Abstract
Little is known about the immuno-inflammatory response to Tocilizumab and its association with outcome in critically-ill SARS-CoV2 pneumonia. In this multicenter retrospective cohort of SARS-CoV-2 patients admitted to three intensive care units between March and April 2020, we matched on gender and SAPS II 21 Tocilizumab-treated patients to 42 non-treated patients. Need for mechanical ventilation was 76% versus 79%. IL-6, C-reactive protein, and fibrinogen had been collected within the first days of admission (T1), 3 d (T2) and 7 d (T3) later. Tocilizumab-treated patients had persistently higher IL-6 plasma levels and persistently lower C-Reactive protein and fibrinogen levels. Among Tocilizumab-treated patients, baseline levels of inflammatory biomarkers were not different according to outcome. Conversely, C-reactive protein and fibrinogen decrease was delayed in non-survivors. C-Reactive protein decreased at T1 in survivors (45 [30–98] vs 170 [69–204] mg/l, P < 0.001) but only at T2 in non-survivors (37 [13–74] vs 277 [235–288], P = 0.03). Fibrinogen decreased at T2 in survivors (4.11 [3.58–4.69] vs 614 [5.61–7.85] g/l, P = 0.005) but not in non-survivors (4.79 [4.12–7.58] vs 7.24 [6.22–9.24] g/l, P = 0.125). Tocilizumab treatment was thus associated with a persistent both increase in plasma IL-6, and decrease in C-reactive protein and fibrinogen. Among Tocilizumab-treated patients, the decrease in inflammatory biomarkers was delayed in non-survivors.
Introduction
Few interventions for SARS-CoV2 pneumonia aside from systemic steroids have shown a survival benefit, 1 highlighting the importance of host response in pathogenesis. SARS-CoV-2 infection leads to impaired both innate and adaptive immune responses including high production of IL-6, which may participate to organ and notably lung damage. 2 We have recently reported, in a multicentre cohort of patients admitted to the intensive care unit (ICU) for SARS-CoV-2 infection, that elevated IL-6, C-reactive protein (CRP) and fibrinogen levels were associated with poor outcome. 3 Conflicting results of randomized trials evaluating Tocilizumab, an anti-IL-6 receptor (anti-IL6R) mAb, certainly show that a “one-size-fits-all” approach is not adequate.4–10 These trials had very different inclusion criteria as well as dosage and re-injection strategies, which may in part explain discrepancies. Retrospective work has suggested that treatment response is variable depending both on illness severity but also on treatment modalities.11,12 Fine tuning the subset of patients most likely to benefit from such therapies as well as the ideal timing of administration and dosage can only be achieved by a better understanding of their effect on the immune response.
The aim of this study was to describe the immune-inflammatory profile of critically-ill SARS-CoV-2 patients compassionately treated by Tocilizumab, to compare it to that of untreated patients, and to analyze it according to the outcome among treated patients.
Methods
Patients
All patients with laboratory-confirmed SARS-CoV-2 infection, referred to Saint-Antoine and Tenon Hospitals (Paris, France), and subsequently admitted to one of their 3 ICUs between 1 March and 15 April 2020, were retrospectively screened. Only patients with at least one available plasma cytokine measurement available in the ICU were eligible for study inclusion. Laboratory confirmation for SARS-CoV-2 was defined as a positive result of real-time reverse transcriptase–PCR (RT-PCR) assay of naso-pharyngeal swabs or lower respiratory tract samples.
We included all patients having received Tocilizumab, which were matched to controls who had not. Among treated patients, survivors were then compared to non-survivors.
In Saint-Antoine hospital, in the emergency context of the SARS-CoV-2 pandemic, and before the onset of randomized clinical trials, a multidisciplinary committee including physicians from different specialties (Internal medicine, infectious diseases, intensive care medicine, radiology) was created. Based on clinical and biological parameters (oxygen requirements > 6 l/min or a rise equal or superior to 4 l/min in less than 48 h, CRP levels > 100 mg/l and no evidence of bacterial co-infection) this committee proposed compassionate Tocilizumab to SARS-CoV-2 patients with severe disease, and written informed consent was collected for all patients before administration.
Data collection
We evaluated and analyzed the medical history, physical examination, and hematological, biochemical, microbiological and immunological data obtained from included patients. Epidemiological, clinical and laboratory characteristics, treatment and outcome data were obtained from electronic medical records. Data collection forms were anonymized and reviewed independently by two investigators. Data collected upon admission included age, gender, comorbidities (obesity, hypertension, diabetes mellitus, cardiovascular disease, chronic respiratory disease, chronic renal failure with creatinine clearance < 60 ml/kg/min). Disease severity was evaluated using the simplified acute physiology score II (SAPS II), the sequential organ failure assessment score without neurological points (SOFA) and organ support therapy.
Plasma levels of three cytokines (IL-10, TNF-α, and IL-6) were measured in association with other inflammatory markers, including CRP or fibrinogen within the first days of ICU admission (T1), 3 d (T2) and 7 d (T3) after. Cytokines were measured by ELISA (Quantikine ELISA Kit, R&D Systems) on plasma according to the appropriate dilution and following recommendations of the manufacturer. Thresholds detection were 7.8 pg/ml for IL-10, 15.6 pg/ml for TNF-α, and 3.13 pg/ml for IL-6. Organ failure severity was evaluated at the same time-points using organ support therapy and the SOFA score. The latter's variations were assessed as a delta-SOFA defined by the difference between the later time and the earlier time-point values of the score. A negative delta-SOFA was thus an improvement in organ failure severity, a positive one a worsening.
Statistical analysis
Patient characteristics were summarized as median (25th–75th percentiles) for continuous variables with skewed distributions and percentages for categorical variables as appropriate. Patients having received Tocilizumab were matched to untreated control patients base on sex and admission SAPS II score on a 1:2 ratio with a caliper of 0.3. Differences between groups were compared using the Mann-Whitney test for continuous variables or Fisher's exact test for categorical variables. Wilcoxon's paired test was used to analyze variations of biomarker concentrations between different time points. All tests were two-sided, and P values less than 0.05 were considered statistically significant. No imputation was performed for missing data. No patient was lost to follow-up. Statistical analyses were performed using R (https://www.R-project.org/) software, and graphical representations were established using GraphPad Prism 5.04 (Graph Pad Software Inc. ®).
Study approval
The study was performed in accordance with Good Clinical Practice and the Declaration of Helsinki principles for ethical research. The study protocol was approved by the Institutional Review Board of Entrepôt de Données de Santé de l’Assistance Publique – Hôpitaux de Paris (AP-HP) and by the Ethical committee, Comité de Protection des Personnes (CPP Ile de France X, 2020-A02784-35). All patients received information during their hospital stay that data abstracted from their medical charts could be used for research purposes.
Results
Study population
Over a 6-wk period, 150 patients with RT-PCR confirmed SARS-CoV-2 infection were admitted in the three ICUs. Cytokine profile was available for 130 patients. Twenty-one patients were treated with Tocilizumab (8 mg/kg), a median of 10 [8–16] d after symptom onset, and 3 [1–4] d before first cytokine dosage (T1). These 21 treated patients were matched on a 1:2 ratio to 42 matched control patients who did not receive anti-IL6R therapy (Supplemental Figure 1).
Baseline characteristics are summarized in Table 1. The median age in treated and matched controls was of respectively 57 [45–62] versus 59 [47–66] yr old, with a high proportion of men. Comorbidities were frequent and the only significant difference was a higher proportion of patients treated with angiotensin-converting-enzyme inhibitors or angiotensin-receptor-blockers in the control group (10% vs 41%, P = 0.026). Patients had severe respiratory disease with a PaO2/FiO2 ratio of 133 [93–169] vs 140 [114–224] mmHg (P = 0.171), requiring mechanical ventilation in 76% versus 79% patients (P > 0.999) upon admission, and prone positioning in 33% versus 52% (P = 0.245) during ICU stay. Although there was a trend for control patients to having more frequently received steroids, this was not significant (5% vs 24%, P = 0.127). First cytokine dosage (T1) was performed a median of 14 d after first symptoms in both groups.
Study population admission characteristics. Patients treated with Tocilizumab were matched on a 1:2 ratio to untreated control patients based on sex and SAPS II upon admission.
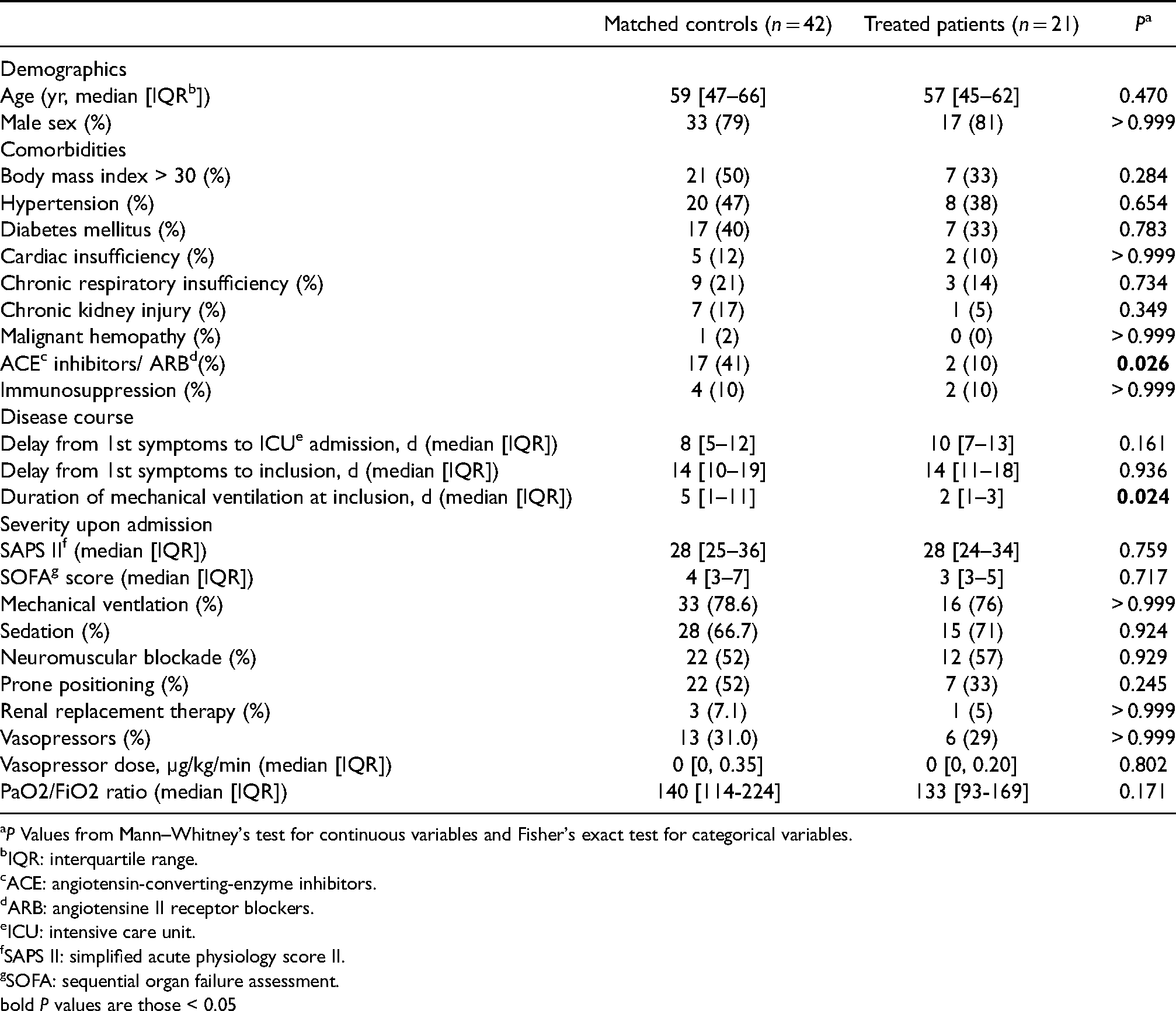
P Values from Mann–Whitney's test for continuous variables and Fisher's exact test for categorical variables.
IQR: interquartile range.
ACE: angiotensin-converting-enzyme inhibitors.
ARB: angiotensine II receptor blockers.
ICU: intensive care unit.
SAPS II: simplified acute physiology score II.
SOFA: sequential organ failure assessment.
bold P values are those < 0.05
Median length of follow-up was 52 [43–59] d. Regarding outcomes, improvement in organ dys-function between T1 and T2 (i.e. 3 d) was less important in Tocilizumab-treated patients as compared to matched controls (delta-SOFA 0 [−1;2] vs − 1 [ − 3;0], P = 0.017) (Supplemental Figure 2). During ICU stay, extra-corporeal membrane oxygenation was more frequently required in the treated group (19% vs 0%, P = 0.018). Nevertheless, there was no significant difference in other outcomes, notably in the rate of bacterial infections at T3 (70% vs 58%, P = 0.536), or in d-60 censored mortality (29% vs 19%, P = 0.592) (Supplemental Table 1).
Systemic immune profile in Tocilizumab-treated patients versus matched controls
Whatever the delay between symptom onset and 1st dosage, IL-6 plasma levels were higher in Tocilizumab-treated patients than in matched controls at T1 (1244 [712–2561] vs 72 [32–195] pg/ml, P < 0.001) (Supplemental Figure 3), and this difference persisted at T2 and T3 (Figure 1). No difference was noted for IL-10 but TNF-α levels were lower in treated patients at T3 (Supplemental Figure 4). Both CRP and fibrinogen were lower in treated patients at T1 (CRP 53 [34–167] vs 207 [88–256] mg/l, P = 0.003; fibrinogen 6.61 [5.00–7.65] vs 7.59 [7.05–8.79] g/l, P = 0.001). Again, this difference persisted at T2 and T3 (Figure 2). There was no significant difference between groups regarding blood lymphocyte counts (Supplemental Figure 5).
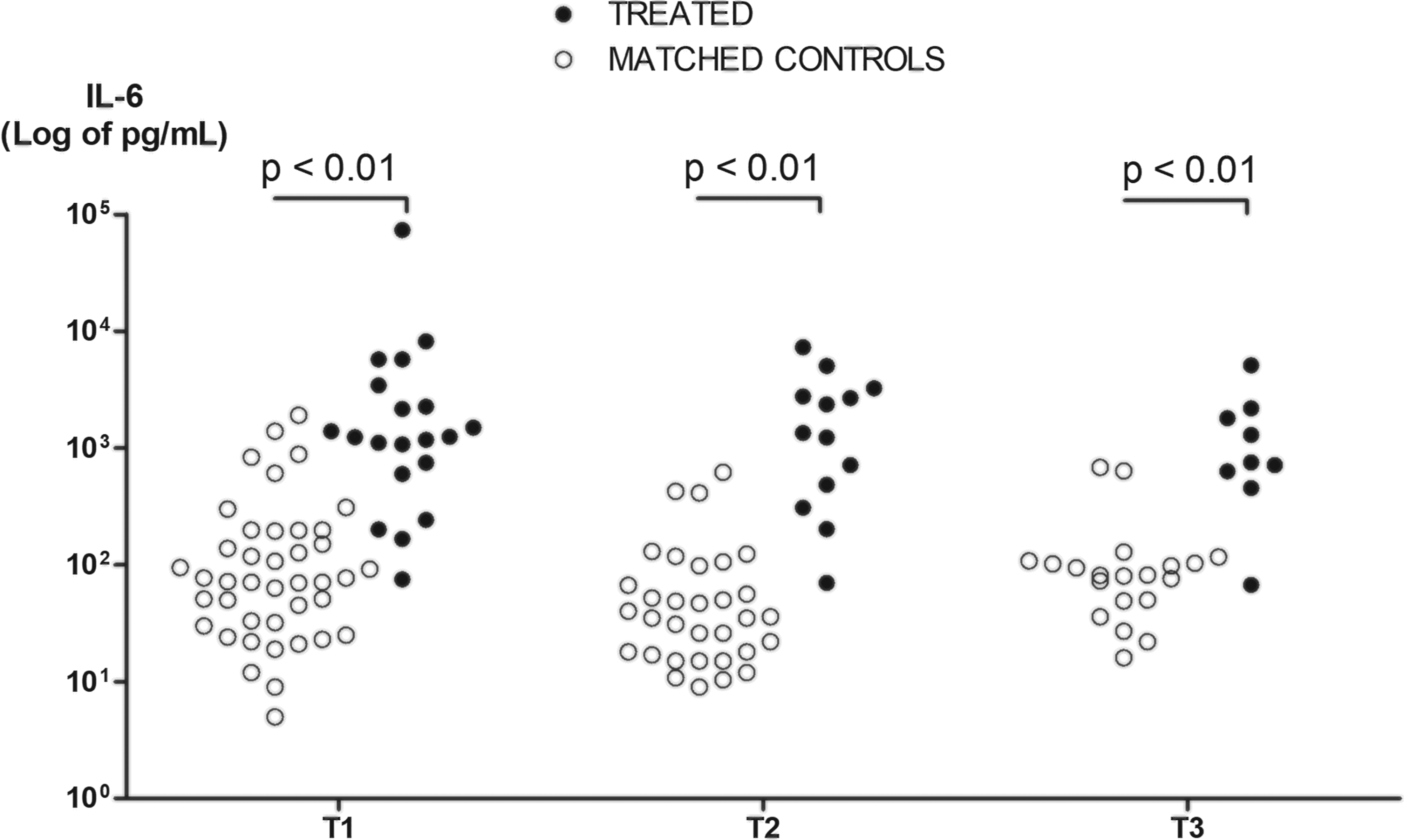
IL-6 plasma level kinetics in Tocilizumab-treated patients and matched controls. IL-6 expressed in log of pg/ml. P Values from a Mann–Whitney test for IL-6 at T1, T2, and T3 between treated patients and matched controls. T1 was the day of 1st cytokine, CRP, fibrinogen and blood lymphocyte count measurement within the first d of ICU admission, T2 was 3 d later and T3 7 d later.
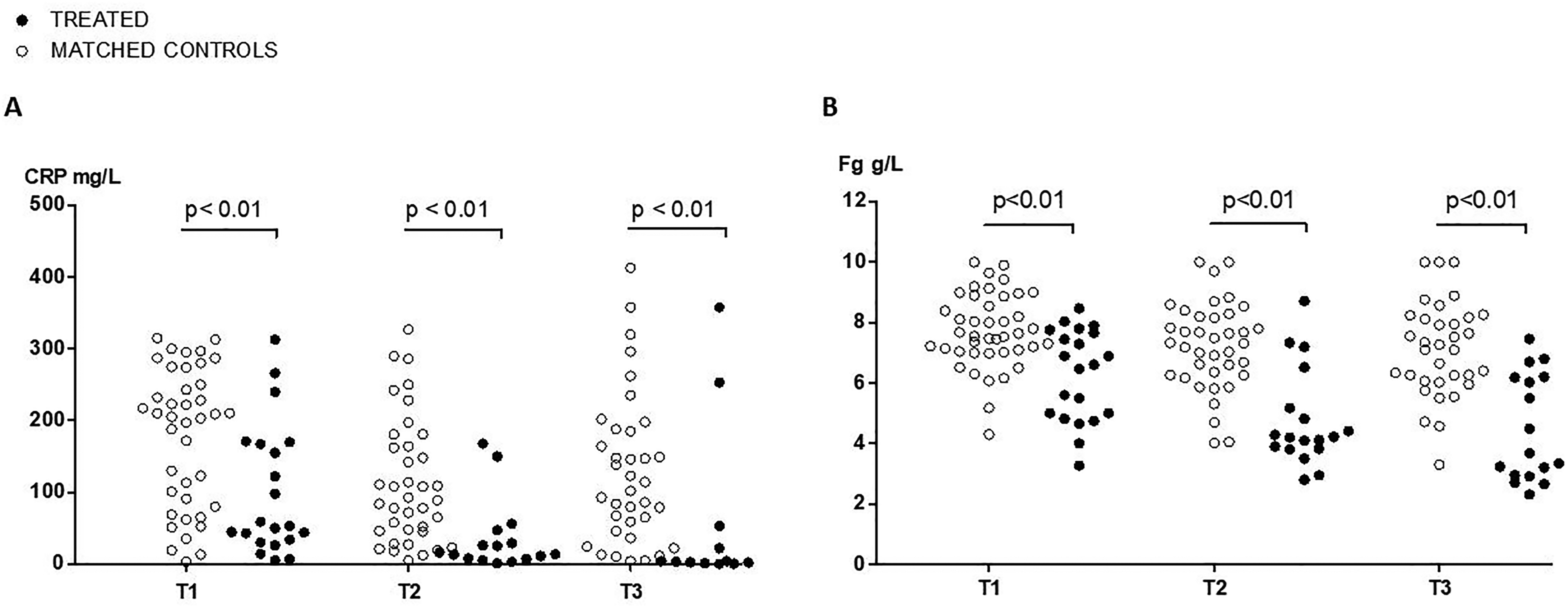
(A) C-Reactive protein and (B) fibrinogen kinetics in Tocilizumab-treated patients and matched controls. P Values from a Mann–Whitney test for C-reactive protein or fibrinogen at T1, T2, and T3 between treated patients and matched controls. T1 was the day of 1st cytokine, CRP, fibrinogen and blood lymphocyte count measurement within the first d of ICU admission, T2 was 3 d later and T3 7 d later.
Immune profile in Tocilizumab-treated patients according to the outcome
Next, we focused on Tocilizumab-treated patients and their systemic immune profile according to the outcome. The kinetics of CRP and fibrinogen plasma levels amongst the 15 (71%) survivors and 6 (29%) non-survivors, are presented in Figure 3. Pre-treatment levels were not different between survivors and non survivors (CRP 170 [69–294] vs 277 [235–288] mg/l, P = 0.09; fibrinogen 6.14 [5.61–7.85] vs 7.24 [6.22–9.24] g/l, P = 0.211). However, although time between anti-IL6R injection and first subsequent dosage (T1) was not different between survivors and non-survivors (2 [1–4] vs 4 [1–5], P = 0.580), the kinetic of inflammatory biomarkers was different according to the outcome. When compared to pre-treatment levels, CRP significantly dropped at T1 in survivors (45 [30–98] vs 170 [69–204] mg/l, P < 0.001) but did not in non-survivors (198 [98–278] vs 277 [173–296], P = 0.219) (Figure 3(A)). CRP decreased later in non-survivors at T2 (37 [13–74 vs 277 [173–296], P = 0.031). Similarly, when compared to pre-treatment levels, fibrinogen significantly decreased at T2 in survivors (4.11 [3.58–4.69] vs 6.14 [5.61–7.85] g/l, P = 0.005) but not in non-survivors (4.79 [4.12–7.58] vs7.24 [6.22–9.24] g/l, P = 0.125) (Figure 3(B)). Although pre-treatment baseline IL-6 plasma levels were not available, levels between survivors and non-survivors were not different at T1 (1238 [675–1825] vs 5747 [1173–8176] pg/ml, P = 0.197) or T3 (713 [261–1547] vs 1470 [660–4375], P = 0.286) (Supplemental Figure 6).
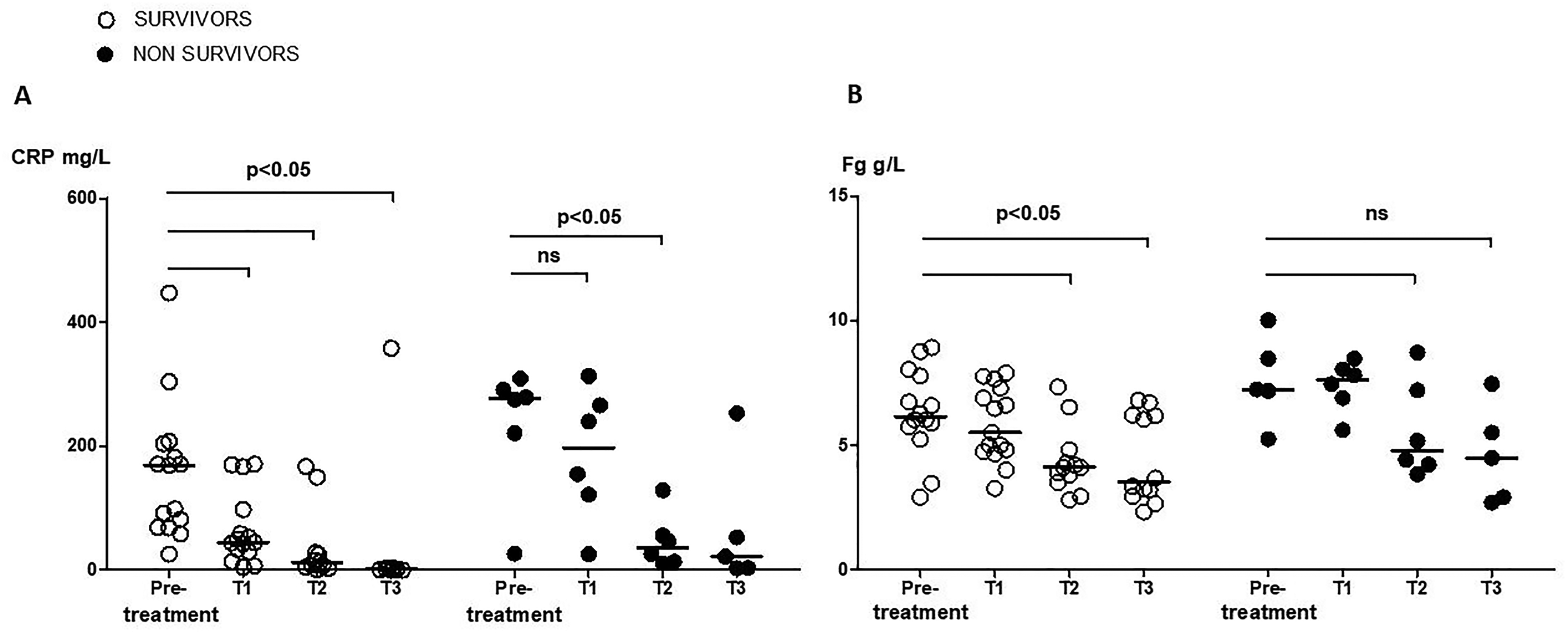
(A) CRP and (B) fibrinogen plasma level kinetics amongst patients treated with Tocilizumab, according to survival. Survival censored at d-60. P Values from a Wilcoxon's paired test for CRP and fibrinogen between different time points amongst respectively survivors and non-survivors in treated patients. Pre-treatment was the day of anti-IL6R injection, T1 was the day of 1st cytokine, CRP, fibrinogen and blood lymphocyte count measurement available in the ICU, T2 was 3 d later and T3 7 d later.
Discussion
The main findings from this matched cohort analysis of critically ill SARS-CoV-2 patients treated with Tocilizumab are as follows: (1) Anti-IL6R treatment, as compared to matched controls, was associated with a significant and persistent increase in plasma IL-6 levels and a significant and persistent decrease in plasma CRP and fibrinogen levels; (2) Amongst Tocilizumab-treated patients, pre-treatment baseline CRP and fibrinogen levels could not discriminate survivors and non-survivors; (3) Following Tocilizumab treatment, CRP and fibrinogen level decrease was delayed in non-survivors.
Both elevated IL-6 plasma levels at a single time-point and their persistence have been identified as predictive for mortality in SARS-CoV-2 patients,3,8,13,14 prompting interest in anti-IL-6R therapy.4–9 Nevertheless, the results of eleven randomized controlled trials evaluating anti-IL-6R in SARS-CoV2 have yielded conflicting results.5,7–10,15–20 Only two trials found a survival benefit,7,10 while the others did not,5,8,9,15–20 including one that was stopped early because of an increased risk of death in the Tocilizumab-treated arm. 19 Similarly, recent meta-analyses show discordant findings.21–24 These discrepancies suggest a “one size fits all” approach is not optimal. Moreover, study methodology varied widely regarding both use of biomarkers as inclusion criteria and timing, indication and dose of re-injection, with restrospective data suggesting a variable treatment effect according to disease severity, timing and dosage of treatment.11,12 Whether identifying a subset of responders, optimal timing or need for higher doses or re-injection would impact survival remains an important unresolved question.
We found that Tocilizumab treatment results in increased circulating IL-6 levels, in accordance with recent prospective data. 19 Whether this is due to reduced clearance by lack of receptor-binding or to a rise in production by a positive feedback remains unknown. 25 Nevertheless, this observation raises the interest of downstream biomarkers to both screen patients most likely to benefit from IL-6 receptor blockade and to evaluate response to treatment, more so as the systemic inflammatory profile of SARS-CoV2 patient has been described as more complex than a single biomarker can capture. 26 We focused on CRP and fibrinogen as global markers of inflammation produced by the liver in response to IL-6 receptor engagement and JAK-STAT pathway activation.27,28 We recently reported that high CRP levels at ICU admission were associated with organ failure worsening within the next three days in patients not treated with anti-IL-6R. 3 Our finding that CRP decreases in all Tocilizumab-treated patients is consistent with the work by Veiga et al. 19 but not by Soin et al. 20 This discrepancy is not explained by steroid use, as more than 70% of patients from both studies received steroids. Regarding the subset of responders, only four of the randomized trials used elevated CRP as an inclusion criteria.8,10,16,19
We further assessed whether kinetics of the immunoinflammatory profile after Tocilizumab administration were associated with outcome. Here, among Tocilizumab-treated patients, baseline pre-treatment levels of CRP and fibrinogen could not discriminate survivors and non-survivors, consistent with two uncontrolled cohorts of Tocilizumab-treated patients.29,30 Regarding kinetics, Toniati et al. found no difference between improvers and non-improvers, but evaluation was done only at a single and very late timepoint, ten days after treatment. 29 In contrast, we analyzed variations within the first week after Tocilizumab injection and found that CRP and fibrinogen drop was delayed in non-survivors. Although we found no significant difference in IL-6 levels except at T2, higher levels 24 h after Tocilizumab injection were associated with mortality in another small cohort. 31 This is of importance, especially as administration of a second dose of anti-IL6R was very heterogenous in randomized trials. Two did not allow re-injection,8,19 one planned it systematically 12 h after the first 16 and the remaining eight allowed it between 24 h and 7 d later.5,7,9,10,15,17,18,20 In these latter trials, decision to administrate a second dose was based on the absence of clinical improvement or according to treating physician decision, and approximatively 30% of patients received it. No trial included biomarkers as a criterion to inject a second dose. Our data suggests that the timing of inflammation control induced by Tocilizumab, especially in the first few days, may be critical and associated with survival, which could help better target patients most likely to benefit from a second dose of treatment. Of note, differences in inflammatory responses after anti-IL6R treatment between survivors and non-survivors are unlikely due to secondary infection in this cohort, as bacterial infection rate was similar between groups, as previously reported. 32
This work has some limitations. There was some heterogeneity in the timing between symptom onset and 1st cytokine dosage as well as anti-IL6R treatment. All treated patients received Tocilizumab before 1st cytokine dosage, at a median of 3 d before and pre-treatment interleukine 6 levels were thus not available. Although these baseline levels have been associated with outcome in patients not treated with anti-IL6R, 3 this was not the case in other uncontrolled series of patients having received such treatments.31,33 Due to the inclusion period early in the pandemic, few patients received steroids. Finally, this study was not designed to assess safety or efficacy of anti-IL6R treatment in SARS-CoV-2 infection, as the small sample size may result in limited statistical power, and results regarding global outcome should be analyzed with caution.
Conclusions
In this multicenter matched-cohort analysis of critically-ill SARS-CoV-2 patients, Tocilizumab treatment was associated with a decrease in CRP and fibrinogen levels, but this decrease was delayed in non-survivors. In light of recent randomized trials, these findings could help refine the subset of patients most likely to benefit from such treatments, as well as the optimal timing, dosage, or need for re-injection.
Supplemental Material
sj-docx-1-ini-10.1177_17534259211064602 - Supplemental material for Delayed inflammation decrease is associated with mortality in Tocilizumab-treated critically ill SARS-CoV-2 patients: A retrospective matched-cohort analysis
Supplemental material, sj-docx-1-ini-10.1177_17534259211064602 for Delayed inflammation decrease is associated with mortality in Tocilizumab-treated critically ill SARS-CoV-2 patients: A retrospective matched-cohort analysis by Tomas Urbina, Jean-Rémi Lavillegrand, Marc Garnier, Arsene Mekinian, Jerome Pacanowski, Nathalie Mario, Guillaume Dumas, Geoffroy Hariri, Antoine Pilon, Lucie Darrivère, Muriel Fartoukh, Bertrand Guidet, Eric Maury, Judith Leblanc, Yannick Chantran, Olivier Fain, Karine Lacombe, Guillaume Voiriot and Hafid Ait-Oufella in Innate Immunity
Footnotes
Acknowledgments
We are indebted to Pierre Aucouturier, Guillaume Lefevre, Jerome Guéchot, Michel Vaubourdolle, Jean-Claude Buzzi, and Marion Fages for their help and l’Entrepôt de Données de Santé de l’Assistance Publique – Hôpitaux de Paris (AP-HP) for their help in data extraction.
Availability of data and material
The dataset used for the analysis is available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
