Abstract
Innate immunity is the first line of defense against pathogen infections. Extracellular ATP (eATP) is one of the most studied danger-associated molecular pattern molecules that can activate host innate immune responses through binding with and activating purinergic receptors on the plasma membrane. The detailed actions of eATP on fish innate immunity, however, remain poorly understood. In this study, we investigated bacterial pathogen-induced ATP release in head kidney cells of the Japanese flounder Paralichthys olivaceus. We also examined the actions of eATP on pro-inflammatory cytokine and immune-related gene expression, the activity of induced NO synthase (iNOS), and the production of reactive oxygen species (ROS) and NO in Japanese flounder immune cells. We demonstrate that ATP is dynamically released from Japanese flounder head kidney cells into the extracellular milieu during immune challenge by formalin-inactivated Edwardsiella tarda and Vibrio anguillarum. In addition, we show that eATP administration results in profound up-regulation of pro-inflammatory cytokine gene expression, iNOS activity, and inflammatory mediator production, including ROS and NO, in Japanese flounder immune cells. Altogether, our findings demonstrate that eATP is a potent signaling molecule for the activation of innate immune responses in fish.
Introduction
Extracellular ATP (eATP) is a primary signaling molecule for the regulation of numerous biological processes in different tissues and organs. 1 In particular, ATP release is induced under injury, pathogen infection or other pathophysiological conditions.2,3 Increasing evidence has demonstrated that eATP is an endogenous, danger-associated molecular pattern molecule, and a potent immunomodulator that regulates the migration and phagocytosis as well as the release of inflammatory mediators/factors such as cathepsins, cytokines including IL-1β, IL-6, IL-8, IL-10, TNF-α, C-C motif chemokine ligands (CCL2, CCL3, and CCL20) and a C-X-C motif chemokine (CXCL2); furthermore, the production of NO and reactive oxygen species (ROS) in the innate immune cells of mammals is also regulated by eATP through the activation of different members of purinergic P2 receptors located on the cell membrane.2,4–6 Although the role of eATP in mammalian innate immunity has been well established,7,8 relatively little is known regarding lower vertebrates, such as fish.
Our previous study revealed that gap junction protein connexin43 and hemi-channel pannexin1 are important conduits for PAMP-induced ATP release in teleost Paralichthys olivaceus.9,10 We also demonstrated that multiple purinergic P2X and P2Y receptor subtypes are co-expressed in Japanese flounder immune-related tissues and immune cells,11–13 and that the activation of P2X7 receptor leads to increased gene expression of multifunctional pro-inflammatory cytokines IL-1β and IL-6 in Japanese flounder P. olivaceus head kidney cells. 11 In addition, our recent studies revealed that eATP is involved in the NLR inflammasome-mediated innate immune response in Japanese flounder head kidney macrophages (HKMs).14,15 Furthermore, we demonstrated a modulatory role for eATP in regulating caspase activity in Japanese flounder HKMs. 16 Finally, He et al. reported bactericidal activity of eATP in Plecoglossus altivelis. 17 However, the contribution of eATP to fish innate immunity remains incompletely understood.
In the present study, we investigated ATP release in Japanese flounder head kidney cells induced by insults from the bacteria Edwardsiella tarda and Vibrio anguillarum, both of which are severe disease-causing pathogens in the Japanese flounder mariculture industry. In addition, eATP-induced innate immune responses, including pro-inflammatory cytokine and immune-relevant gene expression, iNOS activity, and NO and ROS production in the Japanese flounder immune cells, were measured to obtain a comprehensive overview of the functions of eATP in fish innate immunity. Our findings demonstrate that eATP is a conserved and potent signaling molecule in the activation of innate immunity in fish.
Materials and methods
Animal maintenance and tissue sampling
Japanese flounder P. olivaceus from a local farm in Tianjin, China, were transported to the laboratory and maintained in an aerated running seawater system at 21°C for 2 wk to acclimate to laboratory conditions before experiments. The fish were clinically examined and only healthy animals without any pathological signs were selected for use in experiments. For tissue collection, the fish were euthanized with 0.25 g/l tricaine methanesulfonate (Sigma-Aldrich) and individual tissues were separated and collected aseptically. This study was performed under NIH guidelines for the care and use of experimental animals.
Japanese flounder HKMs and peripheral blood leukocytes preparation and cell culture
Japanese flounder head kidney primary cells were prepared as described previously. 18 Japanese flounder HKMs and peripheral blood leukocytes (PBLs) were isolated by discontinuous Percoll density (1.020/1.070 and 1.070/1.077, respectively, GE Biosciences) gradient centrifugation. After centrifugation at 3000 r/min for 40 min at 4°C, the white interface fraction was collected and washed three times with cold PBS. Cell viability was examined by Trypan blue exclusion test, which showed greater than 95% viability. Japanese flounder HKMs and PBLs were then cultured in 24-well or 96-well plates (Corning) with RPMI 1640 medium supplemented with 10% FBS (Invitrogen) and 1% penicillin–streptomycin liquid at 21°C overnight before experimentation. P. olivaceus head kidney cells were seeded into 24-well plates (ThermoFisher Scientific) and were cultured in DMEM-F12 medium supplemented with 10% FBS, penicillin (100 IU/ml), and streptomycin (100 μg/ml) at 21°C under 2.5% CO2 atmosphere.
RNA purification and cDNA synthesis
Total RNA was isolated using an RNeasy Kit (Qiagen) and treated with DNase I (Invitrogen, amplification grade) to remove any residual genomic DNA contamination according to the manufacturer’s protocol. Quality of extracted RNA was examined on a 1.2% formaldehyde-denatured agarose gel stained with ethidium bromide and photographed on a GelDocTM XR system (Bio-Rad). RNA quantity was assessed with a NanoDrop 2000 UV/Vis spectrophotometer (ThermoFisher Scientific). Total RNA (1 μg) from each source was reverse transcribed into first-strand cDNAs using a SuperScript III ribonuclease H- reverse transcriptase Kit (Invitrogen) according to the manufacturer’s instructions. Control experiments using mock transcribed (without transcriptase) samples as template amplified no PCR products (data not shown), confirming that there was no genomic DNA contamination.
Measurement of bacterial challenge-induced ATP release
The bacteria E. tarda and V. anguillarum were cultured from single colonies with marine Luria-Bertani medium. Bacteria were pelleted by centrifugation, resuspended in PBS, and inactivated in 0.5% (vol/vol) formalin at 37°C for 2 h followed by 4°C for 3 h. Inactivated bacteria were washed 4 times with PBS, resuspended and adjusted to 2.5 × 107 cells/ml in DMEM-F12 medium. P. olivaceus head kidney primary cells (2.5 × 106/well) cultured overnight were pre-incubated with or without 100 μM carbenoxolone (CBX, Sigma-Aldrich), a gap junctional protein inhibitor, for 2 h and were then stimulated with E. tarda or V. anguillarum in the presence or absence of 100 μM CBX at indicated times. After the challenge, the culture supernatant was centrifuged at 13,000 r/min for 10 min, collected and kept on ice for analysis of ATP levels. ATP release induced by bacterial pathogen challenges in Japanese flounder head kidney cells was measured in a Tecan infinite® M200 PRO multimode microplate reader (Switzerland) using an ATP Bioluminescence Assay Kit CLS II (Roche) according to the manufacturer’s instructions. ATP levels were recorded as bioluminescence with arbitrary units. ATP release in the culture medium before bacterial challenge was set to “1”, and the data are therefore presented as normalized values.
eATP-induced cytokine and immune-related gene expression in Japanese flounder HKMs and PBLs
To test the effects of eATP on cytokine and immune-related gene expression, Japanese flounder HKM and PBL cells were treated with 200 or 1000 μM ATP (cell culture grade, Sigma-Aldrich) for the indicated times, and eATP-induced gene expression changes in IL-1β, IL-6, IL-11, TNF-α, G-CSF, IFN, Mx, and NF-κ B p65 subunit (p65) were quantified by quantitative real-time PCR (qRT-PCR). For this aim, one microliter of cDNAs from each source was amplified in a MyiQ™ Two-Color Real-Time PCR Detection System (Bio-Rad) with corresponding primer pairs (Table 1) in a total volume of 25 μl using SYBR PrimeScript Ex Taq™ II kit (TaKaRa) under the following conditions: initial denaturation at 95°C for 30 s, 40 cycles at 95°C for 5 s, 60°C for 30 s, followed by dissociation curve analysis (55°C to 95°C: increment 0.5°C for 5 s). β–Actin was used as an internal reference gene. Agarose gel electrophoresis analyses were performed at the end of each qRT-PCR to validate specificity of amplification. The identities of all the qRT-PCR products were further verified by DNA sequencing. The results are expressed as fold changes in the target gene normalized to the reference gene and as relative to expression in untreated controls. Data are presented as the means ± SEM from triplicate experiments.
Sequences for primers used in real-time PCR analysis.

f: forward; r: reverse.
eATP-induced iNOS activity and NO production measurements
The Japanese flounder HKM cells were cultured overnight at a density of 1 × 106 cell per well in a 96-well plate before experiments. The supernatants were removed and replaced with fresh medium in absence of serum. The cells were then incubated with 0 (controls), 200, 500, or 1000 µM ATP for indicated times. After 4, 8, 12, 24, 36, 48, and 72 h of incubation, the supernatants were collected and kept on ice. Following a brief spin, the iNOS activity and NO production in the culture supernatant were measured at 530 nm and 550 nm, respectively, using a NOS typed assay kit and an NO assay kit (Nanjing Jiancheng Bioengineering Institute, China), respectively, according to the manufacturer's instructions. Each condition was repeated in triplicate, and the NO concentration of the samples was calculated from a standard curve.
eATP-induced ROS production measurement
ROS production in Japanese flounder HKM cells induced by eATP administration was quantified using a commercial kit from the Nanjing Jiancheng Bioengineering Institute (Nanjing, China) following the manufacturer’s procedure. Briefly, Japanese flounder HKMs cultured overnight in 24-well plates (2.65 × 106 cells/well) were stimulated with different concentrations of ATP in the presence of 5 µM carboxymethyl-2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) for indicated amounts of time. In parallel experiments, the cells were also incubated with the same amount of DCFH-DA in the absence of ATP stimulation (controls). After the treatments, the cells were washed three times with cold PBS and were then pelleted by centrifugation. Fluorescence emission at 530 nm with arbitrary units was measured using a Tecan infinite® M200 PRO multimode microplate reader. Triplicate experiments were performed, and the data are presented as the means ± standard deviation.
Lactate dehydrogenase assay
Cytotoxicity of different doses of ATP treatment to Japanese flounder HKMs was evaluated by quantification of plasma membrane damage that results in the release of lactate dehydrogenase (LDH) into the cell culture medium. After ATP treatment, the culture medium was collected, clarified by centrifugation, and utilized for LDH assay with an LDH cytotoxicity assay detection kit (Beyotime Institute of Biotechnology, China) following the manufacturer's instructions. LDH concentration in the medium was detected at a wavelength of 490 nm. LDH activity released into the media of treated HKM cells was compared with total activity (100% LDH release, positive controls) in the cells lysed with 1% Triton X-100. Cell culture medium alone served as a blank control, and OD values were subtracted from the readings of the samples and the positive controls. Cytotoxicity was normalized to total LDH activity in the cell lysates and was presented as: cytotoxicity (%) = [(sample − blank)/(positive control − blank) × 100].
Statistical analysis
Statistical differences were detected using either Student’s t-test or one-way ANOVA followed by Duncan’s analysis using SPSS software version 11.0. Differences were considered significant at P < 0.05.
Results
ATP is released from bacteria-challenged Japanese flounder head kidney cells
Our previous studies demonstrated that ATP is dynamically released upon LPS stimulation in both Japanese flounder head kidney cells and gill cells,9,10 suggesting that bacterial insults may induce ATP release in P. olivaceus. We therefore selected severe disease-causing agents, E. tarda and V. anguillarum, as representative bacterial pathogens to examine pathogen-induced ATP release using a luciferase/luciferin bioluminescence assay system. Overall, significant ATP release was observed as early as 3 min after both bacterial pathogen challenges, and the increased ATP release persisted through the end of experiments in Japanese flounder head kidney cells (Figure 1). However, compared with E. tarda, V. anguillarum challenge induced greater ATP release (6.38-fold vs. 3.79-fold). In addition, bacteria-induced ATP release was significantly reduced by pre-treatment with the gap junction inhibitor CBX. Pre-incubation with CBX did not affect the basal ATP release (data not shown).
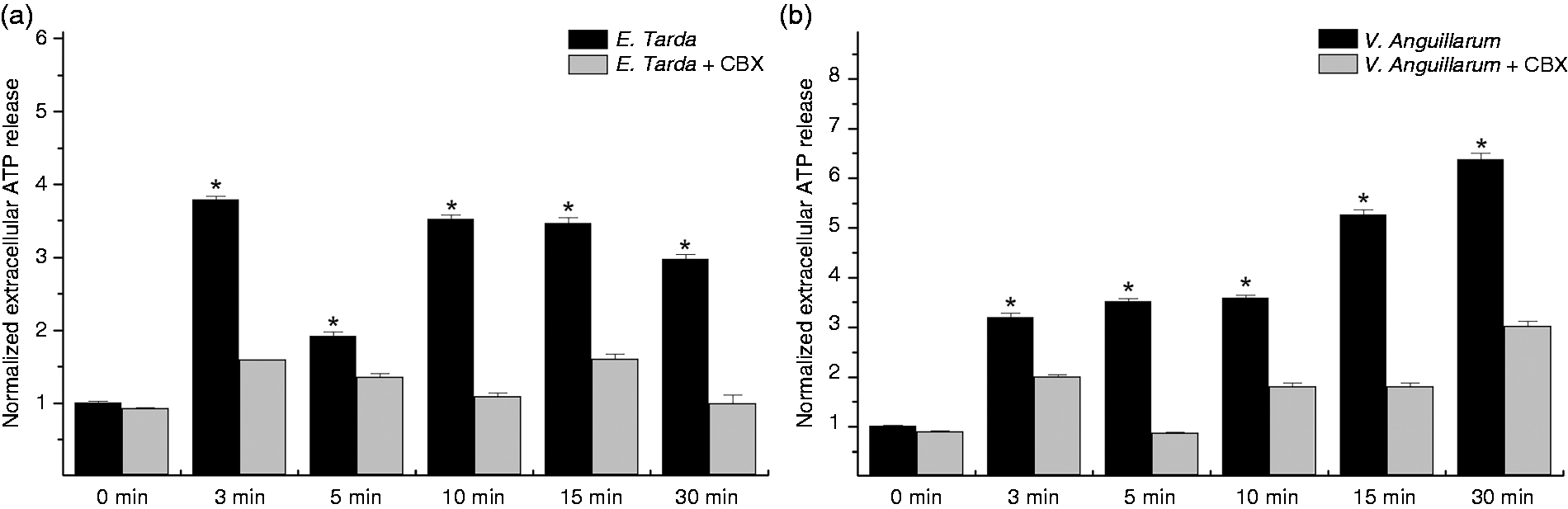
Bacterial pathogen-induced ATP release in Japanese flounder head kidney cells. ATP release in Japanese flounder primary head kidney cells induced by formalin-inactivated E. tarda (a) and V. anguillarum (b) was measured by Tecan Infinite® M200 PRO multimode microplate reader using an ATP Bioluminescence Assay Kit CLS II. Treatment of cells with E. tarda or V. anguillarum resulted in significant increases of ATP release, which were substantially attenuated by pre-incubation with 100 μM of the gap junction channel inhibitor CBX. Data are normalized to basal ATP release from unchallenged control cells (normalized to 1) and are presented as the mean ± SEM of one representative experiment performed in triplicate. Significant differences are indicated by asterisks at P < 0.05 between bacterial-challenged groups and CBX pre-incubated bacterial-challenged groups at the same time point. Similar results were obtained in other two separated experiments.
eATP up-regulates pro-inflammatory cytokine and immune-related gene expression in Japanese flounder immune cells
eATP is an important signaling molecule in stimulating host innate immune response in mammals. To further our understanding of the actions of eATP in Japanese flounder innate immunity, we examined transcriptional responses of pro-inflammatory cytokines, chemokines and immune-related genes in response to different dosages of ATP stimulation in both HKM and PBL cells by qRT-PCR. Generally, exposure to eATP caused significant and quick up-regulation of most examined genes in a dose- and cell type-dependent manner (Figures 2 and 3). Notably, CSF was robustly induced 2 h following ATP treatment in both HKMs and PBLs. In addition, low concentrations of ATP (200 μM) induced much more IL-6, G-CSF, TNF-α, and p65 gene expression in HKM cells (Figure 2) and IL-1β, IL-6, and TNF-α gene expression in PBL cells (Figure 3). However, even though maximal IL-6 expression was induced by low concentrations of ATP in both types of immune cells, the highest expression of IL-6 was induced earlier in PBLs than in HKMs (8 h vs. 12 h after ATP treatment). Furthermore, high concentrations of ATP (1000 μM), which usually activates P2X7R, induced much more IL-1β, Mx, and IFN mRNA production in HKM cells (Figure 2) and IL-11, CSF, and p65 mRNA production in PBL cells (Figure 3). Interestingly, even though the antiviral myxovirus resistance gene (Mx), a commonly used reporter of type-I IFN induction, was up-regulated by both low and high concentrations of ATP treatments in HKM cells, it was down-regulated in PBL cells in response to ATP treatment.
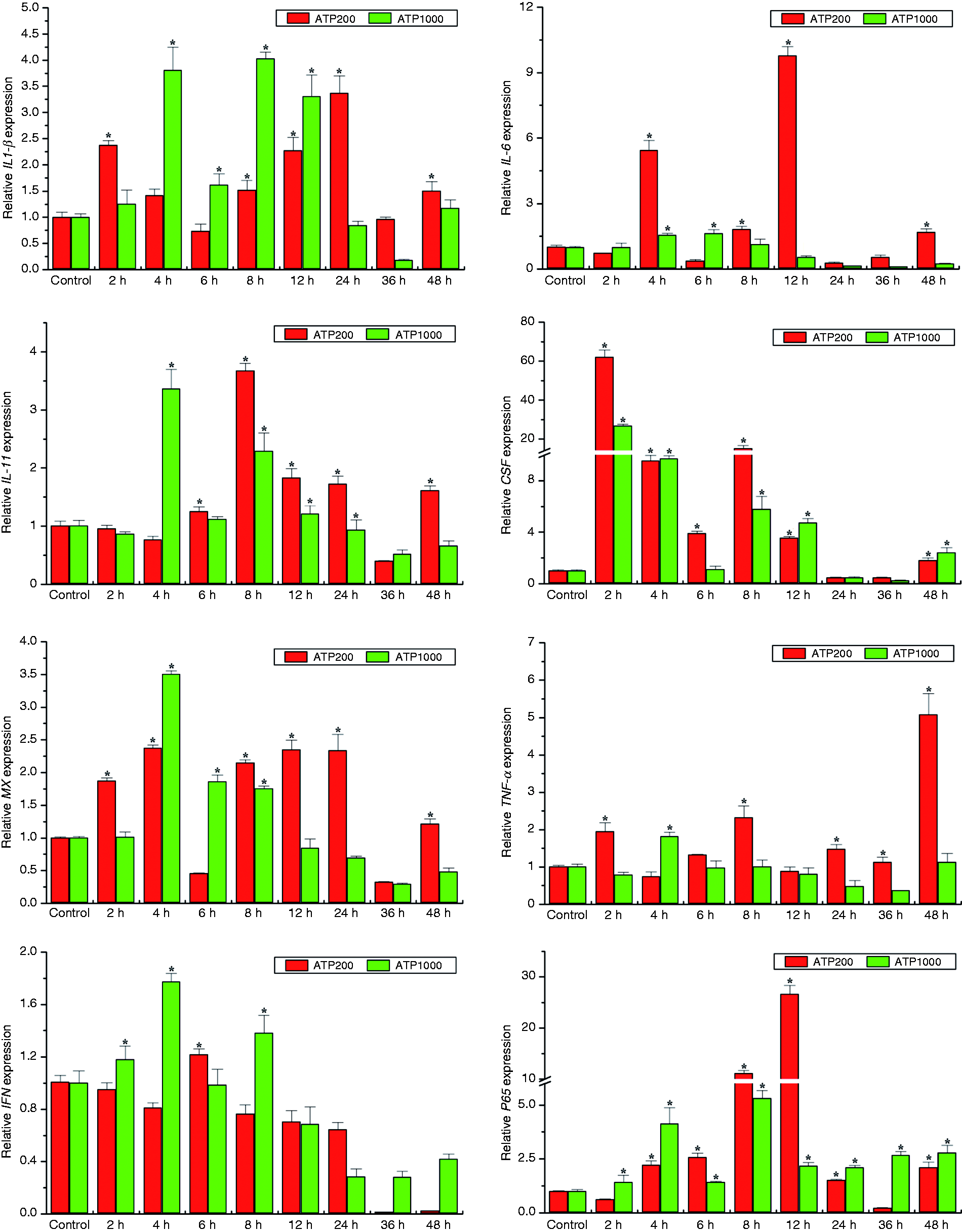
eATP modulates expression of pro-inflammatory cytokines and immune-related genes in Japanese flounder HKM cells. IL-1β, IL-6, IL-11, TNF-α, G-CSF, IFN, Mx, and p65 gene expression levels upon 200 or 1000 μM ATP stimulation were analyzed by qRT-PCR with β-actin as a reference gene and are presented as fold change relative to untreated control groups (normalized to 1). Results are expressed as the mean ± SEM of one representative experiment performed in triplicate. Asterisks (*) mark the significant increase of target gene expression compared with the untreated control groups (P < 0.05). Similar results were obtained in other two separated experiments.
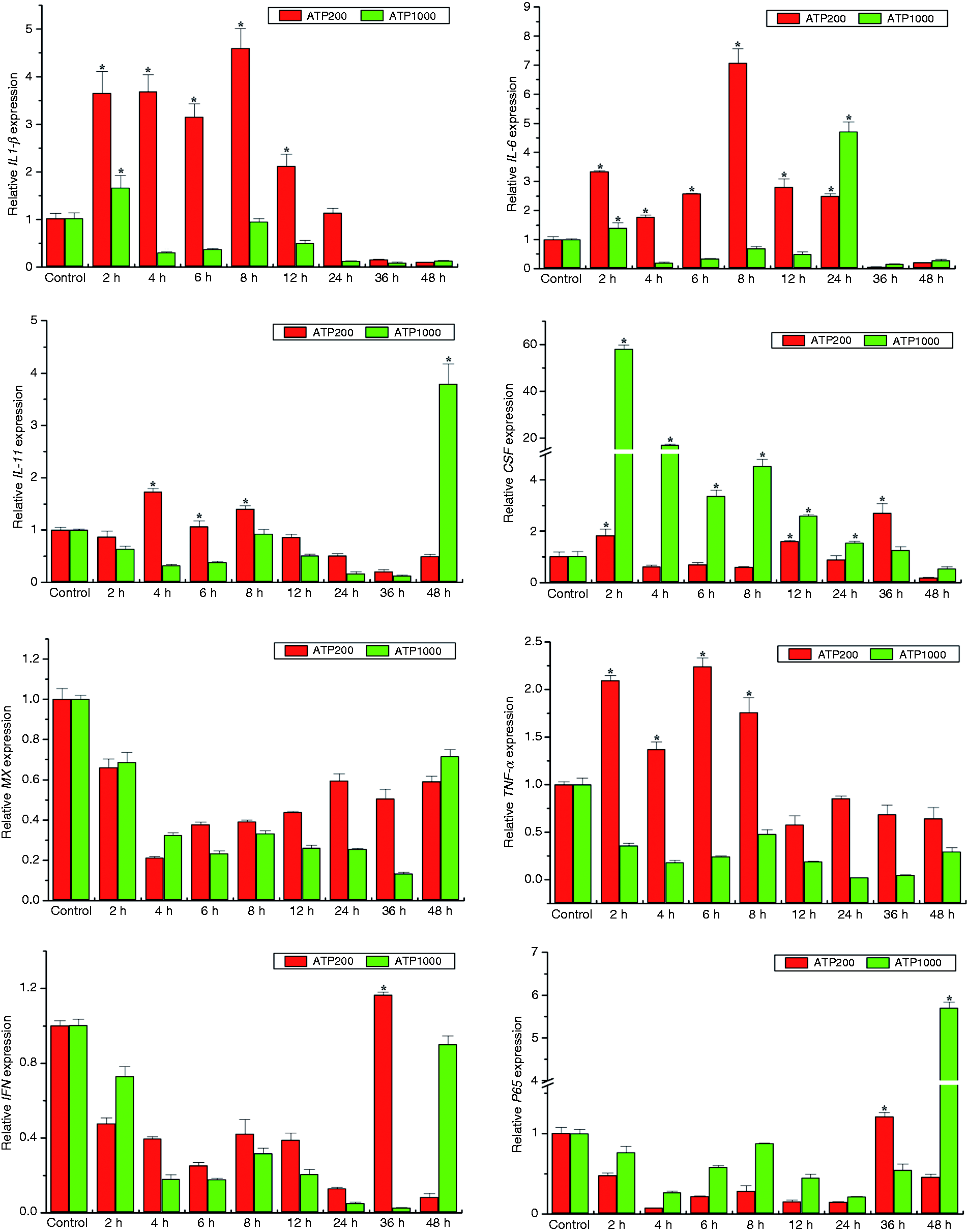
Effects of eATP treatment on gene expression of pro-inflammatory cytokines and immune-related genes in Japanese flounder PBL cells. IL-1β, IL-6, IL-11, TNF-α, G-CSF, IFN, Mx, and p65 gene expression changes following ATP administration were analyzed by qRT-PCR with β-actin as a reference gene. Data were normalized to the untreated control groups (set to 1) and presented as the mean ± SEM of one representative experiment performed in triplicate. Asterisks (*) mark the significant increase of target gene expression compared with the untreated control groups (P < 0.05).
eATP-induced iNOS activity and NO and ROS production in HKMs
Macrophages play pivotal roles in fish innate immunity. 19 NO and ROS are essential inflammatory mediators in immune defense system in fish. To understand the effects of eATP on Japanese flounder innate immunity, Japanese flounder HKMs were stimulated with different doses of ATP, and eATP-induced iNOS activity, and NO and ROS production were measured.
Figure 4 illustrates effects of induction times and ATP dosage on iNOS activity and NO production in Japanese flounder HKM cells. iNOS activity was elevated 4 h after exposure to higher concentrations (500 and 1000 μM) of ATP stimulation. Maximal iNOS activity was induced at 48 h by 1000 μM ATP treatment, while lower concentrations (200 μM) of ATP treatment failed to increase iNOS activity at any time point during the experiment (Figure 4a). Associated with the up-regulated iNOS activity, NO production was slightly increased 8 h after higher concentrations (500 and 1000 μM) of ATP stimulation, and maximal increase was observed 72 h following 1000 μM ATP treatment (Figure 4b). Consistent with the findings of iNOS activity under lower concentrations (200 μM) of ATP treatment, levels of NO production were unchanged in response to 200 μM ATP treatment compared to untreated controls.
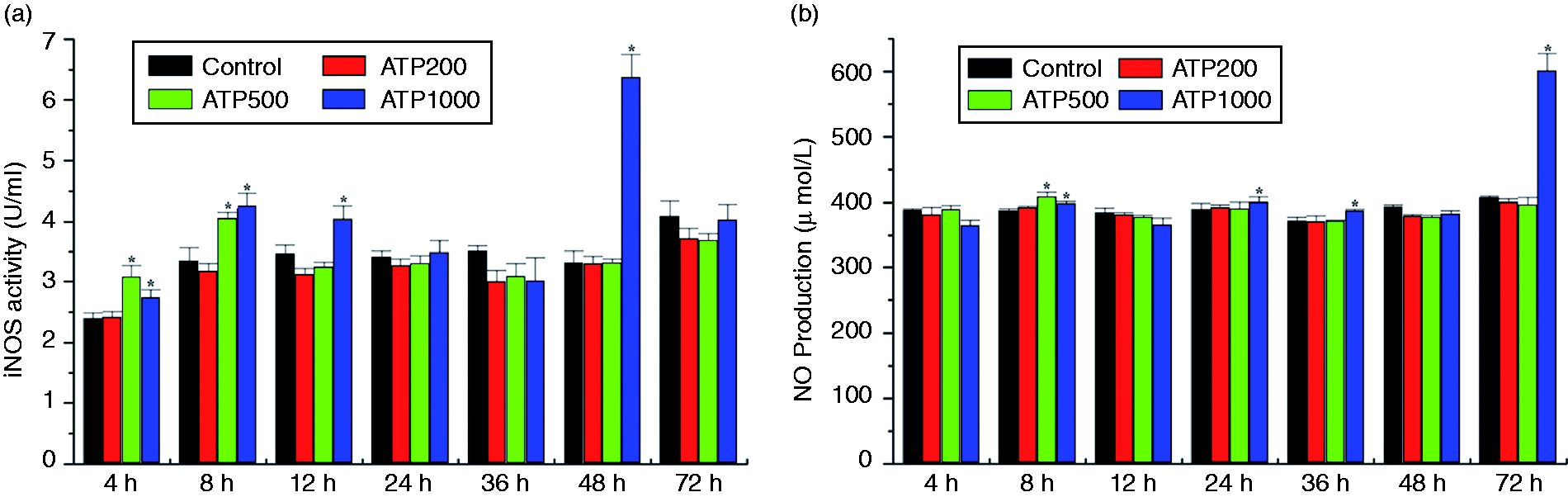
eATP-induced iNOS activity and NO production in Japanese flounder HKM cells. Japanese flounder HKM cells cultured overnight were incubated with 200, 500, or 1000 μM ATP at the indicated time points. After treatment, culture medium was collected and centrifuged. iNOS activity (a) and NO production (b) in the culture supernatant were determined by NO Synthase (NOS) typed assay kit and NO assay kit, respectively. NO concentration in samples was calculated from a standard curve. Values are presented as the means ± standard deviation (n = 3). Asterisks (*) mark the significant increase of iNOS activity (a) or NO production (b) compared with the untreated control groups at the same time point (P < 0.05).
eATP-elicited production of ROS in HKM cells is shown in Figure 5. ROS production was also induced in a dose-dependent manner. Upon 500 or 1000 μM ATP stimulation, ROS was induced immediately (5 min) after ATP administration. Maximum ROS production was observed 15 min after 200 μM ATP addition, while higher doses of ATP (1000 μM) stimulation led to additional increases in ROS production after 2 h of treatment.
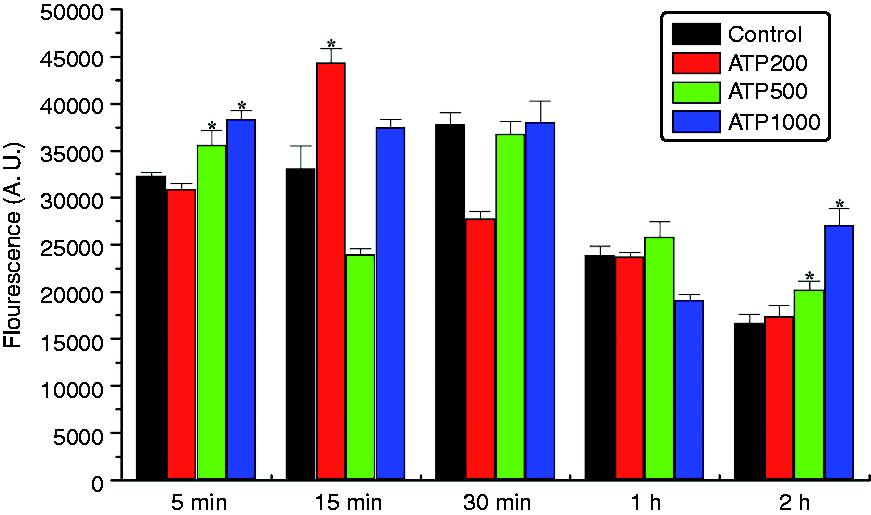
eATP-induced ROS production in Japanese flounder HKM cells. Japanese flounder HKM cells (2.65 × 106 cells/well) cultured overnight were incubated with or without ATP in the presence of 5 µM DCFH-DA for indicated amounts of time. After treatment, cells were washed three times with cold PBS and then pelleted by centrifugation. Fluorescence emission at 530 nm with arbitrary units was measured using a Tecan infinite® M200 PRO multimode microplate reader. Data are expressed as the means ± standard deviation (n = 3). Asterisks (*) mark the significant increase of ROS production compared with the untreated control groups at the same time point (P < 0.05).
Effect of ATP treatment on cell lysis
To determine whether ATP stimulation can lead to necrotic cell death, we examined the release of the cytosolic enzyme LDH under our experimental conditions. We found that under all experimental conditions, LDH release in response to ATP treatment was less than 5% of total cell LDH release produced by 1% Triton X-100 treatment (i.e. LDH release was about 3.69% of total cell LDH release after 1 mM ATP treatment for 72 h in the HKMs), suggesting that ATP concentrations used in our studies did not cause significant cellular toxicity.
Discussion
As an important danger-associated signaling molecule, eATP is widely recognized to have critical roles in the activation of host innate immunity through action on specific cell surface-expressed P2X receptors (P2XRs) and P2Y receptors (P2YRs) in mammals. 20 For example, eATP regulates the gene expression of transcription factors via intracellular signaling pathways in immune cells, 6 and performs multiple roles in stimulation of lymphocyte proliferation, ROS generation, and cytokine and chemokine secretion. 21 However, pathogen-induced ATP release as well as the functions of eATP in fish innate immunity remain incompletely understood. In this comparative immunological study, we performed a comprehensive examination of eATP actions on the innate immune responses in Japanese flounder P. olivaceus, an economically important flat fish cultured in the eastern coast of China and neighboring countries.
In normal tissues, eATP levels are low and tightly regulated. 22 However, ATP is rapidly released from the intracellular space to the extracellular environment under inflammatory or infectious conditions, leading to significant increases in eATP concentration. Our previous study showed that PAMP stimulation induces significant ATP release in fish. 9 We recently showed the engagement of pannexin1 channel in bacterial infection-induced ATP release in tilapia (Oreochromis niloticus). 23 In the present study, we further demonstrated that ATP is dynamically released in Japanese flounder head kidney cells in response to stimulation with the formalin-inactivated bacteria E. tarda and V. anguillarum. Interestingly, even though both E. tarda and V. anguillarum are Gram-negative bacteria, greater ATP release was induced by V. anguillarum, possibly due to their divergent membrane components. Our previous study demonstrated that pannexin1 channel protein expressed in Japanese flounder head kidney tissue, possesses an important role in PAMP-induced ATP release. 9 We also revealed that the abundant expressed gap junction protein connexin43 is an additional important conduit for immune challenge-induced ATP release in Japanese flounder. 10 To examine whether pannexin1 and connexin channels are involved in pathogen-induced ATP release, we pre-treated Japanese flounder primary head kidney cells with the gap junction inhibitor, CBX, and found that both E. tarda- and V. anguillarum-induced ATP release were significantly reduced, suggesting their engagement in pathogen-induced ATP release.
eATP is an important pro-inflammatory signaling molecule in activating host innate immunity under stress, injury or infectious conditions in mammals through initiation of purinergic signaling.2,7 The production of pro-inflammatory cytokines is a key marker of the innate immune response. eATP-evoked mRNA expression and release of pro-inflammatory cytokines IL-6, TNF-α, and chemokine CCL2 has been observed in mouse primary microglia. 24 IL-1β and IL-6 are also induced by 2′(3′)-O-(4-benzoylbenzoyl)adenosine-5′-triphosphate (BzATP, a potent agonist for P2X7R) stimulation in human macrophages. 25 Our previous study showed that activation of P2X7R by eATP up-regulates pro-inflammatory cytokine IL-1β and IL-6 gene expression in Japanese flounder head kidney cells. 11 We also previously revealed a role for eATP in the regulation of caspase1 activity, 16 and NLR family caspase activation and recruitment domain (CARD) containing 3 (NLRC3)-mediated immune signaling in the Japanese flounder. 14 These findings suggest that eATP may serve as an important signaling molecule in activation of innate immune responses in fish. However, it remains unclear whether eATP is involved in regulating expression of other cytokines. Herein, we show that eATP quickly and broadly up-regulates gene expression of several pro-inflammatory cytokines, chemokines and immune-related genes, including IL-1β, IL-6, IL-11, TNF-α, G-CSF, IFN, Mx, and p65 in Japanese flounder immune cells. The significant up-regulation of multiple cytokines and immune-related genes suggests that eATP plays a critical role in induction of early innate immune responses in fish.
IL-1β is a potent pro-inflammatory cytokine that plays pivotal roles in regulating innate immune responses. eATP is an essential signaling molecule for IL-1β maturation and release through assembly and activation of the NLR inflammasome.26,27 Herein, we show that eATP also induces significant IL-1β mRNA production in both HKMs and PBLs, suggesting that eATP may not only involve in IL-1β posttranslational processing, but also in the induction of IL-1β mRNA expression. In addition, IL-1β is also a pleiotropic cytokine that stimulates the expression of several other chemokines and cytokines, such as TNF-α, IL-6 and acute phase proteins associated with inflammation, thus initiating a self-amplifying cytokine network. 28 Therefore, elevated expression of IL-1β induced by eATP may further promote gene expression of other cytokines.
Granulocyte CSF (G-CSF), which is a dominant growth factor that controls the production, maturation, and function of granulocytes, is usually maintained at low concentrations in tissues. 29 Notably, G-CSF was markedly induced (approximately 60-fold compared to untreated controls) 2 h after ATP stimulation in both HKM and PBL cells. This substantial induction of G-CSF indicates that eATP may serve as an important signaling molecule involved in proliferation and differentiation in Japanese flounder immune cells.
In addition to G-CSF, upon ATP treatment, p65, a critical subunit of the transcription factor NF-κB that plays a key role in regulating immune response to infection, was also potently up-regulated (approximately 28-fold compared to untreated controls) in HKM cells. Similarly, potent activation of NF-κB by eATP has previously been reported in mammalian cells. 30 As activation of NF-κB induces cytokine and chemokine gene expression, up-regulated expression of cytokines in our study may be induced by enhanced expression of NF-κB, a hypothesis that needs to be clarified in the future.
Type I IFNs are potent mediators of the innate immune response via their induction of downstream antiviral immune mechanisms. Myxovirus-resistant protein (Mx) is one of the most studied IFN-stimulated proteins that specifically prevent growth in vivo and in vitro, of certain classes of virus. 31 Consistent with this finding, Mx was also up-regulated in HKM cells; however, in PBL cells, IFN-γ was down-regulated at most time points following ATP stimulation, which was associated with decreased expression of Mx gene. This finding indicates that eATP plays an important role in regulating expression of the antiviral genes IFN and Mx in fish.
eATP-induced innate immune responses are typically attributed to P2X7R activation. However, eATP can also activate other P2X and P2Y purinergic receptors. 32 Interestingly, maximum induction of most examined genes in response to eATP treatment was dose and cell-type dependent, suggesting that different concentrations of eATP may activate different P2 receptor(s) in the HKM and PBL cells. Indeed, we showed previously that different P2X and P2Y receptor subtypes, including P2X7R, P2X4R, P2X2R, P2Y2R, and P2Y12R, are co-expressed in Japanese flounder immune cells.11–13,33
In the present study, maximal gene expression of IL-1β, IL-6 and TNF-α was elicited by low concentrations of ATP treatment in PBL cells, indicating that low concentrations of ATP are more potent in induction of these genes, and P2X2 and P2X4 receptors, but not P2X7 receptor, may involve in this process. Maximal gene expression of IL-11, CSF, and p65 in PBLs and IL-1β, Mx, and IFN in HKMs was induced by high concentrations (1 mM) of ATP treatment, indicating that high concentrations of ATP are more potent in induction of these genes, and P2X7R with an estimated EC50 of 743 ± 299 μM for ATP may implicate in these processes. 11 It was worth to mention that IL-11, CSF, p65, IL-1β, Mx and IFN gene expression could also be induced by BzATP treatment (see Supplementary Figure 1 online), a potent agonist for P2X7R, supporting a role of P2X7R in regulating of these gene expression. In addition, as ATP is a key signaling molecule in the activation of NLR inflammasomes, which are responsible for pro-inflammatory cytokine production and release, NLRs may also engage in eATP-induced cytokine gene expression in fish.
NO and ROS are important biological active molecules in fish immune defense system.34–38 We therefore examined whether eATP regulates ROS and NO production in Japanese flounder HKM cells. NO is a free radical gaseous signal generated via NO synthase (NOS). In our study, we found that inducible NOS (iNOS) activity was enhanced by eATP treatment, and this up-regulation of iNOS was correlated with increased production of NO. We further showed that eATP also elicits significant ROS production in Japanese flounder HKM cells, indicating that eATP-mediated purinergic signaling is implicated in regulation of NO and ROS production in fish immune cells. It has been reported that eATP activates P2 receptors, leading to increasing intracellular [Ca2+] that stimulates NOS to produce NO in endothelial cells. 39 In addition, ROS and NO production in HKM cells also differs in response to differential doses of ATP stimulation and duration.
Multiple P2X and P2Y receptor subtypes are often expressed on the same cell type and couple to diverse signaling pathways. 6 Previous studies have also demonstrated the involvement of P2X4R, P2X7R, pannexin1, and P2Y2R in eATP-elicited ROS production.5,40 Different P2X receptors are activated by different concentrations of eATP in a range of millimole to micromole. Our previous studies confirmed co-expression of multiple P2XRs in Japanese flounder HKM and PBL cells. 12 Using electrophysiological recording, we also showed that the Japanese flounder P2X7R has an estimated EC50 of 743 ± 299 μM for ATP. 11 Interestingly, maximum ROS production in HKM cells was achieved by 200 μM ATP stimulation, while long-lasting ROS production was obtained by 1000 μM ATP application. This observation suggests that P2XR subtypes with different ATP binding affinities could account for this difference. For example, P2 receptors (i.e., P2X2R and P2X4R) activated by low threshold eATP concentrations (in a micromolar range) may perform an important role for eATP-induced ROS production at early stage, while P2X7R activated by high concentration eATP (in a millimolar range) may require for ROS production in later stages in Japanese flounder HKM cells. Again, as Japanese flounder immune cells co-express multiple P2X and P2Y receptors,12,33 currently we cannot exclude the involvement of P2Y receptor(s) in eATP-induced ROS and NO production in the Japanese flounder. Thus, eATP-induced innate immune responses in Japanese flounder immune cells may result from the activation of a combination of different P2 receptor(s). Using small interfering RNA to down-regulate the endogenous expression of individual P2 receptors in the immune cells will be helpful to determine the contribution of each P2 receptor in the eATP-induced innate immune responses in Japanese flounder.
In summary, we have shown that ATP is dynamically released during bacterial pathogen challenge in Japanese flounder head kidney cells. Our findings also confirm that eATP is a potent and conserved extracellular signaling molecule in the activation of fish innate immunity. Further investigation of the molecular mechanisms that regulate eATP-mediated innate immune responses in Japanese flounder will improve our understanding of the purinergic signaling pathway(s) in fish innate immunity.
Supplemental Material
INI804635 Supplemental Material - Supplemental material for Extracellular ATP is a potent signaling molecule in the activation of the Japanese flounder (Paralichthys olivaceus) innate immune responses
Supplemental material, INI804635 Supplemental Material for Extracellular ATP is a potent signaling molecule in the activation of the Japanese flounder (Paralichthys olivaceus) innate immune responses by Shuo Li, Xiaoli Chen, Jiafang Li, Xuejing Li, Tianxu Zhang, Gaixiang Hao and Jinsheng Sun in Innate Immunity
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 31572645), the Natural Science Foundation of Tianjin (Grant No. 18JCZDJC33600), the University Yong Talent Cultivation Program provided by Tianjin Municipal Education Commission, the Science Promotion Program for Young Scholars provided by Tianjin Normal University (Grant No. 52XC1503), the Tianjin Development Program for Innovation and Entrepreneurship, and the Innovation Team of Tianjin Fisheries Research System (Grant No. ITTFRS2017007).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
