Abstract
Circular RNAs (circRNAs) are involved in the regulation of various diseases, including periodontitis. The objective of this study was to analyze the biological role and regulatory mechanism of circ_0066881 in LPS-induced periodontal ligament cells (PDLCs). Circ_0066881, microRNA-144-5p (miR-144-5p) and retinoid acid-related orphan receptor A (RORA) levels were determined using reverse transcription-quantitative PCR (RT-qPCR) assay. Cell viability detection was performed by Cell Counting Kit-8 assay. Cell apoptosis was assessed through flow cytometry and caspase-3 activity assay. The protein analysis was completed via Western blot. Inflammatory cytokines were measured by ELISA. The target interaction was validated by dual-luciferase reporter assay and RNA immunoprecipitation (RIP) assay. The level of circ_0066881 was down-regulated in periodontitis tissues. Overexpression of circ_0066881 relieved LPS-induced cell viability inhibition and apoptosis or inflammation promotion in PDLCs. Circ_0066881 could bind to miR-144-5p. The protective function of circ_0066881 was achieved by sponging miR-144-5p in PDLCs. Circ_0066881 acts as a miR-144-5p sponge to mediate the RORA level. Inhibition of miR-144-5p attenuated LPS-induced cell injury via targeting RORA. All these results demonstrated that circ_0066881 partly prevented LPS-evoked cell dysfunction in PDLCs through miR-144-5p-mediated up-regulation of RORA.
Introduction
Periodontitis is a chronic inflammatory disease evoking the irreversible damages in the whole periodontium. 1 Periodontitis is highly prevalent around the world and it has been considered as a risk factor of cardiovascular diseases. 2 Periodontal ligament (PDL) tissues are important to maintain periodontal homeostasis and development. 3 LPS can promote the release of inflammatory cytokines in periodontal ligament cells (PDLCs). 4 Exploring the underlying mechanism of LPS-induced injury of PDLCs is essential for the better understanding of periodontitis progression.
Circular RNAs (circRNAs) are known as covalently closed-loop conformation with loss of 3’ and 5’ ends. 5 A variety of circRNAs play key roles in different human diseases.6,7 Recently, circRNAs have been found to be implicated in the pathogenesis of periodontitis and the differentiation of PDLCs. 8 CircRNA expression profile showed that many circRNAs were dysregulated in periodontitis tissues, and circ_0066881 was down-regulated with significant difference. 9 However, the involvement of circ_0066881 in the progression of periodontitis is not clear.
CircRNAs can interact with the binding sites of microRNAs (miRNAs) to act as miRNA sponges, thereby affecting the levels of downstream genes. 10 Li et al. asserted that microRNA-144-5p (miR-144-5p) was highly expressed in chronic periodontitis patients. 11 Retinoid acid-related orphan receptor A (RORA) is a key inflammatory regulator, and it can attenuate LPS-induced inflammation and organ injury. 12 Also, RORA was shown to protect against LPS-induced production of pro-inflammatory cytokines periodontitis. 13 Moreover, circ_0081572 suppressed LPS-mediated cell apoptosis and inflammation in periodontitis via sponging miR-378h to up-regulate RORA. 14 It remains unknown whether circ_0066881 can regulate RORA level through targeting miR-144-5p.
In this study, we focused on the function of circ_0066881 in periodontitis and the regulatory mechanism with miR-144-5p/RORA axis. The roles of circ_0066881 in regulating the biological processes of LPS-treated PDLCs were investigated. Circ_0066881 was hypothesized as a miR-144-5p inhibitor to regulate RORA expression. The circ_0066881-based molecular pathogenesis in LPS-induced periodontitis was disclosed.
Materials and methods
Tissue collection
PDL tissues were obtained from the gingival sulcus of periodontitis patients (n = 31) at Pingxiang People’s Hospital, and normal tissues were collected from healthy individuals (n = 31). All samples were instantly conserved in liquid nitrogen. These subjects have signed the written informed consent files, and the present study was authorized by the Ethics Committee of Pingxiang People’s Hospital.
Cell culture and treatment
The primary PDLCs were isolated from periodontal ligament tissues of healthy individuals with orthodontic treatment, as previously described. 14 PDLCs were maintained in DMEM (Gibco, Carlsbad, CA, USA) with supplement of 10% FBS and 1% antibiotics (Gibco). Cells were cultured in 5% CO2, 37°C incubator. To induce the periodontitis-like injury, PDLCs were disposed with 1 μg/ml LPS (Sigma, St Louis, MO, USA) for 24 h.
Cell transfection
Transient transfection was carried out via Lipofectamine™ 3000 reagent as per the instruction book of Invitrogen (Carlsbad, CA, USA). Circ_0066881 sequence was constructed into the pcD5-ciR vector (GENESEED, Guangzhou, China), generating pcD5-ciR-circ_0066881 vector (circ_0066881). The miRNA mimics (miR-144-5p, miR-NC), inhibitors (in-miR-144-5p, in-miR-NC), small interfering RNAs (si-RORA, si-NC) were synthesized by RIBOBIO (Guangzhou, China).
Reverse transcription-quantitative PCR (RT-qPCR) assay
Total RNA was extracted from tissues and cells using RNAiso Plus Reagent (Takara, Shiga, Japan). Reverse transcription was performed by PrimeScript™ RT Master Mix (Takara) and PCR detection was prepared by TB Green® Premix Ex Taq™ II reagent (Takara), according to the users’ manuals. The collected data were analyzed using the 2−ΔΔCt method. 15 GAPDH and U6 acted as reference genes for level normalization. The stabilities of circ_0066881 and linear CD80 were detected by RT-qPCR after total RNA was digested with RNase R (GENESEED) for 1 h or cells were incubated with actinomycin D (Sigma) for increasing times (0 h, 4 h, 8 h, 16 h, 24 h). PARIS™ Kit (Invitrogen) was used for RNA isolation from nucleus and cytoplasm, followed by level quantification of circ_0066881, GAPDH and U6. The specific primers were exhibited in Table 1.
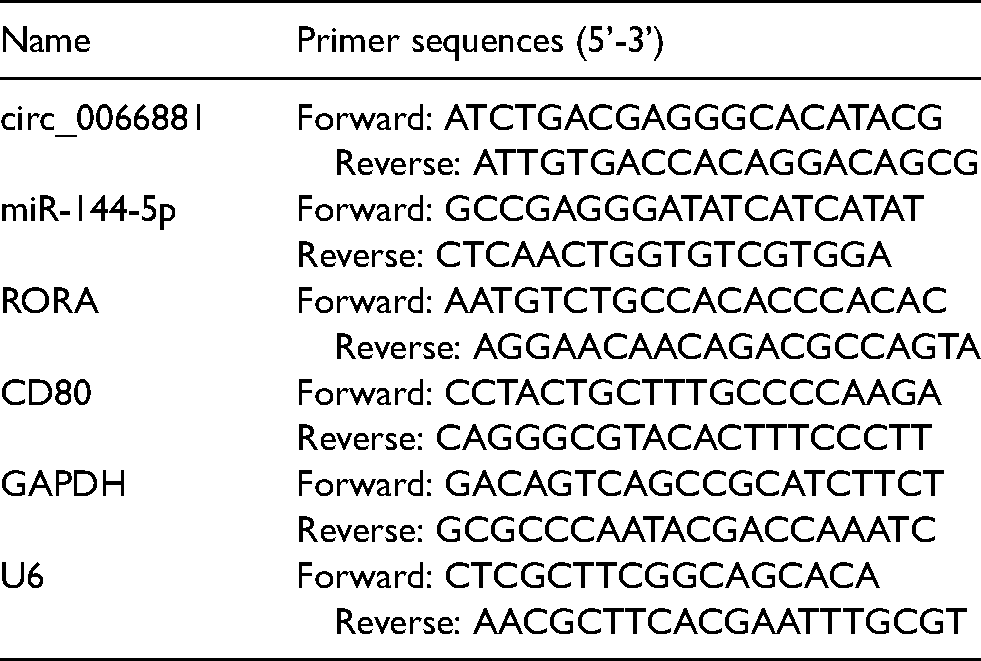
Primer sequences used for RT-qPCR.
Cell counting Kit-8 (CCK-8) assay
The 48-well plates were seeded with 1 × 105 PDLCs, then LPS treatment and cell transfection were performed for 24 h. Subsequently, the plates were added with 10 μl/well CCK-8 solution (Sangon, Shanghai, China) and incubated for 2 h. After the absorbance was detected at 450 nm under the microplate reader, cell viability (%) was calculated by the ratio of viable cells.
Flow cytometry
Annexin V-fluorescein isothiocyanate (Annexin V-FITC) Kit (Sangon) was used for assessment of apoptosis. PDLCs were collected into a new tube, then 4 × 105 cells were re-suspended in 300 µl Binding Buffer. Cells were stained with 10 µl Annexin V-FITC and 10 µl propidium iodide (PI) for 20 min in the dark. The apoptotic cells were then determined via a flow cytometer (BD Biosciences, San Diego, CA, USA).
Caspase-3 activity assay
Cell apoptosis was further evaluated through determining the activity of caspase-3. PDLCs were washed with PBS (Invitrogen), then 1 × 106 cells were collected, and the caspase-3 activity was measured following the operating procedures of EnzChek® Caspase-3 Assay Kit (Invitrogen).
Western blot
Radioimmunoprecipitation assay lysis buffer (Sangon) was applied for total protein extraction, followed by Western blot detection referring to the issued description. 16 The primary Abs targeting B-cell lymphoma-2 (Bcl-2; ab32124, 1:1000), Bcl-2-associated X (Bax; ab182733, 1:1000), RORA (ab256799, 1:1000), GAPDH (ab181603, 1:1000) were provided by Abcam (Cambridge, UK). Goat Anti-rabbit IgG H&L (HRP) secondary Ab (Abcam, ab205718, 1:5000) was incubated at 25°C for 1 h, followed by analysis of protein bands through ECL luminescence reagent (Sangon) and Image J software (NIH, Bethesda, MD, USA).
ELISA
Cell supernatants were harvested for the determination of inflammatory cytokines. The concentrations (pg/ml) of IL-1β, IL-6, IL-8 and TNF-α were measured using IL-1β Human Uncoated ELISA Kit, IL-6 Human ELISA Kit of High Sensitivity, (Invitrogen), IL-8 Human ELISA Kit and TNF-α Human ELISA Kit. All kits were commercially purchased from Invitrogen.
Dual-luciferase reporter assay
The wild type (WT, with miR-144-5p binding sites) and mutant-type (MUT, with mutated miR-144-5p binding sites) circ_0066881 and RORA 3’UTR sequences were cloned into the pmirGLO plasmid (Promega, Madison, WI, USA). Novel reporters (circ_0066881 WT, circ_0066881 MUT, RORA 3’UTR WT and RORA 3’UTR MUT) were co-transfected with miR-144-5p or miR-NC for 48 h, then PDLCs were collected for luciferase activity analysis through the Dual-Luciferase Reporter Detection Kit (Promega).
RNA immunoprecipitation (RIP) assay
The circ_0066881 and miR-144-5p binding was analyzed via Imprint® RNA Immunoprecipitation Kit (Sigma). PDLCs were lysed with RIP lysis Buffer, then 2 × 106 cells were incubated with magnetic beads of anti-Argonaute-2 (anti-Ago2) or anti-immunoglobulin G (anti-IgG) group at 4°C overnight. The unincubated group was defined the Input group. After RNA isolation, circ_0066881 and miR-144-5p levels were examined using RT-qPCR.
Statistical analysis
After three independent experiments, data were displayed as the mean ± standard deviation. Data were analyzed through SPSS 22.0 (SPSS Inc., Chicago, IL, USA), followed by difference analysis via Student’s t-test and analysis of variance (ANOVA) followed by Tukey’s test The relations between targets were determined by Pearson’s correlation coefficient. There was a significant difference when P < 0.05.
Results
Circ_0066881 was down-regulated in periodontitis tissues
Circ_0066881 is produced from exon 3 of CD80 gene with length of 318 bp (Figure 1A). Compared to normal controls, circ_0066881 level was significantly reduced in periodontitis tissues (Figure 1B). RNase R (Figure 1C) and actinomycin D (Figure 1D) decreased the expression of linear CD80 but did not influence circ_0066881 level in PDLCs, indicating that circ_0066881 was highly stable relative to linear RNAs. Additionally, abundant circ_0066881 and GAPDH were detected in the cytoplasm while U6 was detected in the nucleus (Figure 1E). Circ_0066881 was mainly localized in the cytoplasm of PDLCs.
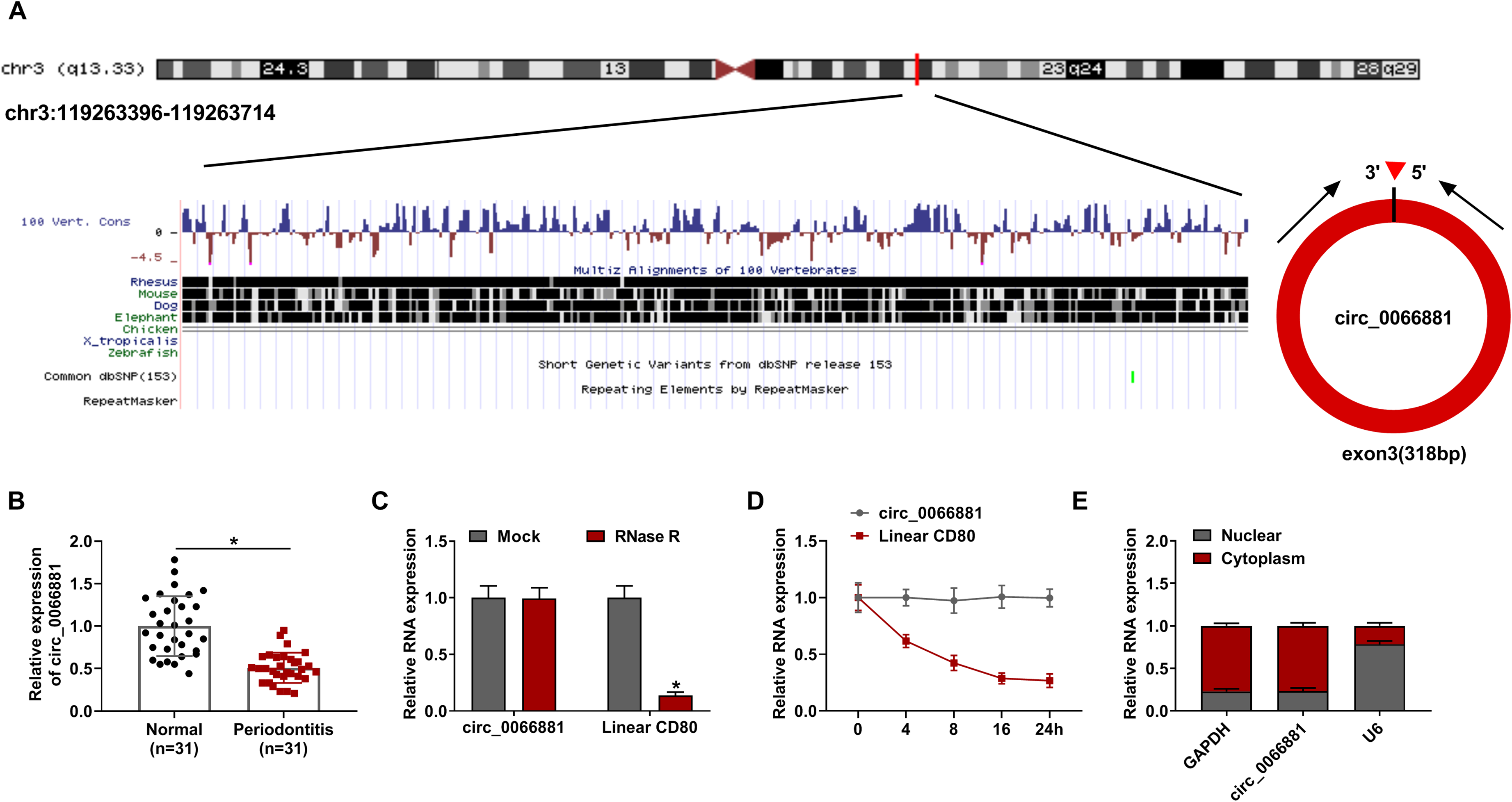
Circ_0066881 was down-regulated in periodontitis tissues. (A) The genetic information of circ_0066881. (B) The circ_0066881 level was determined using RT-qPCR in periodontitis samples and control samples. (C-D) The circ_0066881 stability was assessed after treatment of RNase R (C) and actinomycin D (D). (E) Circ_0066881, U6 and GAPDH expression detection was performed via RT-qPCR in nucleus and cytoplasm. *P < 0.05.
Circ_0066881 mitigated LPS-induced cell apoptosis and inflammatory response in PDLCs
LPS treatment has inhibited circ_0066881 expression in PDLCs, contrasted with the control group (Figure 2A). Transfection of circ_0066881 could elevate the level of circ_0066881 by about 6-fold changes, compared with vector group (Figure 2B). Moreover, circ_0066881 level was higher in LPS + circ_0066881 group than that in LPS + vector group of PDLCs (Figure 2C). CCK-8 assay revealed that cell viability was repressed by LPS, which was recovered after circ_0066881 transfection (Figure 2D). The data from flow cytometry (Figure 2E) and caspase-3 activity assay (Figure 2F) demonstrated that LPS-induced cell apoptosis was alleviated by overexpression of circ_0066881. The protein detection showed that circ_0066881 down-regulated the expression of pro-apoptotic Bax and up-regulated the level of anti-apoptotic Bcl-2 in LPS-treated PDLCs (Figure 2G). The concentrations of IL-1β, IL-6, IL-8 and TNF-α were enhanced after treatment with LPS, then circ_0066881 up-regulation reversed these effects (Figure 2H-K). Thereby, cell dysfunction caused by LPS was lightened by circ_0066881 in PDLCs.
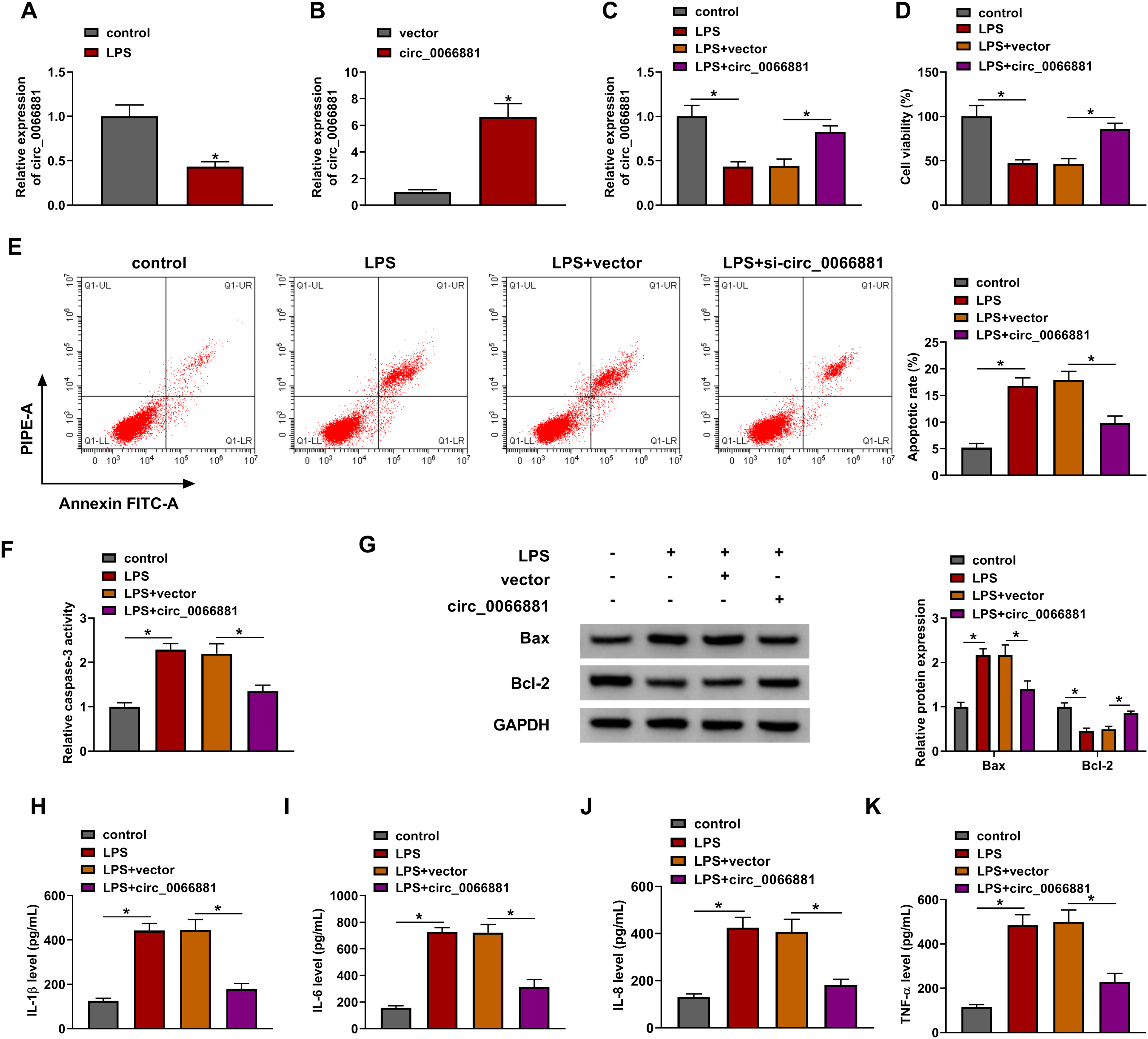
Circ_0066881 mitigated LPS-induced cell apoptosis and inflammatory response in PDLCs. (A-B) Circ_0066881 level analysis was conducted by RT-qPCR in LPS-treated PDLCs (A) and PDLCs with transfection of vector or circ_0066881. (C-K) PDLCs were treated with control, LPS, LPS + vector or LPS + circ_0066881. (C) The circ_0066881 detection was performed via RT-qPCR. (D) Cell viability examination was performed by CCK-8 assay. (E-G) Cell apoptosis assessment was performed by flow cytometry (E), caspase-3 activity assay (F) and Western blot for protein analysis (G). (H-K) IL-1β, IL-6, IL-8 and TNF-α concentrations were measured via ELISA. *P < 0.05.
Circ_0066881 interacted with miR-144-5p
The online starbase (http://starbase.sysu.edu.cn) predicted that circ_0066881 sequence contained the miR-144-5p binding sites (Figure 3A). The miR-144-5p expression was obviously increased after miR-144-5p transfection in PDLCs (Figure 3B). The luciferase activity of circ_0066881 WT group was inhibited by up-regulation of miR-144-5p, but that of circ_0066881 MUT group was not affected in PDLCs (Figure 3C). The results of RIP assay manifested that circ_0066881 and miR-144-5p were enriched in Anti-Ago2 group relative to Anti-IgG group (Figure 3D). There was an inhibitory effect of circ_0066881 on the miR-144-5p level in PDLCs (Figure 3E). RT-qPCR showed that miR-144-5p was highly expressed in periodontitis samples contrasted with the normal samples (Figure 3F). By performing the analysis of Pearson’s correlation coefficient, we found that circ_0066881 expression was negatively associated with miR-144-5p level (r = −0.5847, P = 0.0006) (Figure 3G). Circ_0066881 directly targeted miR-144-5p.
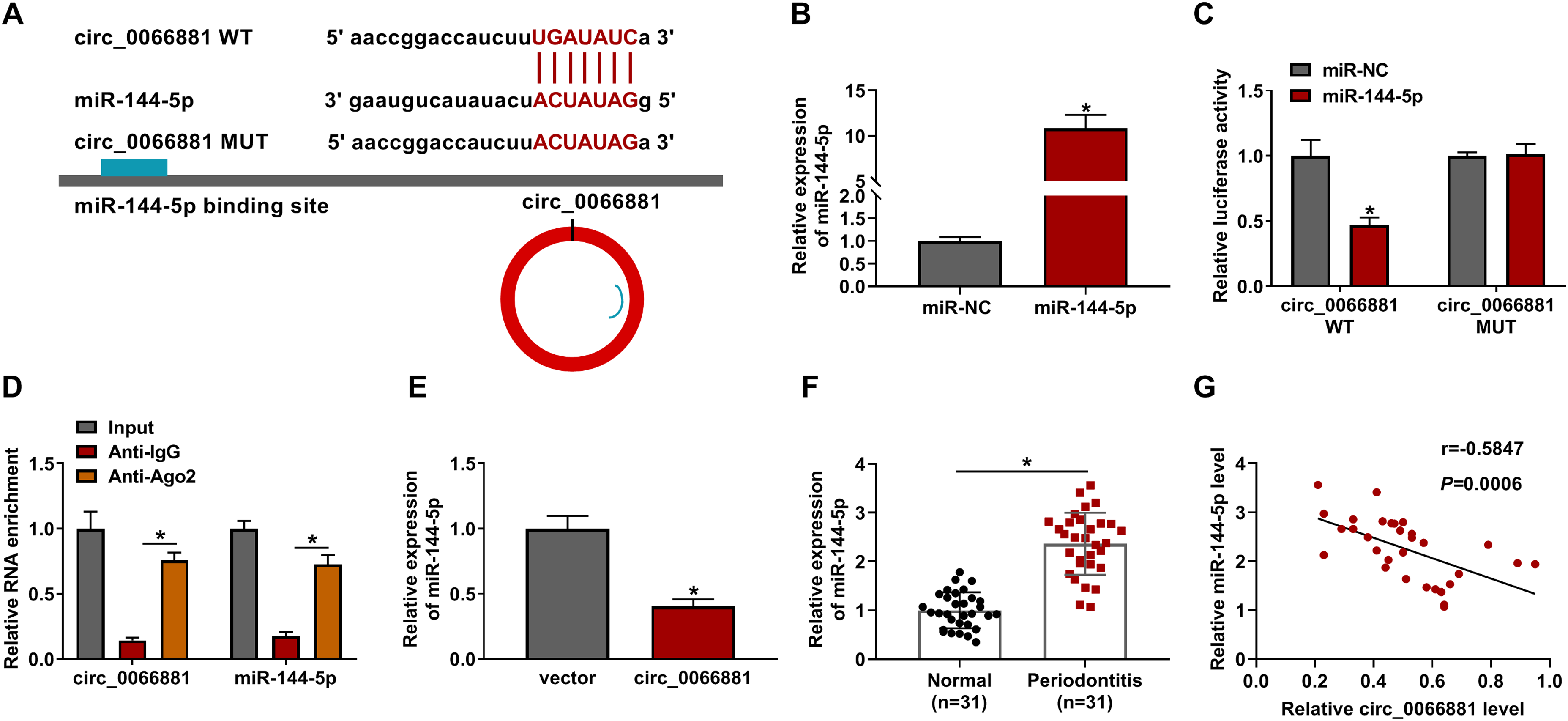
Circ_0066881 interacted with miR-144-5p. (A) Starbase exhibited the binding site of miR-144-5p in the circ_0066881 sequence. (B) The miR-144-5p level was examined after transfection of miR-NC or miR-144-5p. (C-D) The binding analysis between circ_0066881 and miR-144-5p was performed using dual-luciferase reporter assay (C) and RIP assay (D). (E) The effect of circ_0066881 on the miR-144-5p level was evaluated by RT-qPCR. (F) Circ_0066881 was quantified by RT-qPCR in periodontitis and normal samples. (G) The relationship between circ_0066881 and miR-144-5p was analyzed through Pearson’s correlation coefficient. *P < 0.05.
Circ_0066881/miR-144-5p axis regulated LPS-induced cell injury in PDLC
LPS induced the evident up-regulation of miR-144-5p compared with control group in PDLCs (Figure 4A). In addition, circ_0066881-mediated expression inhibition of miR-144-5p was restored by miR-144-5p transfection (Figure 4B). Cell viability promotion by circ_0066881 was attenuated following miR-144-5p overexpression in LPS-treated PDLCs (Figure 4C). The collective data from apoptosis rate (Figure 4D), caspase-3 activity detection (Figure 4E-F) and protein analysis (Figure 4G) affirmed that miR-144-5p mimic eliminated circ_0066881-induced repression of cell apoptosis. With the level elevation of miR-144-5p, the function of circ_0066881 in preventing the release of inflammatory cytokines was abolished in LPS-treated PDLCs (Figure 4H-K). Thus, circ_0066881 acted by targeting miR-144-5p in LPS-induced PDLCs.
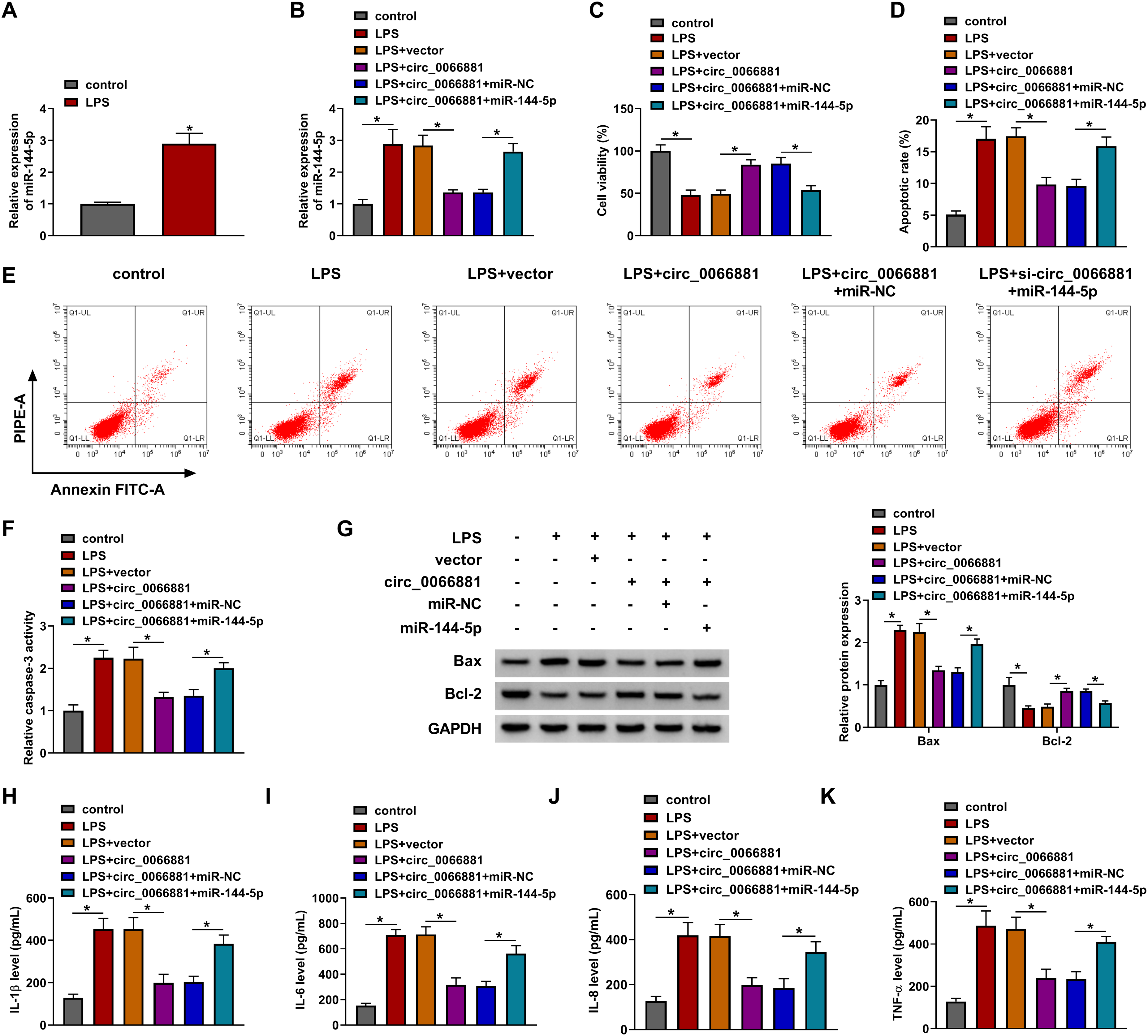
Circ_0066881/miR-144-5p axis regulated LPS-induced cell injury in PDLC. (A) The miR-144-5p level was measured by RT-qPCR in LPS-treated PDLCs. (B-K) PDLCs were treated with control, LPS, LPS + vector, LPS + circ_0066881, LPS + circ_0066881 + miR-NC, LPS + circ_0066881 + miR-144-5p. (B) RT-qPCR was used to detect the miR-144-5p expression. (C) CCK-8 assay was used to assess cell viability. (D-G) Flow cytometry (D-E), caspase-3 activity assay (F) and Western blot (G) were used to determine cell apoptosis. (H-K) ELISA was used to assess inflammatory response. *P < 0.05.
Circ_0066881 resulted in the up-regulation of RORA by sponging miR-144-5p
Targetscan (http://www.targetscan.org) showed the site binding between miR-144-5p and RORA 3’UTR sequences (Figure 5A). Dual-luciferase reporter assay validated that miR-144-5p could bind to the 3’UTR of RORA (Figure 5B). The mRNA and protein levels were down-regulated in periodontitis tissues relative to normal controls (Figure 5C-D). Moreover, circ_0066881 up-regulated RORA protein expression and miR-144-5p abrogated this regulation (Figure 5E). There was a negative relation (r = −0.5671, P = 0.0009) between miR-144-5p and RORA mRNA levels but a positive relationship (r = 0.5718, P = 0.0008) between circ_0066881 and RORA levels (Figure 5F-G). The miR-144-5p level was inhibited by in-miR-144-5p compared with in-miR-NC transfection in PDLCs (Figure 5H), and si-RORA-mediated knockdown of RORA was great (Figure 5I). Thus, circ_0066881 regulated RORA level via targeting miR-144-5p.
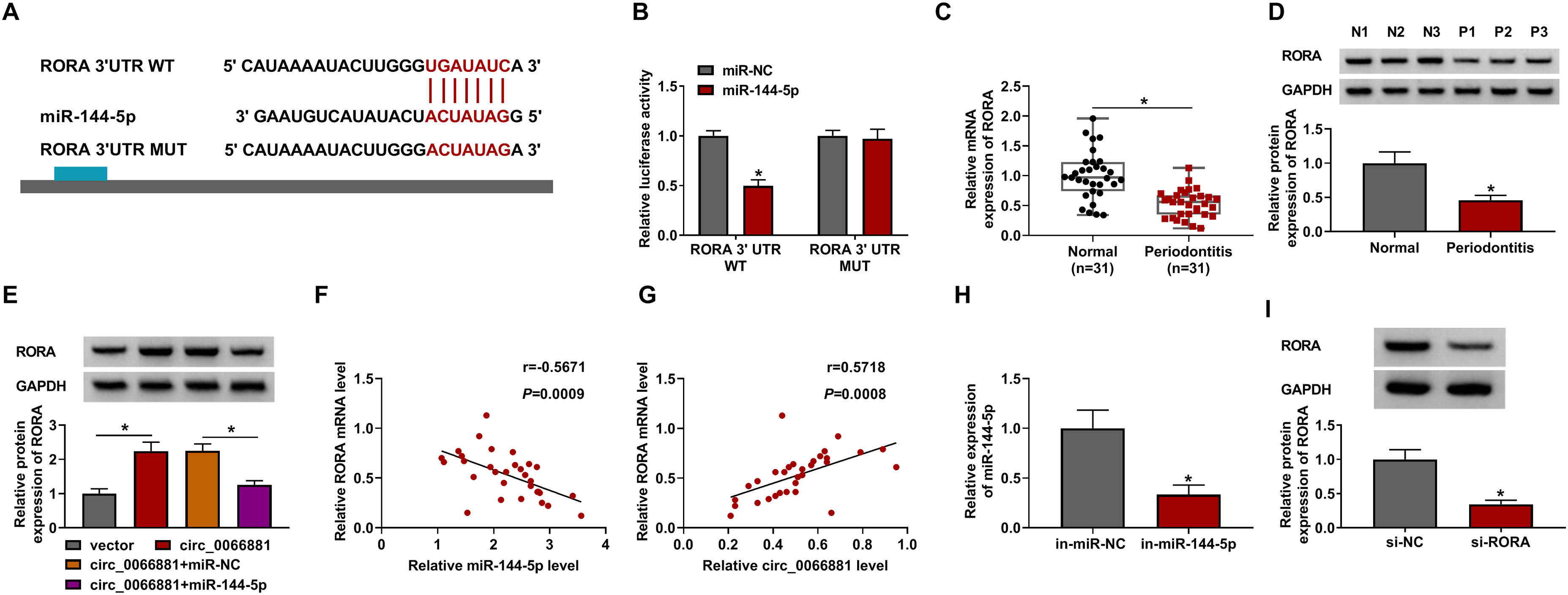
Circ_0066881 resulted in the up-regulation of RORA by sponging miR-144-5p. (A) Targetscan indicated the binding site between RORA 3’UTR and miR-144-5p. (B) Dual-luciferase reporter assay was applied to validate the interaction between RORA 3’UTR and miR-144-5p. (C-D) RORA mRNA and protein levels were detected by RT-qPCR and Western blot in periodontitis and normal tissues. (E) RORA protein expression was measured by Western blot in vector, circ_0066881, circ_0066881 + miR-NC and circ_0066881 + miR-144-5p groups. (F-G) Pearson’s correlation coefficient was used for analyzing the relation between RORA and miR-144-5p (F) or circ_0066881 (G). (H-I) Transfection efficiencies of in-miR-144-5p (H) and si-RORA (I) were assessed using RT-qPCR and Western blot in PDLCs. *P < 0.05.
Down-regulation of miR-144-5p inhibited LPS-induced cell dysfunction in PDLCs via increasing the RORA expression
The protein expression of RORA was lower in LPS-treated PDLCs than that in control cells (Figure 6A). Meanwhile, si-RORA has counteracted the promoting effect of in-miR-144-5p on RORA protein level in LPS-treated PDLCs (Figure 6B). LPS-caused cell viability reduction (Figure 6C) and apoptosis acceleration (Figure 6D-G) were reverted by miR-144-5p inhibitor, whereas these influences were offset by siRNA of RORA. ELISA assay indicated that RORA knockdown promoted the release of IL-1β, IL-6, IL-8 and TNF-α relative to alone in-miR-144-5p transfection in LPS-treated PDLCs (Figure 6H-K). All in all, miR-144-5p inhibition up-regulated the level of RORA to protect against the LPS-induced cell damages.
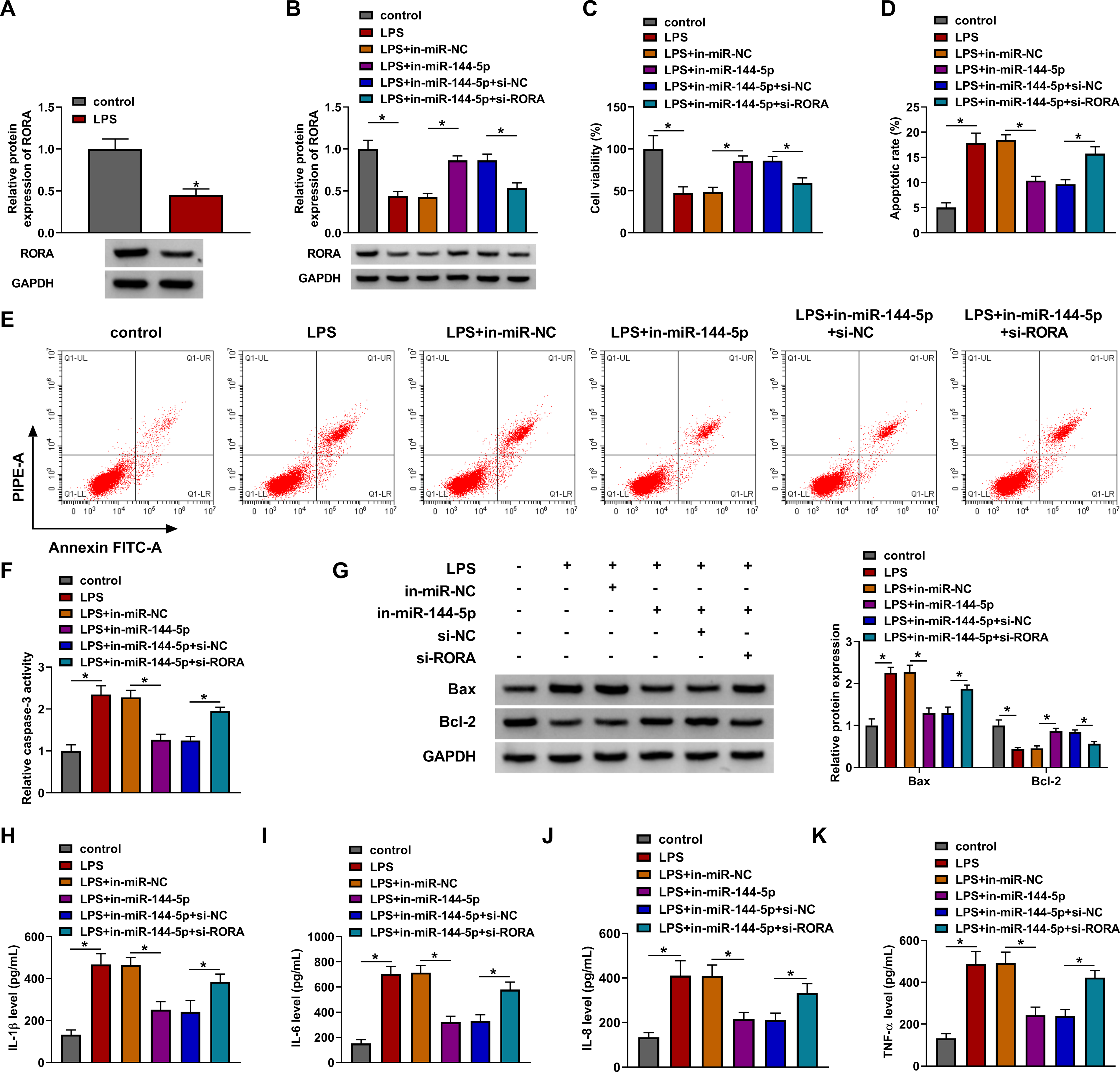
Down-regulation of miR-144-5p inhibited LPS-induced cell dysfunction in PDLCs via increasing the RORA expression. (A) The protein level of RORA was examined using Western blot in LPS-treated PDLCs. (B-K) PDLCs were treated with control, LPS, LPS + in-miR-NC, LPS + in-miR-144-5p, LPS + in-miR-144-5p + si-NC or LPS + in-miR-144-5p + si-RORA. (B) RORA protein quantification was performed through Western blot. (C) Cell viability was determined through CCK-8 assay. (D-G) Cell apoptosis was evaluated through flow cytometry (D-E), caspase-3 activity assay (F) and Western blot (G). (H-K) The inflammatory cytokine release was analyzed through ELISA. *P < 0.05.
Discussion
Through a series of experiments, this study unraveled that circ_0066881 attenuated LPS-mediated cell damages in PDLCs via inhibiting miR-144-5p and up-regulating RORA. The function and mechanism of circ_0066881 were firstly disclosed in periodontitis.
The differentially expressed circRNAs have pivotal roles in the progression of periodontitis. 9 For instance, overexpression of circRNA CDR1as relieved LPS-induced cell proliferation inhibition in PDLCs. 17 Zheng et al. stated that circCDK8 up-regulation induced apoptosis and differentiation of PDLCs. 18 Also, circ_0081572 or circ_0085289 suppressed the development of periodontitis through regulating miR-378h/RORA or miR-let-7f-5p/SOCS6 axis.14,19 Herein, circ_0066881 was identified as a down-regulated circRNA in periodontitis. Furthermore, we found that cell viability inhibition and cell apoptosis acceleration caused by LPS were alleviated with expression elevation of circ_0066881. Moreover, circ_0066881 partly prevented the release of pro-inflammatory cytokines in LPS-stimulated PDLCs. Altogether, circ_0066881 could protect PDLCs from LPS-induced cell dysfunction in periodontitis.
CircRNAs act as essential regulators in various inflammatory diseases by depending on miRNA sponging function. Ye et al. reported that circ_103516 enhanced the inflammatory response in inflammatory bowel disease through interacting with miR-19b-1-5p. 20 Circ_0000950 exacerbated neuron apoptosis and inflammation in Alzheimer’s disease via directly absorbing miR-103. 21 CircRNA_103765 was validated as a pro-inflammatory factor in Crohn’s disease by sponging miR-30 family. 22 In this study, we discovered the target interaction between miR-144-5p and circ_0066881. In addition, circ_0066881 directly suppressed miR-144-5p level and the alleviate regulation of circ_0066881 in LPS-induced cell injury was attributed to inhibit the expression of miR-144-5p.
Small miRNAs can regulate the progression of periodontitis via affecting the downstream targets. MiR-218 provided the protective effect for periodontitis rats against osteoclast differentiation and inflammation reaction by down-regulating MMP9 level. 23 MiR-214 facilitated necroptosis through targeting ATF4 in PDLCs treated with high glucose and LPS. 24 Our results showed that LPS-induced apoptotic and inflammatory damages were reversed by miR-144-5p inhibitor via increasing RORA level, implying that miR-144-5p aggravated the progression of periodontitis in vitro via targeting RORA. More importantly, circ_0066881 induced up-regulation of RORA through sequestering miR-144-5p. Thus, circ_0066881 functioned as a protector against the development of periodontitis by regulating miR-144-5p/RORA pathway.
However, there are certain limitations in this research. Firstly, all results were collected from cell experiments in vitro and further assays in vivo are necessary to provide more support for the current conclusion. Secondly, the function of circ_0066881 was partly counteracted by miR-144-5p/RORA axis and other miRNA/mRNA networks for circ_0066881 remain to be explored. Thirdly, the molecular regulatory mechanism of periodontitis is complicated and exploring the downstream signaling pathway of RORA may contribute to the understanding of circ_0066881 mechanism in periodontitis.
Conclusion
In conclusion, circ_0066881/miR-144-5p/RORA axis regulated the periodontitis progression in vitro. LPS down-regulated circ_0066881 expression to inhibit RORA level via targeting miR-144-5p, consequently reducing cell viability and enhancing cell apoptosis or inflammation in PDLCs (Figure 7). Circ_0066881 is promising to be a useful biomarker for treatment of periodontitis, which may be a novel perspective in future research.
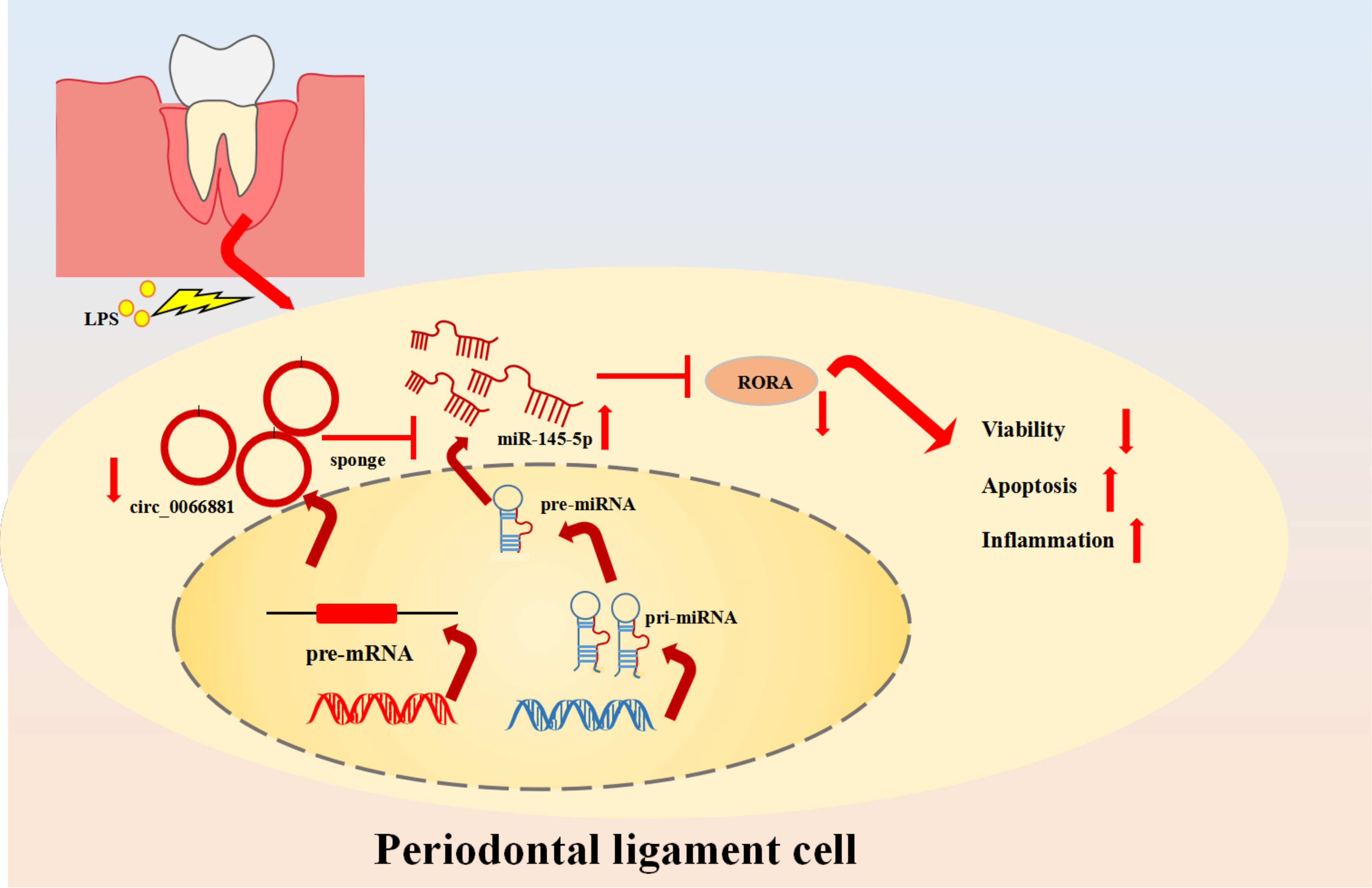
The graphic abstract of this study.
Footnotes
Ethics approval and consent to participate
The present study was approved by the ethical review committee of Pingxiang People’s Hospital. Written informed consent was obtained from all enrolled patients.
Consent for publication
Patients agree to participate in this work
Availability of data and materials
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and technology plan of Jiangxi Provincial Health Commission,《Comparison of periodontal health effects of different types of orthodontic appliances in orthodontic treatment》, (grant number SKJP220200096).
