Abstract
Here, the regulatory role of autophagy is examined in both an LPS-induced lethal endotoxic shock mouse model and cecal ligation and puncture (CLP) mouse model. Autophagy-inhibitor 3-methyladenine (3-MA) and autophagy-enhancer rapamycin were administrated to mice challenged with LPS or CLP. Animals challenged with LPS or CLP combined with 3-MA displayed increased survival after endotoxemia, but LPS combined with rapamycin worsened the endotoxic shock of the mice. Among the different organs studied, the lungs and intestines exhibited significant differences among LPS alone, LPS combined with 3-MA and LPS combined with rapamycin. LPS combined with 3-MA attenuated the inflammatory damages of these organs as compared with LPS alone. In contrast, LPS combined with rapamycin increased damage in these organs. Consistently, serum inflammatory mediators TNF-α and IL-6 were decreased by the treatment of LPS combined with 3-MA as compared with LPS alone, while administration of LPS combined with rapamycin increased the serum TNF-α and IL-6 levels. Similar results were found in mouse bone marrow-derived macrophages exposed to LPS. Moreover, the regulatory effect of autophagy to endotoxic shock is dependent on the TLR4 signaling pathway. Our results demonstrate the central role of autophagy in the regulation of endotoxic shock and its potential modulation for endotoxic shock treatment.
Introduction
Endotoxic shock remains a significant cause of death despite the use of various therapeutic approaches. 1 Sepsis is characterized by dysregulation of inflammation following primarily bacterial infection. The specific reasons for uncontrolled inflammation and death in some septic patients are not clear. 2 Autophagy, an evolutionarily conserved homeostasis process regulating biomass quantity and quality, plays a critical role in the host response to sepsis. 3 A central role of autophagy in the regulation of the lung response to endotoxemia and sepsis and its potential modulation by nutrition has been demonstrated. 4 Sepsis-induced autophagy is initiated by binding of PAMPs within the microbial structure to PRRs, such as TLRs. This in turn activates different intracellular events and leads to increased autophagic activity by promoting the conversion of LC3-I to LC3-II.5,6
Both cell and animal models suggest that autophagy is induced in the early stage of sepsis. This appears to involve predominantly the liver and the heart. The late-stage exhaustion of autophagic activity is associated with inflammatory dysregulation, histological changes, mitochondrial dysfunction and apoptosis. Experimental induction of autophagy has increased the survival rate among septic animals. 7 Blocking autophagy accelerated apoptosis and increased mortality in concordance with the insufficient autophagy process in CD4+T cells in the murine sepsis model, suggesting that T-cell autophagy plays a protective role against apoptosis and immunosuppression in sepsis. 8 Autophagy-related protein 7 (Atg7) is involved in inflammasome activation in Pseudomonas aeruginosa abdominal infection. Loss of Atg7 results in increased production of IL-1β and pyroptosis, consistent with enhanced inflammasome activation. Furthermore, aeruginosa flagellin is a chief trigger of inflammasome activation in the sepsis model. 9 However, neutrophil autophagy primes neutrophils for increased neutrophil extracellular trap (NET) formation, which is important for proper neutrophil effector functions during sepsis. 10 Interestingly, autophagic suppression may result in a positive outcome. Inhibition of autophagy by chloroquine ablates the release of high mobility group box 1 (HMGB1) from macrophages and endothelial cells in Balb/C mice, which in turn prevents degradation of the repressor protein NF-κBI/IkB. Consequently, NF-κB-induced cytokine release is inhibited. 11 Nevertheless, whether the process of autophagy is generally beneficial or harmful to the immune defense and other cell functions during sepsis is still not well defined.
To characterize the functional implication of autophagy in the systemic response to endotoxemia, we compared the effects on endotoxic shock between the autophagy-inhibitor 3-methyladenine (3-MA) and autophagy-enhancer rapamycin. Surprisingly, administration of 3-MA protects mice from endotoxic shock, while application of rapamycin worsens the survival of endotoxic mice. Moreover, this protection effect of 3-MA is dependent on the TLR4 signaling pathway.
Materials and methods
Mice
Male C57/BL6 mice 5–6 wk of age were purchased from the Centers for Disease Control, Wuhan (Hubei, China). C57/B6.129-Tlr2tmIkir/JNju (TLR2–/–) and C57BL/10ScNJNju (TLR4–/–) mice were purchased from the Biomedical Research Institute (Nanjing, China). Mice were kept in specific pathogen-free (SPF) facilities and all the experiments in this study were performed according to the guidelines for animal use in research set by Tongji Medical College, Huazhong University of Science and Technology (China).
Reagents and Abs
LPS, 3-MA and rapamycin were purchased from Sigma-Aldrich (St. Louis, MO, USA). Abs against LC3B I/II and β-actin were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Induction of experimental endotoxic shock and determination of LD50
According to our previously described investigation, 12 the animals were injected i.p. with different doses of Escherichia coli 0111:B4 LPS (Sigma-Aldrich, St. Louis, MO, USA). After 90 min and 180 min, mice were anesthetized with methoxyflurane and bled by cardiac puncture, and serum TNF-α, IL-10 and IL-6 levels were determined by ELISA. Survival over 48 h was evaluated and LD50 was determined. Experimental groups were designed as follows: (1) PBS, (2) LPS, (3) LPS combined with rapamycin (2 mg/kg), (4) LPS combined with DMSO (0.01 ml/g) and (5) LPS combined with 3-MA (15 mg/kg).
Cecal ligation and puncture (CLP) model
CLP was performed as previously described. 13 About 75% of the cecum was ligated and two through and through punctures were made in the cecum using a 21-gauge needle, resulting in high-grade sepsis. Mice in the sham group underwent the same procedures except for the CLP. 3-MA (20 mg/kg), rapamycin (2 mg/kg) or DMSO (0.01 ml/g) were administered intraperitoneally 30 min before CLP.
Histological and immunohistochemical analysis
For the histological examinations, the lungs and small intestine were removed and then fixed overnight in 4% (w/v) paraformaldehyde in PBS and embedded in paraffin. Sections (3 µm) were cut and stained with hematoxylin and eosin (H&E). The histological score of H&E-stained sections of the colon was determined by a pathologist in a blinded fashion, according to a previously described method. 13 Immunohistochemical staining was performed by established technique. 13 The sections were stained in PBS containing Ab against LC3 BI/II, then incubated with FITC- and rhodamine-conjugated secondary Ab and examined by an Olympus confocal microscope.
Generation of mouse bone marrow-derived macrophages (BMs) and stimulation with LPS
Mouse BMs were propagated from bone marrow cells as described previously. 14 Recombinant cytokine M-CSF in vitro experiments were obtained from PeproTech (London, UK). BMs were stimulated with LPS (1 µg/ml) for 24 h. Experimental groups were designed as follows: (1) mock group, (2) LPS (1 µg/ml) alone group, (3) LPS combined with 3-MA (5 mM) and (4) LPS combined with rapamycin (500 ng/ml) group. Thereafter, the supernatants were harvested and stored at –80℃ for ELISA.
Western blotting
Lung tissue protein extraction was performed prior to Western blotting assay conducted as described previously. 15 The primary Abs included rabbit anti-LC3B (Sigma-Aldrich, USA), anti-Beclin1 (Sigma-Aldrich, USA), and anti-GAPDH (Servicebio, China). After incubating with secondary Ab conjugated with HRP, immunoreactivity was developed by an enterochromaffin-like system (Pierce).
ELISA
Blood samples were collected by cardiac puncture and placed at room temperature 30 min before centrifugation. The serum was stored at –80℃ until analyzed. Quantification of cytokines TNF-α, IL-6 and IL-10 in sera and cell culture supernatants was performed utilizing an ELISA kit (BioLegend, San Diego, CA, USA). All ELISA experiments were performed according to the manufacturer’s instructions.
Statistical analysis
Results are shown as mean ± SD. Statistical significance of differences were analyzed by a one-way ANOVA followed by Dunnett post-hoc test (for more than two groups’ comparisons) or Student t test (for two groups’ comparisons). The software package GraphPad Prism 6 (GraphPad Software, San Diego, CA, USA) was used for data analysis. Values of P < 0.05 were considered significant. Mice survival differences between groups were determined using the log-rank test.
Results
Determination of LPS LD50 of mice with septic shock
We first checked the dose of LD50 of LPS in C57BL/c mice. The mice were challenged with 25 mg/kg, 27 mg/kg and 30 mg/kg LPS (i.p. injection), and it was found that the 27 mg/kg dose of LPS resulted in the death of about 50% of the mice (Figure 1a). Consistently, histology of the lungs and intestines displayed the expected pathological changes (Figure 1b, c). Therefore, we used 27 mg/kg of LPS as LD50 for the following experiments. After treating the mice with 27 mg/kg LPS, serum TNF-α, IL-6 and IL-10 levels were determined by ELISA, as shown in Figure 1b (TNF-α and IL-10 levels peaked at 1.5 h after LPS administration, while IL-6 peaked at 3 h after LPS application (Figure 1d)).
Determination of LPS LD50 in C57BL/6 mice. The mice were challenged i.p. with different doses of LPS to monitor the survival rates and organ injury to determine the LD50. (a) Mice were challenged with PBS, 25 mg/kg, 27 mg/kg and 30 mg/kg LPS, respectively, and the survival curves were plotted. (b) Twenty-five mg/kg, 27 mg/kg and 30 mg/kg LPS were injected i.p., and the lungs and intestines (c) were taken for pathological investigation. (d) Twenty-seven mg/kg LPS was injected i.p., and serum TNF-α, IL-6 and IL-10 were determined by ELISA at different time points; the levels of cytokines are summarized by the plots. Magnification: ×200. The experiments were replicated at least twice.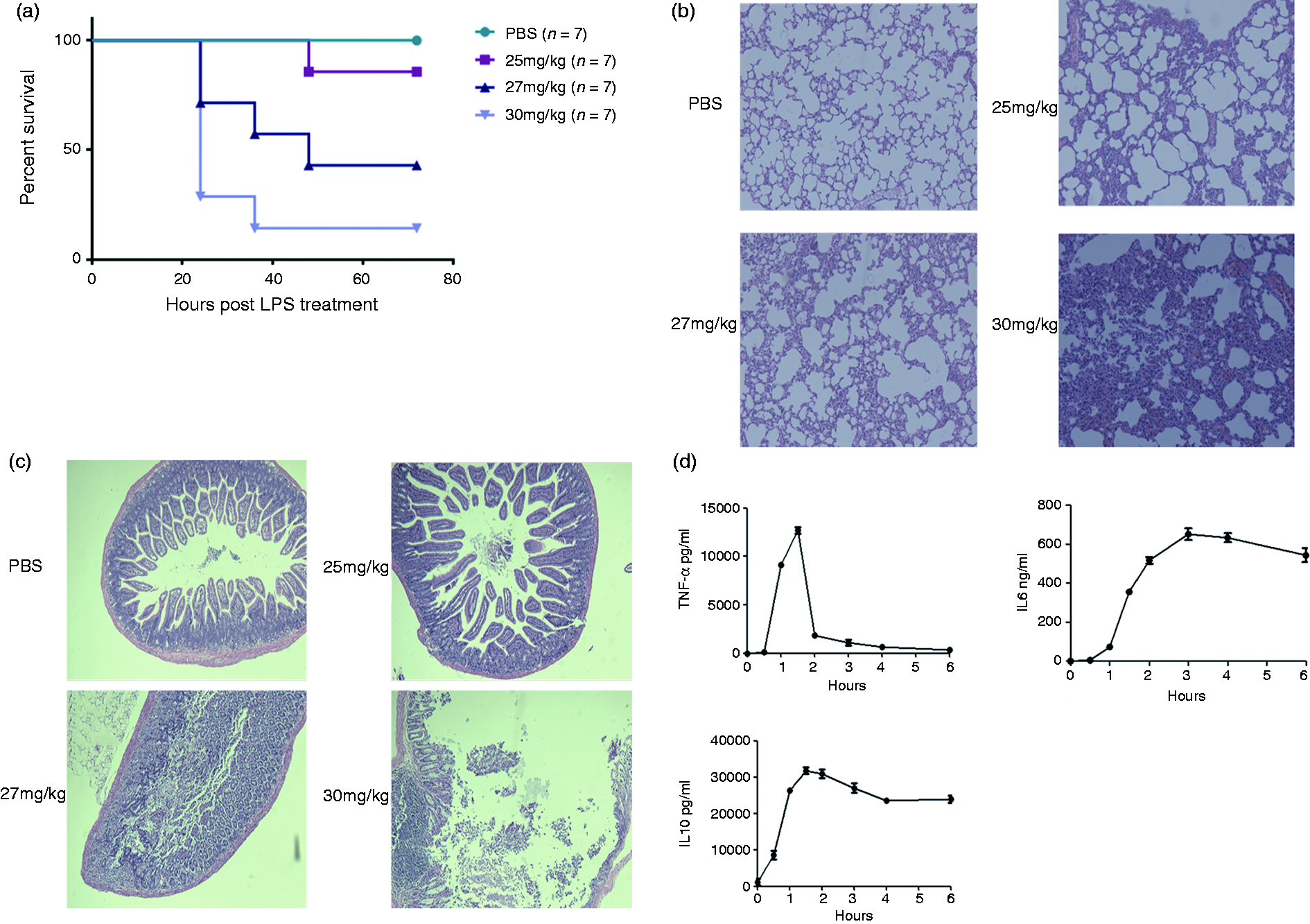
Inhibition of autophagy significantly protected mice from endotoxic shock
We next examined the effect of the autophagy-inhibitor 3-MA. For this purpose, mice were challenged with PBS, LPS alone, LPS combined with rapamycin (2 mg/kg), LPS combined with DMSO, and LPS combined with 3-MA (15 mg/kg). It was found that application of LPS combined with 3-MA significantly protected the mice from endotoxic shock as compared with LPS alone (Figure 2a). In contrast, treatment of mice with LPS combined with rapamycin resulted in mice susceptible to LPS (Figure 2a). In support of this observation, pathological analysis also exhibited that LPS combined with 3-MA significantly attenuated damage to the lungs and intestines (Figure 2b and c). On the contrary, treatment of mice with LPS combined with rapamycin exacerbated the inflammation of lungs and intestines (Figure 2b and c). The CLP model was also used to investigate the effect of 3-MA on septic shock. Mouse survival differences between mice subjected to CLP and control mice were compared. The median survival of CLP group was 2 d, and application of 3-MA significantly prolonged the median survival (4 d, Figure 2d).
Mice challenged with LPS combined with autophagy-inhibitor 3-MA or LPS combined with rapamycin showed different survival and organ damage. (a) Mice (n = 8 per group) were challenged with PBS, 27 mg/kg LPS alone, 27 mg/kg LPS combined with 3-MA, 27 mg/kg LPS combined with rapamycin, and 27 mg/kg LPS combined with DMSO, respectively, and the survival curves were plotted. Lungs (b) and intestines (c) were taken for pathological analysis. (d) The survival rates after cecal ligation and puncture (CLP) in mice. 3-MA, rapamycin or DMSO were administered i.p. 30 min before CLP. Mice survival differences between groups were determined using the log-rank test. Magnification: ×200. *P ≤ 0.05. The experiments were replicated at least twice.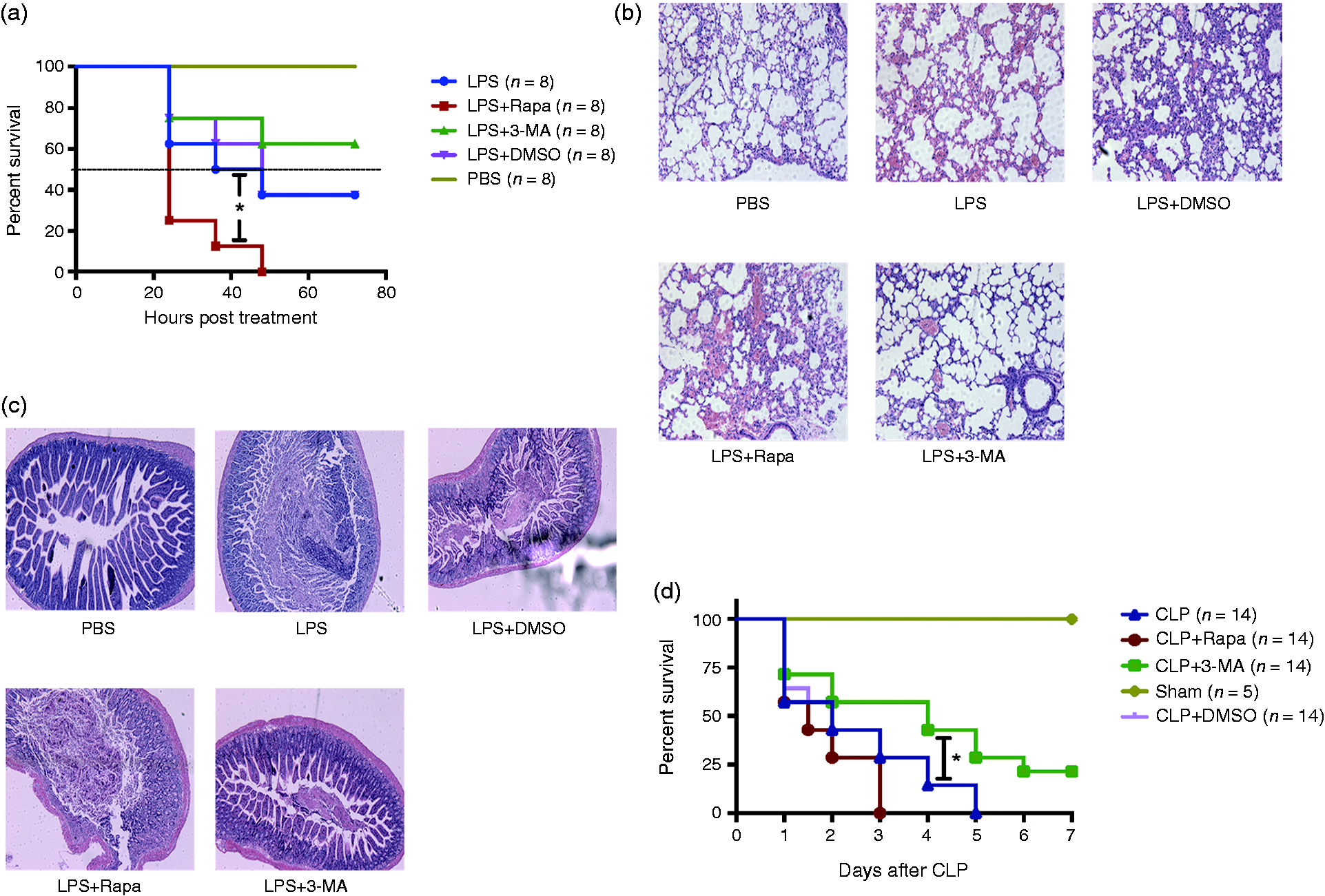
Administration of 3-MA decreased the production of serum TNF-α and IL-6 levels in mice with endotoxic shock
To further demonstrate that treatment of mice with 3-MA protects them from endotoxic shock, we determined the serum inflammatory cytokines of the mice challenged with LPS combined with 3-MA. To this end, blood was harvested at 1.5 and 3 h after mice had been injected i.p. with PBS, LPS alone, LPS combined with 3-MA, or combined with rapamycin, DMSO, respectively, and the cytokines levels were determined with ELISA. In line with the above mice survival results, LPS combined with 3-MA led to dramatically decreased production of inflammatory cytokines TNF-α and IL-6 as compared with LPS alone (Figure 3a).
Cytokine levels in serum of endotoxic shock mice and in supernatant of macrophages stimulated with LPS. (a) Mice were injected i.p. with PBS, 27 mg/kg LPS alone, 27 mk/kg LPS combined with autophagy-inhibitor 3-MA, 27 mg/kg LPS combined with rapamycin, and 27 mg/kg LPS combined with DMSO, respectively. The cytokine levels in serum were determined by ELISA and summarized by plotting. (b) BMs were stimulated with LPS (1 µg/ml) alone, LPS + 3-MA (5 mM), and LPS + rapamycin (500 ng/ml), respectively, and unstimulated BMs were used as mock controls. After 24-h culture, the supernatants were harvested to examine cytokine levels by ELISA, which were summarized by plotting. Values were means ± SD, *P ≤ 0.05, ** P ≤ 0.01, ***P ≤ 0.001. The experiments were replicated at least twice.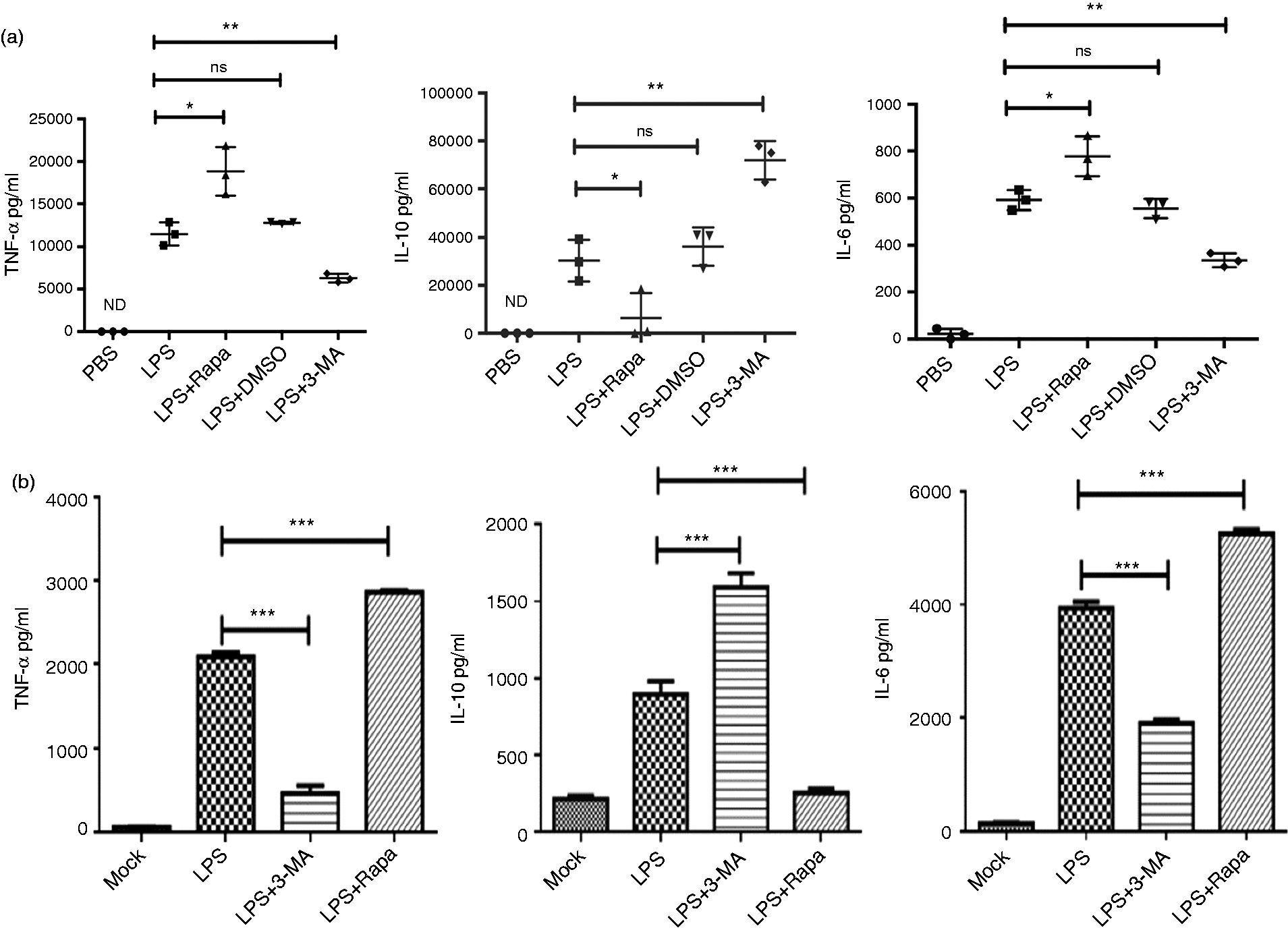
BMs exhibited a distinct production of inflammatory cytokines on LPS combined with 3-MA or rapamycin
Based on the above results, we next sought to investigate whether application of 3-MA or rapamycin regulates the production of inflammatory cytokines in BMs stimulated with LPS (1 µg/ml). The macrophages produced decreased amounts of inflammatory cytokines TNF-α and IL-6 in the presence of LPS combined with 3-MA (5 mM) as compared with LPS alone, while LPS combined with 3-MA increased the production of IL-10. In contrast, stimulation of macrophages on LPS combined with rapamycin (500 ng/ml) exhibited an increase of secretion of TNF-α and IL-6 as well as a decrease of IL-10 (Figure 3b). These results demonstrated that regulation of autophagy of macrophages on LPS stimulation can change the cytokine production pattern of macrophages.
Regulated autophagy resulted in endotoxic mice displaying distinct organ damage
Given the fact that regulated autophagy of macrophages exhibited different cytokine production profiles, we then examined the expression of autophagy-related protein LC3 BI/II after administration of 3-MA and rapamycin. We first conducted the immunochemistry of the lungs and intestines of endotoxic mice to detect the expression of LC3 BI/II. Expectedly, application of 3-MA inhibited the expression of LC3 BI/II of lungs and intestines as compared with LPS alone. In stark contrast, treatment with rapamycin enhanced the expression of LC3 BI/II in the lungs and intestines of the mice challenged with LPS (Figure 4a). To further confirm the autophagy-regulatory effect of 3-MA and rapamycin on the autophagy-related protein LC3 BI/II, Western blotting was used to detect the expression of LC3 BI/II by macrophages stimulated with LPS in the presence of 3-MA or rapamycin. The expression of LC3 BI/II was significantly lower in the presence of 3-MA than with treatment with LPS alone; on the contrary, LPS in the presence of rapamycin dramatically enhanced the expression of LC3BI/II as compared with LPS alone (Figure 4b).
LPS combined with autophagy-inhibitor 3-MA or LPS combined with rapamycin inhibited or enhanced autophagy, respectively, in endotoxic shock mice. (a) Mice were injected i.p. with PBS, 27 mg/kg LPS alone, 27 mg/kg LPS combined with 3-MA, 27 mg/kg LPS combined with rapamycin, and the lungs and intestines (b) were taken for immunohistochemical analysis of LC3BI. Magnification: ×200. Image-Pro Plus 6.0 was used to analyze the integrated optical density. (c). BMs were treated with LPS (1 µg/ml) combined with 3-MA (5 mM) or LPS combined with rapamycin (500 ng/ml) versus the mock group, respectively. The proteins were extracted from these cells for Western blot analysis on autophagy-related proteins beclin-1 and LC3 BI/II. The relative intensity of the bands of beclin-1 and LC3 BI/II are shown by semiquantified analysis utilizing the software Image J. Values were means ± SD, *P ≤ 0.05, **P ≤ 0.01. The experiments were replicated at least twice.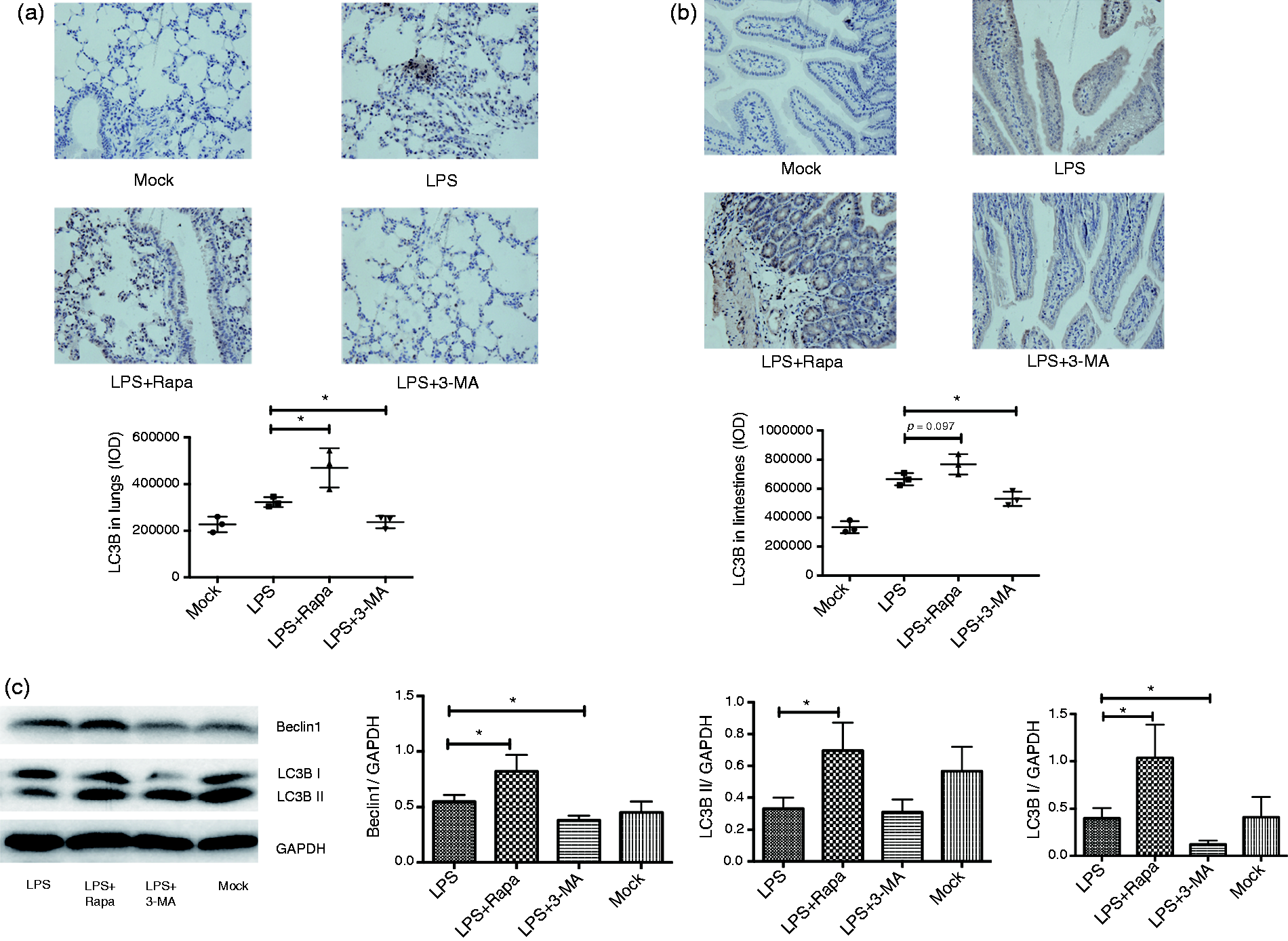
TLR4 signaling pathway is involved in autophagy-regulated TNF-α secretion of macrophages upon LPS stimulation
Because the pro-inflammatory cytokine TNF-α plays a critical role in the process of endotoxic shock, we explored the possible molecular mechanisms by which the autophagy-regulators 3-MA and rapamycin regulate TNF-α production. For this purpose, macrophages derived from TLR4–/–, TLR2–/– and wild type mice were incubated with LPS (1 µg/ml) combined with 3-MA (5 mM), LPS (1 µg/ml) combined with rapamycin (500 ng/ml), LPS (1 µg/ml) alone as well as a unstimulated negative control (mock group) for 24 h, respectively. Thereafter, the supernatants were harvested to examine the levels of TNF-α by ELISA. Inhibition of the autophagy of macrophages from wild type and TLR2–/– mice with 3-MA led to significantly decreased TNF-α production as compared with LPS alone (Figure 5). In contrast, the TNF-α amount produced by TLR4–/– macrophages treated with LPS combined with 3-MA exhibited no significant difference compared with TLR4–/– macrophages treated with LPS alone or LPS combined with rapamycin (Figure 5). These results indicated that the TLR4 but not TLR2 signaling pathway is necessary for modulation of the autophagy to regulate TNF-α production by macrophages.
Autophagy-inhibitor 3-MA inhibiting TNF-α production by macrophages is dependent on the TLR4 signaling pathway. BMs were prepared from TLR4–/–, TLR2–/– and wild type mice, respectively. These macrophages were stimulated with LPS (1 µg/ml) alone, LPS combined with 3-MA (5 mM), and LPS combined with rapamycin (500 ng/ml), respectively. The supernatants were harvested after 18 h to determine TNF-α levels by ELISA. The results were summarized by plotting. Values were means ± SD, **P ≤ 0.01, ***P ≤ 0.001. The experiments were replicated at least twice.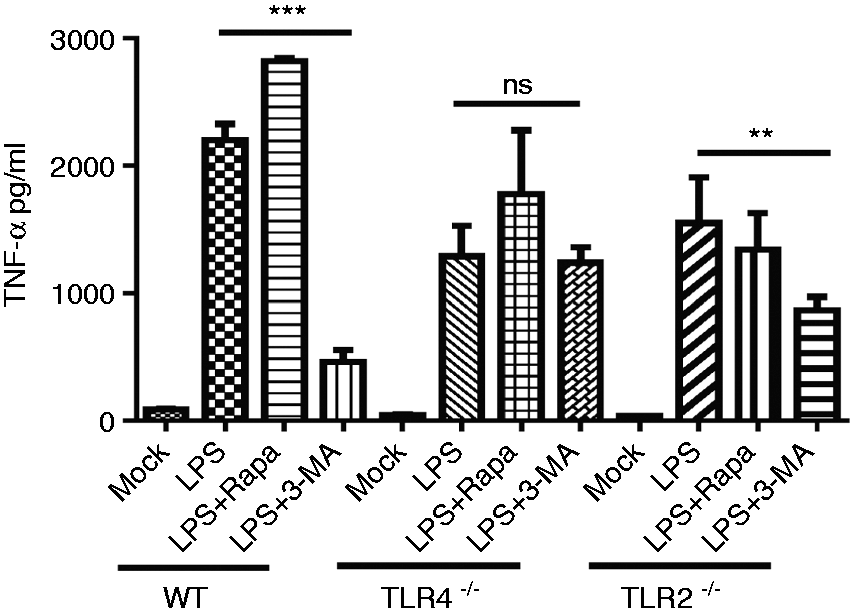
Discussion
In the present study, we investigated the effects and mechanisms of autophagy on experimental endotoxic shock induced by LPS in the mouse. We demonstrated that treatment with 3-MA, an autophagy inhibitor, ameliorated the severity of organ damage caused by endotoxic shock, improved the survival of the mice with endotoxic shock, and decreased the serum levels of inflammatory cytokines. This protection of endotoxic shock was associated with suppression of inflammatory mediators including IL-6 and TNF-α secreted by macrophages. In contrast, rapamycin, an autophagy enhancer, had the opposite effects on the LPS challenge.
The treatment of sepsis should be based on the understanding of its pathogenesis. The pathogenesis of sepsis, however, is not fully understood. Antibiotics kill bacteria in blood plasma, but insufficiently penetrate erythrocytes for killing bacteria there. Constant bacterial reservoirs in erythrocytes decrease antibacterial and immune therapy effectiveness and may be one of the factors that make sepsis therapy so problematic. 16 Therefore, it is important to understand the mechanisms underlying the pathogenesis of sepsis and develop new therapeutic strategies.
3-MA is used to block autophagy in vivo. 17 LC3-II levels are used as evidence of autophagy. 18 Thus, we employed 3-MA to inhibit autophagy in the context of endotoxic shock in mice and evaluated its inhibiting effect on autophagy by LC3BI/II expression. Our results confirmed the effects of 3-MA on autophagy in the setting of in vivo and in vitro. Our novel finding is that serum inflammatory mediators such as TNF-α and IL-6 were significantly reduced in endotoxic shock mice as well as in macrophages stimulated with LPS after application of 3-MA, as compared to LPS alone. Compared to genetic ablation of an autophagy-related gene such as LC3B–/–, 19 pharmacologic inhibition of autophagy could be clinically used for patients. Notably, in addition to inhibition of autophagy, application of 3-MA has other biomedical effects. In line with our results, it has been reported that the N-terminal 8-amino-acid N8 peptide derived from p22phox blocks the Rubicon-p22phox interaction, profoundly suppressing reactive oxygen species and inflammatory cytokine production. Consequently, treatment with Tat-N8 peptide or mimetic compounds from in-silico screening dramatically reduced the mortality of CLP-induced polymicrobial sepsis in mice. 20 However, augmenting autophagy has protective roles for kidney injury and the heart.21–24
Autophagy is a highly conserved mechanism of eukaryotic cells implicated in cell homeostasis and elimination of intracellular pathogens. Functional polymorphisms in genes encoding for autophagy have been associated with susceptibility to inflammatory and infectious diseases. 25 Regarding the association between genetic polymorphism and clinical course/outcome in severely septic patients, previous studies have demonstrated that areas of the ATG16L1 gene, implicated in autophagy, are correlated with an increased risk for septic shock and multiple organ failure development in the case of ventilator-associated pneumonia. 25 Similarly, IRGM(+313), an autophagy-related polymorphic locus, influences the outcome in severely septic patients, with a possible involvement of autophagy in sepsis exacerbation. 26 These findings support the notion that autophagy is involved in endotoxic shock.
Rapamycin was originally isolated from the soil bacterium Streptomyces hygroscopicus as an antifungal compound and was later shown to be a strong immunosuppressant with broad antiproliferative effects in mammalian cells. 27 Induction of autophagy by rapamycin or rapalogs has been tested in various model systems. Treatment with rapamycin strongly induces autophagy in yeast even in the presence of nutrients. 28 Rapamycin regulates the mechanistic target of rapamycin (mTOR) signaling pathway, which plays a crucial role in regulating autophagy. 29 Autophagy inductions could be beneficial or detrimental depending on the type or stage of disease. 30 We observed that at the earlier acute stage of endotoxic shock, enhancing autophagy using rapamycin worsened the endotoxic shock in mice. However, in addition to promoting autophagy, rapamycin has a strong immunosuppressant effect, and this could be responsible for such a detrimental outcome. However, enhancing autophagy with activated protein C and rapamycin protects against sepsis-induced acute lung injury. 31
What is the mechanism by which autophagy regulators (i.e. 3-MA and rapamycin) regulate the production of TNF-α by LPS-stimulated macrophages in the context of endotoxic shock? To answer this question, we examined the effects of these autophagy regulators on TLR4 and TLR2 signaling pathways, which are critical to producing TNF-α in macrophages. LPS-induced activation of TLR4 is followed by a subsequent overwhelming inflammatory response, a hallmark of the first phase of sepsis. Our results indicated that 3-MA and rapamycin may interfere with the TLR4 signaling pathway to regulate the TNF-α production in LPS-activated macrophages. Likewise, autophagy causes PELI3 degradation during TLR4 signaling, thereby impairing the hyperinflammatory phase during sepsis. 32 LPS induced the activation of mTOR in wild-type peritoneal macrophages, but not in IFN regulatory factor 1 (IRF-1) knockout macrophages. In contrast, overexpression of IRF-1 alone increased the activation of mTOR and consequently decreased autophagic flux. Furthermore, the inhibitory effects of IRF-1 mTOR activity were mediated by NO. Therefore, there could be a novel role for IRF-1 and NO in the regulation of macrophage autophagy during LPS stimulation in which IRF-1/NO inhibit autophagy through mTOR activation. 33 In our study, we observed that lack of TLR4 signaling results in the defect of autophagy activity in regulating the production of TNF-α by macrophages. Future studies are warranted to investigate the molecular mechanisms underlying how autophagy regulates the generation of pro-inflammatory mediators by LPS-stimulated macrophages in the setting of endotoxic shock.
In conclusion, we present strong evidence that autophagy plays a crucial role in the process of TNF-α and IL-6 secretion from macrophages in the context of LPS-induced experimental endotoxic shock in mice. Therefore, manipulation of autophagy might have therapeutic implications in patients with endotoxic shock.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (91542110 to M. Fang), the Ministry of Science and Technology of China (grant no. 2013CB530505) and the National Natural Science Foundation of China (81373167 to M. Fang).
