Abstract
Hirsutella sinensis, cultured in vitro, is an attractive substitute for Cordyceps sinensis as health supplement. The aim of this study was to demonstrate whether H. sinensis mycelium (HSM) attenuates murine pulmonary fibrosis induced by bleomycin and to explore the underlying molecular mechanisms. Using lung fibrosis modle induced by intratracheal instillation of bleomycin (BLM; 4 mg/kg), we observed that the administration of HSM reduced HYP, TGF-β1 and the production of several pro-fibrosis cytokines (α-smooth muscle actin, fibronectin and vimentin) in fibrotic mice lung sections. Histopathological examination of lung tissues also demonstrated that HSM improved BLM-induced pathological damage. Concurrently, HSM supplementation markedly reduced the chemotaxis of alveolar macrophages and potently suppressed the expression of inflammatory cytokines. Also, HSM influenced Th1/Th2 and Th17/Treg imbalance and blocked the phosphorylation of mTOR pathway in vivo. Alveolar epithelial A549 cells acquired a mesenchymal phenotype and an increased expression of myofibroblast markers of differentiation (vimentin and fibronectin) after treatment with TGF-β1. HSM suppressed these markers and blocked the phosphorylation of mTOR pathway in vitro. The results provide evidence supporting the use of HSM in the intervention of pulmonary fibrosis and suggest that HSM is a potential therapeutic agent for lung fibrosis.
Introduction
Pulmonary fibrosis (PF) is chronic lung damage followed by multi-pathogenesis, which leads to the loss of pulmonary function with dyspnea and hypoxemia, and finally respiratory failure and death. 1 Although, the prevalence and mortality of PF has recently increased, effective cure is lacking owing to its complicated pathological mechanism. Most patients pass away owing to PF within 5 yr of diagnosis. 2 Some kinds of pharmacological therapies have been proposed to treat PF; however, owing to their side effects and lack of clinical support, these therapies were dismissed.3,4 Because of these difficulties, traditional Chinese medicine has been considered to be an alternative for the treatment of PF disease.
There are several vital pulmonary indicators, such as collagen, hydroxyproline (HYP) and pro-fibrosis cytokines. Collagen, as a primary component of extracellular matrix (ECM), and HYP is a characteristic of collagen fiber. 5 In addition to the collagen containing only about 1% HYP, all the others do not include any HYP. 6 Therefore, lung HYP levels can be used as a reliable biochemical indicator of collagen content. TGF-β1 is one of the major fibrotic cytokines during the induction and development of PF. It is known to induce excessive production and deposition of collagen and ECM, and furthermore contribute to the recruitment of inflammatory cells.7,8 The expression of α-smooth muscle actin (α-SMA) increased during fibroblasts differentiate into myofibroblasts; meanwhile, both fibroblasts and myofibroblasts are primary sources of ECM proteins, such as fibronectin and collagen. 9
Moreover, the immunological balance also influences lung fibrosis. IFN-γ produced by Th1 cells can inhibit TGF-β expression, inhibit collagen deposition and relieve PF. However, mononuclear macrophages, B cells and T cells secrete IL-10, resulting in the activation of Th2 cells. Inhibition of the inflammatory cells leads to secretion of pro-inflammatory factors, which move the Th1/Th2 balance response toward a Th2 cells response, thereby promoting PF. 10
The mammalian target of rapamycin (mTOR) signaling pathway plays a core role in regulating cell proliferation, motility and survival. 11 Previous reports have shown that mTOR stimulation is associated with fibrotic diseases, including cardiac, liver and kidney fibrosis. Gui et al. proved that mTOR activation in alveolar epithelial cells and autophagy in the pulmonary is associated with the development of PF. 12 Moreover, accumulating evidence supports the speculation that the mTOR signaling pathway is closely linked with PF.
Cordyceps sinensis, a Chinese endemic species, is distributed in the Qinghai-Tibet plateau of Tibet, Qinghai, Sichuan, Yunnan, Gansu provinces and alpine meadow of the rim (Nepal, Bhutan, etc.). Medical entomogenous fungus, whose mycelia grow inside the larvae of lepidoptera moth, form a complex of the larva body and dry stroma. 13 Cordyceps sinensis has been shown in recent years to be an antioxidant and an inhibitor of angiogenesis and has been used to cure respiratory diseases.
Researchers have proved that C. sinensis can ameliorate fibrosis in the liver, pulmonary and kidney through inhibition of TGF-β1 expression and promotion of collagen degradation.14,15 The growth of C. sinensis natural fungus is unusual owing to its limited ability to grow on Tibetan plateau and southwestern China. Hence, Hirsutella sinensis culturing in vitro became an attractive substitute for the preparation of health supplements. In previous studies, we showed that H. sinensis mycelium (HSM) relieves PF by suppressing inflammation and the function of Treg cells. Huang et al. showed that C. sinensis decreased the expression of fibrogenic cytokines in lung fibroblasts. 9 In addition, HSM extract plays the role of an anti-inflammatory effectively owing to its capacity to inhibit both canonical and non-canonical inflammasomes. 9 Cordyceps sinensis pre-treatment protection against pulmonary injury may be associated with Smad and Akt phosphorylation. 16 The PI3K/Akt/mTOR signaling pathway plays a vital role in lung fibrosis, thereby instigating us to explore whether the effects of HSM on PF is associated with another underlying mechanism, including key molecules in the mTOR pathway.
The aim of the current study was to investigate whether intra-gastric administration of HSM attenuates fibrosis in mice and explore the underlying molecular mechanisms of this protective effect. Our results show that HSM can effectively regulate the function of immunocytes, suppress inflammation and ameliorate PF. Moreover, the effect of HSM on the formation of lung fibrosis may be associated with the inhibition of phosphorylation of mTOR p70S6K.
Materials and methods
Animals
Six–eight-wk-old C57BL/6J mice (male) weighing between 18 g and 22 g were purchased from the Animal Model Research Institute of Nanjing University (Nanjing, China) and fed for at least 1 wk before use. Procedures for animal care and use in the study were approved by the Ethics Committee of Animal Experiments of Nanjing University. The animals were kept under light–dark cycles of 12 h each at 21–23℃.
Preparation of the HSM
The HSM was originally obtained from Nanjing Zhongke Group (Nanjing, China), The HSM solution was prepared by dissolving HSM powder into saline at a concentration of 40 mg/ml, at 37℃ for 2 h, cooled to room temperature and centrifuged (Thermo Fisher Scientific, Waltham, MA, USA) at 500 g for 10 min at 4℃. Then, transport the supernatant to 5 mm filter (Millipore, Billerica, MA, USA) for sterilization and stored at –20℃ until use.
PF induced by bleomycin
Forty-five mice were randomly divided into four groups: six mice in normal control group (saline), six mice in HSM control groups (saline + 200 mg/kg HSM), 13 mice in bleomycin (BLM) group (4 mg/kg BLM + saline), 13 mice in BLM + HSM groups (4 mg/kg BLM + 200 mg/kg HSM). The HSM solution was prepared as described above. Lung fibrosis was induced by intratracheal instillation of BLM (Nippon Kayaku, Tokyo, Japan) at a single dose of 4 mg/kg on d 0 in 50 µl sterile saline. Control groups were treated with the same volume of sterile saline. The mice received HSM (200 mg/kg) or saline intra-gastrically daily for 21 d 2 h before BLM treatment. One animal died during the course, and the others were sacrificed at the end of 21 d after BLM or saline treatment. Bronchoalveolar lavage fluid (BALF) samples were obtained by washing the right lungs twice with 1 ml PBS, and stored on ice. Subsequently, the samples were centrifuged (500 g, 10 min, 4℃), the supernatants were stored at –80℃ for later analysis, the cells were re-suspended for flow cytometric analysis. The left lungs were removed for overnight (18 h) fixation with paraformaldehyde or storage at –80℃.
ELISA
TGF-β1 in the BALF of mice was measured using ELISA kit (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions.
Flow cytometric analysis
Peritoneal macrophages were washed and fixed with 4% paraformaldehyde, followed by staining with PE-conjugated F4/80 and FITC-conjugated CD11c. Mouse spleen cells were isolated, passed through a 200-mesh sieve, centrifuged and re-suspended in red blood cell lysing buffer for 2 min. For intracellular IFN-γ, IL-4 or IL-17 staining, cells were incubated with 5 ng/ml PMA (Invitrogen, Carlsbad, CA, USA), 1 ng/ml ionomycin (Enzo Life Sciences, Solna, Sweden) and 10 ng/ml brefeldin A (Enzo Life Sciences) for 4 h. After washing, cells were stained with CD4-FITC (eBiosciences, San Diego, CA, USA). After permeabilization using the Cytofix/Cytoperm buffer (BD Biosciences, San Diego, CA, USA), cells were stained with IFN-γ–PE and IL-4–PE (eBiosciences). For intracellular Foxp3–PE staining, the cells were stained with CD4–FITC and CD25–APC (eBiosciences) after permeabilization. The fluorescence was measured by FACS Calibur (BD Biosciences), and data analyzed by FlowJo software (TreeStar, Ashland, OR, USA).
Quantitative real-time PCR
Total RNA was extracted by TRIzol reagent (Invitrogen). One µg RNA was reverse-transcribed into cDNA using a ReverTra Ace-a-RNeasy kit (Toyobo, Osaka, Japan). Quantitative real-time PCR was performed on the Applied BioSystems Step-one plus PCR machine (Bio-Rad, Hercules, CA, USA) using SYBR Green PCR Master Mix (Invitrogen). Dissociation curve was analyzed for exclusion of amplification plot. The primer used in the PCR amplification were synthesized by Invitrogen and the sequences were as follows: mouse GAPDH,5’-AACGACCCCTTCATTGAC-3’(forward) and 5’-TCCACGACATACTCAGCAC-3’(reverse); mouse α-SMA:5’-CCCAGACATCAGGGAGTAATGG-3’(forward) and 5’-TCTATCGGATACTTCAGCGTCA-3’(reverse); mouse vimentin: 5’-CGTCCACACGCACCTACAG-3’(forward) and 5’-GGGGGATGAGGAATAGAGGCT-3’(reverse); mouse fibronectin: 5’-ATGTGGACCCCTCCTGATAGT-3’(forward) and 5’-GCCCAGTGATTTCAGCAAAGG-3’ (reverse); mouse mTOR: 5’-ACCGGCACACATTTGAAGAAG-3’(forward) and 5’-CTCGTTGAGGATCAGCAAGG-3’(reverse); human GAPDH: 5’-AGAAGGCTGGGGCTCATTTG-3’(forward) and 5’-AGGGGCCATCCACAGTCTTC-3’(reverse); human fibronectin: 5’-CGGTGGCTGTCAGTCAAAG-3’(forward) and 5’-AAACCTCGGCTTCCTCCATAA-3’(reverse); human vimentin: 5’-GACGCCATCAACACCGAGTT-3’(forward) and 5’-CTTTGTCGTTGGTTAGCTGGT-3’(reverse).
Histology and immunohistochemistry
Lung tissue sections were fixed with 4% paraformaldehyde and then were embedded in paraffin. Paraffin sections (3 µm) were dehydrated by a gradient series of ethanol, and processed for histology and immunohistochemistry staining. Masson’s trichrome staining was performed and observed by optical microscope. Immunostaining was carried out as follows: blocking with 5% BSA, incubated with primary Abs overnight at 4℃ and then incubation with secondary Abs for 1 h at room temperature (20–25℃). The bound secondary Abs were visualized with DAB substrate. Primary Abs used in the experiment included anti-α-SMA (1:200; Abcam, Cambridge, UK) and anti-p-P70S6K (1:200; Cell Signaling Technology, Danvers, MA, USA).
Cell viability analysis
A549 cell line was purchased from Cell Bank, China Academy of Sciences. A549 cells were cultured in DMEM complete medium [supplemented with 10% (v/v) heat-inactivated FBS, 1% penicillin streptomycin] at 37℃ under 5% CO2. Cells were plated in 96-well plates at a density of 2 × 103 cells/well, followed by treatment with HSM (100–800 µg/ml) and/or TGF-β1 (10 ng/ml) for 24 h. The cell viability was assessed by a Cell Counting Kit 8 (Dojindo Laboratories, Kumamoto, Japan) according to the manufacturer’s protocol.
Western blotting
Western blotting was performed as described previously. The primary Ab including anti-mTOR, anti-p-mTOR, anti-p70S6K, anti-p-p70S6K, anti-GAPDH and anti-tubulin Abs were purchased from Bioworld Technology (St. Louis Park, MN, USA) and Cell Signaling Technology.
Detection of HYP in lungs
Briefly, 30–100 mg lung tissue samples were cut into pieces, and then hydrolyzed in 1 ml lysis buffer solution at 100℃ for 20 min. Impurities were absorbed by activated carbon. The pH of the supernatant was adjusted according to manufacturer's protocol. The absorbance of the colored products was measured at 550 nm. HYP was then estimated following the manufacturer's instructions. The kit was purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China).
Statistical analysis
All data in this experiment are presented as mean ± SEM. The statistical analysis in our research was completed by the GraphPad Prism 5 software and based on Turkey test or t-test using. P < 0.05 was considered statistically significant. Experiments were repeated at least three times, and each data point represents the mean of at least three samples.
Results
Lung injury induced by BLM was attenuated by HSM
In the present study, we selected administration of BLM as a model of lung fibrosis to explore whether HSM has a protective effect on PF.
We performed Masson’s and immunohistochemical staining of lungs from each group. As observed by hematoxylin and eosin staining and Masson staining, BLM-treated mice histologically showed serious pulmonary injury and fibrosis than in the control group. HSM alleviates the injury and fibrosis and decreases the deposition of collagen (Figure 1b). α-SMA was mildly expressed in vascular and airway muscle cells in the control group (Figure 1b). By contrast, the expression of α-SMA was significantly increased in the BLM-treated group (Figure 1b), indicating that a significant number of myofibroblasts were trans-differentiated.
BLM-mediated pulmonary fibrosis in C57BL/6 J mice was attenuated by HSM. (a) Experimental design for the in vivo study. HSM (200 mg/kg) or normal saline (NS) was administered (intra-gastrically) 2 h before BLM (4 mg/kg) injection and repeated daily for 21 d. (b) Hematoxylin and eosin staining, Masson staining and immunostaining performed with α-SMA. Original magnification, ×20. (c) HYP was detected as a sign of collagen content in lungs from control, HSM treatment, BLM treatment and BLM treatment with HSM (200 mg/kg) groups. (d) TGF-β1 levels in BALF of mice treated as in (a) were assessed by ELISA. (e) Effects of HSM on the activation of Smad1/2 signaling pathways and the phosphorylation level of Smad1/2 were analyzed by Western blot. p-Smad1/2 relative band intensity (normalized to GAPDH) was analyzed. (f) Expression of α-SMA, vimentin and fibronectin at mRNA levels were determined using real-time quantitative PCR and expressed as fold expression relative to control. *P < 0.05, **P < 0.01, ***P < 0.001.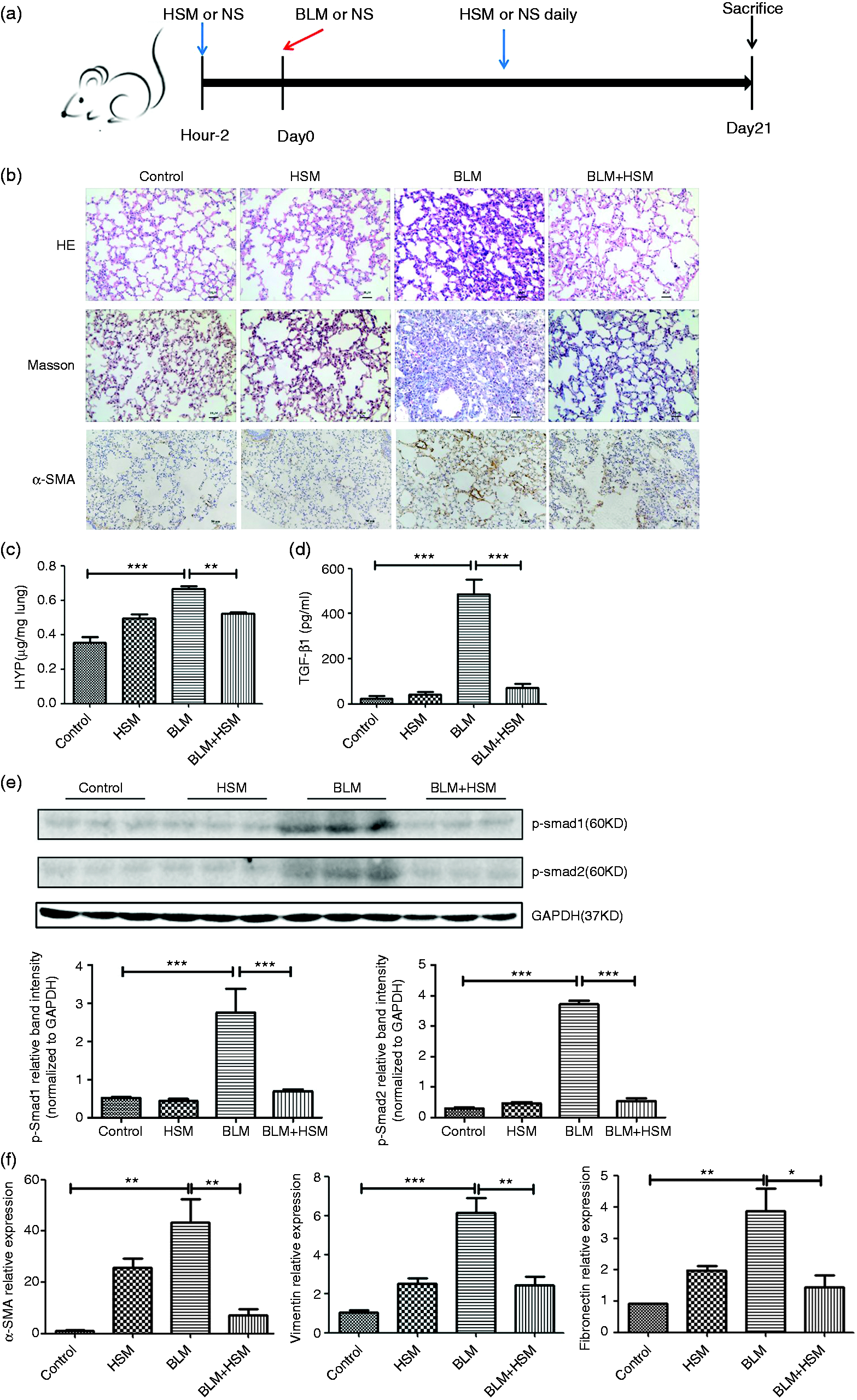
Furthermore, the lung HYP content in the lung of BLM-treated mice was higher than that in the control group (Figure 1c). Treatment with HSM significantly reduced the lung HYP content in the lung.
TGF-β1 is regarded as a key factor of the pro-fibrogenic molecules. Thus, we assayed the TGF-β1 protein level in the BALF, which was found to be significantly higher in BLM-treated mice than that in the control group. The TGF-β1 levels were suppressed by HSM treatment (Figure 1d). To explore whether TGF-β1 is activated or not, we performed Western blot analysis, which showed that HSM decreased the level of p-Smad1/2 (Figure 1e), suggesting that HSM impeded the activation of TGF-β1 induced by BLM in the lung sections.
Furthermore, we examined the mRNA expression of several classic pro-fibrotic mediators (including α-SMA, fibronectin and vimentin) (Figure 1f). The expression of these pro-fibrotic cytokines was significantly increased compared with those in the control group and decreased by HSM treatment. Together, these results suggested that HSM improved BLM-induced lung fibrosis in wild type C57BL/6 J mice.
HSM suppresses the chemotaxis of alveolar macrophages and the expression of inflammatory mediators in lungs of BLM-treated mice
Alveolar macrophages are ideally placed to initiate a strong inflammatory response to the pathogenic material. Hence, we assessed the percentage of alveolar macrophages in each groups’ BALF by flow cytometric analysis. The results showed that CD11c+ F4/80+ alveolar macrophages significantly increased compared with those of the control group. However, the HSM treatment group obviously decreased the percentage of the macrophage population (Figure 2a, b), indicating that HSM hindered the chemotaxis of alveolar macrophages during PF.
HSM suppresses the chemotaxis of alveolar macrophages and the expression of inflammatory mediators in the lung of BLM-treated mice. (a) The percentage of CD11c+ F4/80+ alveolar macrophages in each groups’ lungs was determined by flow cytometry and analyzed by FlowJo software. (b) Data are shown as mean ± SEM. (c) IL-1β and IL-6 mRNA expression levels were determined using real-time quantitative PCR and expressed as fold expression relative to the control. *P < 0.05, **P < 0.01, ***P < 0.001.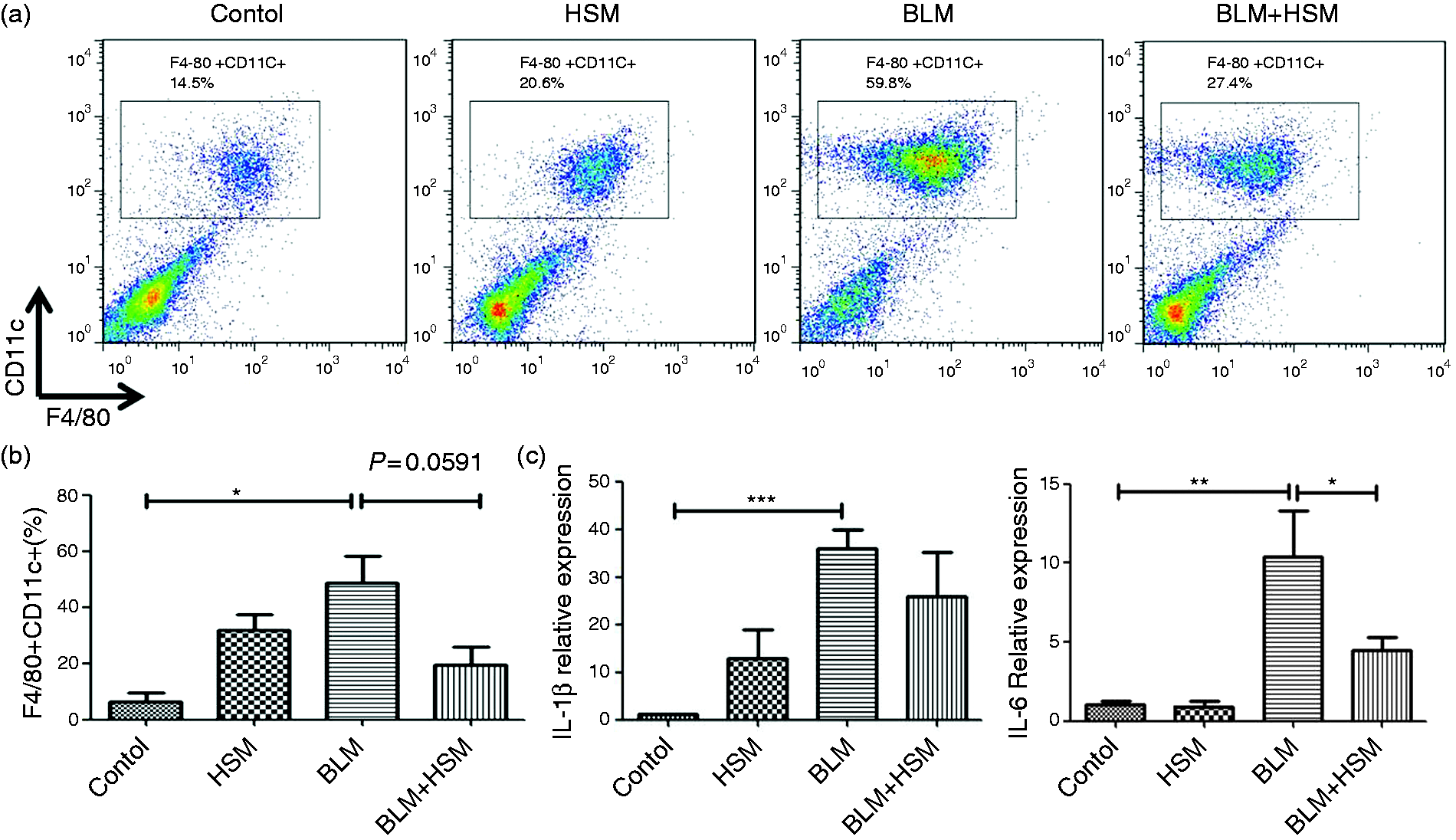
To further confirm the possibility that the anti-PF effect of HSM was associated with the inhibition of early inflammation in the lung, we examined the expression of inflammatory mediators, including IL-1β and IL-6 by Q-PCR (Figure 2c). The expression of these inflammatory cytokines in BLM group was significantly higher than those in control group and decreased by HSM treatment (Figure 2c).
HSM influenced the immune imbalance in PF mice induced by BLM treatment
Compared with controls, mice treated with BLM exhibited a higher proportion of Th2 (Figure 3b) and Treg (Figure 3c) cells, but lower Th1 (Figure 3a) and Th17 (Figure 3d) cells. HSM treatment reversed the imbalance, indicating that BLM can disrupt the balance of Th1/Th2 expression in the spleen and HSM may ameliorate the BLM-induced PF in mice by regulating the Th1/Th2 improving immunity.
HSM influenced immune imbalance in pulmonary fibrosis mice induced by BLM treatment. The percentage of (a) CD4+IFN-γ+Th1 cells, (b) CD4+IL-4+Th2 cells, (c) CD4+CD25+Foxp3+Treg cells and (d) CD4+IL-17+Th17 cells in the spleen of each group were detected by flow cytometry and analyzed by FlowJo software. Data are shown as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001.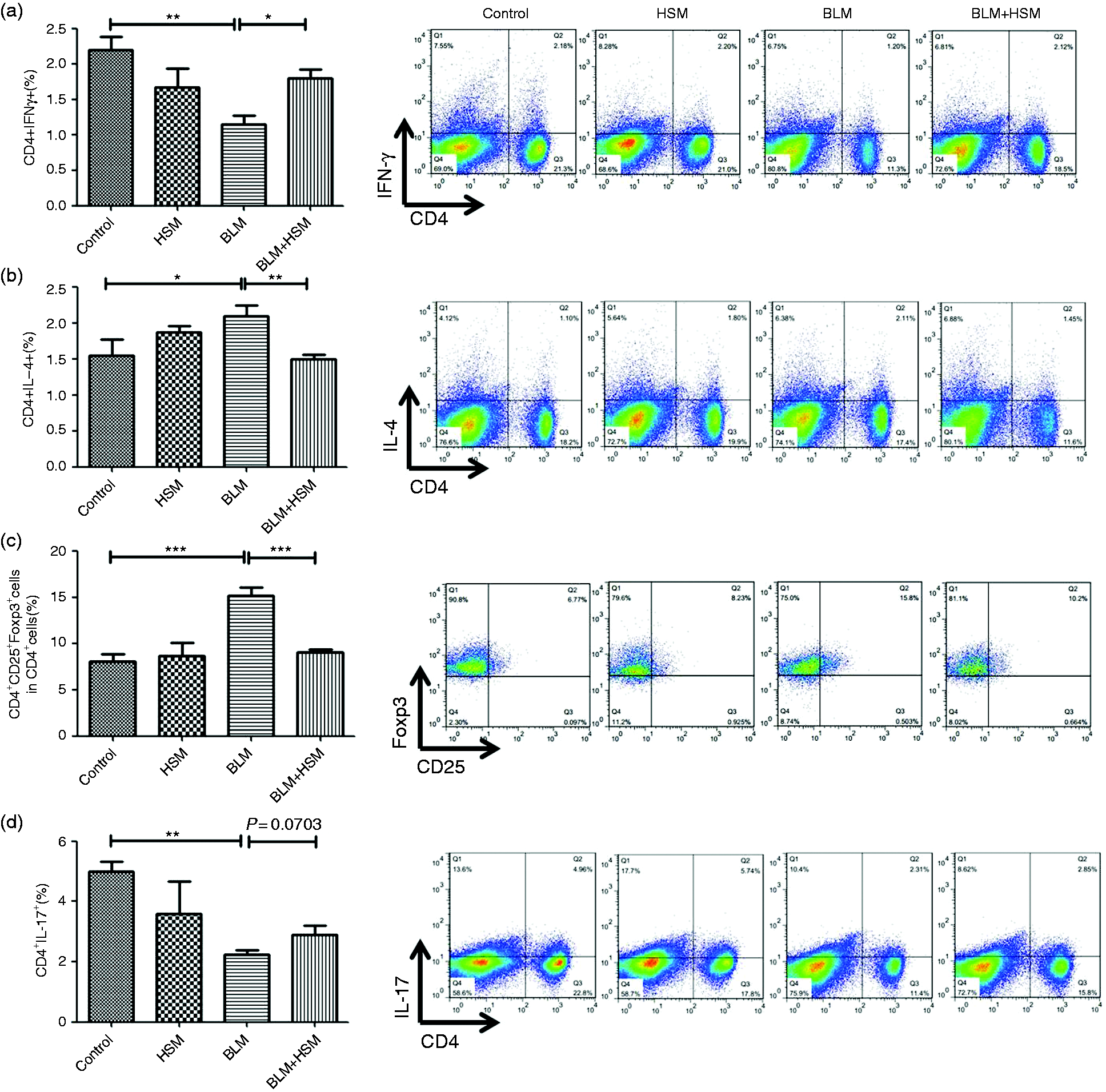
HSM alleviates mTOR overactivation in fibroblast foci of BLM-treated mice
Fibroblast foci have been recognized as an indicator of PF. Therefore, the mTOR activation in fibroblast foci during PF was investigated. As shown in Figure 4(a, b), HSM significantly alleviated mTOR overactivation both in mRNA and protein level. Then we performed immunohistochemical staining of p-p70S6K from each group lung sections (Figure 4c). p-p70S6K in the control group was found to be mildly expressed in vascular and airway muscle cells, whereas the protein expression remarkably improved in lungs from BLM-treated mice. HSM inhibited the phenotype, indicating that HSM alleviated overactivation of the mTOR signaling pathway. Western blotting also indicated that p-p70S6K was weakly expressed in the control lung tissue (Figure 4d). However, the protein was strongly increased in BLM-treated lung tissue, and HSM significantly suppressed the expression. We concluded that the mTOR signaling pathway was overactivated in the fibroblast foci in BLM-treated PF lung sections, and HSM alleviates PF may be associated with suppressing the overactivation in fibroblast foci.
HSM alleviates mTOR overactivation in fibroblast foci of BLM-treated lung tissues. (a) Expression of mTOR at mRNA levels were determined using real-time quantitative PCR and expressed as fold expression relative to control. **P < 0.01, ***P < 0.001. (b) mTOR overexpression was reduced by HSM treatment. Western blot analysis for p-mTOR evaluation in each control lung sections. (c) Immunohistochemical staining of p-pp70S6K showed a remarkable increase of p-p70S6K in PF lung tissues vs. that in the control group. Original magnification,×20. (d) mTOR/p70S6K signaling pathway overactivation was suppressed by HSM. Western blot analysis for p-p70S6K evaluation in each control lung sections. p-S6: phosphorylation of p70S6K; S6: p70S6K.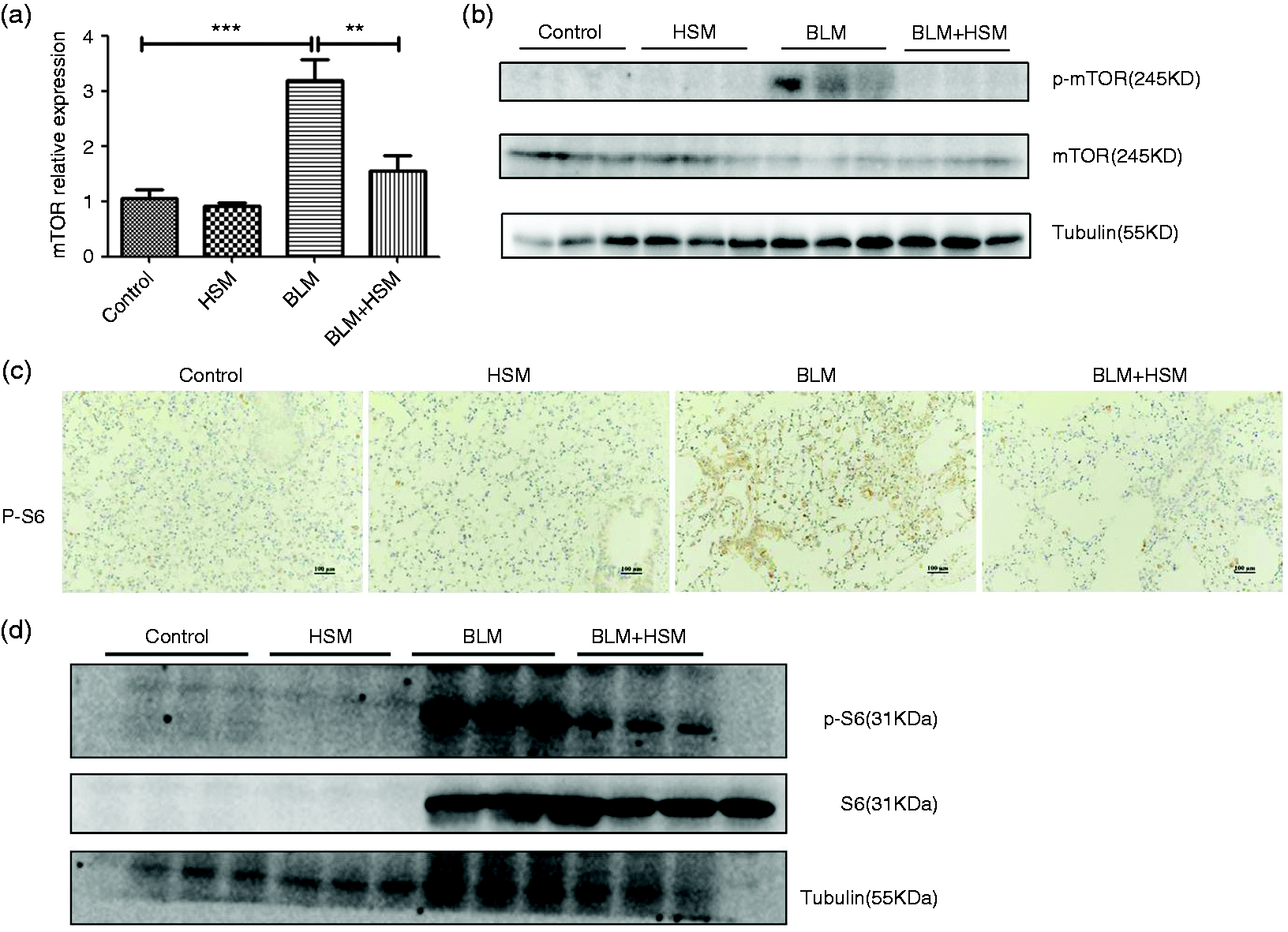
HSM suppresses mTOR activation stimulated by recombinant TGF-β1 in A549 cells during fibroblast–myofibroblast trans-differentiation
We first assessed the cellular toxicity of HSM in A549 cells through CCK-8 assay. As shown in Figure 5(a), up to 800 µg/ml 24-h treatment with HSM did not reduced cell viability obviously in resting A549 or TGF-β1-activated A549 cells. Resting A549 cells normally displayed a layer of cobblestone-like morphology but turned into spindle fibroblast-like appearance after stimulation by recombinant TGF-β1 (10 ng/ml) for 24 h. After treatment with HSM for 24 h, the spindle fibroblast-like phenotype was found to be reversed (Figure 5b). The expression of fibronectin and vimentin in TGF-β1-activated A549 cells was significantly increased compared with those in control group. Strikingly, the expression of these cytokines was reduced by HSM treatment (Figure 5c), indicating that TGF-β1-induced myofibroblast activation had been inhibited.
HSM suppressed mTOR overactivation stimulated by recombinant TGF-β1 in A549 cells. (a) Resting or TGF-β1 (10 ng/ml)-activated A549 cells were treated with various concentrations of HSM or an equal volume of complement culture for 24 h (n = 6). Cell viability was determined by CCK-8 assay. (b) Phenotypic change of A549 cells in the presence of TGF-β1 (10 ng/ml) or HSM (200 µg/ml) for 24 h. Original magnification, ×20. (c) The expression levels of fibronectin and vimentin were assessed by real-time quantitative PCR and preformed as fold expression compared with the control group. *P < 0.05, **P < 0.01, ***P < 0.001. (d) A549 cells were treated as in (b), protein levels of p-mTOR and p-p70S6K were detected by Western blotting. Data were expressed as mean ± SEM. p-S6: phosphorylation of p70S6K; S6: p70S6K.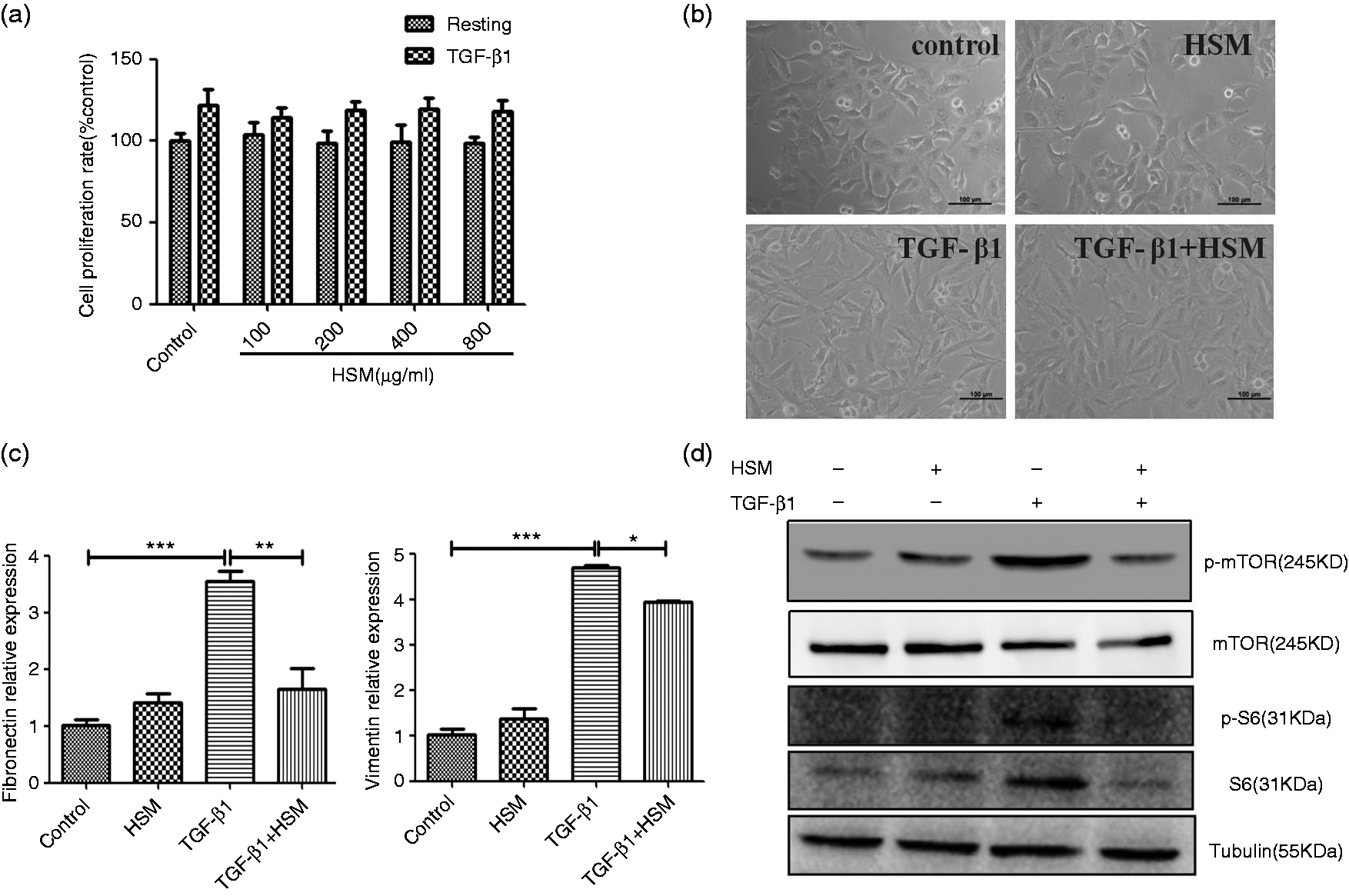
To observe mTOR activation during the fibrosis process in vitro, we assessed the expression of p-mTOR and p-p70S6K protein in resting A549 or activatedA549 cells by Western blotting. After 24 h stimulation, p-p70S6K expression was elevated, while HSM treatment obviously reduced the expression of p-p70S6K, indicating that overactivated mTOR signaling stimulated by TGF-β1 was suppressed by HSM treatment (Figure 5d).
Discussion
Administering BLM to rodents has become the most widely used model for experimental PF, which induces lung injury through two distinct phases. 17 The first alveolitis phase involves alveolar injury, which is associated with hazardous factors, resulting in the inflammation of lungs, an influx of inflammatory cells, i.e. macrophages, and abnormal proliferation of alveolar epithelial cells. The end fibrosis stage involves fibroblasts activation, ECM remodeling and massive collagen deposition.18–20
Although several studies have revealed some molecular mechanism of HSM on lung injury, the composition of HSM is highly complicated. Herein, we attempted to explore whether HSM can relieve PF through other mechanisms.
We demonstrated that HSM possess anti-fibrotic activities, as shown by inhibiting the amount of HYP in pulmonary section, the expression of pro-inflammatory cytokines and pro-fibrosis cytokines, and improvement in the injured organ. In addition, HSM effectively inhibits TGF-β1-induced myofibroblast differentiation and myofibroblast marker expression (vimentin and fibronectin) in A549 cells. These results are consistent with previous studies.
The primary characteristic of alveolitis is the accumulation of inflammatory cells within the lung parenchyma. The inflammatory cells accumulate and release cytokines, resulting in epithelial–mesenchymal transition, fibroblast activation and migration into the areas of acute lung injury, eventually leading to secretion of ECM proteins and massive collagen deposition. 21 The current findings show that HSM reduced the alveolar macrophages’ cell counts in BALF following BLM administration. In addition, reduced expression levels of inflammatory cytokines were also observed after treatment with HSM in lung fibrotic mice. Consistent with these results, Huang et al. showed that HSM reduces IL-1β and IL-18 expression levels in the lungs of BLM-treated mice, as well as inhibits ATP-induced IL-1β and IL-18 secretion in LPS-primed macrophages. 9
TGF-β1-induced pro-fibrosis cytokine expression may be partially mediated by the PI3K/Akt/mTOR pathway. 22 However, hitherto, there have been no reports regarding the association between the effects of HSM on lung fibrosis with the mTOR pathway. In this study, we first determined that the inhibitory effects of HSM on BLM-induced PF and TGF-β1-mediated myofibroblast differentiation and myofibroblast marker expression may be associated with suppressing the mTOR activation.
Our study also revealed that the immune balance plays a pivotal role in PF, which is not in the focus of most researchers. Th1 and Th2 polarization is an indication that alveolitis has entered the end fibrosis stage. Moreover,Th1/Th2 balance can be modulated by Tregs; a reciprocal balance occurs between Treg cells and Th17 cells.Th17 cells represent a novel subset of the specific Th lineage and modulate an early lung inflammation in experimental PF. 23 For differentiation, Th17 cells require TGF-β in combination with IL-6. Previous studies have also indicated that blocking the function of Th17 cells may result in a rapid increase of Treg cells. 24
Experimental studies and clinical screening have provided the basis for pro-fibrotic roles of growth factors and cytokines. The therapeutic agents targeting these factors have been wildly investigated.25–27 Interestingly, we found that HSM not only down-regulates the pro-fibrotic factors, such as TGF-β1, vimentin and fibronectin, but also suppresses the inflammatory cytokines, and influences the function of immunocytes. In addition, the effect of HSM on lung fibrosis may be associated with the inhibition of phosphorylation of mTOR/p70S6K. Therefore, such extensive activities necessitate further study for the potential development of HSM as a therapeutic agent for PF.
Conclusion
mTOR overactivation is involved in the pathogenesis of PF. HSM can effectively ameliorate PF, which is associated with the inhibition of phosphorylation of mTOR p70S6K. Inhibition of mTOR overactivation may become a potential treatment for PF.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81402940 and 31570909) and Natural Science Foundation of Jiangsu Province, China (BK20140615).
