Abstract
Osteoarthritis (OA) is a common joint disease that is characterized by inflammation and cartilage degradation. Death-associated protein kinase 1 (DAPK1) is a multi-domain serine/threonine kinase and has been reported to be involved in the progression of OA. However, its role and mechanism in OA remain unclear. Here, we found the expression of DAPK1 in OA cartilage tissues was higher than that in normal cartilage tissues. The expression of DAPK1 in chondrocytes was up-regulated by IL-1β. Knockdown of DAPK1 promoted cell viability and anti-apoptotic protein expression, while it inhibited the apoptosis rate and pro-apoptotic protein expressions in IL-1β-induced chondrocytes. In addition, DAPK1 inhibition reduced the levels of inflammatory cytokines and expressions of matrix metalloproteinases (MMPs), and increased the expressions of collagen II and aggrecan. The data of mechanistic investigation indicated that the expression of pigment epithelium-derived factor (PEDF) was positively regulated by DAPK1. Overexpression of PEDF attenuated the effects of DAPK1 knockdown on IL-1β-induced cell viability, apoptosis, inflammation, and cartilage degradation. Furthermore, PEDF overexpression restored the activity of the NF-κB pathway and NLRP3 inflammasome after DAPK1 knockdown. Collectively, down-regulation of DAPK1 inhibited IL-1β-induced inflammation and cartilage degradation via the PEDF-mediated NF-κB and NLRP3 inflammasome pathways.
Introduction
Osteoarthritis (OA) is the most common clinical degenerative disease. Its high incidence is positively correlated with aging.1,2 OA is characterized by joint pain, stiffness, and impaired mobility. 3 It not only seriously affects the quality of life of patients, but also brings heavy burdens to family and society. 4 With the rapid growth of the aging population in our country, the impact and burden of OA on the family and entire society have become increasingly prominent.5,6 However, the detailed molecular mechanism underlying the development of OA is still unclear.
The main pathological features of OA are loss of articular cartilage, extracellular matrix (ECM) damage, and chronic inflammation of the synovium of the joints. 4 Chondrocyte, the only cell type presented in articular cartilage tissue, is of great significance to maintain the integrity of the cartilage matrix and the stability of its physical and chemical properties. 7 Inflammatory factors, especially IL-1β, can cause chondrocyte apoptosis. 8 As the main components of the chondrocyte matrix, collagen II and aggrecan maintain the relative stability of chondrocytes, while their synthesis are directly affected by IL-1β. 9 Matrix metalloproteinases (MMPs) play an important role in ECM degradation of cartilage and subsequent cartilage destruction.9,10 During the pathogenesis of OA, IL-1β induces the expressions of MMPs in chondrocytes, thereby exacerbating the degradation of collagen and proteoglycans, which is considered to be one of the key mechanisms in the pathological progression of OA. 11
Death-associated protein kinase 1 (DAPK1), a Ca2+/CaM-dependent serine/threonine protein kinase, is a class of genes encoding enzymes related to cell death.12,13 Initially, studies mainly reported its role in tumour suppression. For example, its expression is reduced in tumour cells, and is closely related to the occurrence, development and metastasis of tumours.14,15 In recent years, researchers have found that DAPK1 also has a regulatory role in inflammation.16,17 DAPK1 is related to cytokine and chemokine signal transduction in OA chondrocytes. 18 Nevertheless, the mechanism of DAPK1 in OA has not been reported.
Pigment epithelium-derived factor (PEDF) is an endogenous secreted glycoprotein with a molecular mass of 50 kDa and is widely expressed in various tissues.19,20 It plays a role in angiogenesis, inflammatory response, oxidant stress, and other biological functions.19,21,22 Recent studies have shown that PEDF plays a regulatory role in the process of chondrocyte and osteoblast differentiation, endochondral ossification and bone remodelling.19,23 Previous study revealed that PEDF expression was significantly up-regulated in advanced OA chondrocytes.24,25 However, the mechanism of action of PEDF in OA remains unclear. Moreover, PEDF expression was proved to be up-regulated by DAPK1 in pancreatic cancer cells. 26 Therefore, the purpose of this study was to observe the role of DAPK1 in IL-1β-induced chondrocytes and the potential relationship between DAPK1 and PEDF. These results may provide an effective clinical method for the treatment of OA.
Material and methods
Tissue samples
Human OA cartilage tissues were collected from 22 patients with OA (age 65.5 ± 9.5 yr) who underwent knee arthroplasty surgery. Normal human cartilage tissues were collected from 15 trauma patients with no history of OA (age 17.6 ± 24.9 yr). All specimens were selected from Yantaishan Hospital, and all procedures were approved by the Research Ethics Committee of Yantaishan Hospital. This study was performed in accordance with the Declaration of Helsinki (1964), and written informed consent was obtained from all patients.
Cell culture and transfection
Human primary chondrocytes were obtained from Shanghai Saibaikang Biotechnology Company (Shanghai, China). Chondrocytes were cultured in DMEM/F-12 (Sigma, St Louis, MO, USA) medium containing 10% FBS (Thermo Fisher Scientific, MA, USA) at a humidified incubato (37°C, 5% CO2). DAPK1 siRNAs, pcDNA-PEDF, and their negative controls were all synthesized by GenePharma (Shanghai, China). For transfection, chondrocytes were placed in 6-well plates with a density of 2.0 × 105 cells/well, and the vectors were transfected into cells using Lipofectamine 3000 reagent (Invitrogen, Carlsbad, CA, USA) for 48 h according to the manufacturer's instructions. Then, cells were cultured for another 24 h with 10 ng/ml IL-1β (Pepro Tech EC, London, UK) before further experiments.
RNA isolation and quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from tissues or chondrocytes by TriZol reagent (Invitrogen, Carlsbad, CA, USA). The cDNA was synthesized and analysed using the PrimeScript RT reagent kit (TaKaRa Biotechnology, Kusatsu, Japan). The PCR was processed by SYBR Premix Ex Taq (Takara Biotechnology, Kusatsu, Japan). GAPDH was used as an endogenous control. The relative expression was analysed using the comparative 2−ΔΔCt method. Primers used in the qRT-PCR were as follows: DAPK1 (Forward): 5’-CAAGACAGGCACGGCAATAC-3’ (Reverse): 5’-GGCTCCCATCAGACAGAGATAC-3’; GAPDH (Forward): 5’-CACCCACTCCTCCACCTTTG-3’; (Reverse): 5’-CCACCACCCTGTTGCTGTAG-3’.
Western blotting
The total protein from tissue and treated or non-treated chondrocytes was extracted using RIPA lysis buffer (Beyotime, Shanghai, China) containing protease inhibitors (Beyotime, Nanjing, China). Then, the total protein was collected after centrifugation at 4°C and 4000 g for 15 min. Protein samples were separated by SDS-PAGE (10% gel) and transferred to a polyvinylidene difluoride membrane (Millipore, Burlington, USA). The membranes were immunoblotted with anti-DAPK1 Ab (sc-136286), anti-Bcl-2 Ab (sc-7382), anti-Bax Ab (sc-20067), anti-cleaved-caspase3 Ab (sc-7272), anti-MMP-1 Ab (sc-21731), anti-MMP-3 Ab (sc-21732), anti-MMP-13 Ab (sc-101564), anti-collagen II Ab (sc-52658), anti-aggrecan Ab (sc-70334), anti-IκB-α Ab (sc-1643), anti-phospho-IκB-α Ab (sc-8404), anti-p65 Ab (sc-71675), anti-phospho-p65 Ab (sc-136548), anti-NLRP3 Ab (sc-518123), anti-ASC Ab (sc-514559), and anti-caspase-1 Ab (sc-56036) (Santa Cruz Biotechnology, CA, USA) overnight at 4°C, followed by TBST wash. Subsequently, the blots were incubated with the HRP-coupled secondary Ab at room temperature (25°C) for 2 h, then washed three times for 5 min in TBST The immunoreactive proteins were visualized using an ECL system (Amersham, UK). ImageJ software (National Institutes of Health, Bethesda, MA, USA) was used to quantify the density of bands.
CCK-8 assay
The cell viability was determined using CCK-8 kit (Beyotime, Shanghai, China). Chondrocytes were seeded into 96-well plates with density of 1.0 × 104 cells/well and incubated for 24 h. A total of 10 μl CCK-8 solution was added after 10 ng/ml IL-1β treatment for 24 h. Then, 2 h later, the absorbance was assessed at 450 nm by a microplate reader (Molecular Devices, USA).
Measurement of NO, Il-6, TNF-α and PGE2
The Griess reaction was used to assess the NO concentration in the culture medium of chondrocytes. IL-6, TNF-α, and PGE2 ELISA kits (R&D Systems, Minneapolis, USA) were used to detect the levels of IL-6, TNF-α and PGE2 in the culture medium of chondrocytes following the manufacturer's instructions. The standard curve of recombinant protein provided by ELISA kit was used to calculate the concentration.
Apoptosis analysis
Chondrocytes (5 × 105 cells/well) were seeded in 6-well plates and stimulated with or without IL-1β for 24 h. Chondrocytes were washed twice by cold PBS (Thermo, MA, USA) and re-suspended in 200 μl buffer. After centrifugation, the supernatant was discarded, and the cells were incubated in buffer containing Annexin V-FITC and PI solution (BioVision, Milpitas, CA, USA) at room temperature in the dark for 1 h. At last, apoptotic cells were measured by a flow cytometer (BD Bioscience, NJ, USA).
Statistical analysis
All experiments were repeated more than three times. SPSS 22.0 software (SPSS, Inc., Chicago, IL, USA) was used for statistical analysis. The results were expressed as mean ± SD. Statistical significance was determined using Student's t test to compare two groups or one-way ANOVA followed by Tukey multiple comparison test for multiple comparision. The results coincided with the normal distribution. P < 0.05 was considered as statistically significant.
Results
DAPK1 was overexpressed in OA cartilage and IL-1β-stimulated chondrocytes
We first assessed whether DAPK1 plays a role in the pathogenesis of OA. The data of qRT-PCR and Western blotting showed that the mRNA and protein expressions of DAPK1 were higher in OA cartilage tissues compared with those in normal cartilage tissues (Figure 1A,B). IL-1β-stimulated chondrocytes play a key role in the progression of OA; thus, we detected the expression of DAPK1 in IL-1β-treated chondrocytes. The results indicated that the mRNA and protein expressions of DAPK1 in chondrocytes were increased by IL-1β stimulation in a dose-dependent manner (Figure 1C,D). These findings suggested that DAPK1 may play a role in the pathogenesis of OA.

DAPK1 was overexpressed in OA cartilage and IL-1β-stimulated chondrocytes. (A) qRT-PCR analysis of DAPK1 mRNA expression in cartilage tissues from normal non-OA patients (n = 15) and OA patients (n = 22). *P < 0.05 vs normal. (B) Representative images and quantitative analysis of DAPK1 protein expression in normal cartilage tissues (n = 3) and OA cartilage tissues (n = 3). GAPDH was used as an internal control. *P < 0.05 vs normal. (C-D) The relative mRNA and protein expressions of DAPK1 in chondrocytes were measured by qRT-PCR and Western blotting after stimulated with 0, 1, 2, 5, 10 ng/ml IL-1β for 24 h. GAPDH was used as an internal control. *P < 0.05 vs 0 ng/ml, **P < 0.01 vs 0 ng/ml.
Knockdown of DAPK1 repressed IL-1β-triggered apoptosis in chondrocytes
To elucidate the biological function of DAPK1 in IL-1β-injured chondrocytes, DAPK1 expression was suppressed by its small interfering RNAs (siRNAs). As shown in Figure 2A,B, DAPK1 siRNAs significantly reduced the mRNA and protein expressions of DAPK1 in chondrocytes. Subsequently, the results of CCK-8 assay revealed that the cell viability of chondrocytes was decreased by IL-1β and subsequently increased by DAPK1 siRNAs (Figure 2C). Meanwhile, IL-1β enhanced the apoptosis rate of chondrocytes, and DAPK1 siRNAs abolished this promotion (Figure 2D). In addition, IL-1β induced a decrease of anti-apoptotic protein, Bcl-2, expression and an increase of pro-apoptotic protein, Bax and cleaved-caspase3, expressions in chondrocytes, while DAPK1 siRNAs attenuated these effects (Figure 2E).
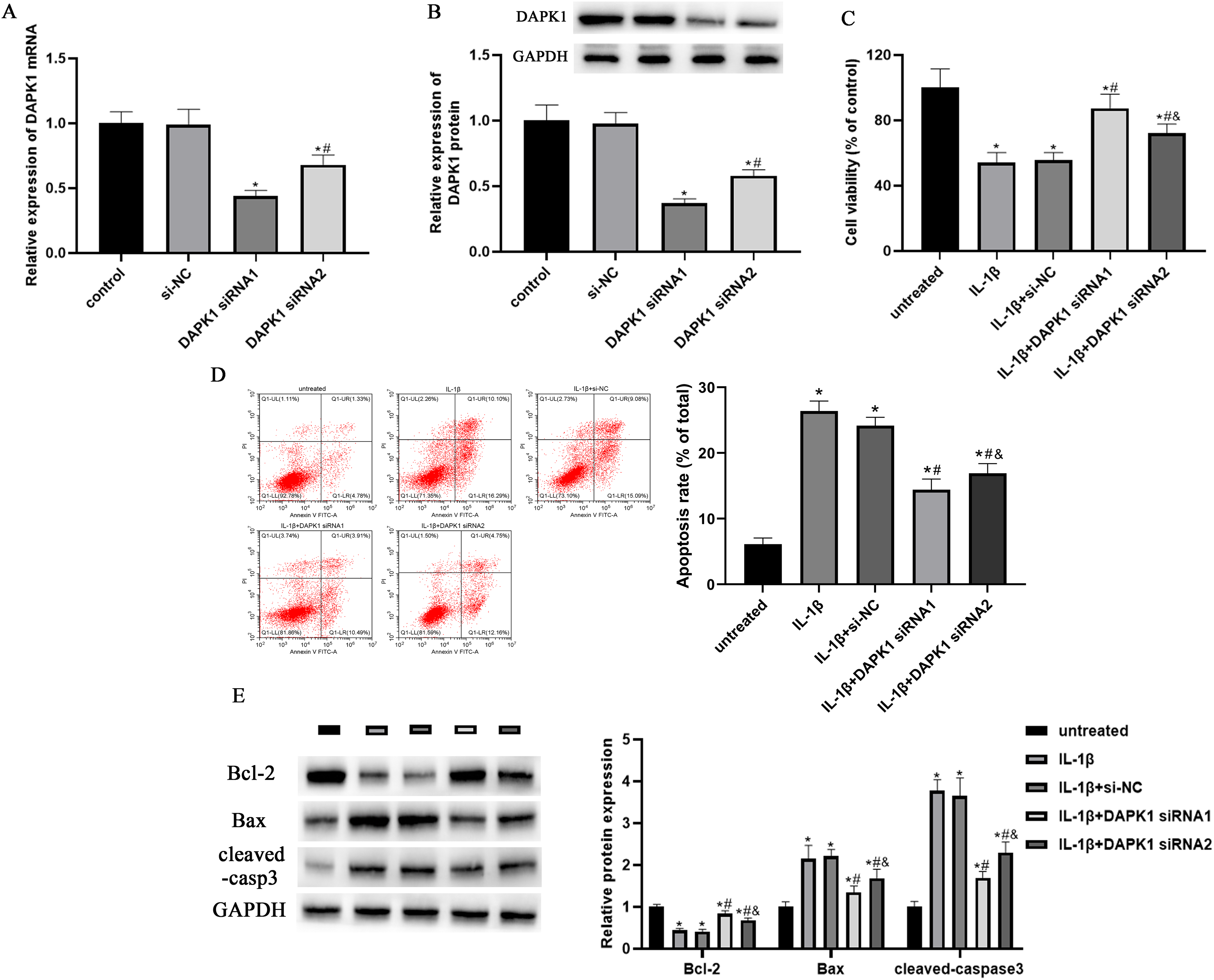
Knockdown of DAPK1 repressed IL-1β-triggered proliferation and apoptosis in chondrocytes. Chondrocytes were transfected with DAPK1 siRNAs (DAPK1 siRNA1 and 2) or siRNA control vector (si-NC) for 48 h. (A) The relative mRNA expression of DAPK1 in chondrocytes was measured by qRT-PCR. *P < 0.05 vs control, #P < 0.05 vs DAPK1 siRNA1. (B) The relative protein expression of DAPK1 was measured by Western blotting. GAPDH was used as an internal control. *P < 0.05 vs control, #P < 0.05 vs DAPK1 siRNA1. (C) After transfecting with DAPK1 siRNAs (DAPK1 siRNA1 and 2) for 48 h, chondrocytes were treated with 10 ng/ml IL-1β. CCK-8 assay was used to detect the viability of chondrocytes. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (D) Annexin V and PI double staining was used to measure the apoptotic rate of chondrocytes. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (E) Representative images and quantitative analysis of Bcl-2, Bax, and cleaved-caspase3 protein expressions in chondrocytes. GAPDH was used as an internal control. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group.
Knockdown of DAPK1 repressed the IL-1β-triggered inflammatory response and ECM degradation in chondrocytes
Matrix degradation and inflammatory damage caused by the combined action of inflammatory factors and MMPs are crucial in the occurrence and development of OA.11,27 Therefore, we explored the role of DAPK1 in the inflammatory response and ECM degradation. ELISA assay results indicated that the levels of pro-inflammatory cytokines, IL-6, TNF-α, PGE2 and NO, in chondrocytes were all elevated by treatment of IL-1β, whereas DAPK1 siRNAs inhibited these effects (Figure 3A-D). Moreover, Western blotting determined that IL-1β up-regulated the expressions of MMP-1, MMP-3 and MMP-13 in chondrocytes, which were suppressed by DAPK1 siRNAs (Figure 3E). Collagen II and aggrecan are the most important components of the ECM. Herein, DAPK1 siRNAs restored the decreased expressions of collagen II and aggrecan in chondrocytes after IL-1β stimulation (Figure 3E).

Knockdown of DAPK1 repressed IL-1β-triggered inflammatory response and ECM degradation in chondrocytes. DAPK1 siRNAs or si-NC was transfected into chondrocytes before IL-1β stimulation. (A-C) The levels of IL-6, TNF-α and PGE2 in chondrocytes were detected using ELISA assay. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (D) The NO concentration of chondrocytes was measured by Griess reaction. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (E) The relative protein expressions of MMP-1, MMP-3, MMP-13, collagen II and aggrecan in chondrocytes were quantified via normalizing to GAPDH. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group.
PEDF attenuated the effects of DAPK1 knockdown on apoptosis in IL-1β-stimulated chondrocytes
PEDF has been proven to contribute to the loss of the cartilage matrix in inflammatory arthritis in vivo.25,28 Our data showed that IL-1β induced an increase of PEDF expression in chondrocytes, while knockdown of DAPK1 blocked this increase (Figure 4A). Then, we investigated whether PEDF participates in the role of DAPK1 in IL-1β-injured chondrocytes. As shown in Figure 4B, the expression of PEDF in chondrocytes was up-regulated by IL-1β, and pcDNA-PEDF enhanced this promotion. After IL-1β stimulation, pcDNA-PEDF reversed the effects of DAPK1 siRNA on cell viability and apoptosis rate of chondrocytes (Figure 4C,D). Moreover, the expression of Bcl-2 was decreased, and the expressions of Bax and cleaved-caspase3 were increased by pcDNA-PEDF in DAPK1 siRNA-transfected cells (Figure 4E).

PEDF attenuated the effects of DAPK1 knockdown on proliferation and apoptosis in IL-1β-stimulated chondrocytes. (A) The relative protein expression of PEDF in IL-1β-stimulated chondrocytes was measured after transfected with DAPK1 siRNAs. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. GAPDH was used as an internal control. (B) After transfected with PEDF overexpression vector (pcDNA-PEDF) or its control vector (pcDNA-NC), the relative protein expression of PEDF in IL-1β-stimulated chondrocytes was detected by Western blotting. *P < 0.05 vs untreated group, #P < 0.05 vs IL-1β group. (C) IL-1β-stimulated chondrocytes were co-transfected with DAPK1 siRNA1 and pcDNA-PEDF. The viability of chondrocytes was detected. *P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (D) The apoptotic rate of chondrocytes was detected. **P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (E) Representative images and quantitative analysis of Bcl-2, Bax, and cleaved-caspase3 protein expressions in chondrocytes. GAPDH was used as an internal control. *P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group.
PEDF attenuated the effects of DAPK1 knockdown on the inflammatory response and ECM degradation in IL-1β-stimulated chondrocytes
With respect to ECM degradation, the results of ELISA demonstrated that pcDNA-PEDF attenuated the inhibitory effects of DAPK1 siRNA on the levels of IL-6, TNF-α, PGE2 and NO in IL-1β-stimulated chondrocytes (Figure 5A-C). Furthermore, the Western blotting analysis indicated that pcDNA-PEDF elevated the expressions of MMP-1, MMP-3 and MMP-13 and reduced the expressions of collagen II and aggrecan in chondrocytes after DAPK1 siRNA transfection (Figure 5E).

PEDF attenuated the effects of DAPK1 knockdown on inflammatory response and ECM degradation in IL-1β-stimulated chondrocytes. IL-1β-stimulated chondrocytes were co-transfected with DAPK1 siRNA1 and pcDNA-PEDF. (A-C) The levels of IL-6, TNF-α and PGE2 in chondrocytes were detected. *P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (D) The NO concentration of chondrocytes was measured. *P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (E) The relative protein expressions of MMP-1, MMP-3, MMP-13, collagen II and aggrecan in chondrocytes were measured. GAPDH was used as an internal control. *P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group.
Knockdown of DAPK1 blocked PEDF-mediated NF-kB pathway and NLRP3 inflammasome activation in chondrocytes
The NF-κB pathway and NLRP3 inflammasome play key roles in the occurrence and development of OA.29–31 It has been proven that PEDF plays a regulatory role in the NF-κB pathway and NLRP3 inflammasome activation. 20 Thus, we detected the activity of the NF-κB pathway and NLRP3 inflammasome in chondrocytes after DAPK1 knockdown. As expected, the phosphorylation of IκB-α and p65, as well as the expressions of NLRP3, ASC and caspase-1 were suppressed by DAPK1 siRNA in IL-1β-induced chondrocytes (Figure 6A,B). Moreover, our data indicated that PEDF partly reversed the effects of DAPK1 siRNA on the NF-κB pathway and NLRP3 inflammasome, suggesting PEDF plays a role in the regulation of DAPK1 in the NF-κB pathway and NLRP3 inflammasome.

Knockdown of DAPK1 blocked PEDF-mediated NF-kB pathway and NLRP3 inflammasome activation in chondrocytes. IL-1β-stimulated chondrocytes were co-transfected with DAPK1 siRNA1 and pcDNA-PEDF. (A) The protein expressions analysis of IκB-α and p65, phosphorylation of IκB-α and p65 in IL-1β-induced chondrocytes. GAPDH was used as an internal control. *P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group. (B) The protein expressions analysis of NLRP3, ASC and caspase-1 in IL-1β-induced chondrocytes. GAPDH was used as an internal control. *P < 0.05 vs IL-1β group, #P < 0.05 vs IL-1β + pcDNA-PEDF group, &P < 0.05 vs IL-1β + DAPK1 siRNA1 group.
Discussion
OA is a chronic joint disease, the main clinical symptom of which is irreversible degeneration of articular cartilage. 32 Current research shows that IL-1β plays a key role in the degradation of OA cartilage matrix. 8 During the pathogenesis of OA, IL-1β induces the overexpression of MMPs in chondrocytes, which leads to disorders of chondrocyte anabolism and catabolism, thereby inhibiting the formation of cartilage matrix. 9 In this experiment, IL-1β was used to induce human chondrocytes in vitro to further study the effect of DAPK1 on OA articular cartilage. Our results showed that DAPK1 expression was not only up-regulated in the cartilage tissue of patients with OA, but also increased in human chondrocytes induced by IL-1β in vitro.
DAPK1 is one of the positive regulators of apoptosis and is widely involved in apoptosis induced by multiple pathways.12,26 Increasing studies have proved that DAPK1 promoted apoptosis in a variety of tumour cells.14,15,26 In this study, we found that knockdown of DAPK1 attenuated IL-1β-induced apoptosis of chondrocytes. Besides, recent studies found that DAPK1 played a dual role in a range of inflammatory regulation.33,34 It has been reported that activating DAPK1 expression improved inflammatory responses in LPS-induced acute respiratory distress syndrome and acute lung injury.14,35 On the contrary, inhibition of DAPK1 expression can reduce the secretion of IL-8 induced by IL-1β in macrophages. DAPK1 deletion inhibits the synthesis of IL-1β and affects the synthesis of NLRP3 inflammasomes.36,37 The results of this study found that knockdown of DAPK1 attenuated the inflammatory response and cartilage degradation of chondrocytes induced by IL-1β.
Previous studies have found that PEDF is significantly up-regulated in advanced OA. 38 It was confirmed that the expression of PEDF in OA chondrocytes was higher than that in normal chondrocytes. 39 The increased expression of PEDF aggravated the loss of cartilage matrix. 28 These studies showed that PEDF played an important regulatory role in OA. In addition, PEDF expression was proved to be positively modulated by DAPK1 in pancreas carcinoma cells. 26 Our data indicated that knockdown of DAPK1 reduced PEDF expression to down-regulate inflammatory response and cartilage degradation in IL-1β-induced chondrocytes.
The NF-κB pathway plays a vital role in the occurrence and development of OA disease.5,32 The activated NF-κB signalling pathway can enhance the expression of the inflammatory cytokine IL-1β, promote the production of MMPs, and ultimately lead to cartilage degradation and joint damage, thereby accelerating the process of OA.6,7,10,40 NLRP3 inflammasome is a protein complex, mainly synthesized by apoptosis-associated speck-like protein containing a CARD (ASC) and caspase-1.30,41 The main function of the NLRP3 inflammasome is to recognize external stimulus signals. The activated NLRP3 inflammasome promoted the activation of caspase-1, further triggered pro-inflammatory mediators (such as IL-1β) to induce inflammatory response.41,42 Recent research reported that NLRP3 inflammasome was activated in the occurrence and development of OA, which is one of the etiologies of OA.29,30,43 It is reported that DAPK1 played an important role in the activation of NLRP3 inflammasome.33,37 Furthermore, studies have found that PEDF activated the NF-κB pathway in skeletal muscle cells and macrophages,20,22 and the NLRP3 inflammasome in myocardial cells and missed abortion tissues.44,45 In this study, our findings demonstrated that knockdown of DAPK1 inhibited IL-1β-activated NF-κB pathway and NLRP3 inflammasome in chondrocytes through PEDF. Currently, inhibiting the activation of NF-κB pathway and NLRP3 inflammasome in chondrocytes is a new treatment strategy to improve OA. 30
However, our study has potential limitations. In our study, we preliminarily explored the role and regulatory mechanism of DAPK1 and PEDF in IL-1β-induced chondrocytes in vitro. Furtherfore, in vivo experiments are needed to further verify the roles of DAPK1 and PEDF in osteoarthritis, thus providing evidence for the clinical treatment of osteoarthritis.
In conclusion, our results revealed that DAPK1 was overexpressed in OA tissues and IL-1β-induced chondrocytes. Decreasing DAPK1 expression attenuated IL-1β-induced apoptosis, inflammation, cartilage degradation, as well as inhibited the activation of NF-κB pathway and NLRP3 inflammasome via regulating PEDF expression (Figure 7). Therefore, DAPK1 might serve as a potential therapeutic target for OA.

Knockdown of DAPK1 inhibits IL-1β-induced inflammation and cartilage degradation in human chondrocytes by modulating the PEDF-mediated NF-κB and NLRP3 inflammasome pathways.
Footnotes
Availability of data and materials
The analysed data sets generated during the present study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Project of Yantai Science and Technology Innovation and Development (2020MSGY083).
