Abstract
Sox2 is an embryonal stem cell Ag essential for early embryonic development, tissue homeostasis and immune regulation. In the current study, one complete Sox2 cDNA sequence was cloned from freshwater bivalve Anodonta woodiana and named AwSox2. Histological changes of testis derived from Bisphenol A (BPA) treatment were analyzed by hematoxylin and eosin staining. Expressions of AwSox2 derived from BPA, LPS and polyinosinic:polycytidylic (Poly I:C) challenge were measured by quantitative real-time PCR. The full-length cDNA of AwSox2 contained an open reading frame of 927 nucleotides bearing the typical structural features of Sox2 family. Obvious degeneration, irregular arrangement of spermatids, and clotted dead and intertwined spermatids were observed in BPA-treated groups. Administration of BPA could result in a dose-dependent up-regulation of AwSox2 expression in the male gonadal tissue of A. woodiana. In addition, expression of AwSox2 was significantly induced by LPS and Poly I:C treatment in the hepatopancreas, gill and hemocytes, compared with that of control group. These results indicated that up-regulations of AwSOx2 are closely related to apoptosis of spermatogonial stem cells derived from BPA treatment as well as enhancement of immune defense against LPS and Poly I:C challenge in A. woodiana.
Introduction
The Sox transcription factors belong to the high-mobility group (HMG) superfamily, which typically bears a DNA-binding domain. Sox proteins are divided into different groups according to their HMG homology.1,2 Sox2, an embryonal stem cell Ag, is essential for early embryonic development, nervous system development, pluripotency of stem cell biology, tissue homeostasis and immune regulation in the vertebrate.3–6 Notably, Sox2 functions have gained great attention in different cancers. In pancreatic cancer cells, over-expression of Sox2 can result in an increase of cell proliferation via cyclin D3 induction. 7 Similarly, cellular proliferation of lung squamous cell is inhibited by Sox2 silencing that is related with up-regulation of tumor suppressor. 8 In glioblastoma tumor-initiating cells of immunodeficient mice, Sox2 silencing can cause a decrease of cell proliferation and loss of tumorigenicity. 9 In breast, prostate, and cervical cancers, up-regulation of Sox2 expression can result in a promoting role of cellular proliferation.10,11 In addition, Sox2 is involved in the evasion of apoptotic signals of prostate cancer, gastric cancer and non-small-cell lung cancer.12–14 Sox2 is regarded as a marker for invasion, migration, and metastasis of melanoma, colorectal cancer, glioma, gastric cancer, ovarian cancer and hepatocellular carcinoma.15–19 Therefore, Sox2 expression is closely associated with immune regulation and body defense in the vertebrate. 20
Comprehensive elucidation of the functions of the Sox family requires full characterization of Sox genes in different phyla. Mollusca is the second largest group behind arthropods in the animal kingdom, and is among the first bilaterians to appear in fossil records. Interestingly, a series of conserved genes are shared between mollusca and vertebrates, but not between arthropod and vertebrates, such as estrogen receptor, Sox, TNF, prostaglandin E synthase and others. 21 To date, only some Sox proteins have been identified and characterized in this group.22–24 In gastropods, SoxB and SoxC were detected in the cerebral and pleuropedal ganglia of abalone Haliotis asinina. 25 SoxB is involved in the formation of neural structures in the limpet Patella vulgate. 22 In the cephalopod Sepia officinalis, three members of Sox from group B and E were isolated, showing diverse expression profiles in early embryogenesis and vasculogenesis. 23 With regard to bivalves, three Sox members have been reported, including SoxH and SoxE of Pacific oyster Crassostrea gigas and SoxB2 of scallop Chlamys farreri. 24 However, a systematic characterization of the Sox2 gene family is very sparse in mollusca. To the best of our knowledge, the sequence information of Sox2 has mainly been identified in vertebrates, such as goldfish, zebrafish, Atlantic salmon, Oryzias latipes, Takifugu rubripes, large yellow croaker, mouse and human. 16
Freshwater bivalves, sedentary filter feeders, represent typical mollusks that inhabit rivers, ponds, lake and estuaries and coastal areas. The Chinese pond mussel, bivalve Anodonta woodiana, functions as a bio-indicator organism. 26 Earlier studies have revealed the ability of A. woodiana to accumulate trace elements and pesticides, 27 as well as its potential to detect genotoxicity. 28 In addition, these mussels have important ecosystem functions such as particle filtration and processing, nutrient release, and sediment mixing. 29 Environmental pollution and eutrophication have caused a great threat to the survival of bivalve A. woodiana. 30 Our previous study demonstrated that 2,4-dichlorophenol, 2,4,6-trichlorophenol and pentachlorophenol can impact on the survival of bivalve A. woodiana. 30 The extensive investigation of A. woodiana as a reliable indicator in eco-toxicological studies has contributed to elucidating its very well-developed immune system, which consists of different defense strategies, such as phagocytosis, antioxidant defenses and antimicrobial pepides. 27 These studies helped to examine the bivalve’s immune response at functional, cellular and molecular levels, therefore advanced the knowledge about the components of the immune system and their related mechanisms. Still, it is necessary to improve the understanding of the innate immune defense mechanism of the freshwater clam. In the current study, a complete sequence of Sox2 was cloned and named AwSox2, and its temporal expressions deriving from Bisphenol A (BPA), LPS and polyinosinic:polycytidylic (Poly I:C) exposure were determined by quantitative real-time PCR.
Materials and methods
Ethics statement
All methods of handling clams were conducted in accordance with the guidelines on the care and use of animals for scientific purposes set up by the Institutional Animal Care and Use Committee of Pingdingshan Medical College, Pingdingshan, China.
Materials
Clams A. woodiana (approximately 2 yr old, shell length, 13.5 ± 1.0 cm) were obtained from the Baihe River of Nanyang, Henan Province, China. BPA was purchased from Sigma-Aldrich (Sigma-Aldrich, St. Louis, MO) and stock solutions were prepared by dissolution in DMSO. Prior to the experiment, animals were maintained in a recirculation system containing filtered freshwater at 24°C for 2 wk in laboratory. After this period, animals were fed twice daily with Chlorella vulgaris. In the process of BPA, LPS and Poly I:C treatments, experiments were conducted in rectangular plastic boxes (120 cm × 60 cm; 30 cm height). Clams were cultured in 50 l artificial pond water containing 48 mg NaHCO3, 33 mg CaCl2·2H20, 60 mg MgSO4·7H20 and 0.5 mg KCl per 1 l deionized water, with a pH of 7.0. 31 In order to determine the tissue distribution of AwSox2, five clams derived from same tank were dissected, and several tissues including foot, gill, hepatopancreas, adductor muscle, heart, hemocytes and mantle were sampled prior to the treatment.
For sex determination of clams, hemolymph was obtained from mantle cavity by a 1 ml hypodermic syringe and used to produce singular slide smears. Slides were investigated by optical microscopy at a magnification of 10× in order to observe the presence of moving spermatozoa to determine the sex of the mussel. Mussels with spermatozoa were considered as male. According to previous investigations,30,32 240 male clams A. woodiana were randomly grouped into BPA-treated groups in which animals were treated with five test concentrations of BPA, and a control group with similar volume of DMSO. Concentration ranges for viability studies were 125 ng/l, 250 ng/l, 500 ng/l, 1000 ng/l and 2000 ng/l for BPA. Valves of clams were opened with a metal plate for max. 5 mm and fixed at this position by the snap pressure of the mussel’s valves. After sex determination, clams were selected to analyze the expressions of AwSox2. Gonads were pooled at 0, 7, 14, 21, 28 and 35 d, then immediately frozen in liquid nitrogen and stored at −80°C until used for quantitative real-time reverse transcriptase PCR (qRT-PCR). After d 35, the rest of the animals were sacrificed according to the regulations of Institutional Animal Care and Use Committee of Pingdingshan Medical College.
To determine the immune responses of AwSox2, LPS and Poly I:C (Sigma-Aldrich, St. Louis, MO) were selected as immune-stimulants in time-course experiments in accordance with previous studies. 32 LPS and Poly I:C were suspended in PBS. According to the above-mentioned culture conditions, 60 clams were treated with PBS in the control group. In total, 120 clams were randomly divided into LPS-treated and Poly I:C-treated groups, inoculated with 100 μl/clam of LPS (2 μg/ml) or 100 μl/clam of Poly I:C (2 μg/ml). The control group was inoculated with a similar volume PBS. Hepatopancreas and gill were dissected at 0, 6, 12, 24, 48 and 72 h from the challenge and the control groups. Hemolymph was extracted from the anterior adductor muscle using a 1 ml syringe and placed in Eppendorf tubes at corresponding times. The tissues were frozen immediately in liquid nitrogen and stored at −80°C.
Histological screening of hematoxylin and eosin (H&E) staining
After being removed from the bivalves, the male gonadal tissue was cut into 0.5 cm2 pieces, fixed in formaldehyde, and then embedded in paraffin. Serial sections were deparaffinized routinely and processed with H&E staining.
Total RNA isolation and reverse transcription
Total RNA of female gonadal tissue, male gonadal tissue, foot, mantle, adductor muscle, heart, hepatopancreas, hemocytes and gill was extracted using TRIzol (Invitrogen Life Technologies, USA) according to the manufacturer’s protocol. Quality of RNA was monitored by 1.2% agarose gel electrophoresis and those with complete rRNA bands were selected to produce cDNA. First-strand cDNA was synthesized using M-MLV First-Strand cDNA synthesis Kit (Takara, China) according to the manufacturer’s instructions and used as the template for PCR reaction.
Cloning of AwSox2 cDNA
The Sox2 fragment was amplified using two degenerate primers Sox2-1 and Sox2-2 (Table 1), designed according to conserved domains of Sox2 of other species including bivalves, gastropods and vertebrates. The PCR products were subcloned into the pMDT-19 (Takara, China), sequenced from both directions (Invitrogen Life Technologies, China) and identified a Sox2 partial cDNA sequence. Highly stringent primers (Table 1) designed from the partial cDNA sequences were used to characterize the 5′ and 3′ region of the AwSox2 cDNA by rapid amplification of cDNA ends (RACE) approaches (Takara, China) according to the manufacturer’s protocol. 5′ Raceouter primer and AwSox2-5-1 (Table 1) were used for the first-round PCR of AwSox2 5' RACE, 3′ Raceouter primer and AwSox2-3-1 (Table 1) for 3′ RACE. Subsequently, 5′ nested PCR was performed by the 5′ first-round PCR product used as template, 5′ Raceinner primer included in the kit and AwSox2-5-2, 3′ nested PCR using 3′ first-round PCR products, 3′ Raceinner primer and AwSox2-3-2 (Table 1). The 5′ RACE and 3′ RACE nest PCR products were cloned, and five clones were sequenced using the method described above.
Description of the primers used in this study. Sox2-1 and Sox2-2 are degenerate primers and used to isolate partial cDNA of AwSox2. 5′ RACE Outerprimer, 5′ RACE Innerprimer, Sox2-5-1 and Sox2-5-2 are used to characterize the 5′ RACE of the AwSox2 in the nest PCR, 3′ RACE Outerprimer, 3′ RACE Innerprimer, Sox2-3-1 and Sox2-3-2 for 3′ RACE of AwSox2. β-Actin primers are selected to isolate internal standard. Sox2-F and Sox2-R primer is used to isolate AwSox2 in real-time PCR, respectively.

Sequence and phylogenetic analysis
The AwSox2 sequence was analyzed and compared using the BLAST program with a GenBank database search (www. ncbi.nlm.nih.gov/blast). The signal peptide was predicted by signal program (http://www.cbs.dtu.dk/services/SignalP). Prediction of protein domain was carried out with the Simple Modular Architecture Research Tool (http://smart.embl-heidelberg.de/). The SECIS element and stem loop structure was predicted by NetPhos 3.0 (http://www.cbs.dtu.dk/services/NetPhos/). Multiple sequence alignments of the AwSox2 gene were performed using the DANMEN analysis program. Prediction of 3-D structure was fulfilled by Swiss-model (http://swissmodel.expasy.org/). Phylogenetic trees constructed from the alignment were generated by the neighbor-joining method using MEGA5.0 software. Reliability of trees obtained was assessed by bootstrapping, using 1000 bootstrap replications.
Quantification of AwSox2 expression by real-time PCR
To determine the mRNA levels of AwSox2 derived from hepatopancreas and other tissues, real-time quantitative PCR was performed with SYBR Premix Ex TaqTM (TaKaRa, China) following the manufacturer’s instructions. Firstly, AwSox2 primers and β-actin primers (Table 1) were designed based on isolated sequences of A. woodiana, respectively, used to isolate target genes in common PCR methods; only one band was detected in the PCR production by agarose gel electrophoresis. PCR products were sequenced and identified as the partial sequence of target genes. Subsequently, real-time PCR was performed using an ABI 7500 Real-Time Detection System (Applied Biosystems, USA) under the following conditions: initial denaturation at 95°C for 30 s and amplifying for 40 cycles (95°C, 5 s; 60°C, 34 s). Based on the constructed standard curve, expression levels of AwSox2 were calculated by 2–ΔΔCT. All data were given in terms of relative mRNA expression as means ± SE. One-way ANOVA was performed on all data and P < 0.05 was considered statistically significant.
Results
Identification of AwSox2
In order to isolate Sox2 from A. woodiana, degenerated primers against the conserved regions of Sox2 family genes were designed. Initially, the partial cDNA sequence of AwSox2 was PCR-amplified from cDNA of the male gonadal tissue of A. woodiana. After BLAST analysis, the sequence data of this fragment exhibited a maximal homology to that of Sox2 sequences derived from bivalve, gastropoda and other vertebrates. The rest of the 5′- and 3′-end sequences were generated by means of RACE strategies and verified by sequencing after the sequence analysis. Finally, a full-length sequence of AwSox2 was cloned and submitted to the National Center for Biotechnology Information’s public database (GenBank accession number, MF805736). AwSox2 cDNA was 1666 bp long, consisting of a 201 bp 5′-flanking region, 538 bp 3′-untranslated region (UTR) that contained a poly(A) tail and an open reading frame (ORF) of 927 nucleotides with an ATG start codon and TAA stop codon. A polyadenylation signal (
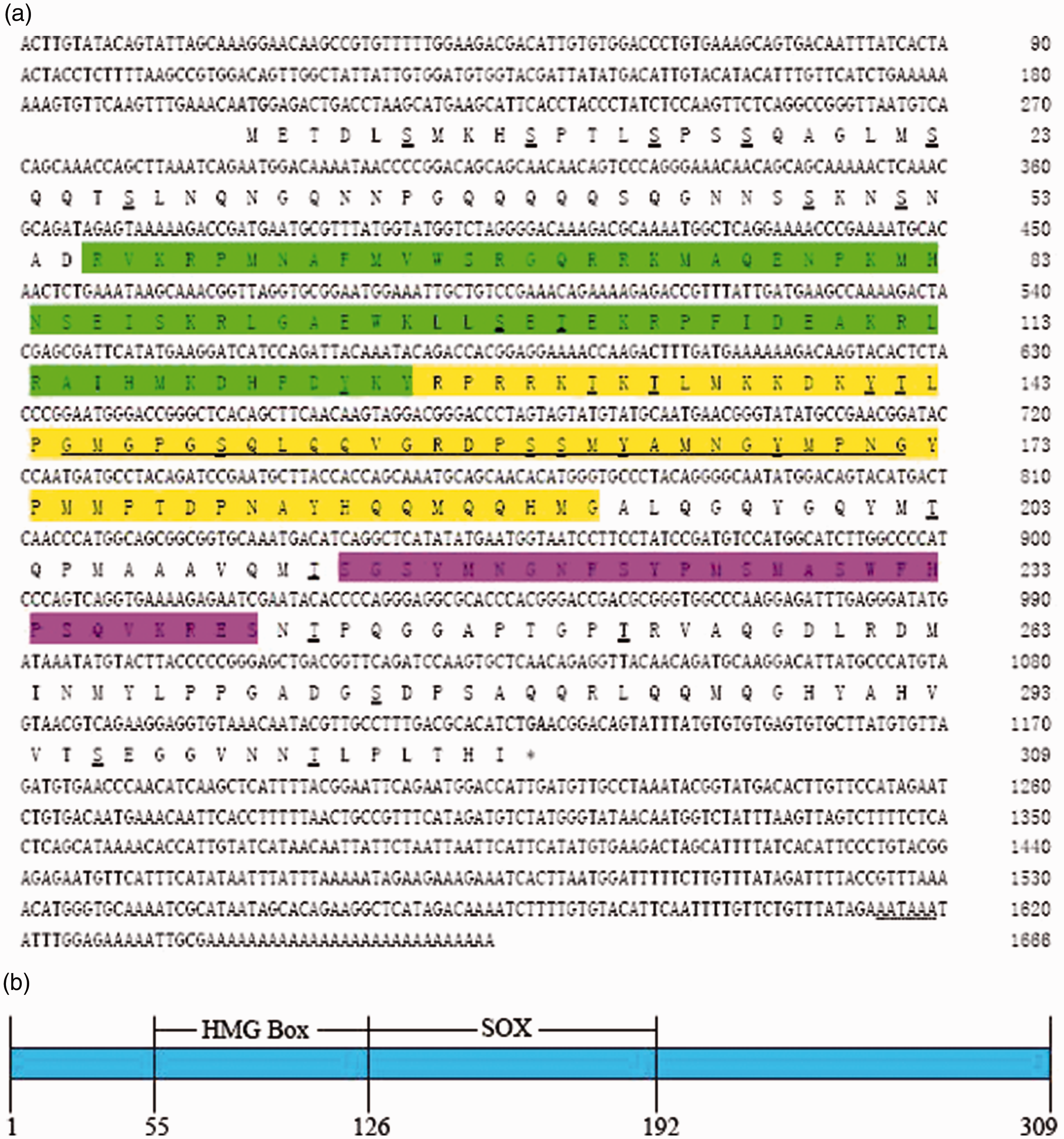
Nucleotide and deduced Aa sequences of AwSox2. The start and stop codons are in bold face. Putative polyadenylation signal “
The ORF of AwSox2 encoded a protein of 309 Aa residues with a predicted molecular mass of 34.51 kDa and an isoelectric point of 10.29. From the SMART program analysis, a well-characterized protein domain, HMG box involved in DNA binding, and the SOX transcription factor involved in up-regulation of nestin (neural promoter), were identified. Two important putative regions, serine-rich and glycine-rich motifs, were also observed in the AwSox2 sequence (Figure 1). In addition, no signal sequence was identified in AwSox2 using the SignaIP 4.1 server. Analysis of potential phosphorylation sites with NetPhos 2.0 showed that AwSox2 contained 17 serine phosphorylation sites, eight threonine phosphorylation sites, and four tyrosine phosphorylation sites (Figure 1).
To validate the similarity, sequence alignments of two conserved domains of predicted Sox2 proteins showed that the HMG box shared a high identity with vertebrate and bivalve, and the SOX transcription factor only with bivalve (Figure 2). The conserved motifs of KKDK and LPG in the SOX transcription factor domain and RPMNAFMVW in HMG domain were also identified in AwSox2 (Figure 2).

Multiple alignments of AwSox2 with other Sox2. Black underlining indicates the Aa stretch of HMGB. The highly conserved motifs present in Sox2 protein are indicated by an asterisk. Crassostrea gigas (XP_011455662.1), Azumapecten farreri (AIT51849.1), Xenopus laevis (XP_018121173.1), Mus musculus (AAH57574.1), Danio rerio (NP_998283.1), Homo sapiens (NP_003097.1).
Analysis of secondary and 3D structures of AwSox2
Two conserved domains of AwSox2 secondary structures, located in the 5-160 and 206-283 regions, showed a high similarity with those of human Sox2 secondary structures (Figure 3a). Seven α-helices were detected in the first domain, three α-helices in the second one (Figure 3a). The 3D structure of AwSox2 showed a high degree of similarity with Sox2 of other species (Figure 3b).
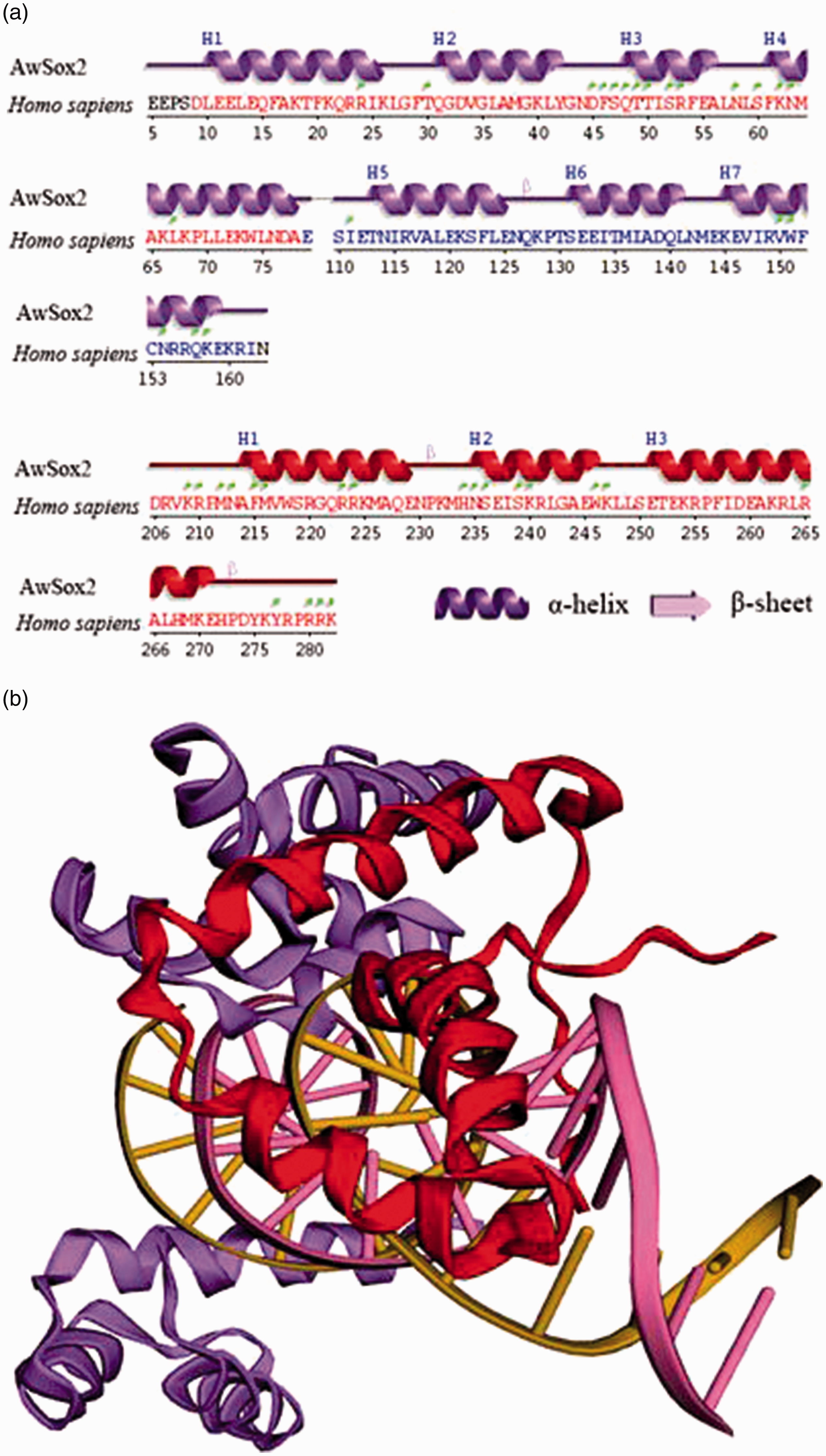
Predicted secondary and 3D structures of AwSox2 deduced Aa. (a) The secondary structure of AwSox2. (b) The 3D structure of AwSox2.
Phylogenetic relationship and identity analysis
BLAST analysis revealed that AwSox2 matched closely with other Sox2 family members. The overall deduced Aa sequence of AwSox2 exhibited 78% identity to bivalve Hyriopsis schlegelii, 55% to bivalve Crassostrea gigas, 53% to bivalve Azumapecten farreri, 39% to gastropoda Aplysia californica, 62% to Oligochaeta Octopus bimaculoides, 45% to fish Danio rerio, 43% to frog Xenopus tropicalis, 45% to Mus musculus and 44% to Homo sapiens (Figure 4).
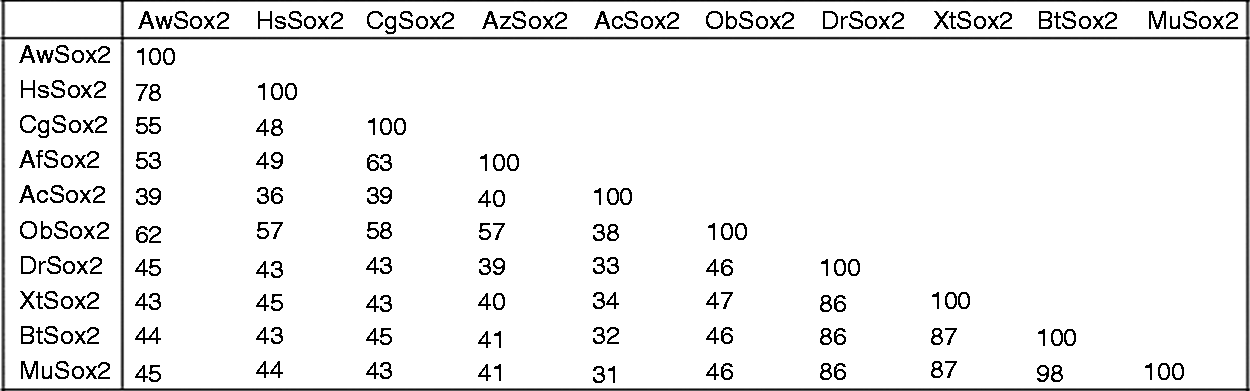
Percentage of identity for pairwise alignments of AwSox2 protein sequences. HsSox2: Hyriopsis schlegelii (AEK27011.1), CgSox2: Crassostrea gigas (XP_011455662.1), AfSox2: Azumapecten farreri (AIT51849.1), AcSox2: Aplysia californica (XP_005093681.1), ObSox2: Octopus bimaculoides (XP_014780771.1), DrSox2: Danio rerio (NP_998283.1), XtSox2: Xenopus tropicalis (NP_998869.1), MuSox2: Mus musculus (AAH57574.1).
To evaluate the evolutionary relationships, a phylogenetic tree was constructed with deduced full-length Sox2 protein of different species available in the public database by the neighbor-joining method. The members of Sox2 family showed a highest degree of Aa identities forming two distinct subgroups supported by high bootstrapping values, the first of which consisted of mammal, bird, amphibian and fish, and the second one of oligochaeta, bivalve, gastropoda and cephalopoda. AwSox2 was belonged to the second cluster in which it was most close to bivalve, then to gastropoda, last to oligochaeta and cephalopoda (Figure 5). With regard of evidenced sequences of bivalves, AwSox2 showed a high relationship of evolution with freshwater clam (Figure 5).
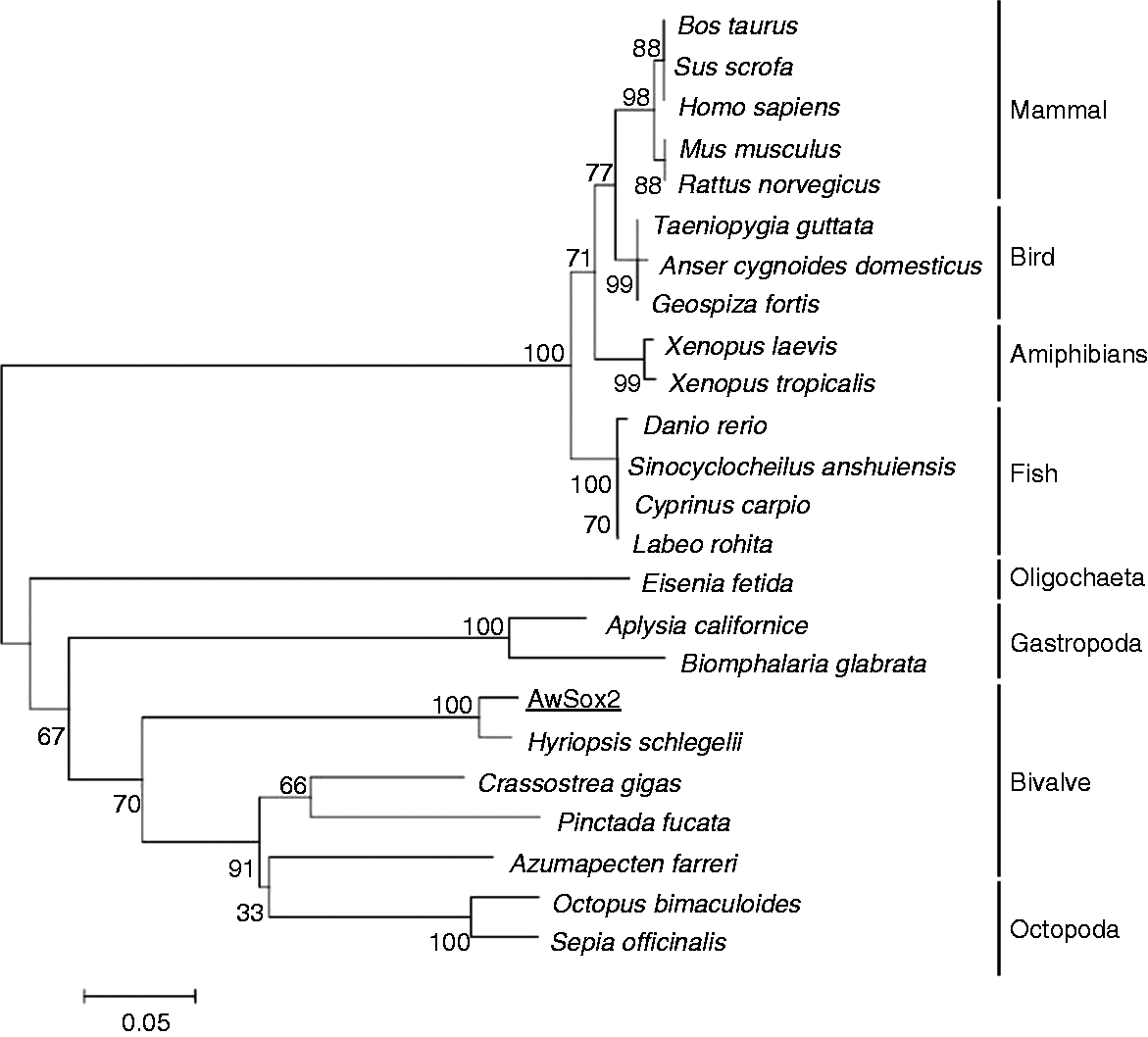
Phylogenetic relationship of AwSox2 to Sox2 from other organisms. Hyriopsis schlegelii (AEK27011.1), Octopus bimaculoides (XP_014780771.1), Sepia officinalis (AGL08098.1), Crassostrea gigas (XP_011455662.1), Pinctada fucata (AGS18764.1), Azumapecten farreri (AIT51849.1), Eisenia fetida (ALC04236.1), Aplysia californica (XP_005093681.1), Homo sapiens (NP_003097.1), Bos taurus (NP_001098933.1), Sus scrofa (ACA58281.1), Cuculus canorus (KFO82025.1), Xenopus tropicalis (NP_998869.1), Danio rerio (NP_998283.1), Mus musculus (AAH57574.1), Rattus norvegicus (NP_001102651.1), Xenopus laevis (XP_018121173.1), Biomphalaria glabrata (XP_013075224.1), Geospiza fortis (XP_005419064.1), Taeniopygia guttata (XP_002196639.1), Anser cygnoides (XP_013030453.1), Sinocyclocheilus anshuiensis (XP_016354450.1), Labeo rohita (AJC01623.1), Cyprinus carpio (XP_018921043.1).
Tissue distributions of AwSox2
The constitutive expression level of AwSox2 was examined in different tissues including female gonadal tissue, male gonadal tissue, foot, mantle, adductor muscle, heart, hepatopancreas, hemocytes and gill (Figure 6). The highest expression of AwSox2 was found in the male gonadal tissue, a moderate level in foot, gill and mantle, a lower level in hepatopancreas, hemocytes, and adductor muscle, and the lowest level in the female gonadal tissue (Figure 6). Compared with that of ovary, a significant increase of AwSox2 expression was observed in the hemocytes, adductor muscle, foot, gill, mantle and male gonadal tissue (P < 0.05) (Figure 6).
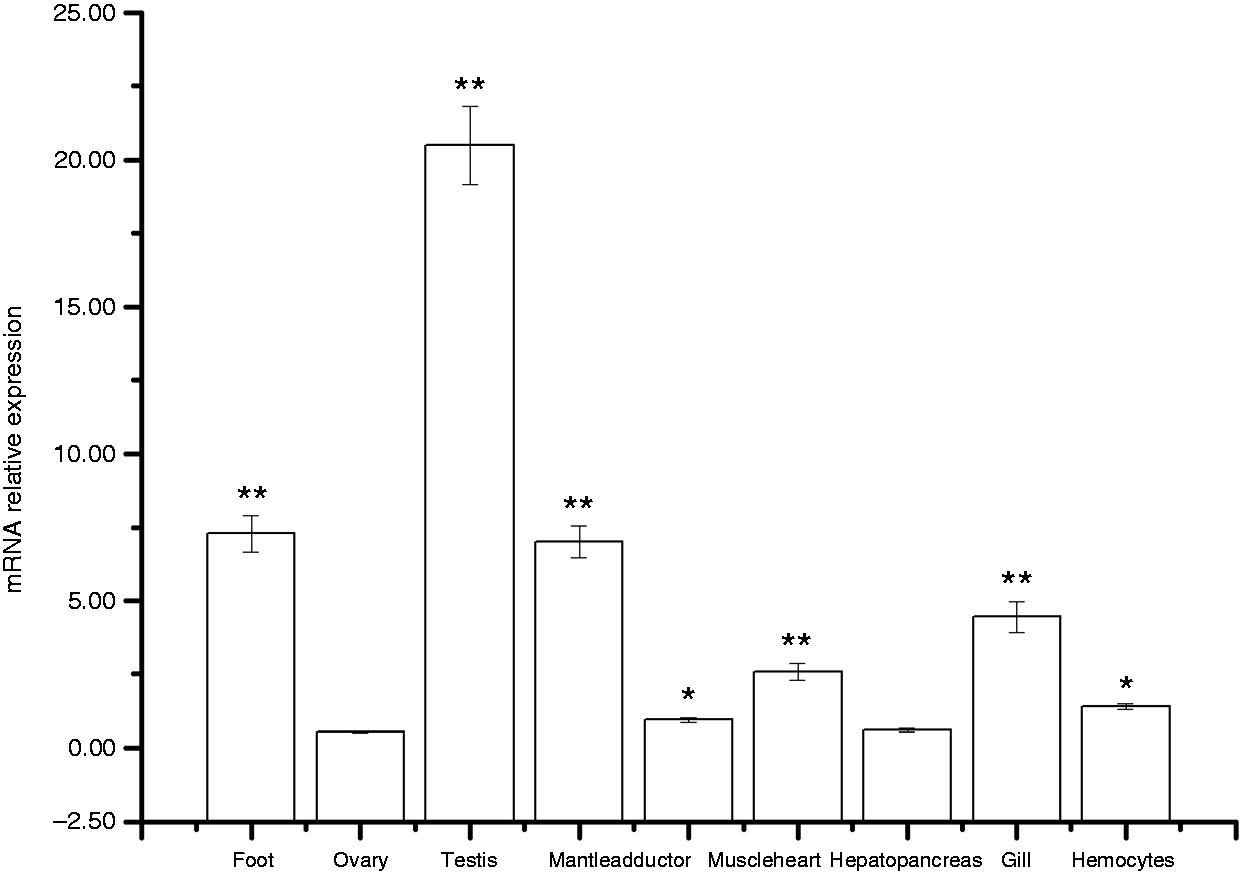
Real-time PCR analysis of AwSox2 transcript from different tissues. n = 5 replicates. **P < 0.05, *P < 0.01 vs. the result of female gonadal tissue.
Male gonadal tissue histology in response to BPA
In male gonadal tissue, normal histological structures were observed in control group characterized by normal spermatogenic series, and formation of spermatids. Treatment with different concentrations of BPA resulted in different levels of destruction of male gonadal tissue (Figure 7). Sections of male gonadal tissue showed remarkable degenerative changes, disorganization and necrosis of the histological structure of seminiferous tubules in the BPA-treated groups (500, 1000 and 2000 ng/l). In addition, obvious degeneration, irregular arrangement of spermatids, clotted dead and intertwined spermatids were observed (Figure 7). No obvious lesions or dead clams occurred in the experiment.
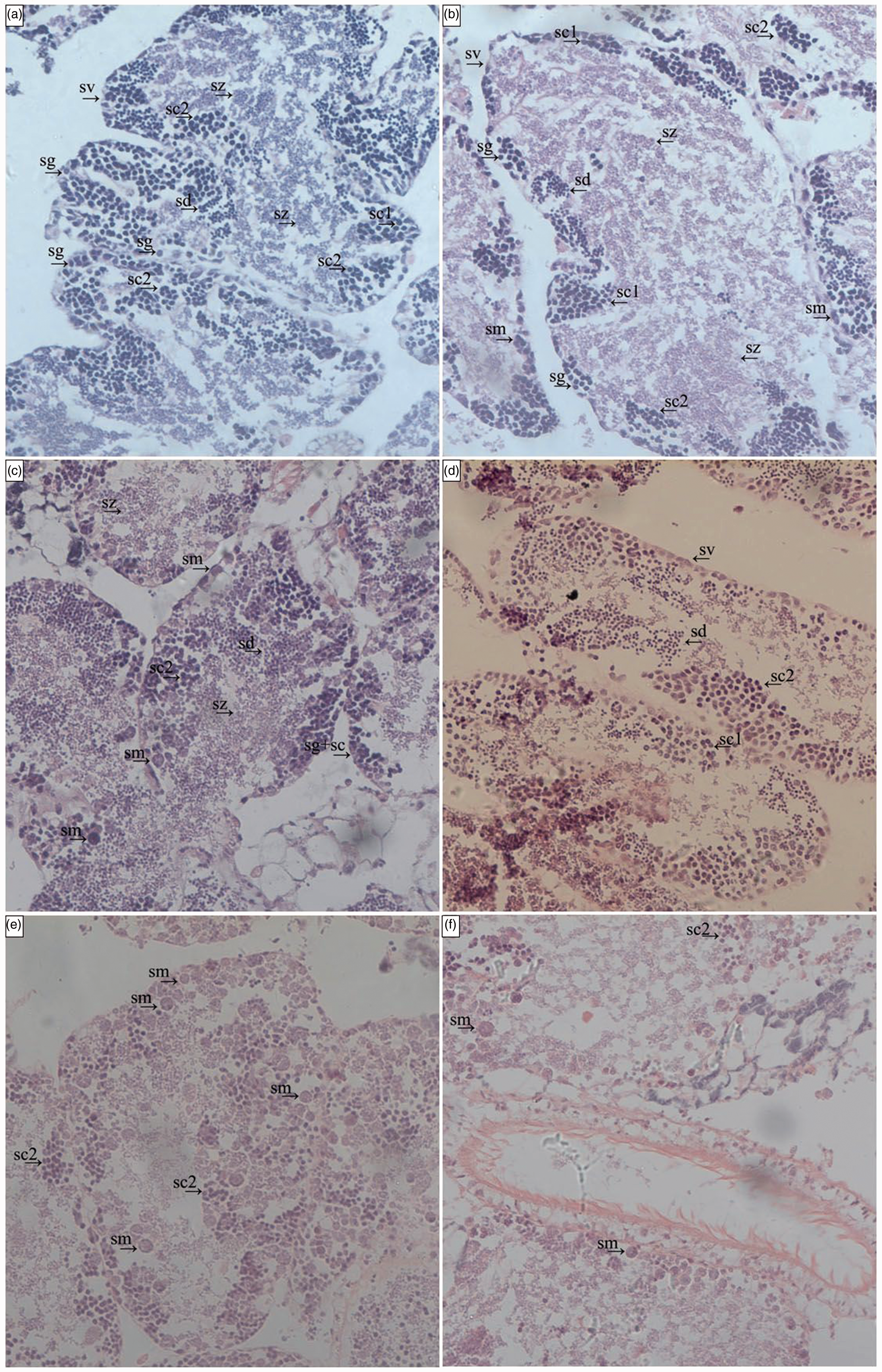
Effects of BPA on the male gonadal tissue of A. woodiana by light microscope investigation (200×). (a) Control group, the male gonadal tissue of control clams showed normal structure. (b) Treated with 125 ng/l BPA group. (c) Treated with 250 ng/l BPA group. (d) Treated with 500 ng/l-BPA group. (e) Treated with 1000 ng/l BPA group. (f) Treated with 2000 ng/l-BPA group. Gn: gonoduct; sd: spermatids; sg: spermatogonia; sc1: primary spermatocytes; sc2: secondary spermatocytes; sm: spermatid morulae; sv: seminal follicle; sz: spermatozoa.
Temporal expressions of AwSox2 in the male gonadal tissue exposed to BPA
In the male gonadal tissue, temporal expressions of AwSox2 were significantly affected by BPA treatment, as identified by using RT-qPCR with β-actin as internal control. Up-regulation of AwSox2 expression in a time- and dose-dependent pattern was observed in the BPA-treated groups (125, 250, 500 and 1000 ng/l) (Figure 8). Compared with that of the control group, the significant up-regulation of AwSox2 expression was only observed at d 35 in the 125 ng/l BPA-treated group, and at d 21, 28 and 35 in the 250 ng/l BPA-treated group (Figure 8). Meanwhile, expression of AwSox2 increased more than 1.59 times (P < 0.01) in 500 ng/l BPA-treated group in contrast to that of the control group (2.22 times, P < 0.01) in the 1000 ng/l BPA-treated group, and 3.09 times (P < 0.01) in the 2000 ng/l BPA-treated group (Figure 8).
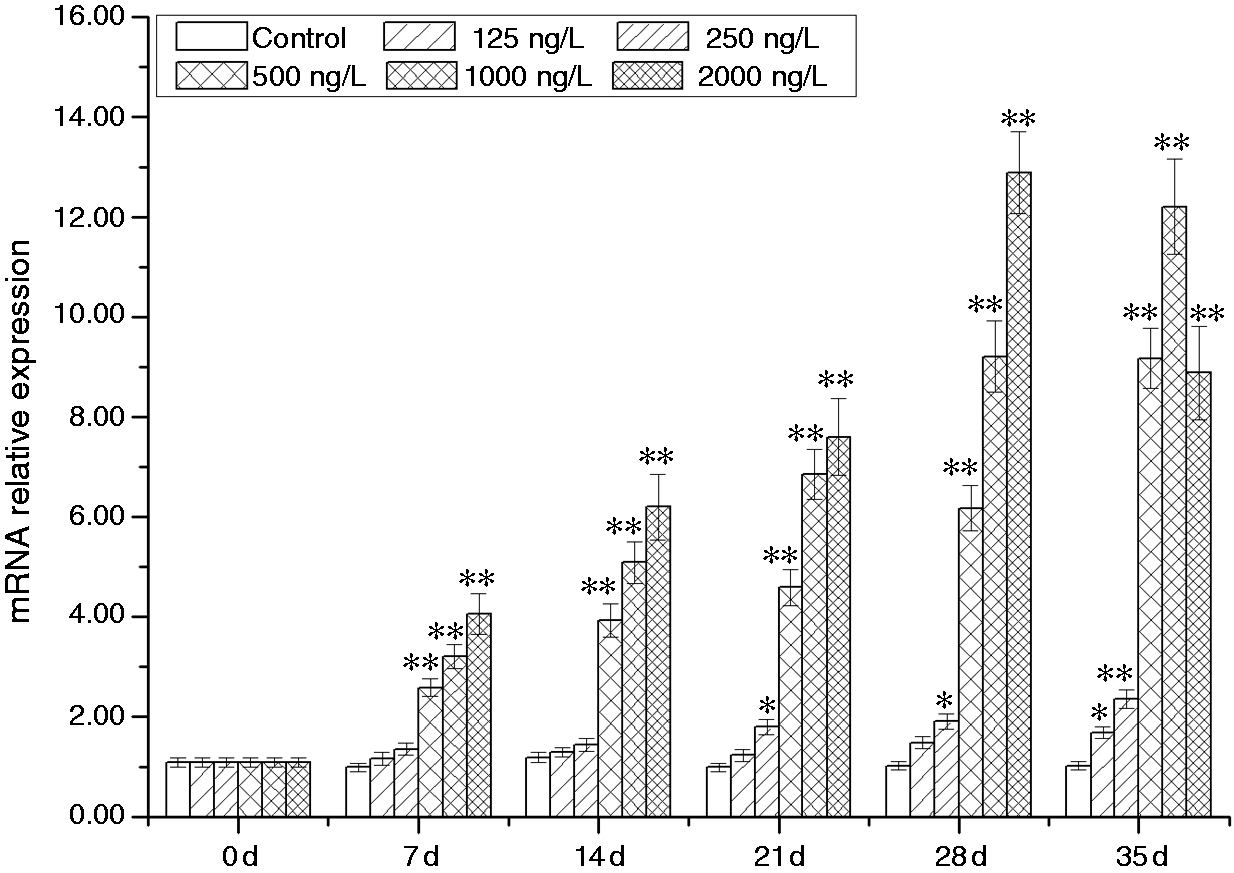
Temporal expressions of AwSox2 in male gonadal tissue after BPA challenge as measured by quantitative real-time RT-PCR. Bars represent means ± SE; n = 5/each group/each time point. **P < 0.05, *P < 0.01 vs. control group at the same time.
Temporal expressions of AwSox2 in hepatopancreas exposed to LPS and Poly I:C
In the hepatopancreas, expression of AwSox2 was significantly induced by LPS and Poly I:C treatment. A time-dependent matter of up-regulation of AwSox2 expression was observed during the whole experiment. Compared with that of control group, AwSox2 mRNA level increased more than 61.22% (P < 0.05) in the LPS-treated group from 12 to 72 h (Figure 9a). In the Poly I:C-treated group, AwSox2 mRNA level increased more than 86.59% (P < 0.05) in contrast to that of control group from 12 to 72 h (Figure 9b).
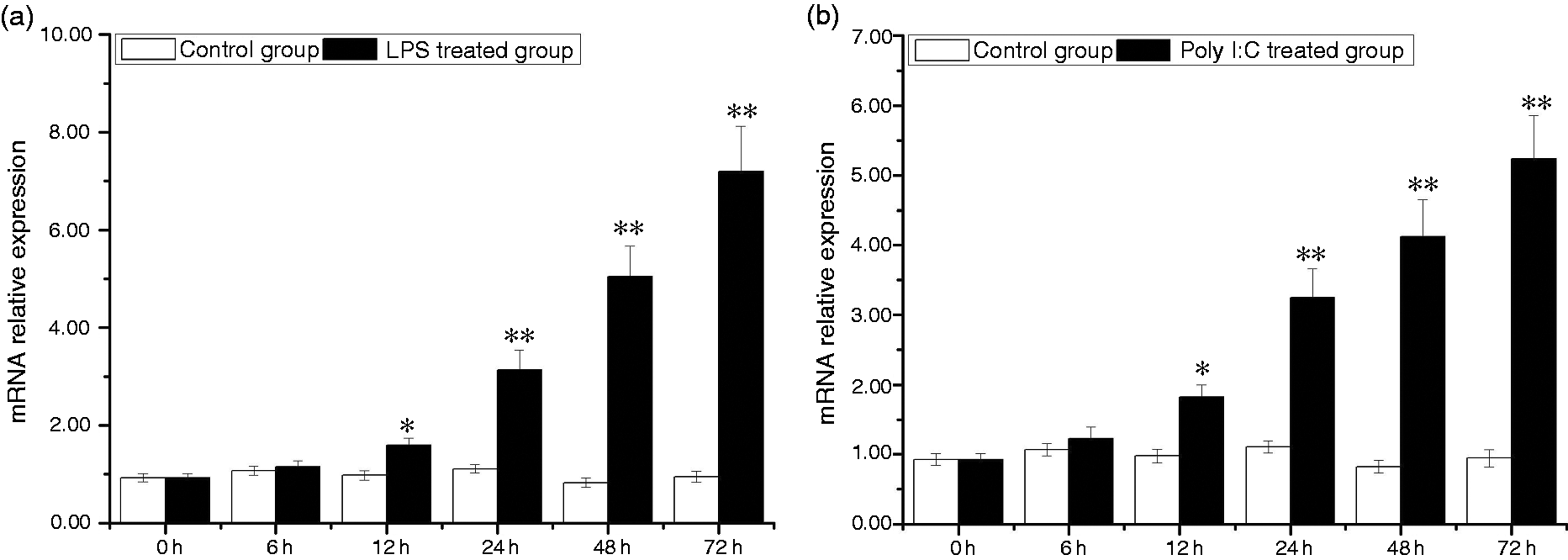
Temporal expressions of AwSox2 in hepatopancreas after LPS and Poly I:C challenge as measured by quantitative real-time RT-PCR. (a) The expression of AwSox2 exposed to LPS treatment. (b) The expression of AwSox2 exposed to Poly I:C treatment. Bars represent means ± SE; n = 5/each group/each time point. **P < 0.05, *P < 0.01 vs. control group at the same time.
Temporal expressions of AwSox2 in gill exposed to LPS and Poly I:C
In the gill, expression of AwSox2 was significantly up-regulated by LPS and Poly I:C treatments. Compared with that of the control group, AwSox2 mRNA level increased more than 77.77% (P < 0.05) in the LPS-treated group from 6 to 72 h (Figure 10a). In the Poly I:C-treated group, AwSox2 mRNA level increased more than 1.27 times (P < 0.01) compared with that of the control group from 6 to 48 h (Figure 10b).
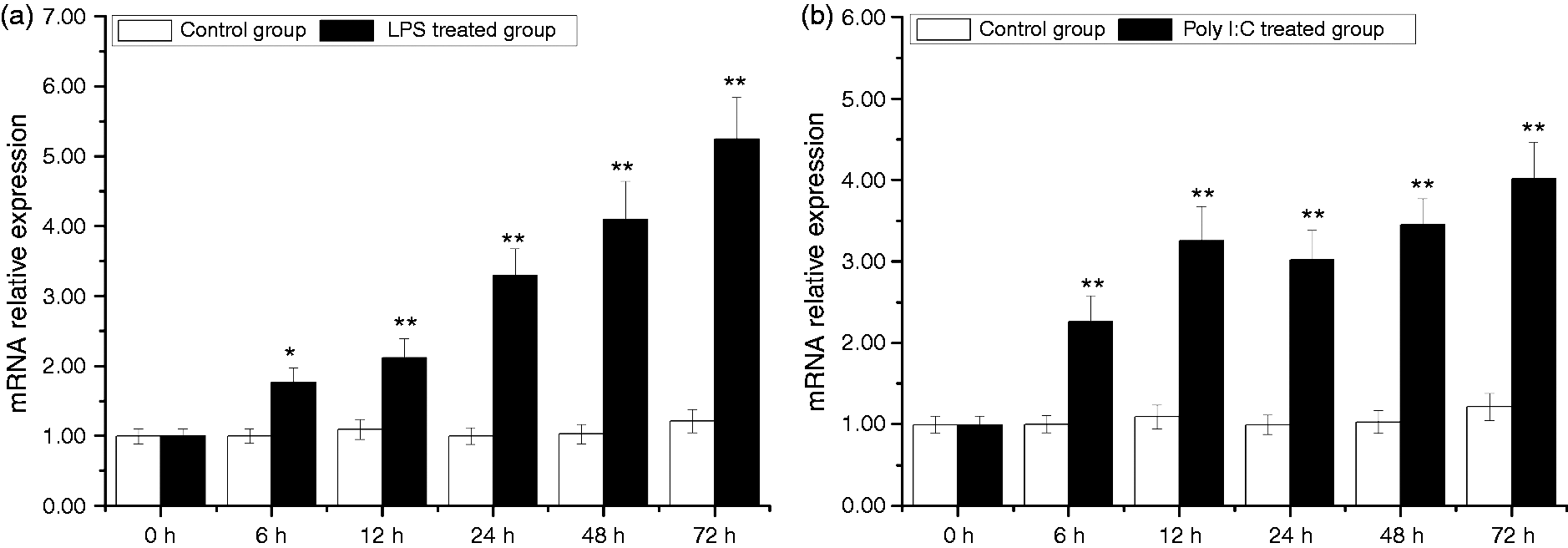
Temporal expressions of AwSox2 in gill after LPS and Poly I:C challenge as measured by quantitative real-time RT-PCR. (a) The expression of AwSox2 exposed to LPS treatment. (b) The expression of AwSox2 treatment exposed to Poly I:C treatment. Bars represent means ± SE; n = 5/each group/each time point. **P < 0.05, *P < 0.01 vs. control group at the same time.
Temporal expressions of AwSox2 in hemocytes exposed to LPS and Poly I:C
In the hemocytes, expression of AwSox2 was up-regulated by LPS and Poly I:C challenges. Compared with that of the control group, AwSox2 mRNA level increased more than 65.55% (P < 0.05) in the LPS-treated group from 12 to 72 h (Figure 11a). In the Poly I:C-treated group, AwSox2 mRNA level increased more than 84.94% times (P < 0.05) compared with that of the control group from 24 to 72 h, respectively (Figure 11b).
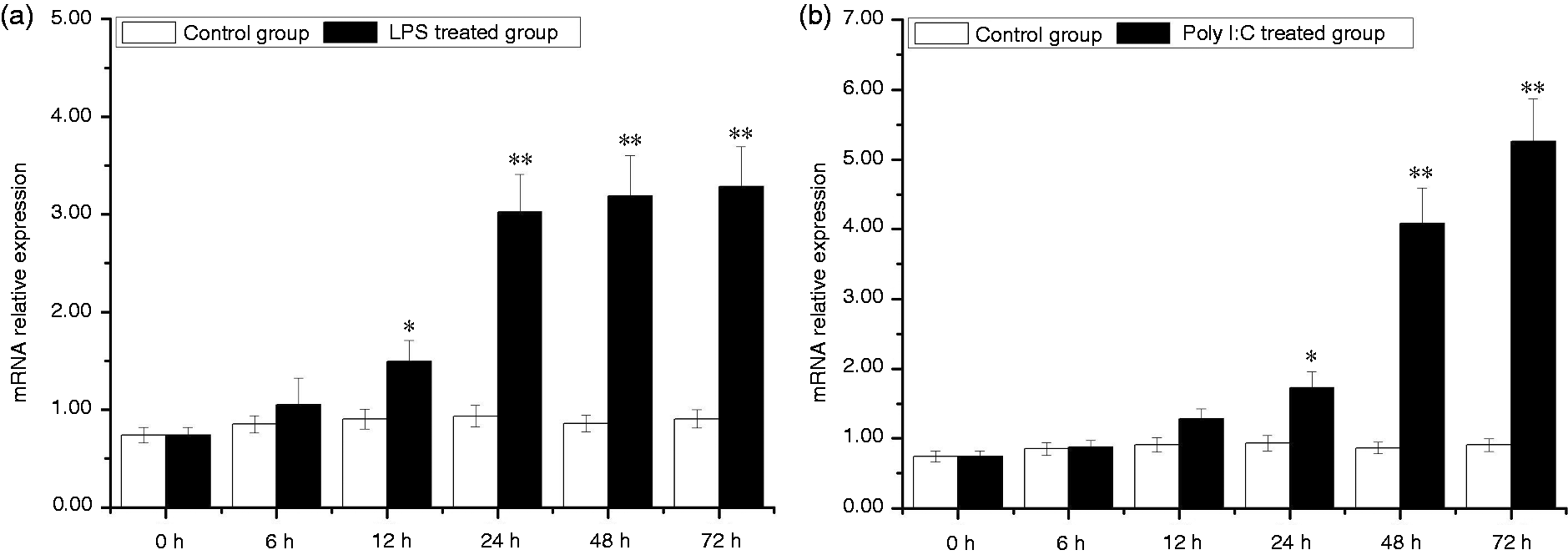
Temporal expressions of AwSox2 in hemocytes after LPS and Poly I:C challenge as measured by quantitative real-time RT-PCR. (a) The expression of AwSox2 exposed to LPS treatment. (b) The expression of AwSox2 exposed to Poly I:C treatment. Bars represent means ± SE; n = 5/each group/each time point. **P < 0.05, *P < 0.01 vs. control group at the same time.
Discussion
In the present study, signatures of HMG box and SOX transcription factor were observed in the deduced protein sequence of AwSox2 which possessed a typical characteristic domain of the Sox2 family. The deduced Aa of AwSox2 HMG showed a high similarity to that of Sox2 derived from mammalian and fish. With that, it is suggested that the AwSox2 likely functions as an important player in the male gonadal tissue of A. woodiana. Alignment of the AwSox2 protein with other vertebrate species indicated that the HMG box is highly conserved across all taxa, in agreement with earlier reports.9,16 Besides the conserved HMG box, the rest of AwSox2 was found to be variable with respect with that of counterparts of other species. These findings assumed that the HMG box domain plays an important role in the recognition of specific target DNA elements, whereas variable regions are likely to play phosphorylation-mediated regulatory roles in various stages of germ cell development.16,19,33
Interestingly, phylogenetic analysis revealed that Sox2 in bivalves formed a distinct hierarchy according to their habitats (marine, brackish and freshwater). The AwSox2 showed the most close relationship to Hyriopsis schlegelii counterpart, which is consistent with the fact that both A. woodiana and H. schlegelii belong to the Unionidae family of freshwater mussels. Meanwhile, it has been shown that evolution of bivalve Sox2 formed different branches with respect of marine, brackish and freshwater. Notably, it appears that the evolutionarily conserved bivalve and gastropoda HMG box domains evolved from a common ancestor. HMG of mollusks showed a higher degree of similarity/identity with the mammalian counterpart, indicating its conserved structural and functional similarity between all taxa.
AwSox2 mRNA was abundantly expressed in the male gonadal tissue that is associated with the development of male gonadal tissue and spermatogenesis. In the bivalve H. schlegelii, the Sox2 mRNA is ubiquitously expressed in all tissues and its expression level in the male gonadal tissue is higher than that in the female gonadal tissue. 34 In addition, the Sox2 expression shows an increasing trend with the testis development stage, which can be divided into three stages: initiation of testis formation (4–11 mo), a stable growth phase (12–24 mo), and a reproductive cell development phase (25–36 mo), indicating that Sox2 functions as important player in the development of male gonadal tissue. 34 In the Zhikong scallop Chlamys farreri, Sox2 mRNA is located in spermatogonia, spermatocytes, spermatids and spermaozoa. 35 Once knockdown of Sox2 expression occurs, spermatogenic failure is observed in the male gonadal tissue characterized by an array of reduced germ cells and a number of apoptosed spermatocytes, suggesting that Sox2 plays an important role in the development of male gonadal tissue and spermatogenesis. 35 In Labeo rohita, the presence of strong expression of Sox2 in spermatogonial stem cells is most likely to be linked with germ cell development and stem cells maintenance of fish, as evidenced earlier in case of human embryonic stem cells. 36 In fish, Sox2 is considered as a biomarker for undifferentiated spermatogonial cells. The absence in the female gonadal tissue suggested that Sox2 transcript is likely not maternally inherited, which is in agreement with previous studies. 37
In addition, a relative higher expression of AwSox2 was observed in foot, mantle and gill, which revealed this expression profile to be likely related with the proliferation of neural stem cells as well as adaption. The foot not only plays an important role in movement of bivalves, but also secretes byssus, which consists of fiber and adhesive coating proteins that play an important role against environmental stresses arising from changes in the salinity, temperature, pollution, pathogen concentration or other factors in water. 38 The shell is formed by three elements secreted by the mantle. It is known that mineralization proteins secreted by mantle tissues mediate the formation of mollusk shell and pearl with the accumulation of ions and micro molecules. 39 Gill is located in the ventilated mantle cavity and direct interacts with the outer environment. Thus, the gill and hepatopancreas are also frequent target organs of environmental conditions.29,30 As a result, foot, mantle and gill are more sensitive organs to external environment changes such as temperature, pH, stress and water flow. Meanwhile, various signals derived from foot, mantle and gill are constantly transmitted into the brain and give a response to environmental changes in order to sustain homeostasis of the body. 40 Like the skin of other organisms, various neural cells are located in these organs. It is assumed that the higher expression of AwSox2 in these tissues is not only associated with regeneration of neural cells, but also with environmental adaption. Generally, bivalves lack lymphocyte-mediated adaptive immunity, and they only possess innate immunity, which is a common character of both vertebrates and invertebrates. 38 The cellular immune reaction including phagocytosis and encapsulation is performed by circulating hemocytes, with subsequent pathogen destruction via enzyme activity and release of oxygen metabolites. 38 Hepatopancreas is the central metabolic organ and plays an important role in detoxification/degradation of xenobiotics and protection from oxidative stress. 30 This organ is closely mediated with the immune response, such as synthesizing a variety of cytokines, and identifying and removing pathogens by phagocytosis. Therefore, elucidation of the role of AwSox2 in hemocytes and hepatopancreas contributes to understanding the immune system of bivalves.
Testicular sections showed remarkable degenerative changes: disorganization, necrosis, apoptosis of the histological structure of seminiferous tubules, and apoptosis and irregular arrangement of spermatids occurred in the BPA-treated groups (500, 1000 and 2000 μg/l), suggesting that administration of BPA strongly influenced the differentiation of sperm cells. In mammalian testis, spermatogonial stem cells (SSCs) are the germline stem cells located along the basement membrane of seminiferous tubules.41,42 SSCs have a strong ability of self-renewing and differentiation in the testicular microenvironment.41,42 In response to differentiation cues, they can be differentiated into one specialized cell lineage, i.e. spermatozoa. Recent studies have shown that these unipotent SSCs can be re-programmed into multipotent SSCs under defined culture conditions.41,42 Several studies have reported that the re-programming of SSCs to multipotent SSCs can be influenced by a few factors. SSCs can self-renew or differentiate into spermatogonial progenitors, which undergo meiosis and eventually differentiate to sperm. 5 The male germinal lineage is considered unipotent because its fate is to produce sperm. However, this lineage appears to exhibit a singular plasticity compared with somatic lineages in its potential to spontaneously re-program in vitro into a pluripotent state. 43 In the current study, the quantity of sperm cells was obviously decreased compared with that of the control group, indicating that administration of BPA could result in a side effect on the self-renewing ability of SSCs and/or differentiation that causes a decrease in the quantity of sperm cells.
In the current study, administration of BPA could result in significant up-regulation of AwSox2 expression in the male gonadal tissue, suggesting that AwSox2 is involved in cell differentiation. In the embryonic stem cell (ESC), it was recognized that up-regulation of Sox2 expression can cause a down-regulation of the expression of several genes (Oct4, Nanog, Utf1, and Fgf4) which are known to be essential for the self-renewal. It is suggested that small increases of Sox2 may trigger cell differentiation. A 0–2-fold increased Sox2 level induced pronounced morphological changes that were associated with cell differentiation. In this process, gene expression analysis demonstrated that pluripotency-associated genes (e.g. Nanog) were down-regulated, whereas genes associated with neuroectoderm, mesoderm and trophectoderm, but not endoderm, were up-regulated. Notably, massive cell apoptosis occurred when Sox2 levels were increased more than five times in the ESC. Thus, it is imperative that the Sox2 is controlled at a perfect level in ESC, even when differentiation is initiated. Currently, relatively little is known about the mechanism by which elevated levels of Sox2 inhibit Sox2, Oct4, Nanog, Utf1, and Fgf4 promoters. 44
Apoptosis is a component of innate immunity and plays a pivotal role in adaptive processes. 45 Apoptosis or programmed cell death is a strongly conserved cellular process throughout evolution. Failure of this normal apoptotic process results in several pathological changes, most notably accumulation of damaged cells such as cancer, or inappropriate cell loss, where the imbalance between cell growth and apoptotic rate leads to abnormal change of tissue. 46 To maintain the balance between cell division and cell death, Sox2 plays a key role in the regulation of apoptosis with respect to various intrinsic and extrinsic stimulations.3–6 The regulation of apoptosis-involved Sox2 is vital for tissue homeostasis, embryo development, development of nervous systems, immune regulation and maturation of blood cells.3–6 Therefore, up-regulation of AwSox2 expressions derived from BPA treatment was associated with cell differentiation.
In the current study, expression of AwSox2 was significantly up-regulated by LPS and Poly I:C treatment in the hepatopancreas, gill and hemocytes, suggesting that AwSox2 is likely involved into the innate immune defense of bivalves. The innate immune response is the acute and first defense against pathogen invasion in animals. Because of the absence of an adaptive immune system, freshwater bivalves strongly depend on their innate immunity to fight against pathogens in the sedimentary environment of the pond and lake, compared with vertebrates.47,48 The innate immune system is complex and regulated by an array of immune-related genes. The immune response to infectious pathogens is mainly determined by changes in the expression of of immune-related genes.47,48 Regarding invertebrates, reports of Sox2 are very limited; Sox2 studies are mainly focused on vertebrates. Sox2 plays an important role in the growth and survival of the glioma-initiating cell/glioma stem cells, as down-regulation of its expression leads to cell cycle arrest and inhibition of tumor growth in glial tumors. 49 Sox2 expression is significantly induced in glial tumors and down-regulated after chemotherapy or radiation therapy.50–52 In vitro, proliferation of Sox2-positive tumor cells can be inhibited by anti-Sox2 T-cell immune response.53,54 In glial tumors, aberrant expression of Sox2 is associated immunogenic events in both children young adults, and these patients mount a cellular immune response to Sox2 in vivo. 55 The presence of Sox2-specific T cells can be detected in circulation in glioma, indicating that the immune response is systemic in nature, in spite of the brain being traditionally considered an immune-privileged site. Importantly, the presence of anti-Sox2 T cells can be detected in the tumor tissue even in a patient lacking such responses in circulating T cells, suggesting that Sox2 is immunogenic and can serve as a target for T-cell immunity. 55 Therefore, we postulated that up-regulation of AwSox2 is related to the immune defense of A. woodiana in the LPS- and Poly I:C-treated groups. Interestingly, Sox genes can bind to other transcription factors to activate or represses target genes. 56 Sox2, Sox3 and Sox19a share a domain with heat shock proteins (HSP) 47 and HSP90. 56 Sox9 can interact with HSP70 in chondrocytes and testicular cell lines. 57 HSP play a vital role in protein homeostasis, cellular stress responses and immune regulation. Sox–HSP interaction should provide a way to shed light on AwSox2 function in our experiment. Great efforts are required to elucidate the potential interaction of Sox genes with HSP.
Temporal expression of AwSox2 in gill exposed to Poly I:C did not follow a dose-dependent pattern, that is related with animals’ adaption. With elongation of Poly I:C treatment, reactive oxygen species (ROS) should increasingly be produced and accumulated in the gill. On the other hand, animals are enhancing their anti-oxidative ability to eliminate ROS. In this process, imbalance in the shift between oxidants and antioxidants should constantly occur.58–60 Therefore, a fluctuation of AwSox2 expression was observed in the Poly I:C-treated group. Significant up-regulation of AwSox2 was observed after 12 h in the LPS-treated group, but only after 24 h in the Poly I:C-treated group, suggesting that different toxic effects and regulating pathways are involved in LPS and Poly I:C challenge. As a powerful stimulator, LPS is known to stimulate immune cells through the activation of transcription factors, resulting in increased pro-inflammatory responses. 61 LPS can stimulate macrophages to release important cytokine mediators as well as to produce large amounts of nitric oxide and ROS. Poly I:C, a double-stranded RNA mimic, is sensed by TLR and ultimately induces production of type 1 IFN and pro-inflammatory cytokines. 62
Notably, AwSox2 up-regulation showed different profiles among BPA-, LPS- and Poly I:C-treated groups, suggesting that complex pathways are likely involved into the regulation of AwSox2 derived from BPA, LPS and Poly I:C challenges. The myriad of data from different pathological conditions, cells types, and models showed a very heterogeneous and confusing picture of how ROS influence the immune system and orchestrate autoimmune and inflammatory diseases.58–60 In early stages of disease development, ROS have a beneficial effect on the prevention of autoimmune diseases by lowering the responsiveness of adaptive immune cells and/or degradation of inflammatory mediators.58–60 In contrast, the high ROS levels often observed in inflammation cause cell and tissue damage, and might directly or indirectly perpetuate the progression of the disease.58–60 Balancing levels of ROS in a temporal and spatial context is essential to sustain homeostasis of the body.58–60 Generally, BPA treatment mainly results in an increase of ROS in the cell, whereas treatments of LPS and Poly I:C can cause a release of pro-inflammation cytokines. Different pathways are involved in the regulation of AwSox2 derived from BPA, LPS and Poly I:C. Therefore, patterns of AwSox2 expression show heterogeneous profiles when exposed to BPA, LPS and Poly I:C.
In conclusion, AwSox2 was cloned from A. woodiana for the first time and its full-length cDNA contained highly conserved motifs and residues of Sox2. After BPA, LPS and Poly I:C challenge, expressions AwSox2 were induced, which were associated with the apoptosis of sperm cell and innate immune response of A. woodiana. This investigation was only the first step toward a better understanding of Sox2 involved in the cell apoptosis and immune response of A. woodiana. Future studies should be undertaken to elucidate the potential interaction of Sox genes with HSP.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of Henan (No. 20A330002, 19B330002, 182300410123).
