Abstract
miR-150 was found to target the 3′-untranslated regions of AKT3, and the AKT pathway was affected by SR protein kinase 1 (SRPK1). However, the expression and significance of miR-150, AKT3 and SRPK1 in acute lung injury (ALI) were not clear. Here, we found that the expression of miR-150 was significantly reduced, while the expression of AKT3 and SRPK1 were markedly increased in LPS-treated A549, THP-1 and RAW 264.7 cells. miR-150 significantly decreased levels of pro-inflammatory cytokines IL-1β, IL-6 and TNF-α, reduced the expression of AKT3, but had no impact on SRPK1 expression compared with the control group in LPS-treated A549, THP-1 and RAW 264.7 cells. AKT3 silencing only reduced the production of pro-inflammatory cytokines and showed no effect on miR-150 and SRPK1 expression. Finally, we observed that miR-150 mimics and/or silencing of SRPK1 decreased the expression of AKT3 mRNA. Besides, over-expression of miR-150 or silencing of SRPK1 also reduced the expression of AKT3 protein, which exhibited the lowest level in the miR-150 mimics plus si-SRPK1 group. However, si-SRPK1 had no effect on miR-150 level. In conclusion, miR-150 and SRPK1 separately and cooperatively participate into inflammatory responses in ALI through regulating AKT3 pathway. Increased miR-150 and silenced SRPK1 may be a novel potential factor for preventing and treating more inflammatory lung diseases.
Keywords
Introduction
Acute lung injury (ALI), a severe respiratory dysfunction, may be induced by a variety of etiologies, such as direct causes (pneumonia, near-drowning and toxic inhalation) and indirect injury to the lung organ (burns and massive blood transfusion), ultimately causing high morbidity and mortality.1–3 Despite great achievements made in improving treatment strategies for ALI, the mortality rate remains high worldwide.4,5 Therefore, it is urgent to explore the molecular pathogenesis of ALI and search for the new therapeutic strategies for ALI.
miRNAs are 19–23 nucleotides long, endogenous double-stranded RNA molecules which play an important role in regulating gene expression by mediating post-transcriptional silencing of target genes, leading to mRNA destabilization and translational inhibition.6–9 miRNAs are reported to modulate the magnitude of inflammatory response by targeting the expressions of cytokine genes, cell surface receptors and inflammation-related signaling molecules. Previously, we demonstrated that miR-150 targeted the 3′-untranslated regions (3′-UTR) of AKT3 to ameliorate LPS-induced inflammatory injury of A549 cells. 10 In addition, SRPK1 silencing affected the AKT signaling pathways. 11 However, the relationship and role of miR-150, AKT3 and SRPK1 are still not fully elucidated in LPS-induced A549 cells and macrophages.
In the present study, we explored the effects of miR-150 in A549 cells and macrophages-related ALI in the presence of LPS. A549, THP-1 and RAW 264.7 cells were stimulated with different concentrations of LPS. Reverse transcription-quantitative PCR (RT-qPCR) and Western blotting were performed to measure the expression of indicated genes. The effects of miR-150, AKT3 and SRPK1 on LPS-treated A549 cells and macrophages were then determined by transfection with miR-150 mimics or AKT3/SRPK1 siRNAs. Cell viability was measured using cell counting kit-8 (CCK-8) assay. The expressions of IL-1β, IL-6 and TNF-α were measured using ELISA. This study aimed to identify miR-150/SRPK1/AKT3 as a novel therapeutic target for treatment of ALI.
Materials and methods
Cell culture
A549, RAW 264.7 and THP-1 cells were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA), and were cultured in RPMI-1640 media supplemented with 10% FBS (Medistech, Herndon, VA, USA), 100 U/ml of penicillin, and 0.1 mg/ml of streptomycin (Sigma-Aldrich, St. Louis, MO, USA). All cells were incubated at 37°C in an atmosphere of 95% air and 5% CO2. Cells (5 × 105/ml) were incubated for the indicated time points in the presence of LPS. Supernatants were collected to measure levels of inflammatory cytokines. Cell pellets were used for RNA and protein extraction.
Cell transfections
miR-150 mimics and its negative control (NC) mimics were synthesized and purchased from GenePharma (Shanghai, China). AKT3 or SRPK1-targeted siRNA and scramble control siRNA duplexes were also obtained from GenePharma (Shanghai, China). Transfection was performed using Lipofectamine 2000 Reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions.
Cell viability assay
Cells at the logarithmic growth stage were seeded into 96-well plates at a density of 5 × 104 cells/well in a total volume of 100 µl per well and cultured for 0, 2, 6, 12 and 24 h post treatment. Then, 10 μl of CCK-8 (C0037, Beyotime, China) reagent were added and incubated in a 37°C, 5% CO2 incubator, and the absorbance was measured at 450 nm using a microplate reader.
RT-qPCR
Total RNA was extracted from cells with TRIzol reagent (Invitrogen, Carlsbad, CA, USA). Total miRNA quantification was achieved by RT-qPCR using the miScript SYBR Green PCR Kit (Qiagen, Hilden, Germany) and the ABI 7500 Real-Time PCR System (Applied Biosystems). The U6 small nuclear RNA served as a reference. The mRNA levels were determined using SYBR Green method, with GAPDH as an internal control. The RT-qPCR results were analyzed with the 2–ΔΔ Ct method.
Western blotting
Protein assay was performed using a BCA protein assay kit (Pierce/Thermo Scientific, Rockford, IL, USA) according to the manufacturer’s instructions. A total of 30 μg proteins were added for one-dimensional SDS-PAGE using the discontinuous buffer system of Laemmli (Bio-Rad Laboratories, USA). The electrophoresed proteins were transferred to a polyvinylidene difluoride membrane. Membranes were blocked with non-fat dry milk, washed, and probed overnight at 4°C with primary Ab followed by HRP-conjugated secondary rabbit or mouse Ab (Cell Signaling Technology). The protein bands were visualized with an electrochemiluminescence (ECL) system (Amersham Biosciences, USA).
ELISA
The ELISA kits were purchased from Invitrogen Corporation (Camerio, CA, USA). ELISA was conducted to measure the levels of IL-1β, IL-6 and TNF-α after treatment according to the manufacturer’s instructions. The experiments were performed at least three times and the optical density was measured at 450 nm using an automated ELISA reader (SPECTRA max M5; Tokyo, Japan).
Statistical analysis
All the reported experiments in this study were repeated in triplicate. Data analyses were conducted using Graph Pad Prism 5.0 (San Diego, CA). Error bars represent SD of three independent experiments. Quantitative data were expressed as mean ± SD. The results were statistically evaluated by one-way ANOVA followed by the LSD post hoc test. P < 0.05 (two-side P value) was considered significant.
Results
LPS induces release of inflammatory cytokines in THP-1 and RAW 264.7 cells
Previous reports indicated that LPS treatment could affect the proliferation and induce release of inflammatory cytokines in A549 cells. Here, we further evaluated the effect of LPS on proliferation in THP-1 and RAW264.7 cells exposed to different concentrations of LPS using CCK-8 assay. As shown in Figures 1(a) and 1(b), when treated with different concentrations of LPS, the proliferation of THP-1 and RAW264.7 cells relative to control were apparently affected in a dose- and time-dependent manner. We determined treatment with 10 μg/ml LPS had a relatively stronger inhibitory effect on cell proliferation, which was selected for further investigation. Next, we explored whether THP-1 and RAW264.7 cells released IL-1β, IL-6, and TNF-α in response to LPS using RT-qPCR and ELISA. We found that the mRNA expression and secretion level of IL-1β, IL-6 and TNF-α began to rise after stimulation, reached a peak at 12 h, and maintained the elevated level at 24 h post stimulation, indicating that THP-1 and RAW264.7 cells time-dependently increased levels of IL-1β, IL-6 and TNF-α mRNA following LPS stimulation (Figure 1(c, d)), accompanied with elevated release of IL-1β, IL-6 and TNF-α molecules (Figure 1(e, f)).
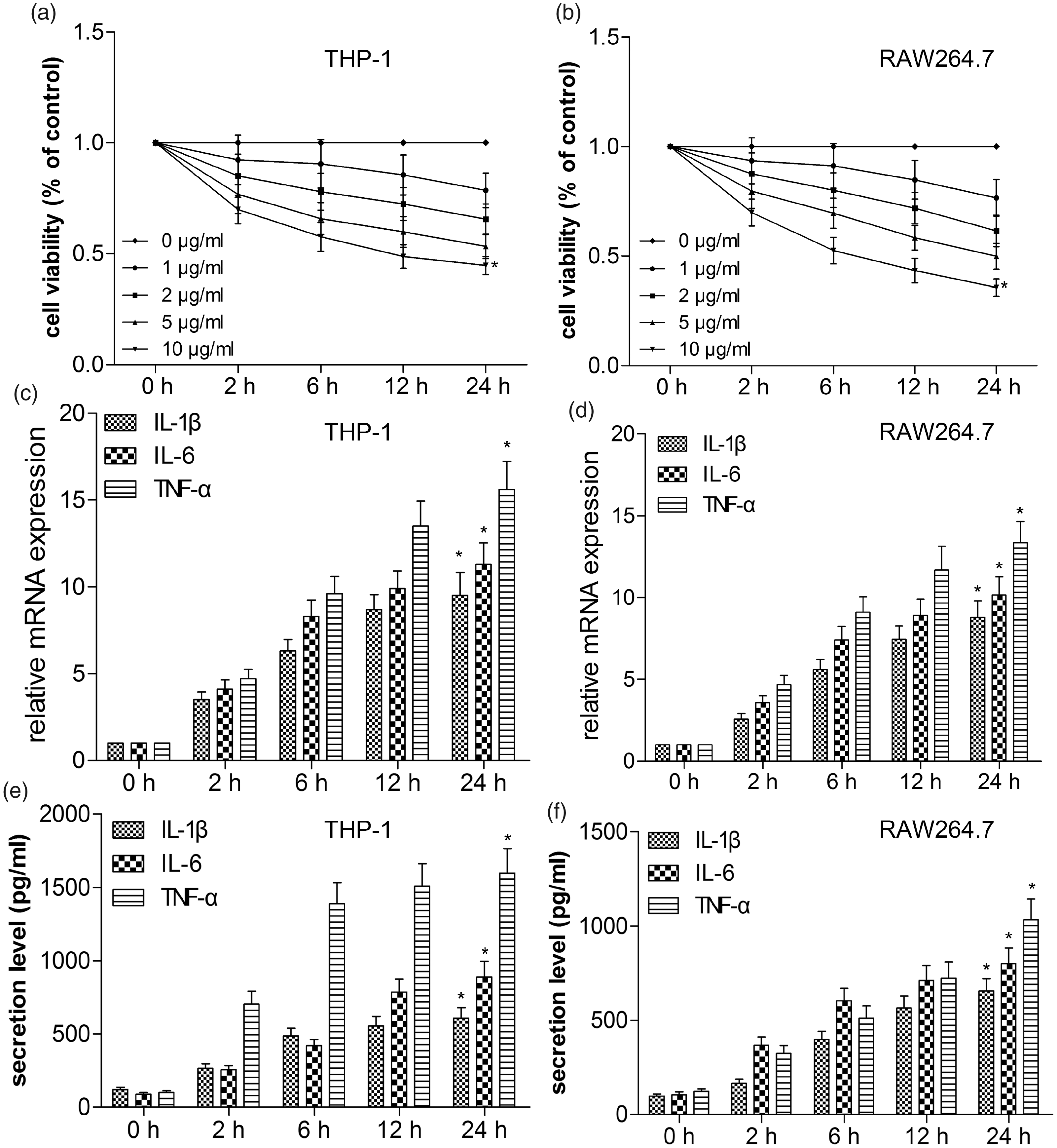
LPS induces release of inflammatory cytokines in THP-1 and RAW264.7 cells. THP-1 and RAW264.7 cells were treated with LPS at different concentrations (0, 1, 2, 5 and 10 μg/ml). THP-1 (A) and RAW264.7 (B) cell viability was measured by CCK-8 assay. The mRNA expressions of IL-1β, IL-6 and TNF-α of THP-1 (C) and RAW264.7 (D) cells were determined by RT-qPCR. Expression was normalized to GAPDH. Protein secretion levels of IL-1β, IL-6 and TNF-α in culture supernatants of THP-1 (E) and RAW264.7 (F) cells were determined using ELISA assays. Values are expressed as the mean ± SD using results from three independent experiments. *P < 0.01 compared with the control group.
LPS reduces miR-150 level and increases the expression of AKT3 and SRPK1 in A549, THP-1 and RAW264.7 cells
We had found LPS stimulation decreased the expression of miR-150 and increased the expression of AKT3 in A549 cells-established ALI model. To examine whether the expression of miR-150 in macrophages was regulated by LPS, THP-1 and RAW264.7 cells were treated with LPS for different time points. We found that LPS time-dependently decreased the expression of miR-150 (Figure 2(a)) in THP-1 and RAW264.7 cells. Because the effects of LPS on cell proliferation and release of inflammatory cytokines were obvious at 24 h post treatment, we further measured the expression of AKT3 and SRPK1 24 h following LPS administration using Western blotting. Compared with the control group, the expression of AKT3 and SRPK1 protein were notably up-regulated by LPS in A549, THP-1 and RAW264.7 cells (Figure 2(b–d)). Previously, we demonstrated that miR-150 inhibited the expression of AKT3 by targeting its 3′-UTR, and SRPK1 affected AKT pathways. Thus, we inferred that miR-150 and SRPK1 may co-regulate inflammatory responses together in ALI through the AKT3 pathway.
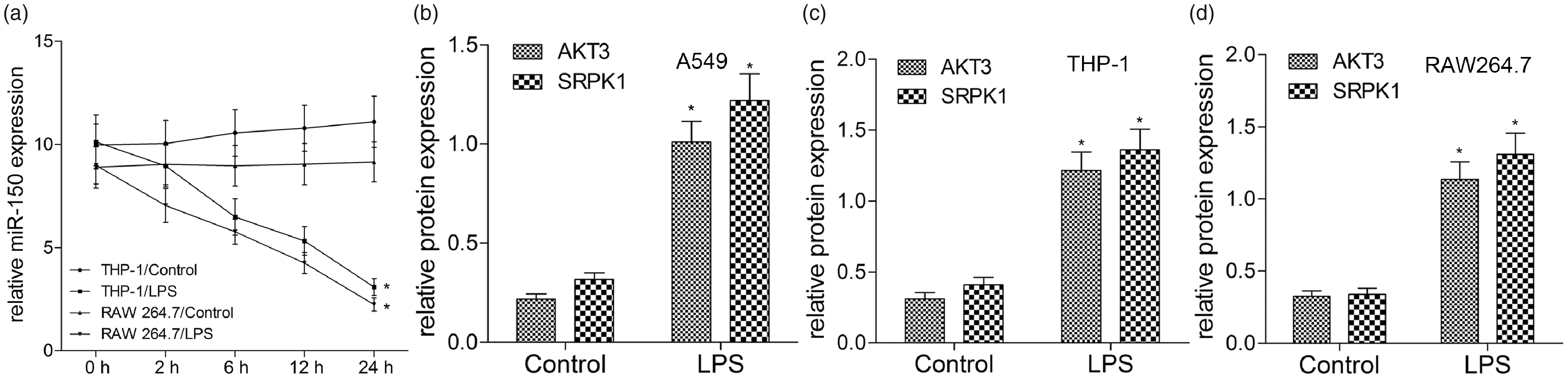
LPS reduces miR-150 level and increases the expression of AKT3 and SRPK1 in A549, THP-1 and RAW264.7 cells. THP-1 and RAW264.7 cells were treated with or without LPS for 2, 6, 12 and 24 h. (A) miR-150 level was examined by quantitative RT-qPCR. A549 (B), THP-1 (C) and RAW264.7 (D) cells were treated with LPS for 24 h, and then cell lysates and supernatants were subjected to Western blot analysis using Abs specific for AKT3, SRPK1 and GAPDH. GAPDH was used as a loading control. Values are expressed as the mean ± SD using results from three independent experiments. Data are presented as fold-change in LPS group relative to control. *P < 0.01 compared with the control group.
miR-150 inhibits inflammatory cytokines and AKT3 rather than SRPK1 in LPS-induced A549, THP-1 and RAW 264.7 cells
Owing to the inhibition effect of miR-150 on inflammatory responses of A549 cells, we continued to investigate its possible role in LPS-induced THP-1 and RAW 264.7 cells. First, miR-150 was prominently up-regulated in THP-1 and RAW 264.7 cells in miR-150 mimics group compared with NC mimics group. ELISA assay revealed that LPS-induced increase of pro-inflammatory cytokines was greatly reversed by over-expression of miR-150 in THP-1 and RAW 264.7 cells (Figures 3(a–c)). Compared with the NC mimics group, over-expression of miR-150 significantly decreased AKT3 expression (P < 0.01, Figure 3(d)), but did not affect SRPK1 level in LPS-induced A549, THP-1 and RAW 264.7 cells (P > 0.10, Figure 3(e)). Together with previous studies, our results suggested that miR-150 could inhibit LPS-induced inflammatory response via inhibiting the expression of AKT3 instead of SRPK1.
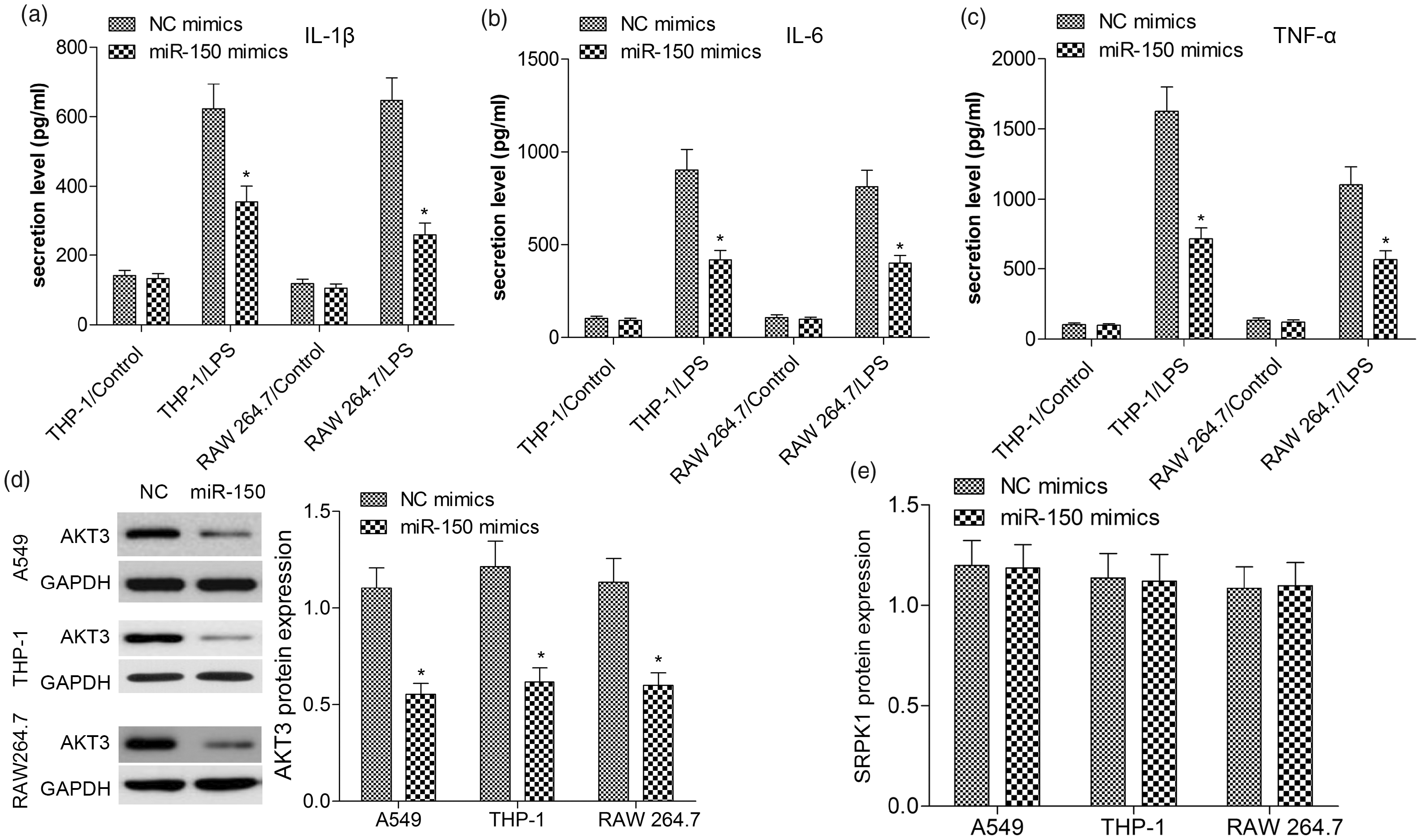
miR-150 inhibits inflammatory cytokines and AKT3 in LPS-induced A549, THP-1 and RAW 264.7 cells. A549, THP-1 and RAW 264.7 cells were co-transfected with 50 nM NC or miR-150 mimics and then treated with LPS for 24 h. The levels of IL-1β (A), IL-6 (B) and TNF-α (C) in the culture supernatants of THP-1 and RAW 264.7 cells were determined by ELISA. The protein levels of AKT3 (D) and SRPK1 (E) were measured by Western blot analysis. Values are expressed as the mean ± SD using results from three independent experiments. *P < 0.01 compared with the control group.
Silencing of AKT3 inhibits inflammatory cytokines in LPS-induced A549, THP-1 and RAW 264.7 cells
In a previous study, silencing of AKT3 expression indeed exerted its anti-inflammatory function in LPS-treated A549 cells. Likewise, we transfected si-AKT3 or si-NC into THP-1 and RAW 264.7 cells in the present study. As presented in Figure 4(a), the si-AKT3 significantly suppressed LPS-induced IL-1β, IL-6, and TNF-α levels in either THP-1 or RAW 264.7 cells in contrast to those in the NC mimics group (P < 0.01, Figure 4(a–c)). However, treatment with si-AKT3 appeared to have no inhibitory effect on miR-150 and SRPK1 expressions using RT-qPCR and Western blotting, respectively (P > 0.10, Figure 4(d, e)). These results indicated that silencing of AKT3 inhibits inflammatory cytokines and AKT3 may be a downstream target of miR-150 or SRPK1.
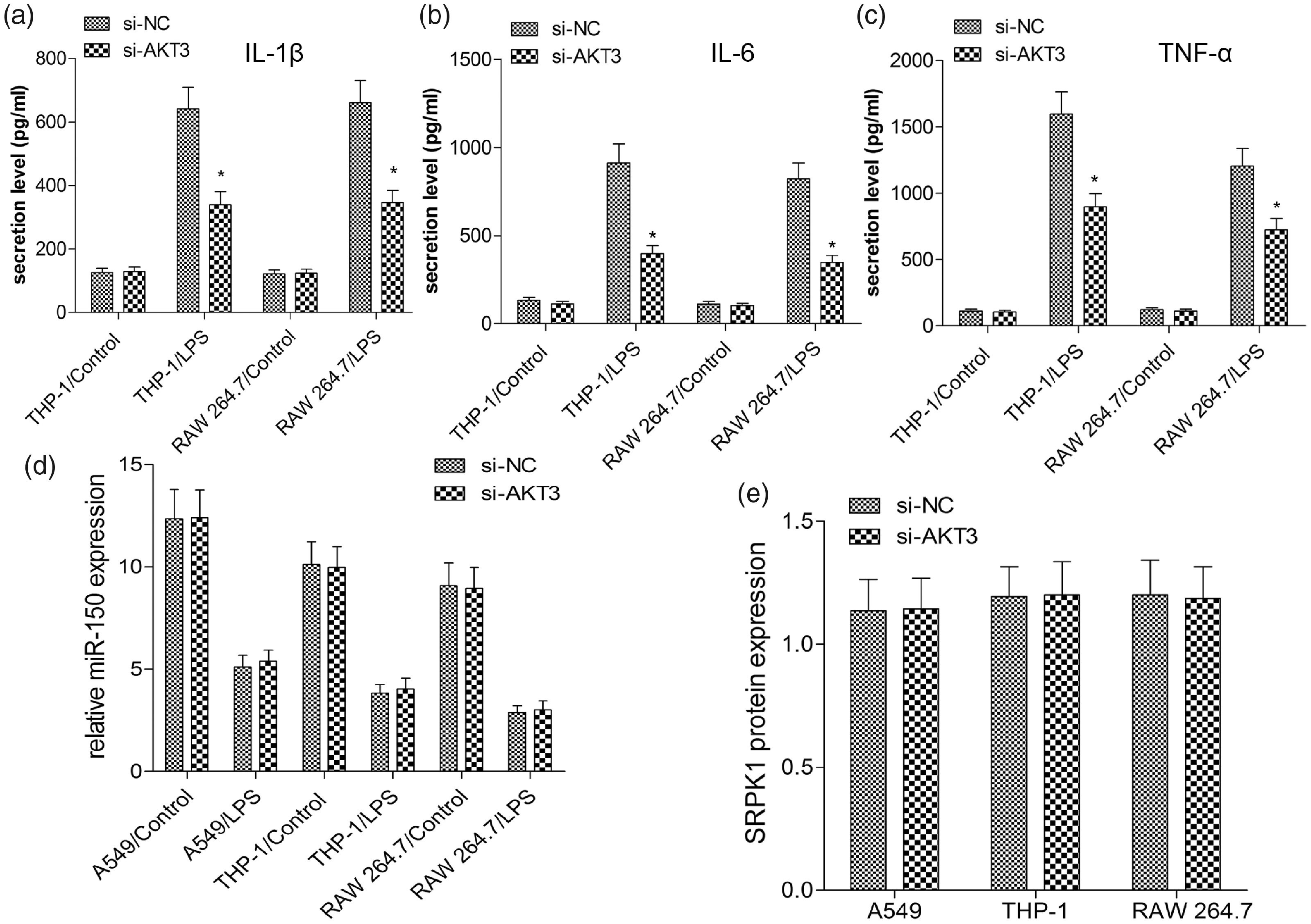
Silencing of AKT3 inhibits inflammatory cytokines in LPS-induced A549, THP-1 and RAW 264.7 cells. A549, THP-1 and RAW 264.7 cells were transfected with 50 nM si-NC or si-AKT3 and then treated with LPS for 24 h. The levels of IL-1β (A), IL-6 (B) and TNF-α (C) in the culture supernatants of THP-1 and RAW 264.7 cells were determined by ELISA. (D) miR-150 level was examined by quantitative RT-PCR. (E) The protein level of SRPK1 was measured by Western blot analysis. Values are expressed as the mean ± SD using results from three independent experiments. *P < 0.01 compared with the control group.
Silencing of SRPK1 inhibits inflammatory cytokines and the mRNA expression of AKT3
To verify the relationship of SRPK1 and AKT3, we co-transfected miR-150 mimics and/or si-SRPK1 into A549, THP-1 and RAW 264.7 cells and treated these cells with or without LPS. As shown in Figure 5(a–c), after 24 h of LPS treatment to activate inflammatory responses, we explored whether SRPK1 was a key factor for the inflammation regulated by miR-150/AKT3. Firstly, inflammation response was reversed after silencing of SRPK1 compared with si-NC (P < 0.001). Then, RT-qPCR showed that silencing of SRPK1 did not affect the level of miR-150 (P > 0.10, Figure 5(d)), indicating they are mutually independent. Subsequently, we found si-SRPK1 suppressed AKT3 mRNA level. Meanwhile, miR-150 mimics further decreased the level of AKT3 mRNA in the presence or absence of si-SRPK1, indicating that miR-150 and SRPK1 were involved in the regulation of AKT3 gene transcription (Figure 5(e)). As shown in Figure 5(f), Western blotting assay identified that down-regulation expression of SRPK1 by si-SRPK1 decreased the AKT3 protein level; further miR-150 mimics aggravated the inhibitory effects of si-SRPK1 on the protein level of AKT3 in comparison with the miR-150 mimics plus si-NC or NC mimics plus si-SRPK1 group. To our knowledge, some factors drive the translocation of SRPK1 from the cytoplasm to the nucleus, which subsequently alters downstream splicing events of pre-mRNAs. Therefore, we thought that SRPK1 regulates AKT3 expression probably at the transcription level, and miR-150 regulates AKT3 expression at the transcription and translation level, by which AKT3 modulates the inflammatory responses in ALI.
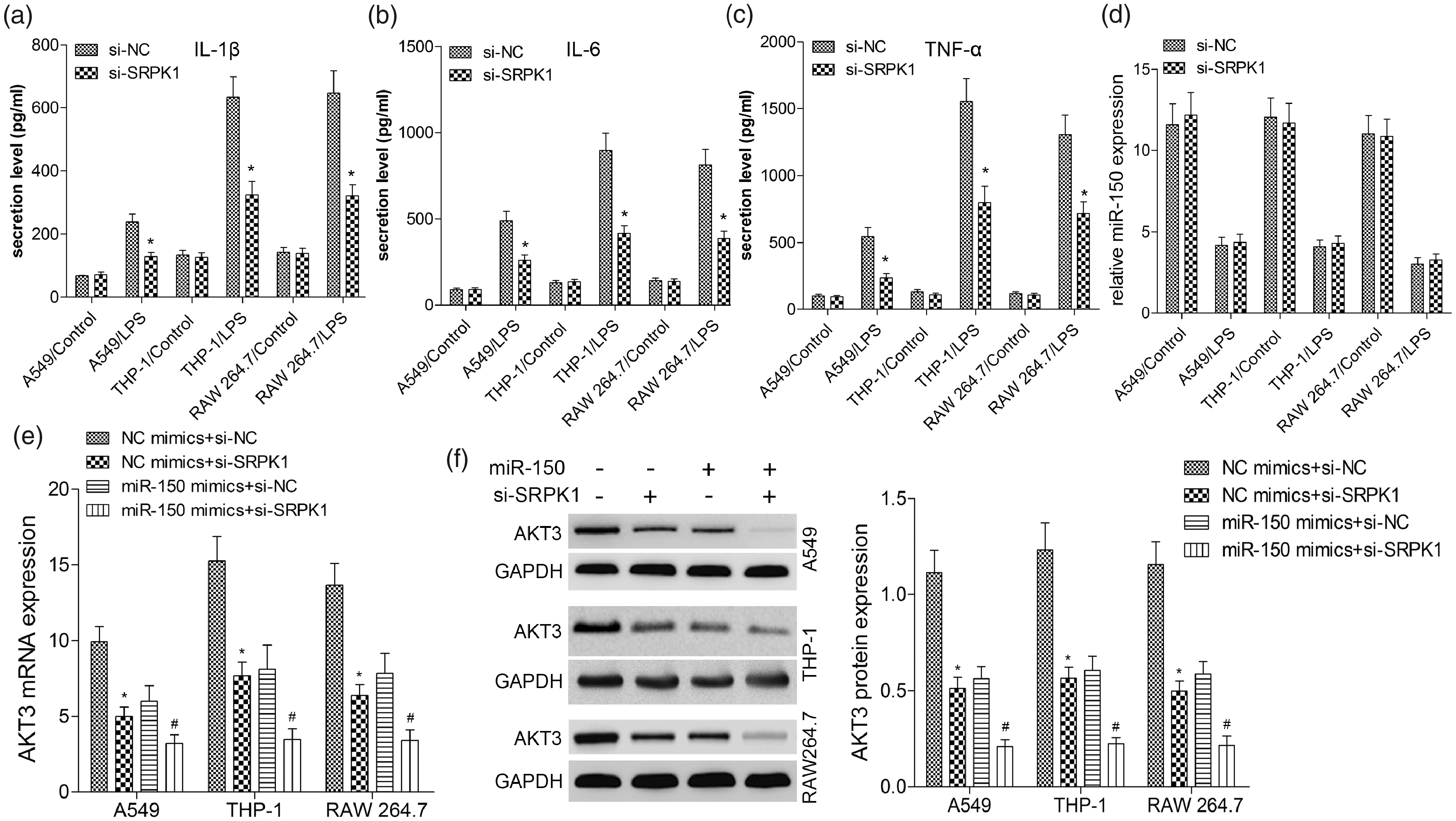
Silencing of SRPK1 inhibits inflammatory cytokines and AKT3 in LPS-induced A549, THP-1 and RAW 264.7 cells. A549, THP-1 and RAW 264.7 cells were transfected with 50 nM si-NC or si-SRPK1 for 24 h and then treated with LPS for 24 h. The levels of IL-1β (A), IL-6 (B) and TNF-α (C) in the culture supernatants of A549, THP-1 and RAW 264.7 cells were determined by ELISA. Then, cells were co-transfected with 50 nM NC or miR-150 mimics plus 50 nM si-NC or si-SRPK1. MiR-150 (D) and AKT3 mRNA (E) level was examined by quantitative RT-PCR. (F) The protein level of AKT3 was measured by Western blot analysis. Values are expressed as the mean ± SD using results from three independent experiments. *P < 0.01 compared with the control group.
Discussion
Inflammation is recognized as a defensive response of the human body to various stimuli or tissue damage. During a normal immune response, immune cells infiltrate the infected area, which enables the body to remove damaged cells, irritants, pathogens and sterile injuries. Among the immune cells, macrophages play an important role in the initiation and regulation of the host’s defense. Macrophages are activated by stimuli to produce a variety of inflammatory mediators. However, when macrophages are over-activated, excessive inflammatory reaction will severely damage various tissues, leading to acute lung inflammation, severe sepsis and pancreatitis. 12 Our earlier study had confirmed that miR-150 was frequently de-regulated in LPS-induced human pulmonary epithelial cell line A549, and functions as an effective inhibitor in ALI progression by targeting AKT3. In the present study, we extended the findings to macrophages (THP-1 and RAW 264.7 cells). The results of these studies validated our previous observations, and showed that IL-1β, IL-6 and TNF-α were induced in macrophages in response to LPS, accompanied by reduced miR-150 expression and increased AKT3 expression, indicating that miR-150/AKT3 also participated in macrophages-related ALI.
AKT, a serine/threonine protein kinase, regulates a variety of cellular responses, and consists of three closely related isoforms, named AKT1, AKT2 and AKT3. 13 The AKT pathway plays an important role in regulating the survival, migration and proliferation of macrophages, and in rapidly orchestrating immune response macrophages.14,15 Of the three AKT isoforms, AKT3 is required for homeostasis and positively correlates with the severity of inflammation and macrophage activation in vitro and in vivo. 16 In recent studies, LPS activated the expression of Akt3 in Kupffer cells during alcoholic liver injury, leading to generation of pro-inflammatory cytokines. 13 Therefore, blocking of AKT3 may be an effective strategy for inhibiting inflammatory responses. Consistently, in this study, AKT3 silencing indeed reduced the production of pro-inflammatory cytokines, while it showed no effect on miR-150 and SRPK1 expressions, suggesting AKT3 may be a downstream target of miR-150 or SRPK1. However, the mechanism by which AKT3 activates the inflammatory reaction was unknown.
SRPK1, an enzyme that phosphorylates splicing factors rich in serine/arginine domains, has been involved in some cellular activities, including cell cycle, innate immune response, chromatin reorganization, and regulation of numerous mRNA-processing pathways as well as inflammation through its interaction with many transcription factors and signaling pathways.17,18 Some factors drive the translocation of SRPK1 from the cytoplasm to the nucleus, which subsequently alters downstream splicing events of pre-mRNAs.19,20 Aberrant expression of SRPK1 has been found to regulate constitutive AKT activation. 21 We previously reported that the activation of PI3K/AKT pathway was inhibited by genetic ablation of SRPK1 in gastric cancer cells. 11 Li ZR et al. demonstrated that SOX2 knockdown decreased SRPK1 expression, which further led to the down-regulation of AKT expression levels in basal cell carcinoma cells. 22 Li et al. found that silencing of SRPK1 inhibits vascular smooth muscle cell apoptosis, and increases cell proliferation and vascular remodeling via the PI3K/AKT signaling pathway. 23 Thus we assumed that SRPK1 was likely to exert its splicing function in the AKT3-related inflammatory pathways. Here, we found that silencing of SRPK1 did not affect the level of miR-150, but indeed suppressed the AKT3 mRNA level. However, miR-150 mimics did not decrease the level of SRPK1, indicating that miR-150 and SRPK1 were separately involved in the regulation of AKT3 gene expression. At the protein level, si-SRPK1 decreased the AKT3 protein level; further, miR-150 mimics aggravated the inhibitory effects of si-SRPK1 on the protein level of AKT3. Therefore, SRPK1 and miR-150 regulate AKT3 expression at the transcription or translation level, through which AKT3 mediates the inflammatory responses (Figure 6).
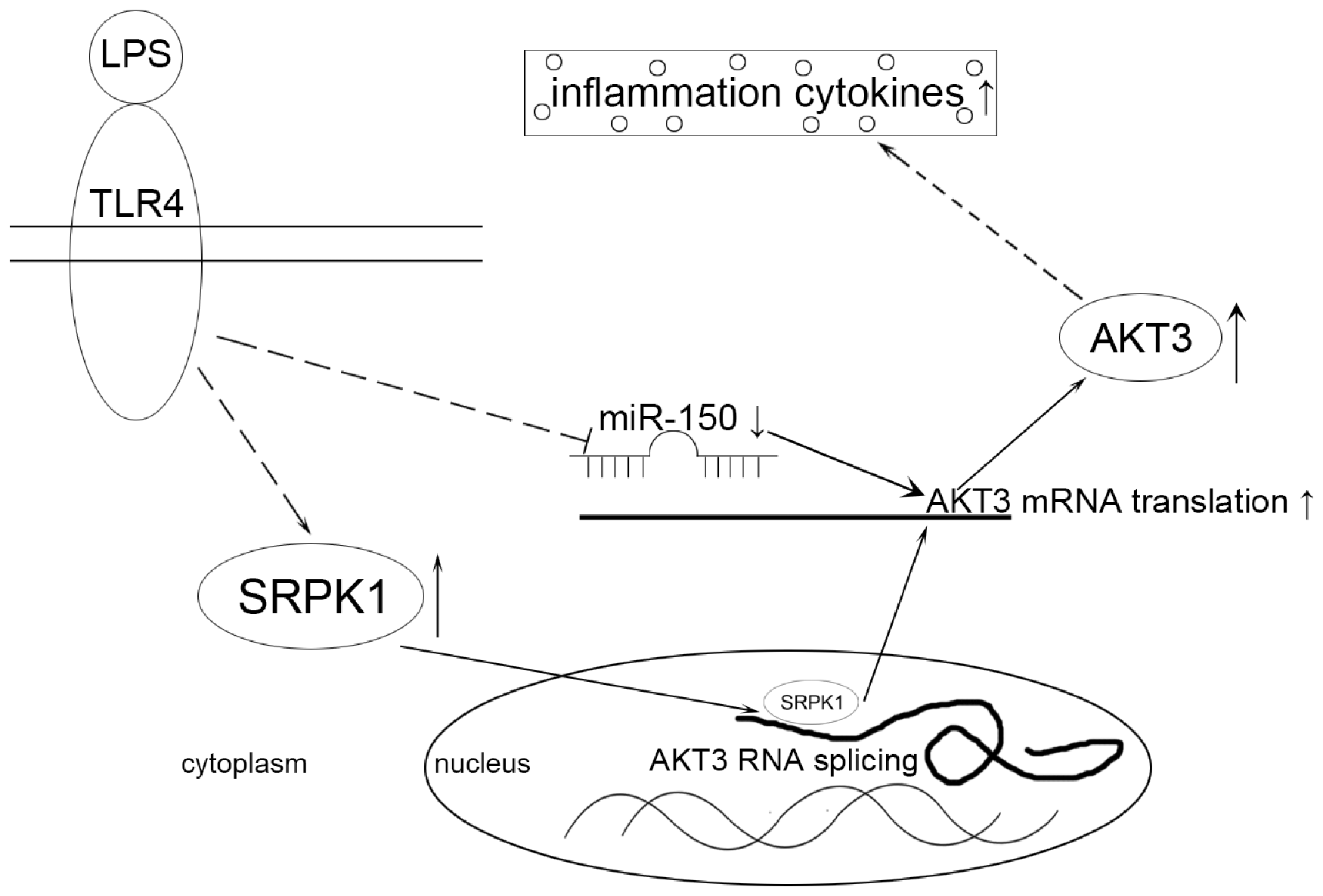
Schematic diagram illustrates the relationship of miR-150, SRPK1 and AKT3 in LPS-induced ALI. LPS up-regulates SRPK1 and down-regulates miR-150. Then SRPK1 was involved in the AKT3 pre-mRNA splicing, and down-regulation of miR-150 promotes AKT3 expression due to its inability to target its 3’-UTR of AKT3 mRNA, which further promotes the progression of inflammation.
In summary, our study identifies miR-150 as an important and novel regulator of LPS-induced and AKT3-mediated inflammation reactions through its cooperation with SRPK1 in ALI. Our results suggest that miR-150 or SRPK1 may serve as a novel therapeutic target for management of ALI.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Medical and Health Science and Technology Development Project of Shandong Province (2019WS453).
Compliance with ethical standards
All experimental animal procedures in this study conformed to the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee of Shandong Provincial Third Hospital.
