Abstract
TLRs have a fundamental role in immunity. We have recently reported that stimulation of TLR2 and TLR5 in freshly isolated and activated human T cells with microbial ligands without concomitant activation through the TCR brings about secretion of neutrophil chemoattractant, CXCL8, and effector cytokine, IFN-γ, respectively. However, the mechanism of TLR signaling in T cells has not been worked out. Here, we show that the Src family kinases, p56lck (Lck) and p59fyn (Fyn), which are essential for activation of T cells through the TCR, are also critical for signal transduction through TLRs in human T cells. The secretion of CXCL8 following stimulation of the model human T cell line, Jurkat, with the TLR5 ligand, flagellin, was reduced in presence of the Src-kinase inhibitor, PP2 and specific inhibitors of Lck and Fyn. These inhibitors suppressed generation of activated JNK and p38, which were both required for TLR-induced CXCL8 production. The Lck-deficient derivative of Jurkat, JCam1.6, responded poorly to TLR2, TLR5 and TLR7 agonists, and did not generate active signaling intermediates. Lck and Fyn inhibitors also reduced TLR5-induced IFN-γ secretion from the activated T cell phenotype-representing T cell line, HuT78, without modulating JNK and p38 activation. These results reveal that TCR and TLRs share key proximal signaling regulators in T cells.
Introduction
TLRs recognize conserved PAMPs and play a fundamental role in inflammation and immunity.1,2 These receptors have been mainly studied in innate immune cells, including macrophages, neutrophils, dendritic cells and epithelial cells lining mucosal surfaces.1,2 However, recent studies suggest that functionally competent TLRs are also present on T lymphocytes and their activation either alone or in conjunction with concurrent TCR ligation brings about secretion of cytokines and chemokines from T cells.3–7 Their ability to activate secretion of chemokines such as neutrophil chemoattractant CXCL8 in the absence of TCR-generated signals suggests that T cells can also directly participate in innate immunity as neutrophils are known to play a vital role in killing bacterial pathogens. 8 We have recently shown that human T cells and model T cell lines, Jurkat and HuT78, readily respond to activation with TLR ligands and these responses are modulated by prior activation of T cell through TCR. 7 These results raised the possibility that signaling intermediates generated through activation of the TCR might participate in regulating TLR responses in T cells. Here, we provide evidence that Src-kinases, Lck and Fyn, which play a vital role in signal transduction through the TCR, are also required for driving TLR-activated innate immune response from human T cells.
Materials and methods
Cells and reagents
The human T cell lymphoma line Jurkat and its Lck-deficient derivative, Jcam1.6, were obtained from the American Type Culture Collection (ATCC). The human cutaneous T lymphocyte line HuT78 was acquired from Cell Repository, National Centre for Cell Sciences, India. PP2 and Lck inhibitor 7-cyclopentyl-5-(4-phenoxyphenyl)-7H-pyrrolo [2,3-d] pyrimidin-4-ylamine were obtained from Sigma Chemical (St. Louis, MO, USA). Fyn inhibitor (SU6656), p38 inhibitor (SB203580) and JNK inhibitor II were purchased from Calbiochem (Billerica, MA, USA). Abs against pJNK, pp38, pIkBα, IkBα and actin were from Cell Signaling (Danvers, MA, USA). Pam3CSK4 and Imiquimod were purchased from Invivogen (New York, NY, USA. Flagellin was isolated from Salmonella Typhi grown in Luria Bertani broth as described by Smith et al. 9 Anti-Salmonella Typhi flagellin-specific monoclonal Ab has been described previously. 10
Stimulation of T cells
T cells (Jurkat and HuT78) were stimulated with TLR ligands at 37℃ in an atmosphere of 5% CO2 for 24 h. As Jurkat responds to TLR activation efficiently in the absence of serum, cell stimulations with this cell line and JCam1.6 were carried out in serum-free medium. 6 CXCL8 and IFN-γ levels were determined in the supernatants by ELISA. In some experiments, cell stimulations were carried out in presence of Src-kinase inhibitor PP2, or specific inhibitors of Lck, Fyn and MAP kinases (p38 and JNK).
Binding of flagellin to T cells
Jurkat cells were washed with serum-free medium and incubated with different concentrations of flagellin for 1 h at 4℃. After extensive washing, cells were lysed and cell-bound flagellin was analyzed by Western blotting with flagellin-specific monoclonal Ab. Bands were visualized using enhanced chemiluminescence (ECL).
Analysis of intracellular signaling
Jurkat and HuT78, in the presence or absence of various inhibitors, and JCam1.6 cells were stimulated with TLR5 agonist flagellin at different time points. Cells were lysed with TKM buffer (Tris HCl 50 mM, KCl 25 mM, MgCl2 5 mM, EDTA 1 mM, NaN3 0.02% and 1 mM PMSF) containing 1% Triton-X 100, boiled with Laemmli sample buffer and run in a 12% SDS polyacrylamide gel. The proteins were transferred to a nitrocellulose membrane and probed with Abs against various signaling intermediates. The blot was developed using ECL reagent.
Densitometry analysis
The intensities of individual bands in the Western blots were quantified using ImageJ software. The arbitrary density unit of each experimental band was calculated by determining the ratio of its intensity and that of a protein band used as a loading control.
Statistical analysis
Student’s t-test with a two-tailed distribution was used to calculate P-values for unequal variances. A P-value <0.05 was considered statistically significant. Data are expressed as mean ± SD. Error bars represent SD.
Results and discussion
Src family kinases are essential for TLR-mediated secretion of CXCL8 and IFN-γ from human T cells
Tyrosine phosphorylation of TCR–CD3 associated immunoreceptor tyrosine-based activation motifs (ITAMs) is a key step in the initiation of intracellular signaling through the TCR, and the Src-family kinases are the key to bringing about these phosphorylations.
11
Our recent study showed that model human T-cell lines, Jurkat and HuT78, mimic freshly isolated and activated human T cells in their response to innate activation with TLR ligands.
7
These cell lines were therefore chosen in the present study as model T-cell lines to analyze possible involvement of Src kinases in the induction of innate immune response from human T cells. As shown previously,6,7 Jurkat readily produced CXCL8 upon stimulation with the TLR5 ligand, flagellin (Figure 1A). This response was inhibited in the presence of Src kinase inhibitor, PP2, in a dose-dependent fashion (Figure 1A), suggesting that one or more Src family tyrosine kinases might be required for the induction of CXCL8 from human T cells activated with TLR ligands. Treatment of this cell line with specific inhibitors of the Src kinases, Lck and Fyn, also resulted in a dose-dependent reduction in CXCL8 secretion in response to TLR5 activation (Figure 1B).12,13 Fyn inhibitor, SU6656, also inhibits Lck. However, its IC50 value for Lck is about 40 times higher than that for Fyn; therefore, the inhibition seen with different concentrations of SU6656 in our experiments is unlikely to be due to targeting of Lck but to specific inhibition of Fyn.
13
The inhibition observed with various inhibitors was not due to any cell death, as determined by staining cells with propidium iodide (data not shown). Consistent with the results obtained with the Lck inhibitor, JCam1.6, a Jurkat-derived T-cell line that lacks Lck and expresses lower levels of Fyn,
14
produced negligible amounts of CXCL8 in response to TLR2 (Pam3CSK), TLR5 (flagellin) and TLR7 (Imiquimod) ligands (Figure 1C). This lack of response was not due to reduced binding of the ligand to cells, as the amount of flagellin found associated with WT Jurkat and JCam1.6 was comparable (Figure 1D). WT Jurkat and JCam1.6 showed comparable levels of surface TLR5 as determined by staining with anti-hTLR5 Ab (data not shown). The inhibitors of Lck and Fyn also produced a dose-dependent reduction in IFN-γ secretion from HuT78 activated with flagellin (Figure 1E). These results established that Lck and Fyn are essential for TLR-induced secretion of CXCL8 and IFN-γ from human T cells.
Src family kinases, Lck and Fyn, are required for TLR-activated CXCL8 and IFN-γ secretion from human T cells. Jurkat cells were stimulated with flagellin for 24 h in presence or absence of varying concentrations of the Src kinase inhibitor, (A) PP2 or (B) Lck and Fyn inhibitors. CXCL8 levels in the supernatant were determined by ELISA. Data are representative of three independent experiments. (C) WT and Lck deficient Jurkat (JCam1.6) cells were activated with TLR2, TLR5 and TLR7 agonists for 24 h following which CXCL8 secretion was measured by ELISA. Data are representative of two independent experiments. (D) WT and JCam1.6 cells were washed free of serum and incubated with flagellin for 1 h at 4℃. The levels of bound flagellin were determined by immunoblotting with flagellin-specific monoclonal Ab. The blot was subsequently probed with anti-actin Ab. Data are representative of three independent experiments. The blot was scanned and band intensities were quantitated by densitometry. (E) HuT78 cells were stimulated with flagellin (10 ng/ml) in presence or absence of inhibitors of Lck and Fyn. Twenty-four h later IFN-γ levels in the supernatants were determined by ELISA. Data are representative of three independent experiments. *P < 0.05, **P < 0.001, ***P < 0.0001. In (A), (B), (C) and (E), CXCL8 and IFN-γ values (ranging from 5 to 17 pg/ml) of unstimulated cells were subtracted from those obtained with TLR-stimulated cells.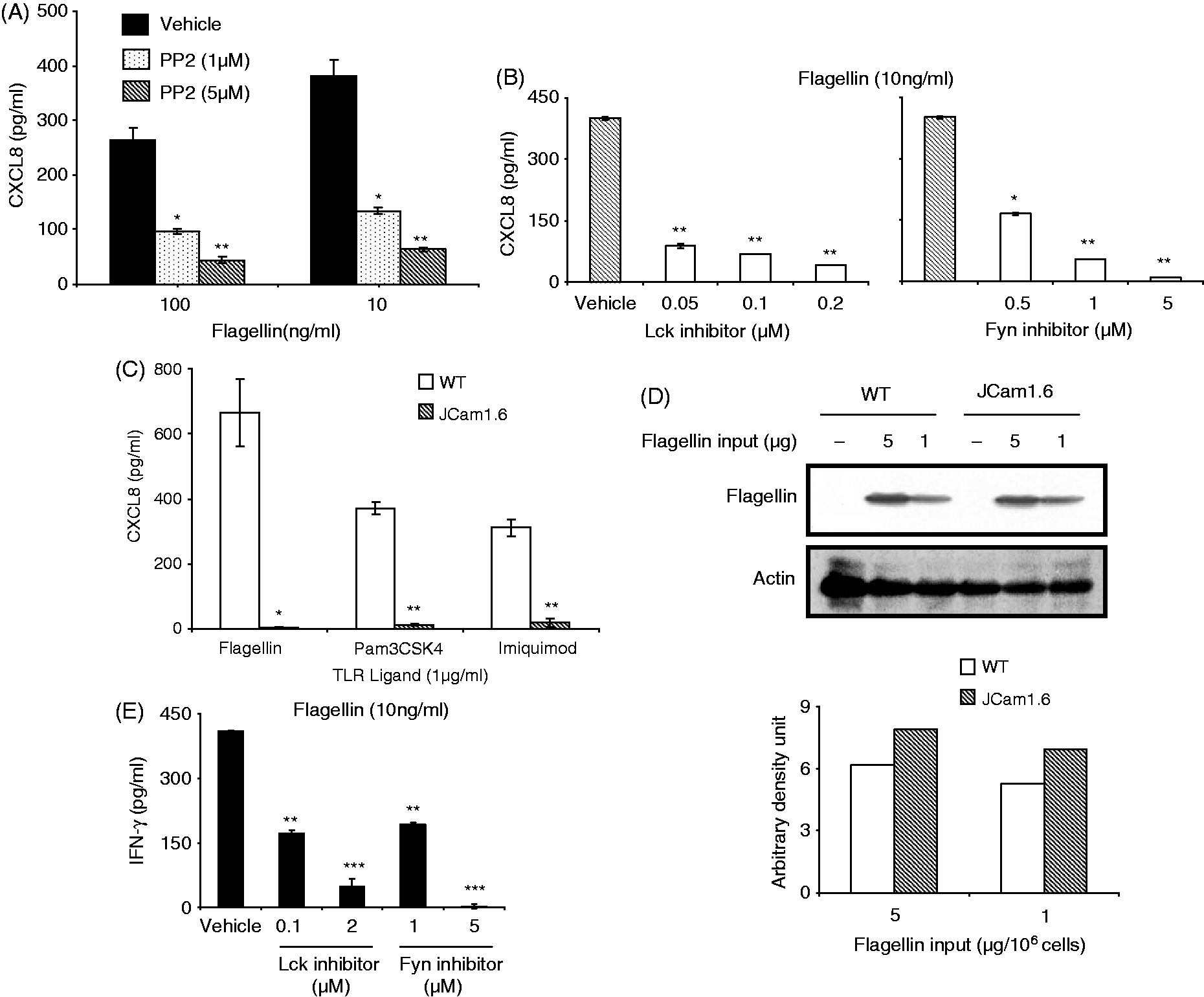
Lck and Fyn regulate TLR-induced CXCL8 and IFN-γ through different mechanisms
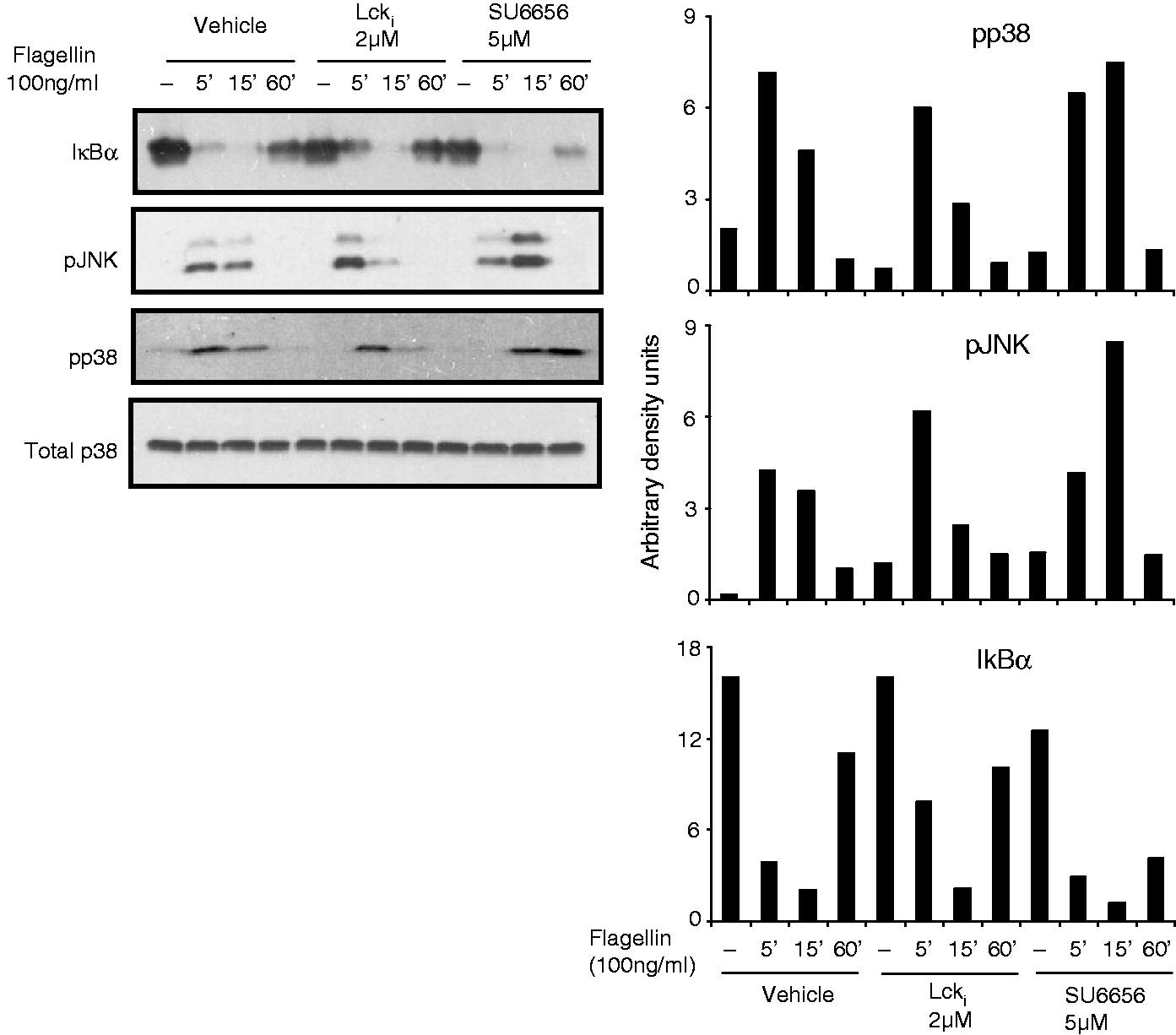
To understand how Lck and Fyn might be regulating intracellular signaling in TLR-activated T cells, the activation of MAP kinases and NF-κB pathway of intracellular signaling was investigated. Flagellin-stimulation of Jurkat resulted in a time-dependent phosphorylation of MAP-kinases, p38 and JNK, and degradation of IkBα, indicative of NF-κB activation (Figure 2A). Treatment of cells with inhibitors of Lck and Fyn reduced activation of both JNK and p38 but did not affect degradation of IκBα (Figure 2A). In contrast, phosphorylation of IKKαβ and IκBα, as well as degradation of IκBα and activation of MAP kinases, was completely abrogated in JCam1.6 (Figure 2B). The significance of JNK and p38 in TLR5-activated CXCL8 production was revealed by suppression of this chemokine in presence of inhibitors of these kinases (Figure 2C). However, in HuT78, the reduction in flagellin-stimulated IFN-γ secretion with Lck and Fyn inhibitors was not associated with reduced activation of MAP kinases or reduced degradation of IκBα (Figure 3).
Lck and Fyn regulate TLR signaling in Jurkat by modulating activation of MAP kinases. (A) Jurkat cells were stimulated with flagellin (100 ng/ml) in the absence or presence of Lck inhibitor (0.1 µM) or Fyn inhibitor (1 µM) for different time points. (B) WT and JCam1.6 cells were stimulated with flagellin (100 ng/ml) for different time points. Cells were washed, lysed and the activation status of various intracellular signaling intermediates of the NF-κB and MAP kinase pathways was checked by Western blotting. The band intensities were measured by densitometry. Data are representative of three independent experiments. (C) Flagellin stimulation of Jurkat cells was carried out in presence or absence of the MAP-kinase inhibitors (p38 and JNK) for 24 h. CXCL8 levels were determined in the supernatants by ELISA. Data are representative of three independent experiments *P < 0.05, **P < 0.001. Lck and Fyn do not modulate activation of MAP kinases during TLR5 stimulation of HuT78. HuT78 cells were incubated with flagellin (100 ng/ml) in the absence or presence of Lck inhibitor (2 µM) or Fyn inhibitor (5 µM) for different time points. The activation of various intracellular signaling intermediates was checked by Western blotting. The band intensities were determined by densitometric analysis. Data are representative of three independent experiments.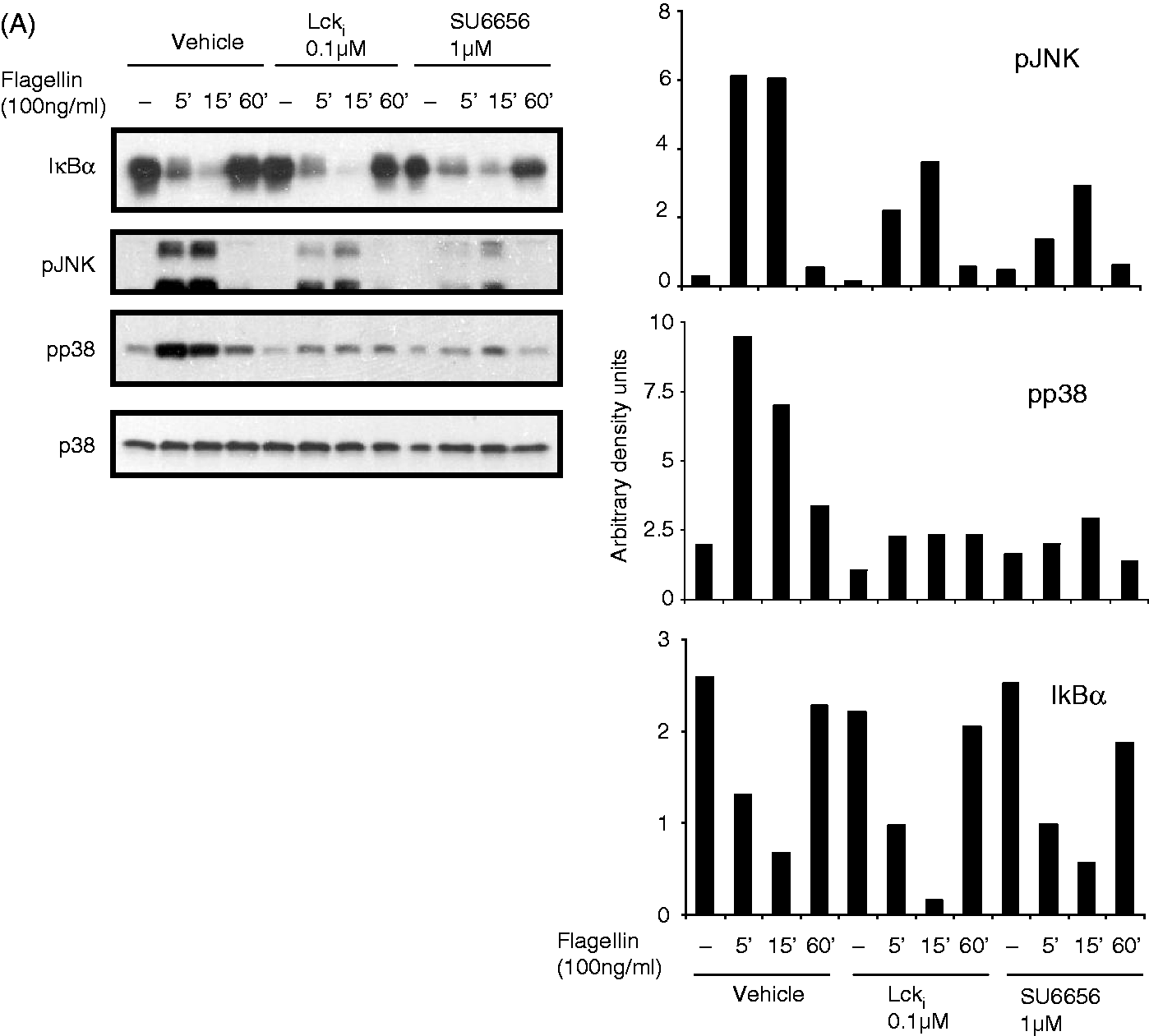
These results show, for the first time, that TLR-activated production of CXCL8 and IFN-γ in human T cells is dependent on the Src-kinases, Lck and Fyn. Analysis using specific inhibitors of these two Src kinases in Jurkat suggested that these kinases regulate production of CXCL8 by regulating activation of JNK and p38. However, experiments with Lck-null Jurkat line, JCam1.6, indicated that Lck or both Lck and Fyn might not only regulate activation of MAP kinases, but also that of NF-κB pathway of intracellular signaling, the latter most likely through a mechanism that is independent of kinase activities of Lck and Fyn. A similar kinase-independent role for Lck has been previously suggested for TCR activation. 15 In HuT78, these Src kinases regulated TLR-induced secretion of IFN-γ without modulating activation of p38 MAP kinase, which has been previously shown to be critical for the induction of this cytokine in response to stimulation with flagellin. 7 These results suggest that Lck and Fyn might regulate secretion of CXCL8 and IFN-γ from TLR-activated T cells through different pathways.
The role of tyrosine phosphorylation and the involvement of the Src kinases therein has been previously suggested in studies on TLR activation in macrophages and dendritic cells, and in cell lines transfected with different TLRs.16–20 Ivison et al. reported that tyrosine phosphorylation of TLR5 in its cytoplasmic tail is required for flagellin-induced CXCL8 secretion in epithelial cells. 17 Tyrosine phosphorylations have also been reported in accessory molecule MD2 and TLR adaptor proteins MyD88, Mal, TRIF and TRAM;21–25 therefore, it is possible that Lck and Fyn regulate TLR responses through tyrosine phosphorylation of TLR and/or adaptor proteins involved in TLR signaling. The exact targets of Lck and Fyn will be identified in future studies.
Lck (as well as Fyn) is normally present in a less active state in resting T cells and becomes activated through dephosphorylation of regulatory c-terminal tyrosine (Y505) followed by autophosphorylation of a tyrosine residue in the kinase domain (Y394) when cells are stimulated through the TCR. 26 This dephosphorylation and phosphorylation were not readily observed during TLR activation of T cells (data not shown); therefore, basal activity of Lck seems to be sufficient to participate in intracellular signaling through TLRs. Several studies have suggested that translocation of TLRs into lipid rafts might be required for transduction of signals through these receptors.27–30 It is therefore possible that in T cells, post-engagement with their ligands, TLRs become readily available for phosphorylation by Lck and Fyn, which are known to be enriched in lipid rafts. Thus, the functioning of these Src kinases during TLR activation might be distinct from that of their functioning during TCR signaling. Understanding the exact mechanism of this mode of functioning might unravel novel aspects of the biology of these kinases in receptor signaling. Taken together, our findings assign a critical role to Lck and Fyn that are central to TCR signaling, in the activation of TLR-driven innate responses from human T cells.
Footnotes
Acknowledgements
We would like to thank Drs. Rahul Pal, Devinder Sehgal, Pushkar Sharma, Praful Tailor and Chinmay Mukhopadhyay for their valuable inputs in the course of this study, and members of the Ayub laboratory for insightful discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Biotechnology, Government of India, through the National Institute of Immunology. N.S. and A.S.A. received research fellowship from the Council of Scientific and Industrial Research, Govt. of India.
