Abstract
Large genome-wide analysis studies (GWAS) and meta-analyses have dramatically increased our knowledge of the genetic risk factors of inflammatory bowel disease (IBD), identifying at least 163 loci. The endoplasmic reticulum aminopeptidase-2 (ERAP2) gene has been reported as a potential candidate gene for IBD. GWAS have also shown the potential associations between ERAP single nucleotide polymorphisms (SNP) loci and susceptibility to several autoimmune diseases, and ERAP1 and ERAP2 polymorphisms are related to HLA class I-associated diseases, including ankylosing spondylitis and Behçet’s disease. Interestingly, these associations were confined to individuals carrying HLA class I-risk alleles. The aim of this study was to investigate the association of ERAP1 and ERAP2 SNPs with IBD in a Spanish population, analysing their possible interaction with specific HLA-C alleles to IBD susceptibility. A total of 367 individuals were divided into 216 IBD cases and 151 controls. SNP genotyping was performed using TaqMan® genotyping assays, whereas HLA-C typing was analysed by sequence-specific oligonucleotide probing. Herein, we report an association of the ERAP1 SNP rs30187 with the HLA-C*07 allele. The existence of shared inflammatory pathways in immunologically related diseases together with the understanding of ERAP1 function may offer clues to novel treatment strategies.
Introduction
Inflammatory bowel disease (IBD) is a chronic inflammatory process that primarily affects the gastrointestinal tract. 1 This term encompasses Crohn’s disease (CD), ulcerative colitis (UC) and intermediate entities termed ‘indeterminate colitis’. Although their exact aetiology remains unclear, it is known that genetic factors contribute to disease susceptibility, along with environmental agents. 2 Genome-wide association studies (GWAS) have significantly advanced our understanding of the importance of genetic susceptibility in IBD.3,4
A single-nucleotide polymorphism (SNP) in the ERAP2 gene, rs2549794, has been reported as a potential predisposing factor for the development of CD, 5 and a potential candidate gene in IBD aetiology. ERAP2 is one of the two human endoplasmic reticulum aminopeptidases that play a critical role, working in concert with ERAP1, in trimming peptides for presentation on HLA class I molecules, which affects antigen presentation to T cells. 6 There are data indicating that ERAP2 forms a repertoire of ligands for HLA class I that is distinct from those generated by ERAP1. 7 However, it has also been reported that ERAP1 and ERAP2 may perform antigenic peptide trimming in a coordinated way. 6 GWAS have shown the potential associations between ERAP SNP loci and susceptibility to several autoimmune diseases, and ERAP1 and ERAP2 polymorphisms are related to HLA class I-associated diseases, 8 including ankylosing spondylitis (AS) and Behçet’s disease (BD). Interestingly, these associations were confined to individuals carrying HLA class I-risk alleles. 9 AS is a chronic, inflammatory rheumatic disease known to be closely associated with HLA-B27, although the precise mechanisms of pathophysiology remain unclear. 10 In this disease, ERAP1 SNP rs30187 has been found to be strongly associated with susceptibility. 11 Interestingly, HLA-B27 positive and negative AS cases differ in their association with the ERAP1 gene, 12 so it is possible that ERAP1 may be involved in the generation of autoimmunogenic peptides prior to HLA-B27 assembly and peptide presentation. In the same way, an epistasis between an HLA-B allele and ERAP1, specifically HLA-B*51, was found to recessively increase BD risk. 13 Similar to AS, many putative susceptibility loci have been described for psoriasis, but the strongest genetic signal of susceptibility resides in the HLA region, mostly to HLA-Cw6. 14 Moreover, a gene–gene interaction between HLA class I molecules and ERAP1 has also been described,15,16 in this case with HLA-C*06:02.
All these studies provide examples of interactions between risk loci observed in GWAS, suggesting that similar analyses may be valuable in other autoimmune diseases. Additional studies have described associations of ERAP SNPs in other disorders. For example, ERAP1 SNP rs30187 has been associated with multiple sclerosis (MS) in both European and US populations, 17 and with type 1 diabetes, 18 and ERAP2 SNPs (rs2549782 and rs17408150) have been associated with the pathogenesis of pre-eclampsia. 19
A recent study that conducted high-density SNP genotyping of the MHC in >32,000 individuals with IBD implicated HLA alleles in both CD and UC, 20 and reported associations of HLA with IBD (class I and II). An important component of NK target recognition is the control of HLA class I molecules by killer immunoglobulin-like receptors (KIR), and some KIR receptors bind to HLA-C molecules. 21 Ser77/Asn80 (C1) and Asn77/Lys80 (C2) amino-acid positions define serologically distinct allotypes of HLA-C. KIR2DL2, KIR2DL3 and KIR2DS2 interact with HLA-C1, whereas KIR2DL1 and KIR2DS1 recognize HLA-C2. For example, HLA-C*06:02 is a ligand of KIR2DL1/S1. Evidence suggests that KIR2DL2 and KIR2DL3 genes in combination with specific HLA-C ligand allele interactions could contribute to genetic susceptibility to CD. 22 Against this background, the aim of this study was to assess the association of ERAP1 and ERAP2 SNP markers with IBD in a Spanish population, analysing the possible association of specific HLA-C allele and ERAP1/ERAP2 polymorphisms with susceptibility to IBD.
Material and methods
Study population
A total of 367 individuals were studied, divided into 216 IBD cases and 151 controls. The patient population included 86 UC and 130 CD patients. The IBD patients were referred to a central specialized Immunology Unit (Hospital Universitario Central de Asturias, Asturias, Spain) from the Gastroenterology Clinics (Hospital San Agustín, Asturias, Spain) for immunology evaluation. Diagnosis of IBD and disease extent was established on the basis of clinical, radiological, and histological data according to standard criteria. 23 The control population consisted of 151 matched unrelated blood donor healthy individuals, collected between January 2015 and May 2015 (Hospital Universitario Central de Asturias, Asturias, Spain). All subjects were Caucasian, originated from Northern Spain and were matched for age and sex. The protocol was approved by the local ethics committee, and all participants provided their written informed consent prior to enrolling in the study.
Genotyping
SNP genotyping was performed using TaqMan® genotyping assays (Applied Biosystems, Foster City, CA, USA): C___3056885_10 for rs30187, C___3282749_20 for rs2549782, C__25649529_10 for rs2248374, C___3282732_10 for rs2549794, and C__26382310_10 for rs2910686. Allelic discrimination analysis was performed with LightCycler 480 SW software version 1.5.1.62 (Roche Applied Science). A random 10% of the samples underwent repeated genotyping for quality control. No discrepant results were observed in repeated samples.
The HLA-C was typed by using Lifecodes KIR-SSO typing kits (Tepnel Lifecodes Corporation, Stamford, UK) based on the Luminex Multi-Analyte Profiling System (xMAP technology) (Luminex Corp., Austin, TX, USA), following the manufacturer’s instructions.
Statistical analysis
The statistical analysis of SNP genotyping data was carried out as follows: (i) allele frequencies were compared between IBD patients and controls by χ2 analysis, and odds ratios (OR) and 95% confidence intervals (CIs) were calculated using PLINK software; 24 (ii) haplotype analysis was performed with Haploview version 4.1 software using the CI method to perform linkage disequilibrium (LD) assessment and define haplotype blocks. 25 Sliding windows of 2–5 SNPs each were considered for association with χ2 analysis using Plink software. 24 Sliding windows analyses assess the frequency of composite genotypes of a fixed number of contiguous SNPs (shifting one SNP at a time) instead of examining haplotypes based on their LD. Allelic frequencies obtained for each SNP were tested for deviations from Hardy–Weinberg Equilibrium (HWE) expectations. A type II error of 0.5, i.e. a statistical power of 0.5, was established. We used the SPSS v.22 statistical software to assess the association of ERAP1/ERAP2 SNPs and HLA-C alleles with IBD susceptibility.
Results
SNP allelic association analysis of the markers included in the study.
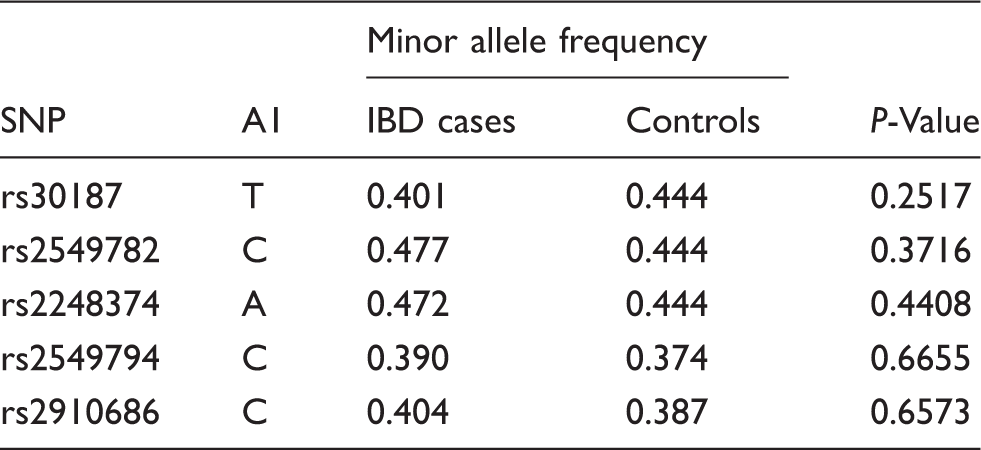
Allele frequencies were compared using χ2 analysis (Haploview software).
The distribution of HLA-C in the study population.
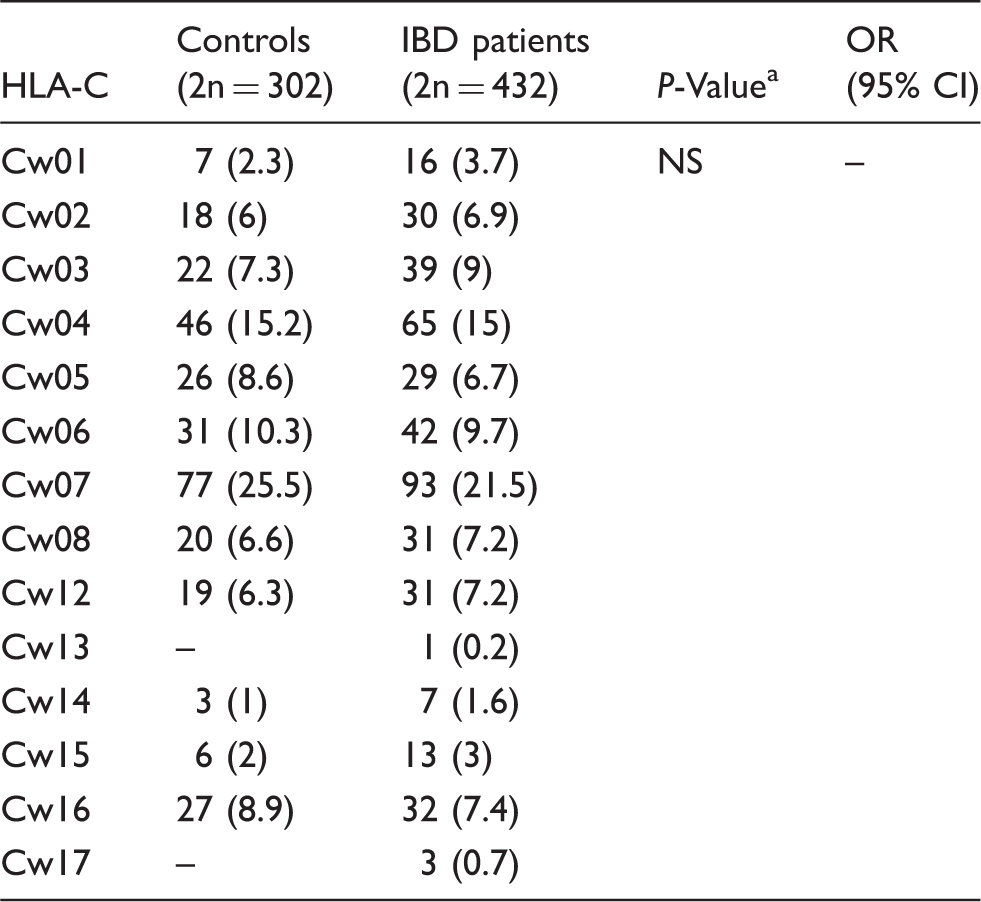
NS: not significant; OR: odds ratio; CI: confidence interval
Two-tailed Fisher’s exact test.
The dependent association of ERAP1 SNP rs30187 and HLA-C*07 with IBD.
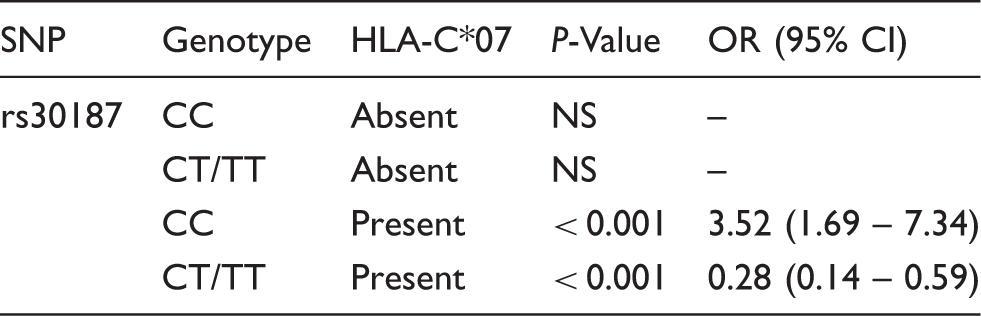
P-Value and odds ratio (OR) estimated for ERAP1 genotypes at rs30187 stratified by the presence of HLA-C*07. Absent: cohort comprised 86 controls and 131 IBD patients; present: cohort comprising 65 controls and 74 IBD patients; NS: not significant; CI: 95% confidence interval.
Discussion
This study examined the association of the ERAP1 and ERAP2 genes with IBD in a Spanish population. We did not observe a direct correlation between SNPs analysed and CD and/or UC. This may indicate that the investigated genes are not related to these phenotypes individually. However, we cannot exclude that more statistical power with a larger sample size is needed to detect potential associations with IBD, particularly if these effects are small. Additionally, we assessed the effects of these genetic variants in combination with HLA-C alleles. Interestingly, we report an interesting association with ERAP1 rs30187 that seems to depend on the presence of the HLA-C*07 allele.
Although GWAS and GWAS meta-analysis have expanded the number of IBD-associated loci to 163 in European ancestry populations, generating new biological hypotheses for disease pathogenesis,
3
only a modest proportion of the expected heritability has been explained: approximately 19–26% for CD and 15–19% for UC.
26
Gene–gene interactions may contribute to the missing heritability of complex phenotypes such as IBD. It appears that gene–gene or gene–environment interactions and other structural variants may be the only explanation for this missing heritability. Research on the role of interactions between genes in complex phenotypes is still limited,
27
but further studies on these interactions could have a significant impact on our understanding of the pathophysiology of IBD and on identifying potential new therapies. The finding that ERAP1 SNP rs30187 is associated with disease in individuals carrying the HLA-C*07 allele is biologically interesting. Inside the ER, ERAP1 trims amino terminal residues of precursor peptides to an optimal length for MHC-I loading (Figure 1a), and its contribution in antigen processing has been extensively established in studies using ERAP-deficient mouse models.
28
Different ERAP1 polymorphisms could change the peptide repertoire, affecting the folding and stability of HLA class I complexes and recognition by CD8+ T and NK cells.29,30 Thus, the pathogenic effect of ERAP1 may be due to a modified interaction between HLA-C/peptide and selective inhibitory KIRs on CD8+ T and NK cells (Figure 1a).
Hypothesis to explain the potential implications of the association between ERAP1 and HLA class I molecules. (a) Antigenic presenting capability of HLA class I molecules could be affected by the ability of ERAP1 to modify the peptides. ERAP1 polymorphism could change the peptide repertoire, affecting the stability of HLA class I/peptide complexes and recognition by CD8+ T and NK cells. (b) Ankylosing spondylitis, Behçet disease, psoriasis and inflammatory bowel disease have a shared immunopathogenetic basis. The identifying of these pathways, together with the understanding of the role of ERAP1, may present clues to common strategies for treating. These conditions share the clinical features of disease localization in sites of interaction between the body and the external environment. At the same time, they are associated with HLA class I molecules. Molecular mimicry between foreign and self-peptides may unleash a cytotoxic T-cell response leading to the autoimmune destruction of self-tissues.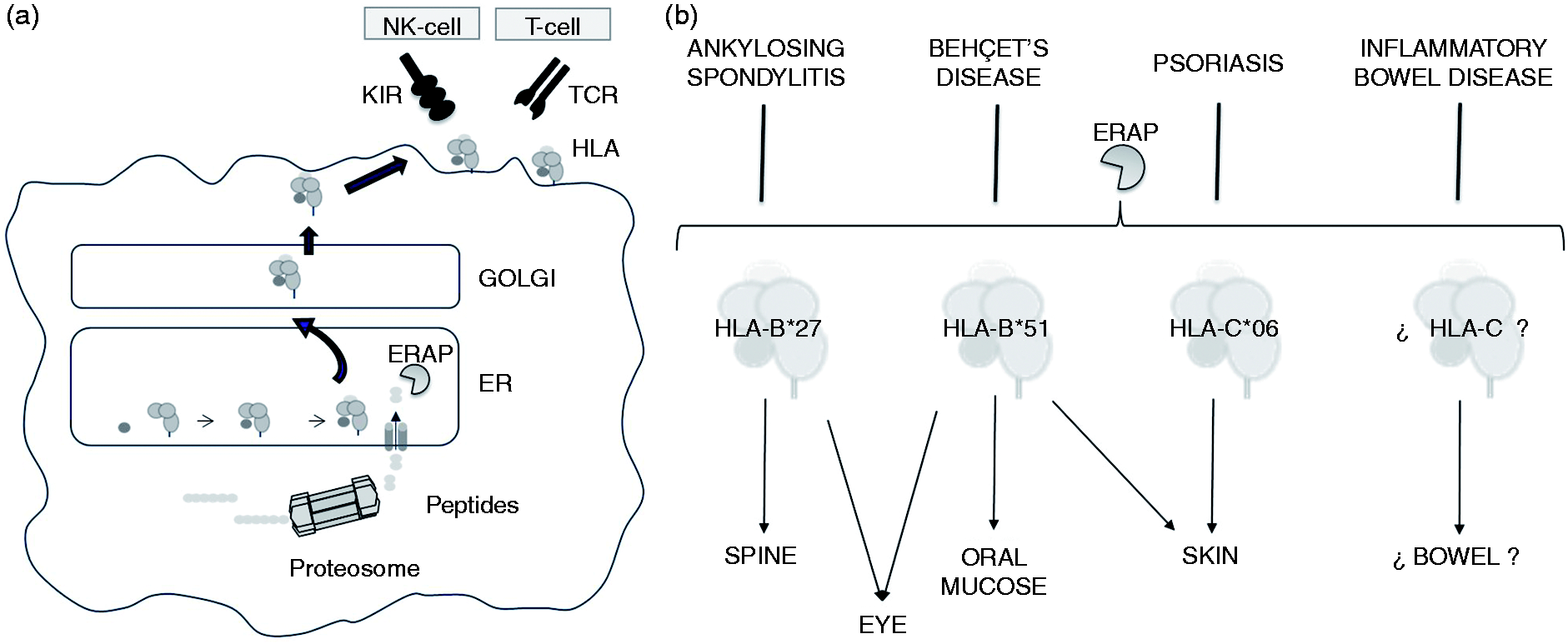
In this context, we extend the concept of ‘MHC-I–opathy’ reviewed by McGonagle et al., 31 based on the immunopathological links between BD and spondyloarthropathies, extending it to IBD (Figure 1b). The association of ERAP1 variants with MHC-I-opathies shows an epistatic relationship with known disease-associated HLA alleles. The interaction between the ERAP1 SNP rs30187 and the HLA-B*27 and HLA-B*40:01 alleles has been demonstrated previously. 32 Similar interactions have been described in BD with HLA-B*51 13 and psoriasis with HLA-C*02. 15 All of these disorders share genetic susceptibility factors. Approximately 70% of IBD risk loci have been shown to be shared with other complex traits, including 66 loci shared with other autoimmune disorders, particularly AS and psoriasis. 3 The existence of shared inflammatory pathways in AS, BD and psoriasis, even IBD, together with the understanding of ERAP1 function may offer clues to common strategies for treating autoimmune disorders. However, the picture may be more complex. Different ERAP1 SNPs have been shown to affect the specificity, as well as the effectiveness, of its enzymatic activity. For example, a Q725 ERAP1 variant increases the risk for BD but is protective for AS, so the same variant can exert opposing effects on different but related diseases. 9
Several limitations in our design should be kept in mind when considering these conclusions. The sample size of our cohort is small compared with other studies that have found significant associations between susceptibility loci and IBD. Moreover, most IBD loci confer risk to both UC and CD but typically show distinct effect sizes in the two disorders. 3 We did not have a large enough sample size to detect any differences between phenotypes. In conclusion, we show that the contribution of the ERAP1 gene to IBD susceptibility extends beyond just ERAP itself in a case–control study. We show the relevance of the ERAP1 gene with respect to its interaction with HLA-C*07 in IBD. These findings emphasize the need for comprehensive and detailed studies in large patient and control cohorts (UC and CD) in which the ERAP1 SNP rs30187 should be analysed and a complete HLA class I genotyping (HLA-B and HLA-C), as well as a complete KIR genotyping, should be performed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Universidad Autónoma de Chile 36/2014 internal grant, and by FONDECYT grant n° 1151048.
