Abstract
Numerous immunosuppressive cells such as myeloid-derived suppressor cells (MDSCs) and inhibitory cytokines identified in melanoma microenvironment have the important role in immune escape. Therefore, in this study we analyzed monocytic (m)MDSCs in peripheral blood of metastatic melanoma (MM) patients. In peripheral blood of 35 MM patients and 30 healthy controls we analyzed percentage of CD14 + HLA-DR- mMDSCs in monocyte gate and the mean fluorescence intensity of Foxp3 in CD25 + CD4 + regulatory T cells by Flow cytometry. Serum levels of transforming growth factor beta, interferon-gamma, interleukin (IL)-6, IL-8, IL-10 are measured by ELISA assays. In this study MM patients have significantly higher percentage of CD14 + HLA-DR- mMDSCs, as well as increased the baseline mMDSC/PBMC subset (NK, T, B cells, monocytes) ratio. Although there is no significant difference in the percentage of mMDSCs between groups of MM patients with different localization of distant metastasis, patients with elevated serum lactate dehydrogenase (LDH) have statistically significant higher percentage of these cells compared to LDH negative patients. Furthermore, in MM patients there is statistically significant positive correlation between values of IL-10 and the percentage of mMDSCs, only. Therefore, therapeutics that target circulating mMDSCs and IL-10 may have a big importance in the improvement of antitumor immunity in MM patients.
Keywords
Introduction
Melanoma, malignant tumor of melanocytes, is considered as highly immunogenic malignancy with local and systemic activation of immune response and significant infiltration of immune cells. Despite this, in tumor microenvironment (TME) of melanoma it has identified numerous immunosuppressive cells and inhibitory soluble mediators that contribute to immune escape.1,2 Myeloid-derived suppressor cells (MDSCs) and regulatory T (Tregs) cells have an important role in establishing an immunosuppressive TME in multiple solid tumors including melanoma. 2 The activation and accumulation of these cells is induced by chronic inflammation in TME and they by production of numerous immunosuppressive inflammatory mediators inhibit anti-tumor immune response and favor tumor progression. 3 The MDSCs are immature myeloid cells that vary in their phenotype and function. Two main subtypes of these cells have been reported: granulocytic (gMDSC) and monocytic (mMDSC). 4 By producing interleukin (IL)-1, IL-6, reactive oxygen species (ROS), nitric oxide (NO), MDSCs inhibit CD3 + CD8 + cytotoxic T cells (CTL), as well as CD3 + CD4 + helper T cells (Th) functions. Also through membrane bound transforming growth factor beta (mTGF-β), MDSCs can suppress natural killer (NK) cells. In addition, MDSCs enhance immunosuppression by activation and expansion of Treg cells.4,5 Immunosuppressive CD4 + CD25highFoxP3 + cells, Treg cells decrease the function of T and NK cells in tumor-bearing hosts, by cell-to-cell contact due to the expression of inhibitory molecules on their surface, as well as upon the secretion of soluble factors such as TGF-β. The accumulation of Tregs in the TME has been shown to correlate with an unfavorable prognosis in numerous malignancies. 6 Additionally, melanoma cells by producing IL-6 and IL-8 recruit MDSCs, Tregs and tumor-associated macrophages (TAMs) to the TME and promote immunosuppression, angiogenesis and metastasis. 7 Also, cancer cells, as well as MDSCs, Tregs and TAMs in TME secrete IL-10, an important immunosuppressive cytokine. It enhances tumor cell survival, proliferation and metastasis by the inhibition of potent antitumor cytotoxic NK and CTL cells. 8 Furthermore, IL-10 together with IL-1, IL-6, TNF, prostaglandin E2 (PGE2) promotes myelopoiesis, skewing differentiation of progenitors into monocytes inducing generation and survival of mMDSCs. 9 Also, IL-10 induces the expression of Foxp3, TGF-β receptor 2 and TGF-β on activated Tregs, and thus stabilizes the phenotype and function of these cells. 10
In this study we have analyzed CD14 + HLA-DR- mMDSCs and Treg cells in peripheral blood of metastatic melanoma patients and their role in the suppression of immune response. Elimination of these immune suppressive cells is one of the major current challenges in immunotherapy of cancer.
Methods
Blood samples
Peripheral venous blood was obtained from 35 metastatic melanoma (MM) patients (stage IV according to 8th modified AJCC/UICC staging system) 11 at the time of diagnosis prior to systemic therapy and 30 healthy controls (HC). The patients and controls were age and sex matched with no evidence of infection, autoimmune or any other disease. The study was approved by the Ethical committee of Institute of Oncology and Radiology of Serbia (No. 5004/2-01) and all patients and HC have signed the informed consent before inclusion in the study. Clinicopathological characteristics of all patients and characteristics of HC enrolled in this study are listed in Table 1. Performance status was evaluated from 0 to 4 by the Eastern Cooperative Oncology Group (ECOG) scale. In order to analyze the percentage of mMDSCs in different groups of MM patients, patients are divided in 3 groups based on the localization of distant metastases. 11 Patients that have metastases in distant skin, the subcutaneous layer or in distant lymph nodes (M1a) and patients with metastases in the lungs (M1b) are included in M1a + M1b group, while the patients with distant metastases in non-central nervous system (CNS) viscera are included in M1c group. The patients in group M1d have metastasized to the CNS. MM patients were also divided into two groups according to lactate dehydrogenase (LDH) serum concentrations. The first group (LDH-) included patients with normal LDH serum values (<460 U/l), whereas the other group (LDH + ) included patients with elevated LDH (>460 U/l).
The characteristics of healthy controls (hc) and metastatic melanoma patients (mm).

Peripheral blood mononuclear cells (PBMCs) isolation
Peripheral blood mononuclear cells were isolated from heparinized blood obtained from MM patients and HC using Lymphoprep (Nypacon, Oslo, Norway) density gradient, centrifuged at 500 g for 40 min, and washed three times in RPMI 1640 culture medium (CM), (Sigma, St Louis, USA) supplemented with 10% fetal calf serum (FCS) (Sigma). After washing PBMCs were immediately used for further analysis.
Flow cytometric analysis
The immuno-phenotype of PBMCs was done as previously described. 12 Briefly, 1.0 × 105 PBMCs in 100 µL RPMI 1640 supplemented with 10% FCS were incubated for 30 min at 4°C with 20 µL of appropriate monoclonal antibodies (mAbs) combination, washed twice with ice-cold phosphate-buffered saline (PBS) and fixed with 1% paraformaldehyde prior to FACS analyses. The marker expression was quantified on FACSCalibur flow cytometer (Becton Dickinson). A total of 50 000 gated events, verified as peripheral blood lymphocytes (PBLs) or monocytes according to their physical characteristics (forward scatter (FSC) and side scatter (SSC)), were collected per sample and analyzed using CellQUEST software. Exclusion of non-specific fluorescence was based on matched IgG1 isotype mAb combinations conjugated with FITC, PE and PerCP (Becton Dickinson). The following combinations of directly labeled mAbs, CD3FITC/CD56PE/CD19PerCP and CD3FITC/CD8PE/CD4PerCP (Becton Dickinson, San Jose, USA), were used for the analysis of NK (CD3-CD56 + ), CTL (CD3 + CD8 + ), Th (CD3 + CD4 + ) and B (CD19 + ) cells in lymphocyte gate. For CD4 + CD25 + Foxp3 + Treg analysis, intracellular Foxp3 (anti-Foxp3PE (Becton Dickinson)) was measured in 1 × 106 freshly isolated, unstimulated PBMCs followed by fixing and permeabilizing with the Permeabilizing Solution 2 (Perm 2) accords to the manufacturer's instructions. Then the cells were analyzed for the surface expression of CD25 and CD4 (CD25FITC, CD4PerCP mAbs (Becton Dickinson)). Finally, the cells were washed and analyzed by flow cytometry directly after preparation. The mean fluorescence intensity (MFI) of Foxp3 was analyzed in CD25 + CD4 + T cells in PBLs for 10 MM patients. In monocyte gate mMDSCs cells were defined as CD14 + HLA-DR- for 35 MM patients. Baseline mMDSC/PBMC subset ratio is represented in indexes calculated as the percentage of mMDSC for each MM patient and HC devides with the percentage of appropriate PBMC subset (CD3-CD56 + NK, CD3 + CD8 + CTL, CD3 + CD4 + Th, CD19 + B cells, monocytes) for 30 MM patients and 25 HC.
The measurement of cytokines by ELISA
Blood samples were obtained from MM patients by venipuncture and clotted at room temperature. The sera were collected following centrifugation and frozen immediately at −80 °C until analysis. The concentrations of TGF-β, IFN-γ, IL-6, IL-8 and IL-10 in sera of the investigated MM patients were determined by commercial uncoated ELISA kits (Invitrogen, Massachusetts, United States), according to manufacturer's instructions, as previously described. 13
Statistical analysis
Significance of difference for parameters between HC and MM patients, as well as between different groups of MM patients was determined by nonparametric Mann-Whitney exact test, while the correlation between serum levels of investigated cytokines and the percent of mMDSCs in MM patients was performed by Spearman's rank correlation. The results were considered statistically significant if p ≤ 0.05. Data and statistical analyses were done in GraphPad Prism.
Results
Decreased percentage of NK cells in mm patients
Analysing the percentage of immune cells in MM patients and HC we show that patients have significantly decreased (p < 0.01, Mann–Whitney exact test) percentage of CD3-CD56 + NK cells compared to HC (Table 2). There is no significant difference (p > 0.05, Mann–Whitney exact test) in the percentage of CD3 + CD8 + CTL, CD3 + CD4 + Th, CD19 + B cells and monocytes between MM and HC (Table 2).
The percentage of peripheral blood mononuclear cell subsets in healthy controls (hc) and metastatic melanoma patients (mm).
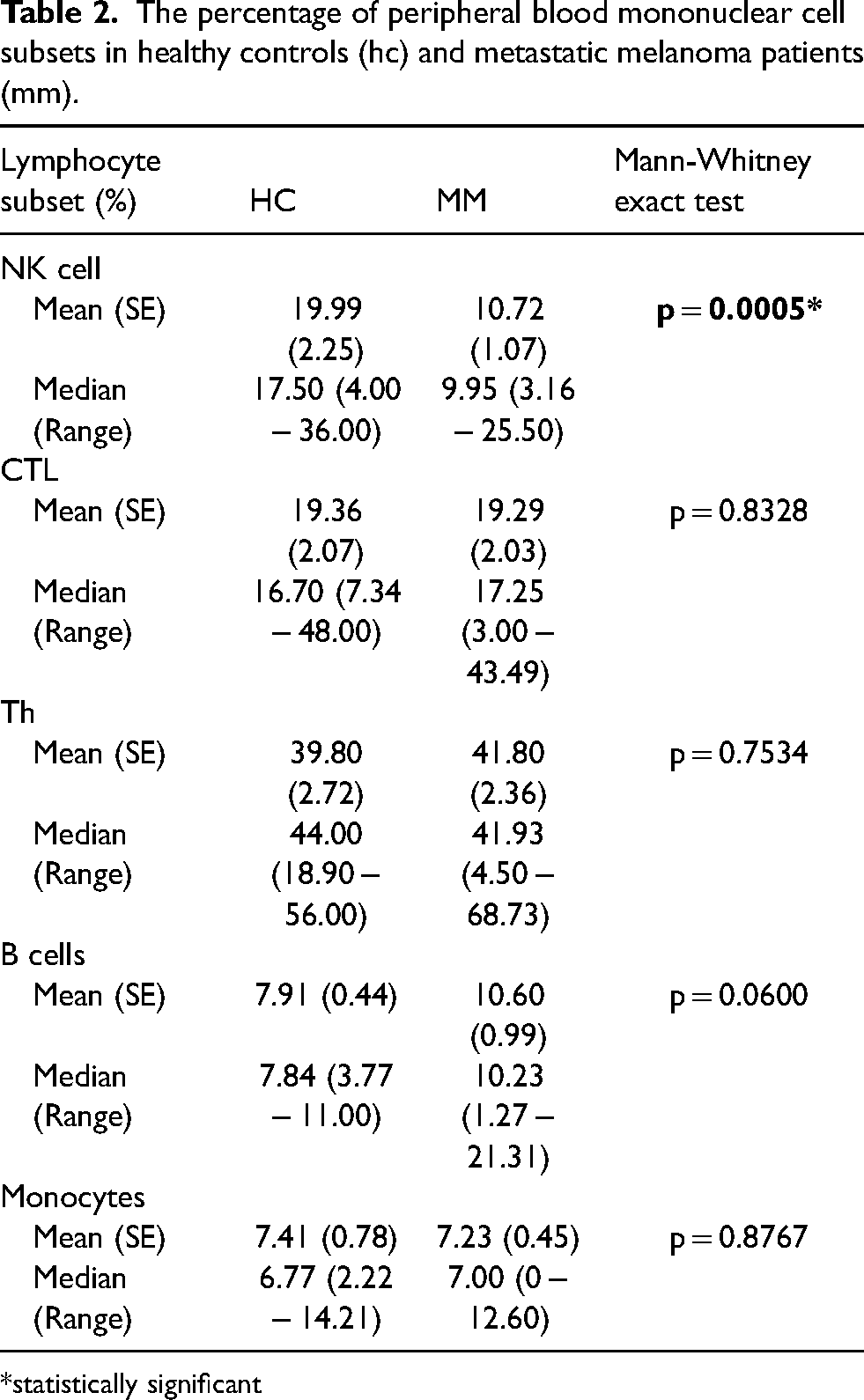
*statistically significant
Increased mMDSCs and tregs in mm patients
Metastatic melanoma patients have significantly higher (p < 0.01, Mann–Whitney exact test) percentage of CD14 + HLA-DR- mMDSCs (Figure 1(a), (c)), as well as MFI of Foxp3 in CD25 + CD4 + Tregs (p ≤ 0.05, Mann–Whitney exact test) compared to HC (Figure 1(b)).

Mm patients compared to HC have significantly higher a) percentage of CD14 + HLA-DR- mMDSCs (**p < 0.01, Mann–Whitney exact test) and b) mean fluorescence intensity (MFI) of Foxp3 in CD25 + CD4 + tregs (*p ≤ 0.05, Mann–Whitney exact test); c) Representative flow cytometric dot plots of mMDSCs analysed in monocyte gate the results are estimated by flow cytometry and shown as mean values ± se for 35 MM patients and 30 HC for mMDSCs analysis and 10 MM patients and 10 HC for Foxp3 analysis.
Increased mMDSC/PBMC subset ratio in mm patients
Analysing the baseline mMDSC/PBMC subset (NK, CTL, Th, B cells, monocytes) ratio we show significantly increased (p ≤ 0.05, p < 0.01 Mann–Whitney exact test) indexes in MM patients compared to HC (Figure 2).

The baseline mMDSC/PBMC subset ratio is significantly increased (*p ≤ 0.05, **p < 0.01 Mann–Whitney exact test) in MM patients compared to HC. Results are expressed in indexes calculated as the percentage of mMDSC for each MM patient and HC devides with the percenatage of appropriate PBMC subset (CD3-CD56 + NK, CD3 + CD8 + CTL, CD3 + CD4 + Th, CD19 + B cells, monocytes). Results are shown as mean ± SE for 30 MM patients and 25 HC.
Increased percentage of mMDSCs in LDH + mm patients
Analyzing the difference between MM patients groups according to a different localization of metastasis (M1a + M1b, M1c, M1d groups) we show that there is no significant difference (p > 0.05, Mann–Whitney exact test) in the percentage of mMDSCs between investigated groups of patients (Figure 3(a)). We also analyse mMDSCs frequency in LDH- and LDH + MM patients and show that LDH + patients have statistically significant higher percentage of these cells compared to LDH- MM patients (p ≤ 0.05, Mann–Whitney exact test) (Figure 3(b)).

a) There is no significant difference (p > 0.05, Mann–Whitney exact test) in the percentage of mMDSCs between groups of MM patients according to localization of distant metastasis (M1a + M1b, M1c, M1d); b) LDH + MM patients have statistically significant higher percentage of mMDSCs (*p ≤ 0.05, Mann–Whitney exact test) compared to LDH- patients. The results are shown as mean values ± SE for 35 MM patients and 30 HC.
Positive correlation between serum il-10 and percentage of mMDSCs in mm patients
We also show in MM patients the statistically significant positive correlation between values of serum IL-10 and the percentage of mMDSCs (p ≤ 0.05, Spearman's rank correlation) (Table 3, Figure 4). On the other hand, there is no statistically significant correlation (p > 0.05, Spearman's rank correlation) between the values of the other investigated cytokines (TGF-β, IFN-γ, IL-6, IL-8) and the percentage of mMDSCs (Table 3).

In MM patients there is statistically significant positive correlation (p ≤ 0.05, Spearman's rank correlation) between serum values of IL-10 estimated by ELISA and the percentage of mMDSCs estimated by flow cytometry in monocyte gate. These parameters were analysed in 35 MM patients.
Correlation between cytokine serum values and percentage of mMDSCs in metastatic melanoma patients.

*statistically significant
Discussions
The tumor cells and numerous other cells in TME by realizing various immunosuppressive factors direct the differentiation of anti-tumor Th1 lymphocytes and monocytes towards immunosuppressive Tregs and mMDSCs, respectively. 14 Also, it is known that mMDSCs and Tregs are important immune modulators that inhibit antitumor immune response of lymphocytes such as NK, CTL, Th and B cells in tumor-bearing animals and cancer patients.14,15 In this study, we show significantly decreased percentage of NK cells in peripheral blood of MM patients compared to controls that is in accord with data in literature. 16 NK cells elicit strong antitumor immune response through secretion of cytotoxic molecules, perforin and granzyme B, as well as by IFN-γ production. However, melanoma cells can develop many mechanisms to evade NK cell antitumor activity, such as upregulating ligands for inhibitory NK cell receptors, secreting inhibitory cytokines and recruiting immunosuppressive cells such as MDSCs and Tregs.17,18 In this study, we show significantly increased percentage of CD14 + HLA-DR − mMDSC cells in the blood of advanced, MM patients compared with healthy individuals that is in accord with previous reports.19,20 Furthermore, the baseline mMDSC/PBMC subset (NK, CTL, Th, B cells, monocytes) ratio is also significantly increased in melanoma patients with distant metastasis compared to HC. These results indicate that MDSCs might have the influence on the important cells of antitumor immune response. MDSCs are potent suppressors of T and NK cell activation and function through various mechanisms, including production of immunosuppressive cytokines such as TGF-β and IL-6, induction of enzymes like arginase 1 (Arg1) and inducible nitric oxide synthase (iNOS) and differentiation of Tregs. 15 Furthermore, MDSCs can inhibit NK cell cytotoxicity and IFN-γ production through cell-to-cell contact mostly mediated by mTGF-β, which has the ability to down-regulate the expression of NK cell activating receptors, NCRs and NKG2D. 21 Additionally, by producing TGF-β, MDSCs induce the expression of FoxP3, the Treg lineage-specific transcription factor. 22 In this study we show significantly higher MFI of Foxp3 in CD4 + CD25 + Treg cells in MM patients compared to HC. Therefore, the data in this study suggest that the immune system in cancer patients could be suppressed due to an augmentation in Treg and mMDSC populations.
It is known that there is a causal relationship between melanoma disease status and the presence of MDSCs and that patients with no active disease or in the regression have physiological frequencies of these cells. 20 Meyer et al. 23 have shown that MDSCs frequencies in ipilimumab-treated stage IV or unresectable stage III melanoma patients were independent of baseline serum LDH levels but tended to increase in patients with metastasis in viscera (M1c) compared to patients with metastases in skin or lymph nodes (M1a). In this study, we only analyzed advanced, Stage IV MM patients before systemic therapy and although there is no significant difference in the percentage of mMDSCs between the groups of MM patients according the localization of metastasis (M1a + M1b, M1c, M1d groups), we show that MM patients with elevated LDH serum values have statistically significant higher percentage of mMDSCs compared to LDH- patients. Baseline serum LDH is an established, independent negative prognostic factor for survival24,25 and it is a part of AJCC classification for stage IV melanoma. 11
Accumulation of MDSCs is mainly influenced by the various cytokines produced in the TME. Therefore, proinflammatory cytokines such as TNF, IL-1β, IL-6 secreted by the tumor cells, as well as IFN-γ, IL-4, IL-10 and IL-13 secreted by tumor-associated T cells affect differentiation of myeloid progenitors and promote suppressor activity of MDSCs. 26 Also, new evidence shows that insulin-like growth factors (IGFs) regulate the phenotype and functions of immune cells and induce immunosuppression. 27 Therefore, they have the ability to increase the production of IL-10 and TGF-β, as well as promote the polarization of the pro-inflammatory M1 macrophages toward the anti-inflammatory, inhibitory M2 macrophages. Furthermore, IGFs via the signal transducer and activator of transcription 3 (STAT3) stimulate MDSCs and Tregs.28,29 Therefore, in this study we show in MM patients the statistically significant positive correlation between the values of serum IL-10 and the frequency of mMDSCs. In support of these results, a previous work demonstrated the positive correlation between plasma IL-10 and CD33+HLADR−CD15− MDSCs in gastrointestinal malignancies. 30 The data obtained in this study reveal novel relationship between individual CD14 + HLA-DR- mMDSCs subset and systemic cytokine IL-10 that suppress immune response in advanced MM patients. However, in this study we did not find the association between the percentage of mMDSCs and the serum values of the other investigated cytokines, TGF-β, IL-6, IL-8 and IFN-γ. The large analysis would be desirable to more precisely define the relationship between these proinflammatory cytokines and immunosuppressive cells, such as MDSCs.
In this study we show that the frequency of CD14 + HLA-DR- mMDSCs is increased in MM patients especially in those patients with elevated serum LDH. Furthermore, we find a positive correlation between the percentage of these cells and serum IL-10, that are the important inhibitors of immune response. The results of this study could be used to identify potential prognostic indicators related to the MDSCs and IL-10 at baseline and over time, these immune components may be targets for new immunotherapies.
Footnotes
Acknowledgments
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (175056 and 451-03-68/2022-14/200043). We wish to thank Mrs. Jasna Popović Basić for excellent technical work.
Conflict of interest
None declared.
Contribution statement
KMM, AV, NTM, MN and VJ conceived the idea of the study. All authors contributed to the design of the research and were involved in data collection. KMM, AV and NTM performed Flow cytometric analysis, while KMM and AV did ELISA assays. KMM, AV, NB, SM and VJ analyzed the data and prepared the manuscript. All authors approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Ministry of Education,Science and Technological Development of the Republic of Serbia number: 451-03-47/2023-01/200111.
