‘missing self’ recognition during normal NK cell development and in recognition of infected and stressed cells. 56 Clr molecules are broadly expressed by normal cells and their regulation can decide the faith of NK cell response. 90 Rat CMV infection rapidly induces a reduction in host Clr11 (ligand of the activating NKR-P1A) expression that may lead to killing by NK cells. The RCTL molecule, encoded by rat CMV, may hence function as a decoy ligand protecting infected cells from NK killing via interactions with the inhibitory NKR-P1B receptor.56,91 A similar LM-strategy was observed for downregulation of ligands for NKR-P1G and NKR-P1F receptors which are highly expressed in gut mucosa lymphocytes.58,92 In that case, LM-downregulation of Clr molecules in epithelial cells, the main cell type in intestine, is therefore an attempt to escape from NKR-P1 receptor recognition. More conformation for this observation awaits specific antibodies against Clr molecules.
NKR-P1F and NKR-P1G are highly expressed in gut mucosa lymphocytes while the inhibitory NKR-P1G receptor is expressed exclusively by the Ly49+ subset. 58 Studies in mice has shown that inhibitory NKR-P1G receptor is involved in mucosal immunity. 93 If LM evolutionally downregulate Clr molecules to escape stimulation of activating NKR-P1F+ NK cells may interrupt the inhibition by NKR-P1B+ NK cells, or upregulation of MHC-I molecules can, in turn, stimulated the MHC-I binding Ly49+ subset. The modulation of Clr molecules indicate that in addition to MHC-I molecules upregulation, several, perhaps many, other NK-ligands are probably involved; meaning that the mechanism machinery of recognizing of LM-infected cells by NK cells may be much more complex.
NK-response variation to different cell types infected with Listeria monocytogenes
Throughout all in vitro infection experiments we mostly used three different rat cell types (R2-MØ and epithelial CC531). Some of those responses were observed to be different (table 2). For instance, NK cells were proliferated against LM-infected R2 cells, but not infected CC531 cells while there was no difference in production of amount of IFN-γ. 76 We assumed that IFN-γ production might be a proper NK-response when LM has crossed the epithelial layer defense in intestine, but in vivo conformation for this notion remains to be proven.
Future studies
Depletion of other NK-subsets with available rat antibodies has not yet been tested. Depletion of STOK27+ NK cells (Ly49− subset) by STOK27 mAb, in the control of LM infection remains to be discovered. The rat anti-NKp46, WEN23 mAb binds solely on NK cells, in contrast to the antibody 3.2.3 we used in our depletion experiments. 10 To exclude the contribution of NKT cells which was also depleted in NK-depletion experiments it is interesting to repeat these experiments with WEN23. Depletion of NK cells in athymic rats have not yet been tested and that will also reinforce our observations. NKG2D ligands: LM likely triggers expression of NKG2D ligands on MØ in response to TLR stimulation that has been shown in mouse by Hamerman and colleges. 27 Examining the NKG2D ligand in rats infected with LM therefore might reveal the role of the receptor in question. In addition, the ligands and function of several above-mentioned rat NK cells are still undiscovered. What contribution they give during LM-infected cells remains to be investigated. Activating NK receptors are likely the key in the establishment of NK cell memory as has been shown in mice by the establishment of CMV-specific Ly49H activation receptor. 94 Further experiments by the rat LM infection model may be able to answer this question.
Concluding Remarks
The mechanisms NK cells explore to recognise infected cells are unquestionably complicated, and the principals that guide this process have not yet been worked out in detail. Our experiments contributed to shed light on some of these mechanisms. Certainly, there are many questions that remains to be answered. Do NK cells play a protective role against LM depending on species, strains, tissues, and route of infection? Are all NK-receptors involved? Which type of receptors play the most important role; activating or inhibitory? Can LM-peptide presentation on MHC-Ib molecules reinforce the NK cell response? And if this is true, what is its relevance for human NK cells? All of which means that further investigations are still required to answer these, and other, questions.
Footnotes
Acknowledgments
In the memory of professor Bent Rolstad who initiated this research project.
Author's declaration
The author has no financial or commercial conflicts of interests.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. NLA pays for the publication fee of this article.
