Abstract
Neutrophils are innate immune cells implicated in the process of killing Mycobacterium tuberculosis early during infection. Once the mycobacteria enter the human system, neutrophils sense and engulf them. By secreting bactericidal enzymes and α-defensins like human neutrophil peptides loaded in their granule armory, neutrophils kill the pathogen. Peripheral blood neutrophils secrete a wide range of cytokines like IL-8, IL-1-β and IFN-γ in response to mycobacterial infection. Thus they signal and activate distant immune cells thereby informing them of prevailing infection. The activated monocytes, dendritic cells and T cells further continue the immune response. As a final call, neutrophils release neutrophil extracellular traps in circulation which can trap mycobacteria in patients with active pulmonary tuberculosis. Extensive neutrophilic response is associated with inflammation, pulmonary destruction, and pathology. For example, inappropriate phagocytosis of mycobacteria-infected neutrophils can damage host cells due to necrosis of neutrophils, leading to chronic inflammation and tissue damage. This dual nature of neutrophils makes them double-edged swords during tuberculosis, and hence data available on neutrophil functions against mycobacterium are controversial and non-uniform. This article reviews the role of neutrophils in tuberculosis infection and highlights research gaps that need to be addressed. We focus on our understanding of new research ideologies targeting neutrophils (a) in the early stages of infection for boosting specific immune functions or (b) in the later stages of infection to prevent inflammatory conditions mediated by activated neutrophils. This would plausibly lead to the development of better tuberculosis vaccines and therapeutics in the future.
Introduction
The World Health Organization reported that 1.3 million tuberculosis (TB) deaths and 10 million new TB infections occurred in 2017. 1 Though Mycobacterium tuberculosis (MTB) was discovered as the causative agent for tuberculosis way back in 1882, 2 Bacillus Calmette--Guérin (BCG) is the most widely used vaccine against TB. 3 But this vaccine has higher efficacy in the absence of prior MTB infection or sensitization with environmental mycobacteria. 4 Hence, TB treatment continues to be a difficult challenge for people worldwide. Rather than trying to understand the complexity and factors causing the virulence of MTB, it is high time we look into the host immunological responses that play an exceptionally significant role in both protection and pathology, since the outcome of MTB infection depends on the host’s ability to mount effective and balanced inflammatory responses. 5 Exposure to MTB may result in asymptomatic clearance of the pathogen or latent infection or clinical disease of varying severity. Discovering the reasons for these different outcomes will result in development of improved vaccines and therapeutic interventions. 6
The outcome of any infection is dependent on the immune status of the host. A strong immune and inflammatory response nullifies the infecting organism. Discovery of the various functions of neutrophils such as neutrophil extracellular trap (NET) formation, phagocytosis, heterogeneity and plasticity has increased and opened new avenues in recent neutrophil research. As a key component of the inflammatory response, neutrophils make important contributions to the recruitment, activation and programming of Ag-presenting cells which initially recognize the pathogen. For example, neutrophils generate chemotactic signals that attract immune cells such as monocytes and dendritic cells (DCs) to the site of infection.7,8 In this way neutrophils act as decision shapers by recruiting and activating the other immune responders against an infectious agent, and MTB is no exception. The importance of neutrophil response in TB is evidenced by the identification of a neutrophil-driven transcriptional signature prominent in the blood of patients with TB. 9
Neutrophils: The indispensable cells
Neutrophils are key mediators of the innate immune response, providing protection from invading mycobacteria. The inverse association observed between the risk of tuberculosis infection and peripheral blood neutrophil count in household contacts of patients with TB10 emphasizes the importance of neutrophils in TB. Being innate cells, neutrophils are expected to generate generic immune responses, but they are putative targets for host-directed therapies in TB. 11
Following activation by mycobacteria, neutrophils execute several specialized functions that include chemotaxis, phagocytosis, generation of reactive oxygen metabolites and activation of other immune cells. All of these processes are important for elimination of the invading microbe. These multifaceted functions of neutrophils are represented pictorially in Figure 1. When such an immune niche is disturbed, there occurs a dramatic increase in susceptibility to infection and/or tissue damage. 12 In spite of their importance in MTB infection, neutrophils are less considered as crucial responders compared with the other components of the human immune system. Among the available data there are controversies, probably because of the implicit difficulties in working with neutrophils. Neutrophils are fragile, short-lived cells and cannot be cryopreserved, making their study in vitro prone to difficulty. Also, isolated or depleted granulocytes are rarely characterized with accuracy in studies examining neutrophil–mycobacteria interactions, including neutrophil-mediated killing. Even so, as a result of advances in recent research, various insights into the role of neutrophils during TB in both animals and humans are available, which are detailed below.
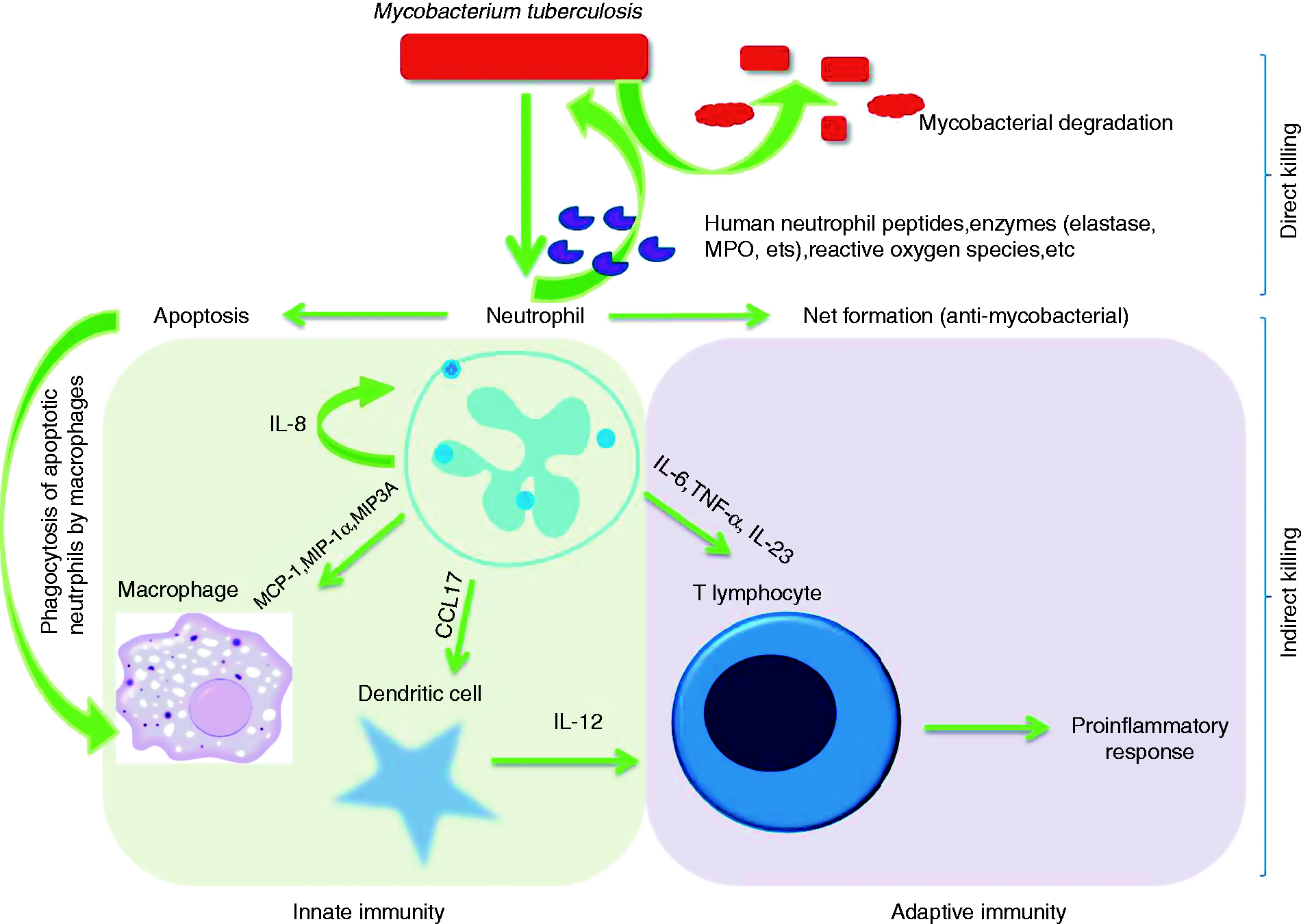
Once M. tuberculosis enters the human system, neutrophils recognize the pathogen and phagocytose it. A cascade of events then starts: in direct killing, neutrophils release lysosomal enzymes, human neutrophil peptides, reactive oxygen species, etc, which directly lyse the mycobacterium. Another mechanism is also activated during the apoptotic phase of neutrophils, wherein they release neutrophil extracellular traps, which can trap the microbe and prevent its further action on the host. In indirect killing, neutrophils secrete cytokines that signal other innate and adaptive immune cells, which in turn become activated and start functioning towards the elimination of the prevailing infection.
Neutrophils: Early responders to mycobacterial infection
The contribution of neutrophils to early defence against mycobacteria is well proved in animal models. For example, in rats recruitment of neutrophils to the lungs through the use of LPS at the time of airborne infection with 200 bacteria decreased downstream CFU in the lung. 13 Neutrophils also serve as the most abundant inflammatory cell type during early murine pulmonary TB, and participate in the clearance of lung bacilli and prevention of dissemination. 14 There are many other studies which show that abrogating neutrophils at the time of infection results in either a negative or neutral host outcome, and that recruiting neutrophils to the site of infection improves outcome.15,16 But this is believed to happen only when neutrophils are recruited early during infection, affirming their role in early immunity. However, the literature is not always consistent. For example, one study presented that neutrophils ultimately do not play an important role in the initial control of mycobacteria. This is because it was observed that neutrophil depletion had no effect on the bacterial load of intravenously infected mice with 106 CFU of MTB, BCG or Mycobacterium fortuitum. 17
Mechanisms of action
Phagocytosis and oxidative burst
Phagocytosis, being a basic function of neutrophils, decides the immediate response to MTB infection and is immediately followed by oxidative burst. Compared with macrophages, neutrophils showed higher levels and intensity of phagocytosis and oxidative respiratory response. 18 During phagocytosis, microbes are engulfed into phagosomes, which rapidly fuse with intracellular granules to form phago-lysosomes. 19 Then, neutrophils, through the generation of reactive oxygen species (ROS) 20 followed by the release of these preformed oxidants and proteolytic enzymes from granules, 21 contribute to the control of pathogens such as MTB. 10 Oxidative burst/killing by ROS includes superoxide and hydrogen peroxide, involving the assembly of the NOX2-containing NADPH–Oxidase complex at the phago-lysosomal membranes. 22 Hypochlorous acid and other additional toxic intermediates are generated through myeloperoxidase. 23 Thus, ROS are considered essential bactericides of neutrophils. 24 Contradictory reports prevail about the phagocytic potential and oxidative burst capacity of neutrophils in TB infection. Our group observed a decline in phagocytic potential of neutrophils in those with TB, 25 supporting an early report that granulocytes from patients with pulmonary TB have an impaired ability to phagocytose and undergo oxidative burst. 26 In line with this observation, mycobacteria were shown to retain Rab5a and Syntaxin-4 on the phagosome membrane for blocking the fusion of granules in human neutrophils. 27 Nevertheless, another report details that the phagocytic activity of blood neutrophils was significantly increased in patients with active pulmonary TB prior to treatment. 28 While phagocytosis is believed to be a defence mechanism of the host, in vitro experiments have also shown that human neutrophils quickly phagocytose vast numbers of MTB. Sometimes, depending on mycobacterial virulence, neutrophils fail to destroy them by oxidative killing. Instead, the engulfed MTB is shown to induce ROS in neutrophils, driving them into necrosis. 29 This implies that even if phagocytosis occurs, MTB can still escape the immune response depending on bacterial virulence and immune status of the host. One possible explanation comes from a recent study by Mishra et al., where the authors suggest that MTB exploits neutrophilic inflammation to preferentially replicate at sites of tissue damage that promote contagion. They also report that nitric oxide primarily affects this neutrophilc influx by repressing an IL-1 and 12/15-lipoxygenase-dependent neutrophil recruitment cascade. 30
Neutrophil enzymes
Neutrophils employ an arsenal of proteolytic enzymes present within their granules for the process of bacterial killing. When neutrophils phagocytose pathogens, rapid degranulation occurs as cytoplasmic granules fuse with the phagocytic vacuole and discharge their hydrolases and bactericidal proteins. Given their importance, tailoring the neutrophil enzyme activation and secretion mechanisms would possibly result in shaping the immune response during MTB infection. Some of the important facts about these enzymes are explained here. Neutrophil elastase, proteinase 3, and cathepsin G are three of the important hematopoietic serine proteases stored in large quantities in neutrophil cytoplasmic azurophilic granules. They are synthesized as inactive pre-pro-proteins containing a signal peptide and an amino-terminal pro dipeptide. 31 The controlled activation and release of these enzymes are crucial for any host, as these enzymes have the potential to be highly destructive to normal tissues. Once activated, they act in combination with ROS to help degrade engulfed microorganisms inside phagolysosomes. 32 For example, neutrophil-derived cathepsin G and elastase are said to critically contribute to the deceleration of mycobacterial replication during the early phase of the antimycobacterial response. 33 Accordingly treatment of human neutrophils with elastase has shown to result in the cleavage of caspase 3 and 9, 34 which are major proteins in the apoptotic pathway. In humans, it is evident from our previous work that MTB strains influence neutrophil enzyme secretion. 35 In mice, serine protease activity has been shown to play a protective role within hypoxic regions of lung granulomas, the hallmark of MTB infection. 36 Granuloma was once believed to be an uniquely host-driven response, set to constrain MTB and prevent dissemination of bacilli. But now granulomas are proved to play central role in TB pathophysiology. They form a pathological immunogical niche where neutrophils play a major role. Hypoxia and neutrophils are crucial to the development of lung lesions during TB disease. 37 Yet, the role of hypoxia and hypoxia-induced factors inside granulomas on neutrophil function and TB pathophysiology needs to be addressed.
Apoptosis of neutrophils: Self destruction for the better good
Apoptosis is crucial for the resolution of inflammation, 38 and its failure may lead to tissue damage and pathophysiology. The mycobacteria are destroyed by the process of oxidative burst following enzyme action and phagocytosis, but the by-products of oxidative burst are deleterious to the host. Thus, once phagocytosis and oxidative burst is complete, the infected neutrophils undergo apoptosis and become susceptible to phagocytosis by macrophages, and are finally disintegrated from the system. In this scenario, mycobacteria are proven to induce rapid cell death in neutrophils displaying the characteristic features of apoptosis such as morphologic changes, phosphatidylserine exposure and DNA fragmentation. 39 Experiments prove that activation of oxidative burst in neutrophils by MTB regulates the inflammatory response by induction of apoptosis. 40 More importantly, the potent proinflammatory response activated in human macrophages during MTB infection is augmented by apoptotic neutrophils, 41 establishing neutrophil-mediated macrophage activation.
Human neutrophil peptides
Besides phagocytosis and oxidative burst, several mechanisms prevail that account for the antimycobacterial activity of neutrophils. One such important element is the human neutrophil peptide (HNP) that has mycobactericidal and cytotoxic activity. Neutrophils produce HNPs, also known as α-defensins, constitutively and/or in response to microbial products or pro-inflammatory cytokines. They potentially influence the inflammatory or immune responses by modulating cytokine production or acting like opsonins or chemotactic factors. 42 In TB their role is well documented. An earlier study showed that higher concentrations of HNPs are found at sites of active TB infection. This finding suggests that plasma HNP concentration might be used as marker of disease severity and deterioration of pulmonary function. 43 HNPs are known for their ability to control MTB in vitro and when administered exogenously to animals. 44 In vitro, macrophages take up free HNPs, which ultimately enhances their ability to kill MTB and impair its macromolecular biosynthesis. 45 It has been reported that enhanced restriction of mycobacterial growth is attributed to mononuclear cells that phagocytose the granules or entire apoptotic neutrophils after trafficking their contents (which could contain HNP) to the endosomes. 46 Further research on neutrophil HNPs may lead to the identification of important tools for TB prevention and treatment.
NETosis
Besides destroying extracellular pathogens through phagocytosis and production of lytic enzymes, neutrophils release structures called NETs that can trap and kill microbes. 47 NETs are structures composed of chromatin and granule proteins that are capable of binding and killing extracellular pathogens such as bacteria and fungi. When neutrophils die in vitro they release these NETs, representing an extracellular mechanism of neutrophils to kill microbes. This way of infection containment minimizes injury to the surrounding tissue. Diverse microorganisms, including M. tuberculosis, are NET inducers in vitro. 48 Supporting this fact, studies have observed that plasma NET levels in patients with TB at baseline correlated with disease severity and decreased with antibiotic therapy. 49 Ramos-Kichik et al. demonstrated that although NETs trap mycobacteria, they cannot destroy or kill them. Thus, they suggest that NETs might help in localization of the infectious pathogen, thus preventing mycobacterial spread to other organs. In addition, NETs contain high local concentrations of antimicrobial agents which could make MTB susceptible to the available antimicrobial immune responses. 50 Although NET formation and release benefit the host in most infections, its exact role in the host response during TB is still controversial, demanding further research.
Neutrophils and comrades
Neutrophils can destroy MTB by their own immune machinery as discussed above. Besides this, their anti mycobacterial activity is extended by virtue of their co operation with other immune cells. Some of their well-characterized interactions are briefed here.
Neutrophil–macrophage cooperation
Neutrophil–macrophage cooperation during TB is well established. Neutrophils are the initial cells to arrive at sites of mycobacterial infection and they immediately and profusely engulf bacilli. However, neutrophils are short-lived, and their cellular contents are highly toxic. Therefore, instant removal of these cells from tissues after apoptosis/necrosis is a crucial process that depends usually on monocytes/macrophages. Necrotic neutrophils containing MTB within them are reported to promote mycobacterial survival and proliferation in human monocyte-derived macrophages, but apoptotic neutrophils control mycobacterial growth. 51 This supports an early study that has shown that phagocytosis of apoptotic neutrophils by macrophages is related to decreased viability of intracellular MTB. 46 Apoptotic neutrophils are phagocytosed by macrophages through the process of efferocytosis, leading to several consequences such as removal of neutrophils and prevention of tissue injury, allowing macrophages to utilize neutrophil granule proteins for antimicrobial defence, and altering cytokine production by macrophages. 52
In addition, inflammatory monocytes are attracted towards neutrophils by their secretory products. Monocyte recruitment from blood has been proved to be driven by neutrophil-derived chemokines, cytokines and their granule products. 53 After monocyte recruitment and maturation, cytokines secreted by both neutrophils and macrophages further drive the accumulation and activation of both cell types. 54 For example, Braian et al. reported that there exists close interaction between macrophages and MTB-activated neutrophils. They also found significant secretion of cytokines such as IL-6, TNF-α, IL-1β and IL-10 by macrophages co-cultured with NETs only from MTB-activated neutrophils but not phorbol myristate acetate-activated neutrophils. 55 Our group also reported that during TB, neutrophils show increased secretion of macrophage-attracting chemokines.56,57 From these observations, it is evident that neutrophil–macrophage cooperation is essential for a good downstream immune response in TB.
Neutrophil–DC–T-cell cooperation
During TB infection, in mice and humans, neutrophils help DCs to cross-present MTB Ags to T cells. This “ménage à trois” involving neutrophils, DCs and T cells has been shown to play a major role in the immune response to BCG. 58 Specifically, neutrophils increase the capacity of DCs to activate CD4 cells by presenting MTB to DCs in a more effective form. 59 The neutrophil–DC crosstalk is expected to have a direct impact on the immune response to any infection through the development of an Ag-specific immune response. 60 Regarding T cells alone, depletion of neutrophils during BCG vaccination has been shown to abolish the induction of Th1-specific responses, and eventually resulted in a prohibition of reduction in bacterial load which was otherwise observed in vaccinated animals. 61 It has also been showed that NETs released by human neutrophils can directly prime T cells by reducing their activation threshold. 62 These findings provide evidence for the influential role of neutrophils on T cells during TB.
Neutrophils: The double crosser
Given the multifaceted response of neutrophils in either direct killing of MTB or influencing other immune cells against MTB, howbeit at later stages of TB neutrophilia becomes largely detrimental to the host. 63 Neutrophilia in TB has been shown to be independently associated with increased risk of mortality. 64 Neutrophils which remain crucial immune responders during the initial stages of TB infection become detrimental drivers later. This is because infected/necrotic neutrophils harbour highly toxic contents. Removal of these dead cells is essential to resolve immunopathology, failure of which causes tissue damage. Also, recent studies have revealed that neutrophils play a major role in certain TB-related pathologies. For example in TB-IRIS (immune reconstitution inflammatory syndrome) neutrophils are shown to release granule contents, thereby contributing to pathology. 65 On performing longitudinal whole-blood microarray analysis it was further found that patients with TB Meningitis-IRIS show significantly more abundant neutrophil-associated transcripts from before development of TB Meningitis-IRIS through IRIS symptom onset. 66 Another study reported that during MTB infection, neutrophils recruited by 12 lipoxygenase harbour MTB and lead to disease pathogenesis. The authors suggest that in future, TB therapies aimed at reducing neutrophil recruitment to the lung might be an option to reduce lung pathology. 67 Supporting this fact, a recent report on immune mediators involved in inflammatory granuloma formation during TB demonstrated a major pathologic role for S100A8/A9 proteins in mediating neutrophil accumulation and inflammation associated with TB. 68 The crucial point where neutrophils shift from host protector to perpetrator of damage is of paramount importance both in research and intervention. Future research addressing this concept will help to effectively modify therapy according to the status of host and stage of disease. Also, future host-directed therapies in TB targeting neutrophils will be critical, as early neutrophil responses contain and limit infection, whereas later in disease sustained neutrophil responses damage the host and contribute to spreading the infection. 69
Concluding remarks
To solve this puzzle of neutrophils turning from active adversary of the pathogen to invaders of the host itself, it is high time to intensify neutrophil research to elucidate the intricate mechanisms involved in order to turn them into clinically beneficial tools. Progressive research in this line holds promise for such an understanding of neutrophils and their role in TB.
Footnotes
Acknowledgement
The authors thank the Indian Council of Medical Research for its support.
Author contributions
NH drafted the manuscript. LH corrected the manuscript. SD and ST reviewed the manuscript.
Declaration of conflicting interests
The author(s) declare that there is no conflict of interest regarding the publication of this paper.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
