Abstract
Hepatitis B virus (HBV) infection is a major cause of chronic liver disease that may progress to liver cirrhosis and hepatocellular carcinoma. Host immune responses represent the key determinants of HBV clearance or persistence. Here, we investigated the role of the early activation marker, CD69 and effector cytokines, granzyme B (GrB) and IFN-γ in the exhaustion of innate-like TCR Vα7.2+CD4+T cells, in 15 individuals with chronic HBV (CHB) infection where six were HBV DNA+ and nine were HBV DNA–. The percentage of cytokine-producing T cells and MAIT cells were significantly perturbed in HBV patients relative to healthy controls (HCs). The intracellular expression of GrB and IFN-γ was significantly reduced in MAIT cells derived from HBV-infected patients as compared to HCs, and the levels correlated with the percentage and levels [mean fluorescence intensity (MFI)] of CD69 expression. The total expression of CD69 (iMFI) was lower in CHB patients as compared to HCs. The frequency of CD69+ cells correlated with the levels of cytokine expression (MFI), particularly in CHB patients as compared to HCs. In summary, the polyfunctionality of peripheral T cells was significantly reduced among CHB patients, especially in the TCR Vα7.2+CD4+T cells, and the levels of cytokine expression correlated with functional cytokine levels.
Introduction
Hepatitis B virus (HBV) infection represents one of the most common chronic diseases that can cause liver injury and inflammation, afflicting ∼350 million people worldwide. 1 The natural course of infection can be either acute or chronic. HBV infection, in general, leads to an acute self-limiting infection in adults.1,2 The progression rate to chronic infection is relatively higher when HBV is acquired at birth or early infancy. 1 Chronically infected individuals are at risk for developing cirrhosis, hepatocellular carcinoma (HCC), and liver failure, which accounts for 780,000 to 1 million deaths annually.1,3
Acute HBV infection in adults triggers effective and sustained multi-epitope-specific CD4+ and CD8+ T-cell responses that are capable of controlling viral replication,4,5 although not always leading to HBV clearance. Of note, HBV genome becomes detectable in a fraction of patients who have experienced clearance of plasma hepatitis B surface antigen (HBsAg). 6 On the contrary, during chronic hepatitis B (CHB) infection, virus-specific T cells succumb to exhaustion as their ability to respond to antigenic activation are impaired, leading to sub-optimal cytotoxic functions and cytokine production. Several co-inhibitory receptors including programmed cell death-1 (PD-1), 7 lymphocyte activation gene-3 (LAG-3), T-cell immunoglobulin and mucin-domain containing-3 (TIM-3),8,9 cytotoxic T lymphocyte-associated antigen-4 (CTLA-4) 10 are elevated during many chronic viral infections, including CHB infection. Nonetheless, the role of certain co-stimulatory receptors, especially CD69 in innate-like T cells, has seldom been investigated.
CD69 receptor is a type II integral protein containing extracellular C-type lectin super-family domain,11,12 and its genes are located within the NK gene complex.13,14 CD69 is constitutively expressed across numerous cell types, including platelets and monocytes,15,16 and are up-regulated in activated lymphocytes such as T cells, B cells, NK cells, neutrophils and eosinophils recruited at the sites of infection. 17 Once expressed, CD69 acts as a co-stimulatory molecule, and promotes cellular proliferation, cytokine production and/or cytotoxicity.18,19 Engagement with CD69 will trigger the activation of ERK, and is essential for CD69-mediated cell degranulation. 20
Mucosal-associated invariant T (MAIT) cells are a recently described subset of innate-like T cells characterized by a semi-invariant T-cell receptor (TCR), Vα7.2 (TRAV 1–2), paired with variable β-chains 21 and a C-type lectin-like receptor, CD161.22,23 MAIT cell frequency among healthy adults is estimated to account for ∼5% of the total T-cell pool but enriched up to ∼40% in mucosal tissues such as liver and lungs.24–26 MAIT cells play an important role in innate host defence against bacterial and viral infections,27,28 and are weakened in chronic viral infections, including HIV,29,30 HCV infections 31 and HIV-TB co-infections. 32 Here, we investigated the role of the early activation molecule and co-stimulatory receptor, CD69 on TCR Vα7.2+CD4+ lymphocytes and their association with their exhaustion. We found that the percentage of cytokine-producing T cells across T cells and MAIT cells were significantly perturbed. The intracellular expression of granzyme-B (GrB), and IFN-γ were significantly reduced in MAIT cells among HBV-infected patients relative to healthy controls (HCs), and the levels correlated with the percentage and levels [mean fluorescence intensity (MFI)] of CD69 expression.
Materials and methods
Patients and healthy controls
CHB infection was defined as subjects who tested positive for HBsAg for at least 6 mo. 1 A total of 15 individuals with CHB infection were recruited in the study where six patients were HBV DNA-positive and nine had undetectable serum HBV DNA. HBV viral load was measured using a commercial COBAS AmpliPrep-COBAS TaqMan HBV test (CAP-CTM; Roche Molecular Systems, Inc., Branchburg, NJ, USA). In addition, 11 individuals negative for chronic viral (HBV, HCV and HIV) and Mycobacterium tuberculosis infections were also recruited as HCs. Peripheral blood was obtained from the participants at the outpatient clinic of the University of Malaya Medical Centre (UMMC), Malaysia. Informed consent was obtained from all participants in accordance with the Declaration of Helsinki, and the study was performed according to study protocol approved by the Medical Ethics Committee of the UMMC Kuala Lumpur, Malaysia (MEC201311-0496).
Peripheral blood mononuclear cells
Venous blood was collected (10 ml) from all participants by venipuncture in lithium heparin BD Vacutainer tubes (BD Biosciences, Franklin Lakes, NJ, USA). Peripheral blood mononuclear cells (PBMCs) were purified by a density-gradient centrifugation method using FicollPaque Plus (Sigma-Aldrich, St. Louis, MO, USA) overlay. Cell viability was assessed by 0.4% trypan blue vital staining. Purified PBMCs were subsequently used in the immunophenotyping and cell culture experiments.
MAIT cell activation and intracellular staining
For intracellular cytokine staining, the cells were stimulated with PMA (100 ng/ml) and ionomycin (0.67 µM) for 5 h at 37℃ and 5% CO2 prior to immunostaining. Brefeldin A (10 g/ml) was added for the last 4 h of stimulation. The immunostained samples were washed twice prior to acquisition on a FACS Canto II Immunocytometry system (BD Biosciences).
Multicolour flow cytometry
All Abs were pre-titrated to determine appropriate working concentrations. All Abs were procured from BD PharmingenTM (BD Biosciences) unless and otherwise specified. Immunostaining was performed with two panels: one with FITC-conjugated anti-IFN-γ, PE-Cy7-conjugated anti-CD4, PerCp-Cy5.5-conjugated anti-CD3, APC-conjugated anti-CD161, APC-H7-conjugated anti-CD8, PE-conjugated anti-Perforin (eBioscience, San Diego, CA, USA) and BV421-conjugated anti-GrB; and the other with FITC-conjugated anti-IFN-γ, PE-Cy7-conjugated anti-CD4, PerCp-Cy5.5-conjugated anti-CD3, APC-conjugated anti-CD161, APC-H7-conjugated anti-CD8 and PE-Vio770-conjugated anti-TCRVα7.2 (Miltenyi Biotec, Bergisch Gladbach, Germany). Unstained PBMCs and isotype were used as controls. After the addition of Abs, the cells were incubated at 4℃ in the dark for 30 min and washed twice with wash buffer at 4℃. Lastly, 300 µl washing buffer (PBS, 1% BSA or 10% FBS, 0.1% NaN3) was added to each tube. Flow cytometry analysis was made using FlowJo for Windows, version 10.0.8 (FlowJo LLC, Ashland, OR, USA). The gating scheme is illustrated in Figures 1a and 2a. All cytokine measurements were background subtracted, taking into account the frequency of cells producing cytokines in the absence of PMA stimulation (mock control). For the phenotypic analysis of cytokine-producing cells, only those samples with >10 cytokine-positive events were considered.
33
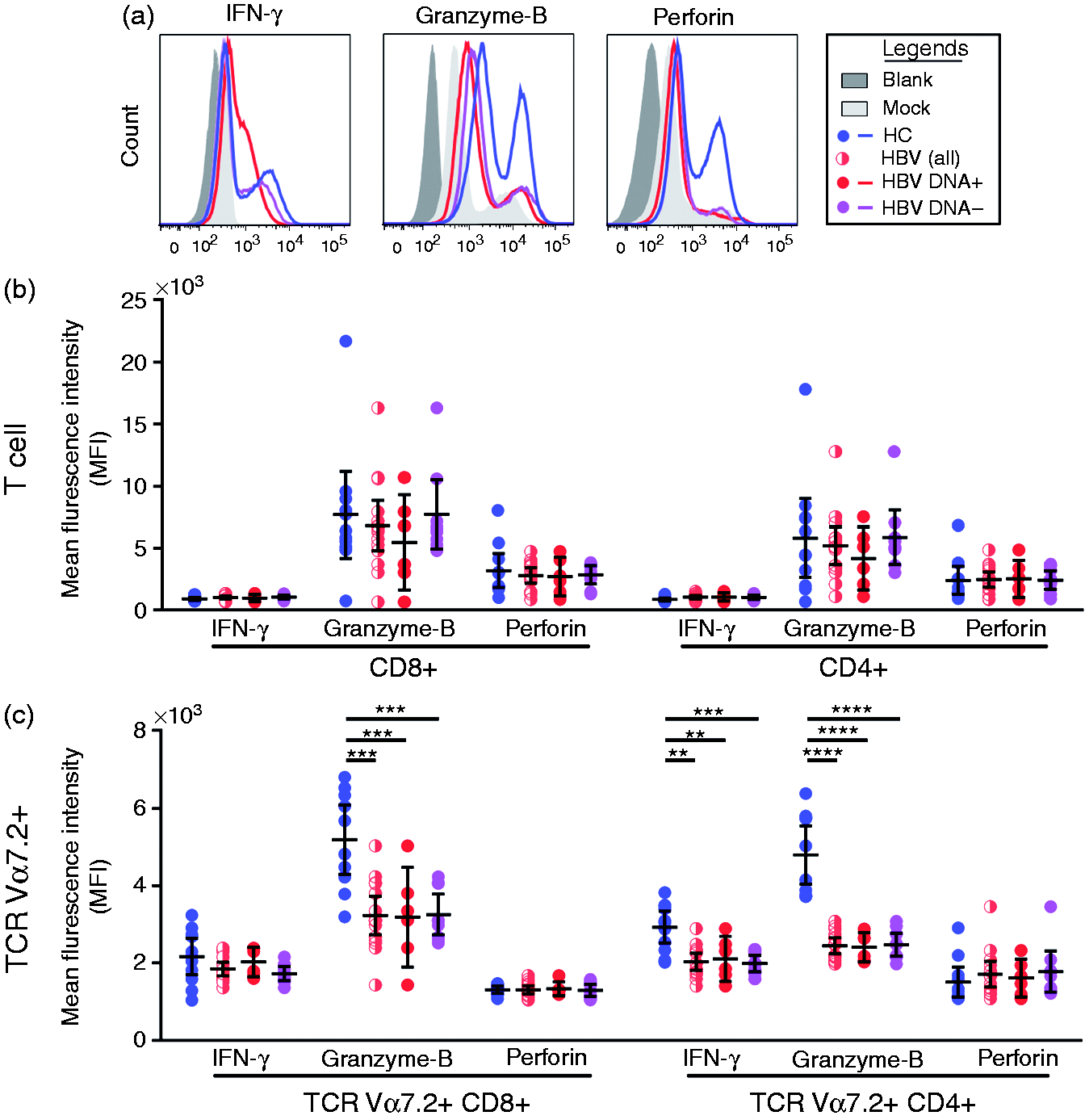
Polyfunctionality of T lymphocytes among chronic HBV-infected patients (CHB) and HCs. (a) Representative FACS plots showing the gating strategies, distribution of CD4+, CD8+ T cells and TCR Vα7.2+ MAIT cells. (b) Comparison of proportion of CD4+, CD8+ T cells and TCR Vα7.2+ MAIT cells among CHB patients with HBV DNA+, HBV DNA– and HCs. (c) Comparisons of cytokine pattern of T lymphocytes between CHB patients with HBV DNA+, HBV DNA– and HCs. Pie chart indicates the proportion of cells producing IFN-γ, granzyme-B (GrB), perforin and in any other combinations. (d) Comparisons of polyfunctionality of T lymphocytes (reflected as proportion of cells producing at least one type of cytokine) between CHB patients with HBV DNA+, HBV DNA– and HCs. P-Value <0.05 (significant correlation; * P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001). Kruskal–Wallis test was used to compare across the three study groups; MN, Mann–Whitney U-test, post-hoc test was used to compare across the two study groups. Expression levels of IFN-γ, GrB and perforin between CHB virus-infected patients with HBV DNA+, HBV DNA– and healthy controls (HCs). (a) Representative histogram showing the MFI of IFN-γ, GrB and perforin. (b) MFI of IFN-γ, GrB and perforin for CD8+ and CD4+ T cells. (c) MFI of IFN-γ, GrB and perforin for TCR Vα7.2+CD8+ and TCR Vα7.2+CD4+ T cells. Kruskal–Wallis test was used to compare across the three study groups; MN, Mann–Whitney U-test, post-hoc test was used to compare across the two groups. P-Value < 0.05 represents significant correlation (*P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001).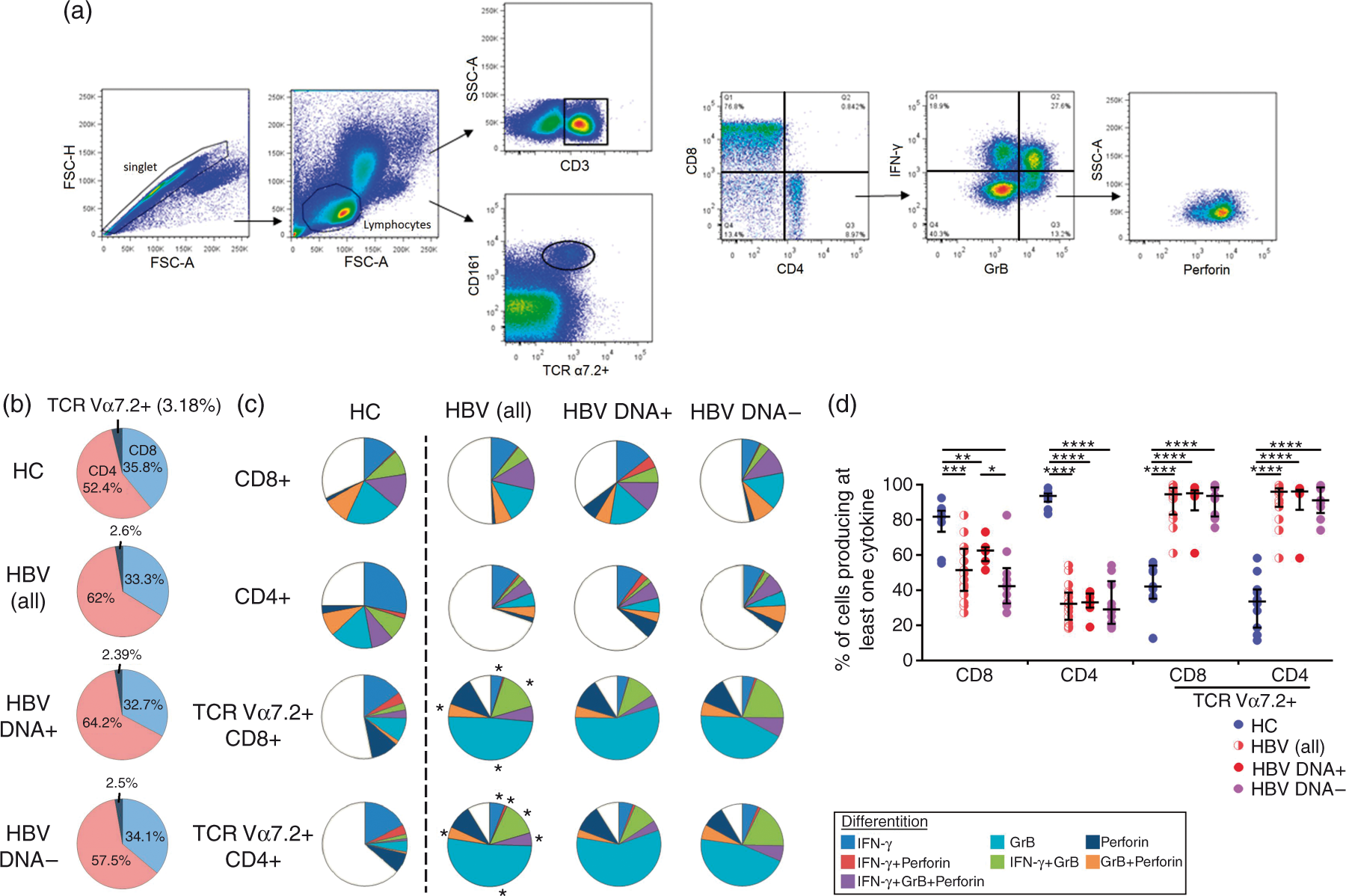
Statistical analysis
We primary compared the percentages and expression levels (mean fluorescent intensity, MFI) of GrB, perforin, IFN-γ and CD69 on different subsets of T cells and MAIT cells across all the three study groups. The percentages and expression levels of cytokines were then correlated with the percentages and expression levels of CD69 by Spearman rank test. Comparisons of categorical variables were tested using χ2test or Fisher’s exact test, whereas continuous variables were tested using the non-parametric Kruskal–Wallis test for multiple group comparisons followed by a post-hoc Mann–Whitney U-test for those that showed significant differences in Kruskal–Wallis test. Correlation between two continuous variables was compared using the Spearman’s rank correlation. Differences were considered significant at P < 0.05. All analyses and graphs were performed using GraphPad Prism 6 software (GraphPad, La Jolla, CA, USA).
Results
Patient cohort characteristics
The three-group, non-randomized study design consisted of a convenience sample of 26 subjects with chronic HBV infection: (1) group 1 (G1): HBV DNA-positive (n = 6); (2) group 2 (G2): HBV DNA-negative (n = 9) and (3) a control group (group 3; G3): HCs (n = 11). Patient HBeAg positivity was 83.3% in G1 and 88.9% in G2, while all patients were positive for HBsAg. There were no significant differences for demographic, laboratory and clinical data across the three study groups (Supplementary Table 1).
Percentage of TCR Vα7.2+CD161+ MAIT cells in the peripheral blood of CHB patients was marginally reduced
We first compared the percentages of CD4, CD8 and TCR Vα7.2+CD161+ MAIT cells on CD3+ lymphocytes (Figure 1b). Unlike CD4+ and CD8+ T cells, the frequencies of TCR Vα7.2+ CD161+ subset among HBV [HBV DNA+ (2.39%) and HBV DNA– (2.5%)] patients were marginally, but significantly (P < 0.05), reduced compared with HCs (3.5%).
Cytokine pattern of T cells and MAIT cells were perturbed among CHB patients
T-Cell exhaustion plays a critical role in T-cell dysfunction in chronic viral infections. 34 In order to characterize functional exhaustion of T cells in the study groups, we performed intracellular staining for GrB, perforin and IFN-γ, and compared their cytokine profile by relative proportion of total cells producing each possible combination of these cytokines. In general, we found that the changes of cytokine pattern for CD8+ and CD4+ T cells were minimal, and there was no significant difference in the cytokine pattern for CD8+ T cells, whereas the relative frequencies for perforin+ CD4+ T cells were higher in CHB [HBV DNA+ 3.4% (P = 0.0005) and HBV DNA– 2.7% (P = 0.0041)] patients compared with HCs (1.01%) (Figure 1c).
The cytokine pattern in TCR Vα7.2+CD161+ MAIT (both CD8+ and CD4+) cells was significantly perturbed with GrB+ and IFN-γ+GrB+ sub-population was profoundly expanded in CHB patients compared with HCs (Figure 1c). In addition, we also found that the polyfunctionality measured as relative frequency of cells producing at least one type of cytokine (GrB, perforin and/or IFN-γ in any combination) were significantly reduced in the T cells of CHB patients (HBV DNA+ CD8: 63.3%; HBV DNA– 42%; CD4: HBV DNA+ 34% and HBV DNA– 28%) compared with HCs (CD8: 81.7%; CD4: 93%). Interestingly, this pattern was reverse across the MAIT cell subset where the relative proportion of polyfunctional CD8+ T cells and CD4+ T cells was expanded in the CHB patients (HBV DNA+ CD8: 95% and HBV DNA– 93%; CD4: HBV DNA+ 96% and HBV DNA– 91%) compared with HCs (CD8: 57%; CD4: 33%) (Figure 1d). Taken together, our data suggested that there was a significant perturbation in T cells and MAIT cells among HBV-infected patients.
Chronic HBV infection led to decreased expression levels of intracellular cytokines relative to HCs
Next, we set out to determine whether the magnitude of T-cell response varied between the CHB groups and HCs. We found no significant difference in terms of magnitude of cytokine response in the CD4+ and CD8+ T-cell populations between CHB patients and HCs. Nevertheless, the level (MFI) of GrB in CD8+ MAIT cells (HBV DNA+ median = 2695; HBV DNA– median = 3218; HCs median = 5985) were significantly lowered in CHB patients compared with HCs. Furthermore, the GrB levels (HBV DNA+ median = 2888; HBV DNA– median = 2846; HCs median = 5256) and IFN-γ levels (HBV DNA+ median = 1680; HBV DNA– median = 1660; HCs median = 1960) in CD4+ MAIT cells were also significantly lowered in CHB patients compared with HCs (Figure 2b). These data suggest that the functional attributes of MAIT cells were significantly impaired in CHB patients.
Early activation marker CD69 was significantly reduced in chronic HBV-infected patients
CD69 is a co-stimulatory receptor and an early activation marker whose function is associated with the cytotoxic potentials of lymphocytes.
19
Given that the frequency of cytokine-producing cells and the levels of cytokines produced were reduced in patients with CHB, we investigated if such impairment was associated with the expression of CD69. We found that the frequency and level of expression of CD69, measured as MFI was generally decreased in HBV-infected patients (except the MFI of CD69 on TCR Vα7.2+CD4+ T cells), although the frequencies of TCR Vα7.2+ cells co-expressing CD69 was remarkably low (Figure 3a, b). In order to reconcile this seemingly contradictory result, we calcualted the intergrated MFI (iMFI) of the CD69 [(%) CD69 × (MFI) of CD69] in TCR Va7.2+CD4+ T cells. Our results showed that the total expression of CD69 (iMFI) was lower in CHB patients (HBV DNA+ = 1652 and HBV DNA– = 1580) as compared with HCs (29,800; P = 0.002 and P < 0.0001, respectively) (Figure 3c). Furthermore, Wilcoxon rank test analysis showed that there was no significant increment in CD69 expression (MFI) across the CHB patients after stimulation with PMA, although the stimulation experiments were performed the same way as in HCs (Figure 3d). Together, our data indicated that T cells of CHB patients represent an exhausted phenotype.
Expression levels of CD69 between CHB virus-infected patients with HBV DNA+, HBV DNA– and HCs. (a) Percentage of T lymphocytes expressing CD69 between CHB patients with HBV DNA+, HBV DNA– and HCs. (b) Expression levels of CD69 (measured as MFI). (c) iMFI of CD69 for TCR Vα7.2+CD4+ T cells. (d) Changes in CD69 expression following PMA stimulation. Kruskal–Wallis test was used to compare across the three study groups; MN, Mann–Whitney U-test, post-hoc test was used to compare across the two groups. Wilcoxon rank test was used to compare the levels of CD69 (MFI) before and after stimulation with PMA. P-Value <0.05 (significant correlation; *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001).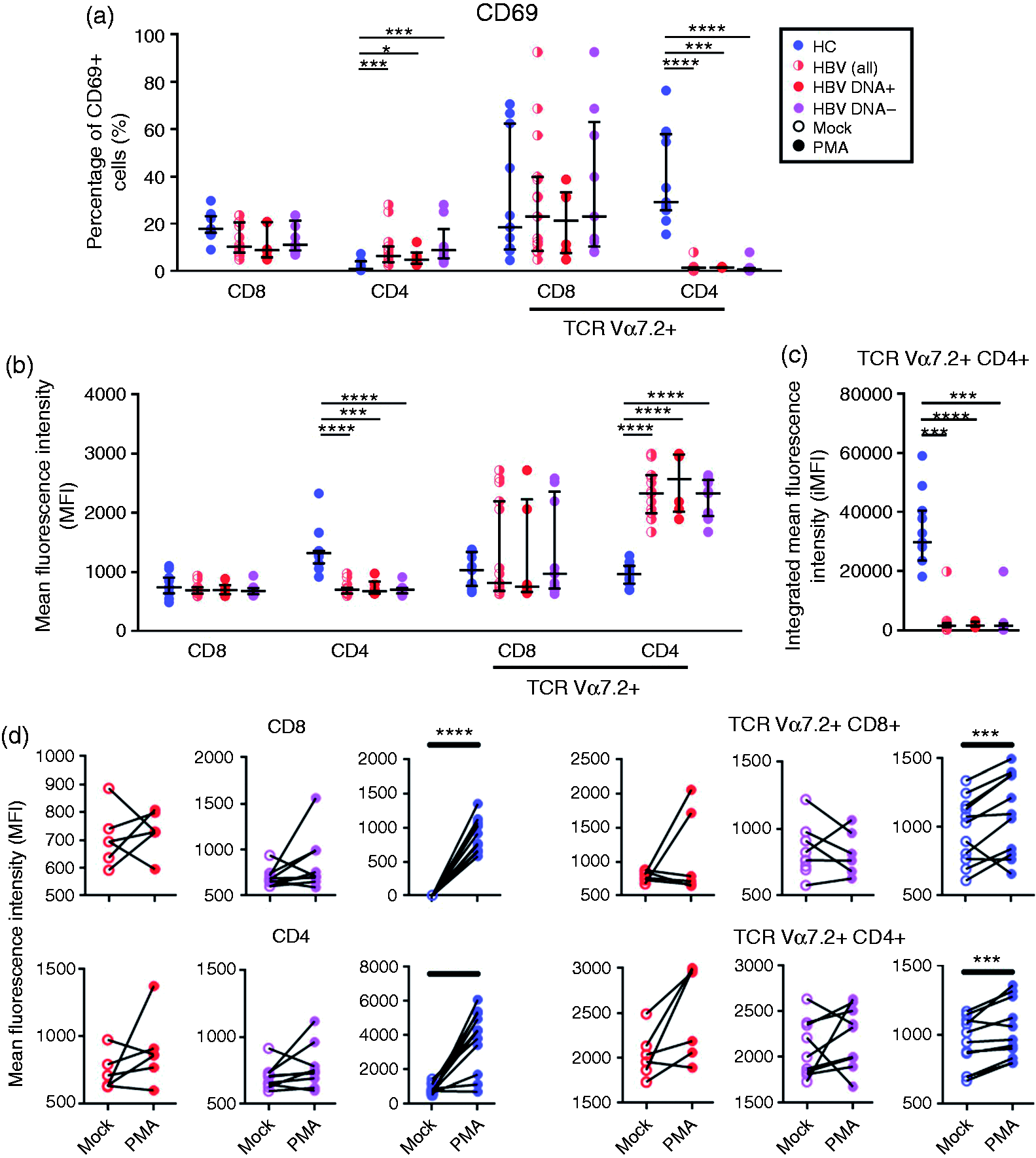
Attrition of CD69 correlated positively with intracellular cytokine levels in T cells across the different study groups
Given that the levels of cytokines and CD69 were lower in CHB patients, we were wondering if there existed any correlation between the levels of cytokines and CD69 expression. Spearman correlation analysis showed that the level of CD69 expression (MFI) positively correlated with the percentage of CD8+ T cells expressing GrB (P < 0.01) and perforin (P < 0.01) (Figure 4a, upper left panel). The MFI of CD69 was also correlated with the percentage of polyfunctional CD4+ T cells (r = 0.703, P < 0.0001) (Figure 4a, upper right panel).
Spearman correlation between CD69 and cytokine pattern, as well as expression levels of intracellular cytokines across the study populations. (a) Correlation between CD69 expression (MFI) and cytokine pattern. (b) Correlations between the frequency of CD69 (%) and cytokine expression levels. Spearman correlation was used to compare the levels of CD69 and cytokines. P-Value <0.05 (significant correlation; *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.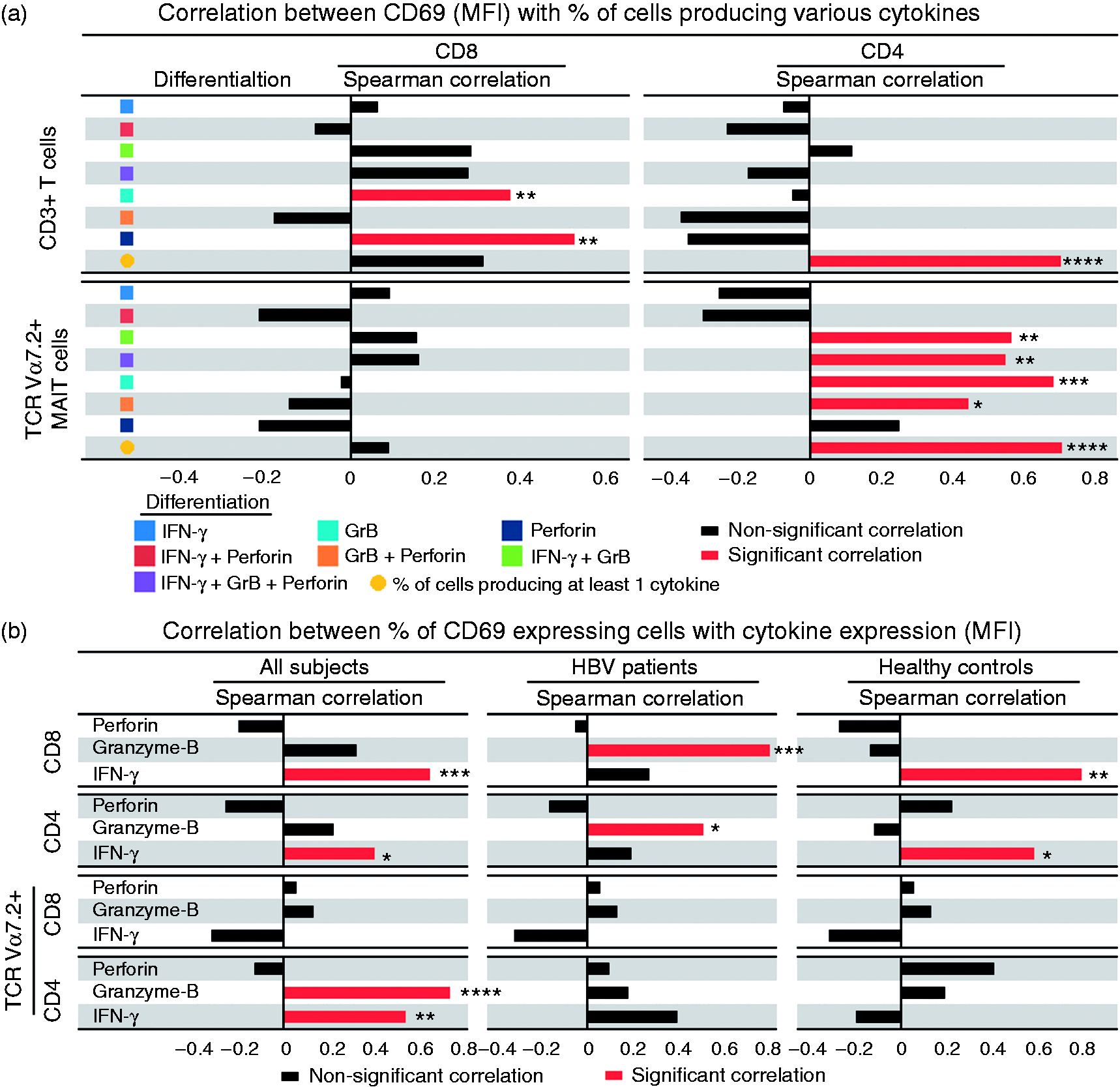
In MAIT cells, there were no association between MFI of CD69 with cytokine pattern in TCR Va7.2+CD8+ but a good correlation with the cytokine pattern, as well as percentage of polyfunctional TCR Vα7.2+CD4+ cells (r = 0.71, P < 0.0001) (Figure 4a, lower right panel). In addition, the frequency of cells expressing CD69 also correlated with levels of cytokine expression (MFI), particularly in CHB patients (Figure 4b) as compared with HCs. Our results suggest that CD69 could play a pivotal role in functional cytotoxic T-cell responses. Hence, attrition of CD69 expression among HBV-infected patients could lead to impaired cytotoxic functions and may likely impact HBV clearance.
Discussion
Up-regulation of immune exhaustion markers such as PD-1, TIM-3, LAG-3 and CTLA-4 have been shown to cause functional impairment of T cells and are implicated in chronic viral infections.35–39 However, the role of reduced levels of the co-stimulatory molecule CD69 in immune exhaustion is far less studied. Given that TCR Vα7.2+ MAIT cells are found in the systemic circulation and are enriched in the mucosa and liver, we postulated that functional exhaustion of MAIT cells may have an association with reduced levels of CD69, an early activation marker. We reported that the relative frequency of polyfunctional cells was significantly reduced in classical T cells of CHB patients, whereas these frequencies were increased in MAIT cells. Further, the proportion of IFN-γ+ and IFN-γ+GrB+-producing cells were significantly increased across the MAIT cell compartment of CHB patients. In spite of an increase in the frequency of cytokine-producing cells, the levels of expression (MFI) were lowered in MAIT cells of CHB patients as compared with HCs. Finally, we also found that the cytokine expression correlated with the expression levels of CD69, underlining the critical role of the co-stimulatory receptor in sustaining functional immune responses against chronic viral infections.
An effective T-cell response capable of viral clearance (of HBV infection) is characterized by active and sustained multi-epitope-specific, polyfunctional CD4+ and CD8+ T-cell responses.2,40 Conversely, chronic HBV infection fails to mount exuberant innate and adaptive immune responses against the virus, resulting in prolonged persistence of the virus in the host. 40 Here, we showed cellular immune responses in CHB patients are functionally impaired, especially in reduced polyfunctionality aspects in the periphery. Further, the functional impairment in TCR Vα7.2+CD4+ appears to be profound as compared with the TCR Vα7.2+CD8+ cells, as reflected by the magnitude of cytokines produced, as well as the expression CD69 levels.
It is widely believed that CD4+ T cells coordinate the diverse arms of the adaptive immune system, providing support for sustained activation, proliferation and survival of cytotoxic CD8+ T and B cells.41,42 However, perforin/GrB-positive cytotoxic CD4+ T cells with direct cytotoxic effector functions have been studied,43–46 and are implicated in the control of several chronic viral infections such as Epstein–Barr virus, 47 HCV 48 and HIV-149 viruses. Furthermore, in a large cohort that consisted of 547 cases with HCC, the frequency of circulating and liver-infiltrating cytotoxic CD4+ T cells was found to be increased during the early stages but gradually depleted as the disease progressed, and this correlated with poor survival, as well as high recurrence rates in the patients. 50 Though the precise role of cytotoxic CD4+ T cells in CHB infection is not completely understood, and given that the TCR Vα7.2+ T cells are enriched across the mucosa, the role of TCR Vα7.2+CD4+ T cells in anti-HBV immunity and their prognostic value should be investigated using a larger cohort.
CD69 is an early immune activation marker that regulates the activation and migration of lymphocytes during T-cell activation. 51 It is present in the cytoplasm of resting T cells, 52 and its surface expression can be observed ∼2 h post-activation on T cells, B cells and NK cells, 17 without the involvement of new mRNA expression or protein synthesis. 52 The importance of CD69 to mount an effective immune response against M. tuberculosis infection has previously been demonstrated. 53 In a cohort of 33 tuberculous pleurisy patients, the authors showed that CD8+ T cells in pleural fluid expressed significantly higher levels of CD69 than that found in the peripheral circulation. Further, CD69 was expressed preferentially on cells with activated memory phenotype (CD45RO+ HLA-DR+), with increased CCR6 (mucosal homing receptor) together with decreased homing receptor for secondary lymphoid tissue (CCR7 and CD62L). 54 This suggests that CD69 might also facilitate the migration of circulating lymphocytes to the sites of infection. Moreover, the authors also showed that CD69 expression was associated with polyfunctionality, in this case CD107a/b, IFN-γ and TNF-α expressing CD8+ T cells. 53 Consistent with the observation here we observed a significant reduction of both CD69 and polyfunctionality, especially in TCR Vα7.2+CD4+ T cells among CHB patients. Further, the levels of CD69 correlated with polyfunctional TCR Vα7.2+CD4+ T cells indicating the pivotal role of CD69 against chronic HBV infection.
There are certain limitations to the current study; the first and foremost being the smaller sample size. Secondly, the study lacks other HBV disease groups such those with HCC and liver cirrhosis as internal controls and for improved and convincing comparisons. Notwithstanding these limitations, we have clearly reported the profound functional impairment in TCR Vα7.2+CD4+ T cells of chronic HBV infection. Future studies to longitudinally investigate the functional role of TCR Vα7.2+CD4+ cells in the pathogenesis of HBV disease and progression using a larger cohort is warranted.
In conclusion, we found that the polyfunctionality of peripheral T cells was significantly reduced among CHB patients. The impairment was most profoundly observed in the TCR Vα7.2+CD4+ T cells and the levels of cytokine expression correlated with the levels of functional intracellular cytokines.
Footnotes
Acknowledgements
The author(s) are grateful to all the participants, clinical, paraclinical and laboratory staff of University of Malaya Medical Centre for assistance with patient recruitment, specimen collection and cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Universiti Malaya Research Grant (UMRG), RP021A-13HTM to Jayakumar Rajarajeswaran and HIR E000063 to Mohamed Rosmawati. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
