Abstract
Our study aimed to assess the influence of common methylprednisolone therapy on innate inflammatory factors in potential brain-dead organ donors (BDDs). The study groups consisted of 50 potential BDDs who received 15 mg/kg/d methylprednisolone and 25 live organ donors (LDs) as control group. Innate immunity gene expression profiling was performed by RT-PCR array. Soluble serum cytokines and chemokines, complement components, heat shock protein 70 (HSP70) and high mobility group box-1 (HMGB1) were measured by ELISA. Surface expression of TLR2 and TLR4 were determined using flow cytometry. Gene expression profiling revealed up-regulation of TLRs 1, 2, 4, 5, 6, 7 and 8, MYD88, NF-κB, NF-κB1A, IRAK1, STAT3, JAK2, TNF-α, IL-1β, CD86 and CD14 in the BDD group. Remarkably, the serum levels of C-reactive protein and HSP70 were considerably higher in the BDD group. In addition, serum amounts of IL-1β, IL-6, TNF-α, HMGB1, HSP70, C3a and C5a, but not IL-8, sCD86 or monocyte chemoattractant protein-1, were significantly increased in the BDD group. Significant differences were observed in flow cytometry analysis of TLR2 and TLR4 between the two groups. In summary, common methylprednisolone therapy in BDDs did not adequately reduce systemic inflammation, which could be due to inadequate doses or inefficient impact on other inflammatory-inducing pathways, for example oxidative stress or production of damage-associated molecules.
Introduction
Experimental and human studies suggest that brain death induces an inflammatory condition that significantly affects graft quality and function. 1 Systemic inflammatory response in brain-dead donors (BDDs) may occur through several mechanisms, the first of which is the ischaemic brain release of circulating inflammatory mediators, which may induce a systemic inflammatory response. Patients with traumatic brain injury have shown a release of IL-6 from the intracranial space to the systemic circulation. 2 In the second, ischaemia of the gut and flow-induced shear stress of endothelial cells leads to inflammatory cytokine release. 3 In the third, the change from aerobic to anaerobic metabolism associated with severe vasoconstriction results in a catecholamine storm and may induce a systemic inflammatory response. 4 As a consequence, activation of innate immunity leads to increased transcription of a range of inflammatory cytokines and chemokines. 5 Furthermore, the damage-associated molecular patterns (DAMPs) induced by potential oxidative injury generate immunostimulatory mature dendritic cells that translate innate to adaptive immunity. 6 Therefore, it has been thought that brain death promotes up-regulation of inflammatory mediators in peripheral organs of donors. 7 Also, it is hypothesised that this Ag-independent inflammation may be the cause of the rate and intensity of host alloresponsiveness after engraftment. 8 Studies on inflammation in brain-dead kidney donors have produced conflicting results. Nijboer et al. reported no differences in expression levels of intercellular adhesion molecules (ICAM)-1 and vascular cell adhesion molecule (VCAM)-1, as well as in IL-6, IL-1β, IL-8, monocyte chemoattractant protein (MCP)-1, inducible NO synthase, TGF-β, E-selectin and heat shock proteins (HSPs) in BDDs compared with live donors. 9 Controversially, Koo et al. and Schwarz et al., in separate studies,10,11 have demonstrated significant increases in ICAM-1 and VCAM-1 in kidney biopsies drawn after cold ischaemia time from BDD compared with living donors (LDs). Biopsies drawn after cold ischaemia show higher expression levels of IL-10, IFN-γ and TNF-α mRNA, but not IL-1β, IL-6 or IL-15 in BDDs compared with LDs. 12 In another study, mRNA expression of IL-10, IL-6 and IL-2 were significantly up-regulated after reperfusion of kidneys from BDDs. 13 Krüger et al. have reported that, consequently, TLR4 was up-regulated in BDDs compared with LDs. They also found that kidney biopsies from BDDs showed significant increase in high mobility group box-1 (HMGB1) in comparison with biopsies from living donors. 14 Some studies have suggested that ischaemia–reperfusion injury mediated by HMGB1 activated TRIF-adaptor independent TLR4 signalling. 15
Many studies have shown that organs derived from BDDs have lower graft survival and higher graft-function loss in comparison with LDs.16,17 Accelerating rejection of renal allografts and inferior graft survival in BDD recipients has led to an increased need for interventions in order to optimise and improve graft quality.8,18,19 Corticosteroids are used to reduce inflammatory organ injury and promote haemodynamic stability after brain death.20,21 Based on comparisons with untreated controls, high-dose methylprednisolone (MP) has become the standard regimen for treatment of BD donors. 21 In vitro and experimental studies have suggested high corticosteroid doses prevent production of IL-1, IL-2 and TNF-α, and IFN-γ synthesis by inhibition of transcription factor NF-κB. 22 One study has suggested that protein–protein interaction between activated glucocorticosteroid receptor and signal transducer and activator of transcription (STAT) proteins, activator protein 1 (AP-1) and NF-κB result in regulation of inflammatory gene expression. 23 A prospective study of humans on liver transplantation has reported decreased tissue expression and serum levels of pro-inflammatory cytokines after steroid therapy in BDDs. 1 Prednisolone treatment reduces circulating IL-6 and complement component 3 (C3) expressions in the kidney but not in the liver. 24 Kotsch et al. demonstrated that MP treatment resulted in a significant decrease in intra-graft mRNA levels of ICAM, MHC class II, Fas ligand, inducible protein-10 and CD68. In addition, decreased levels of several cytokines including MCP-1, IL-2, IL-6, TNF-α and inducible protein-10, were detected. However, there was no significant difference in expression of TLR2 and TLR4. Hence, this study aimed to detailed evaluation of methylprednisolone on inflammation in BDDs.
Materials and methods
Patients
Demographic characteristics of the donors. The parameters used for matching (age, sex, cause of death and used drugs in BDDs) between the two groups are listed in the table.
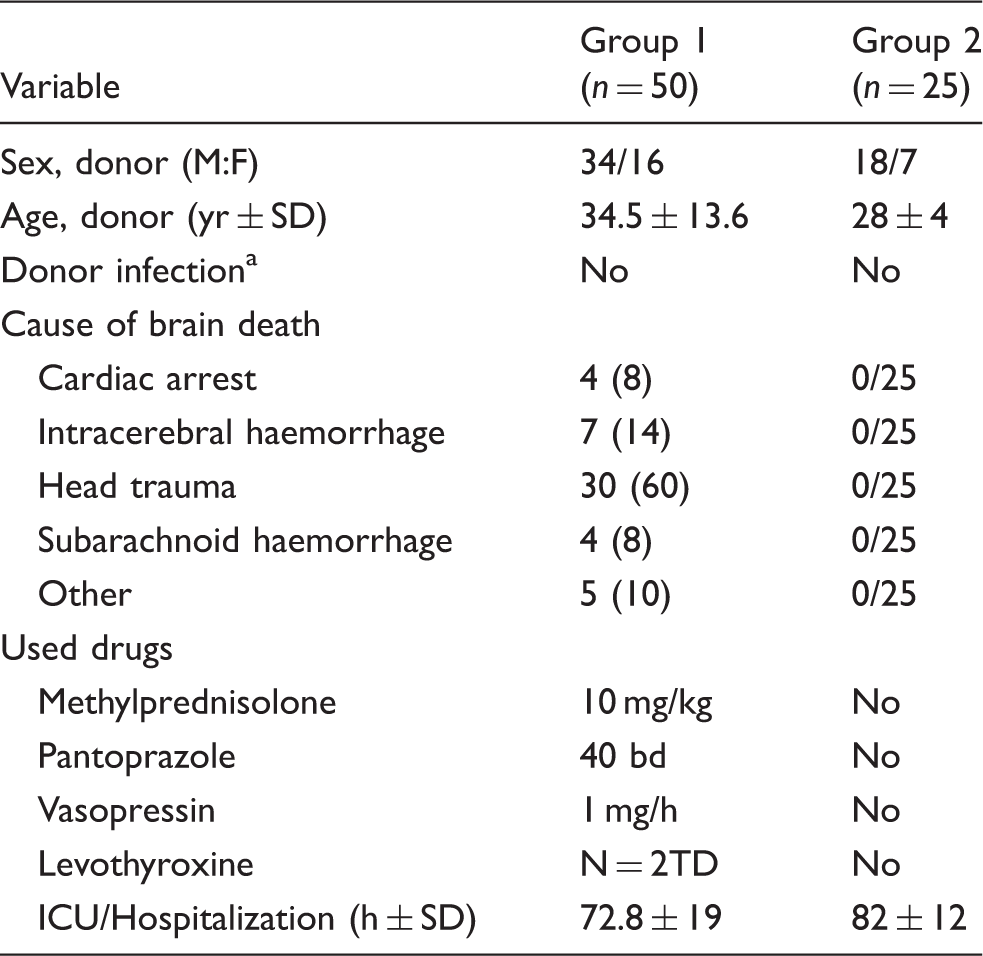
Data are n (%) unless otherwise stated. aDefined as infection of the donor requiring antibiotic treatment.
Isolation of peripheral blood mononuclear cells and plasma
After consent for organ donation and just before donor harvesting, blood samples were collected into EDTA tubes. Plasma samples were aliquoted in cryogenic vials and stored below −80℃ immediately until analysis. Blood mononuclear cells were isolated by Ficoll/Paque density gradient centrifugation and then washed in Hanks solution and used for either flow cytometry or RNA extraction.
RNA extraction
RNA was extracted by Trizol (Lot. 94402; Sigma Aldrich, St Louis, MO, USA), according to the manufacturer’s protocol. Briefly, 600 µl of Trizol per 106 cells was added to each sample and then 150 µl chloroform per 600 µl Trizol was added and mixed vigorously before centrifuging at 12,000 g for 10 min at 4℃. The aqueous layer was then collected carefully to another tube. The extracted solution was mixed with the same volume of isopropanol before incubation at −20℃ for 1 h. The micro-tubes were centrifuged for 10 min at 12,000 g. One hundred microlitres of 70% EtOH was added to the discarded supernatant. The tube was again centrifuged at 7000 g for 10 min at 4℃ and further purified with the RNeasy mini kit (Cat No 74104; QIAGEN, Hilden, Germany), according to the manufacturer’s protocol.
PCR array
For elimination of genomic DNA contaminants, a concentration of 5 µg/μl RNA in a final volume of 10 μl was treated with GE buffer, according to the RT2 First Strand Kit protocol (Cat. No 330401; QIAGEN). The genomic DNA elimination mix was incubated for 5 min at 42℃, and then placed immediately on dry ice for 1 min. The cDNA was then generated using RT2 First Strand Kit. At first, the reverse-transcription mix was prepared as per the following protocol for three reaction: 12 µl 5× buffer BC3, 3 µl control P2, 6 µl RE3 reverse transcriptase mix and 9 µl RNase-free water in total volume of 30 µl. Reverse-transcription mix at 10 µl was added to each tube containing 10 µl genomic DNA elimination and incubated at 42℃ for 15 min. The reactions were stopped immediately by incubating at 95℃ for 5 min. RNase-free water at 166.5 µl was added to each cDNA reaction and was loaded onto RT2 Profiler (QIAGEN) 96-well PCR array plates (330231 PAHS-052ZA) and amplified on the Step One Plus Real-time PCR system (Applied Biosystems, Foster City, CA, USA) for 35 cycles. The resulting threshold cycle (Ct) values were uploaded onto the SABioscience website (http://www.sabiosciences.com/pcr/arrayanalysis.php) and relative gene expression was analysed using 2-ΔΔCt.
Flow cytometry
Surface expression of TLR2 and TLR4 on fresh PBMC samples was determined using flow cytometry. Briefly, 50 μl freshly isolated PBMC was incubated with primary Abs for 1 h on ice. The PBMC samples were stained with the following conjugated Abs in Hanks solution with 2% FBS at 4℃ for 1 h: anti-human CD282 (TLR2) FITC (cat. no. 11-9922; eBioscience, San Diego, CA, USA), anti-human CD284 (TLR4) PE (cat. no. 12-9917; eBioscience), mouse IgG2a K isotype control FITC (cat. no. 11-4724) and mouse IgG2a K isotype control PE (cat. no. 12-4724). Samples were washed three times by Hanks solution, then collected on BD FACS Calibur (BD Biosciences, San Jose, CA, USA) and analysed with FlowJo software version 10 (Tree Star, Ashland, OR, USA).
ELISA
Plasma from all donors was analysed using TNF-α ELISA kit (cat. no. 430207, sensitivity = 3.5 pg/ml; BioLegend, San Diego, CA, USA), IL-1β ELISA kit (cat. no. 437007, sensitivity = 0.5 pg/ml; BioLegend), IL-6 ELISA kit (cat. no. 430507, sensitivity = 1.6 pg/ml; Biolegend), C-reactive protein (CRP) ELISA kit (cat. no. BMS288INSTCE, sensitivity = 3 pg/ml; eBioscience), complement component C3a ELISA kit (cat. no. BMS2089, sensitivity = 0.14 pg/ml; eBioscience), C5a ELISA kit (cat. no. 442107, sensitivity = 2.7 pg/ml; Biolegend), MCP1 ELISA kit (cat. no. 438807, sensitivity = 1.6 pg/ml; Biolegend), sCD86 ELISA kit (cat. no. BMS291INST, sensitivity = 0.1 pg/ml; eBioscience), HMGB1 ELISA kit (cat. no. 194485, sensitivity = 0.1 pg/ml; Life Sciences, Massacchusetts, USA) and HSP70 ELISA kit (cat. no. ab133061, sensitivity = 2.5 IU/ml; Abcam, Cambridge, UK), all according to the manufacturers’ instructions. Briefly, sample and diluted standard were added to prepared wells and incubated at room temperature (RT; 25℃) on a pelate shaker during all incubation steps. Plates were washed with washing solution. Detection Ab solution was added and incubated for 1 h. Plates were washed and incubated at RT. Avidin–HRP solution was added and incubated at RT. Plates were washed and substrate solution was added to sample wells and incubated under dark conditions. Reaction was stopped with stop solution and absorbance was read at 450 nm by multisKan spectrum software.
Statistical analysis
Data in the text or tables are presented as mean ± SD and mean ± SEM in the figures. Two-tailed t-test and Mann–Whitney U-test were used for analysis of normally distributed data and non-normally distributed data, respectively. Data were analysed by Prism 7 software (GraphPad Software; GraphPad Inc., La Jolla, CA, USA). A P-value of 0.05 (two-sided) was considered statistically significant (P < 0.05, corrected for multiple comparisons by GraphPad Prism V7.03 using two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli).
Results
Global gene expression profiles
To determine the effect of brain death on the inflammatory agents of donors, we evaluated selected factors at the time point of organ harvesting for evaluation of effect of MP on innate immune responses. We analysed global gene expression of TLRs 1, 2, 3, 4, 5, 6, 7, 8 and 9, and associated signalling pathway molecules such as IL-1 receptor-associated kinase 1 (IRAK1), MYD88, NF-κB1, NF-κB1A, TNF receptor associated factor 6 and TIR domain-containing adapter molecule 1 (TICAM1/TRIF). Also, PCR array analysis was performed for MCP-1, IL-1β, IL-6, IL-8, IL-18, granulocyte-monocyte colony stimulating factor (GM-CSF) and TNF-α, and for CD markers such as CD14, CD40, CD40L, CD80 and CD86. Moreover, expression of other genes involved in innate immune responses such as ICAM-1, CRP, complement component C3, human leukocyte antigen-A (HLA-A), HLA-E, IL-1 receptor (IL-1R1), JAK2, STAT3 and caspase-1, were determined in two groups (Figure 1a).
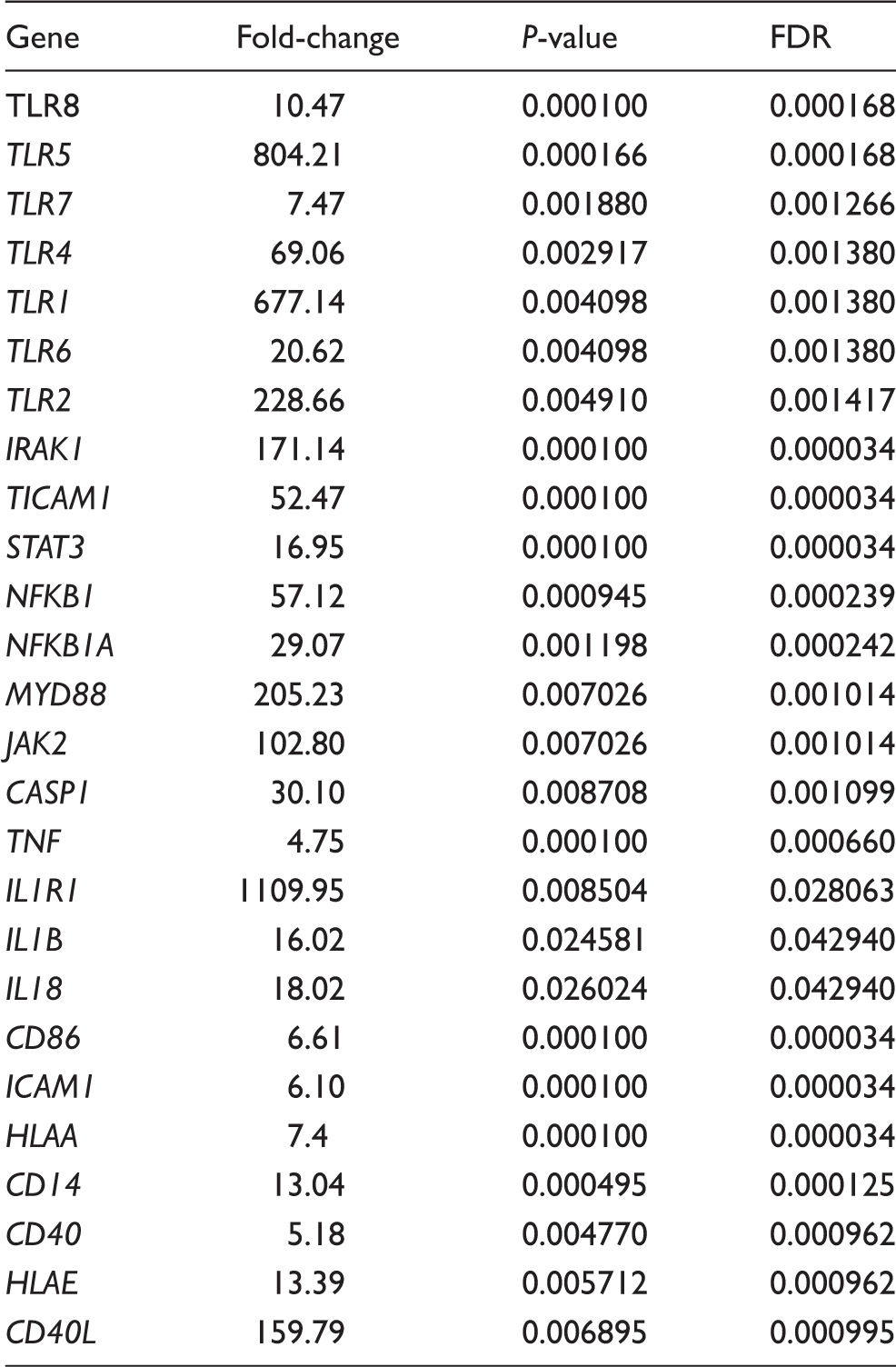
Analysis of integrated gene expression generated by RT2 PCR Array profiler data analysis (SABiosciences, Hilden, Germany). (a) Gene expression data from LDs and BDDs were determined using hierarchical cluster analysis. Columns and rows represent group samples and each gene, respectively. Each cell in the matrix represents the expression level of a gene feature in groups. High and low expression levels of a gene are represented in red and green, respectively.(b) Gene expression evaluated using the RT2 Profiler PCR Array (Biosiences, Qiagen) that represented twofold gene expression or more in the BDDs compared with LDs. We detected a significant increase in expression of TLRs 1, 2, 4, 5, 7 and 8, MYD88, IRAK1, NF-κB1, NF-κB1A, TICAM 1, STAT3, JAK2, TNF, IL-1, IL-1β, caspase 1, TNF-α, IL-1R1, HLA-A, HLA-E, ICAM-1, CD86, CD40, CD40 ligand and CD14 in BDDs compared with LDs. Data are presented as mean ± SEM, and significant values are represented as *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001 (Mann–Whitney U-test). Multiple comparison test for each group of genes corrected with FDR.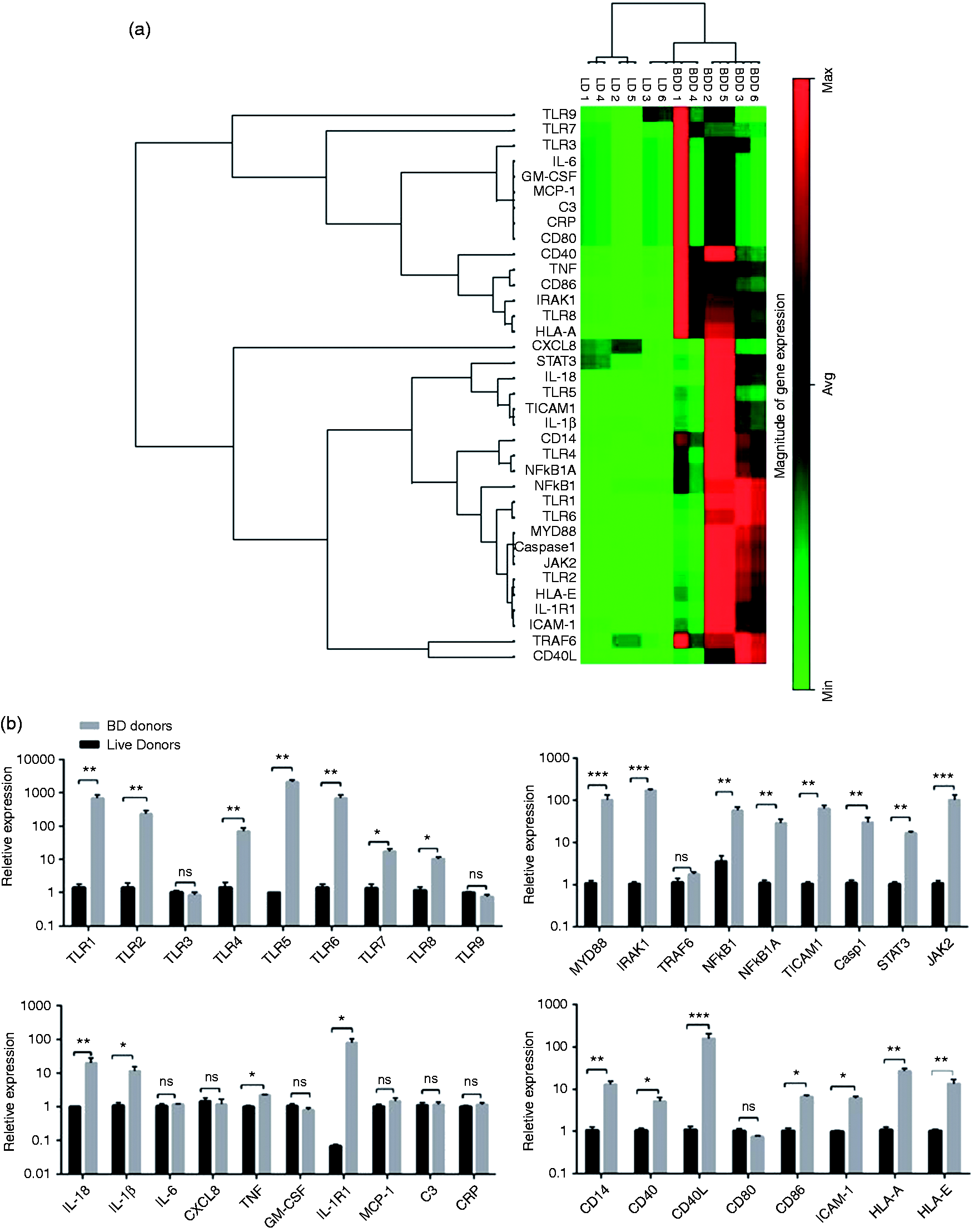
Fold change ≥2 and FDR multiple testing correction.
Increased levels of soluble inflammatory mediators in deceased donors
Inflammatory chemokines and cytokines, as well as C3a and C5a, were investigated in the serum sample drawn from BDDs and LDs. Immunoassay analysis revealed that the level of some soluble inflammatory factors, despite MP therapy, was significantly higher in BDDs (Figure 2). Significantly differences in the upper concentration for IL-1β, IL-6, TNF-α, C3a, C5a, MCP-1 and CRP was observed in BDDs (P < 0.0001). Serum levels of soluble CD86 (sCD86) were not significant between two groups. In addition, concentration levels of HSP70 and HMGB1 showed further increase in the BDDs (P < 0.0001).
Levels of inflammatory mediators in serum. Serum levels of (a) IL-1β, (b) IL-6, (c) TNF-α, (d) C3a, (e) C5a, (f) CRP, (g) MCP-1, (h) sCD86, (i) HMGB1 and (j) HSP70 were measured by immunoassay in two groups. Pro-inflammatory mediators including Il-6, TNF-α, IL-1β, MCP-1, CRP, C5a and HSP70 had a higher amount in the BDD group, especially for IL-6 and HSP70. The level of C3a had a tendency high levels in BDD compared with LDs but was not statistically significant. The concentration of sCD86 was not different between the two groups. Overall, serum levels of inflammatory factors were significantly higher in BDDs than in LDs (Mann–Whitney U-test). Data are presented as mean ± SEM and significant values are represented as *P < 0.05 and ****P < 0.0001.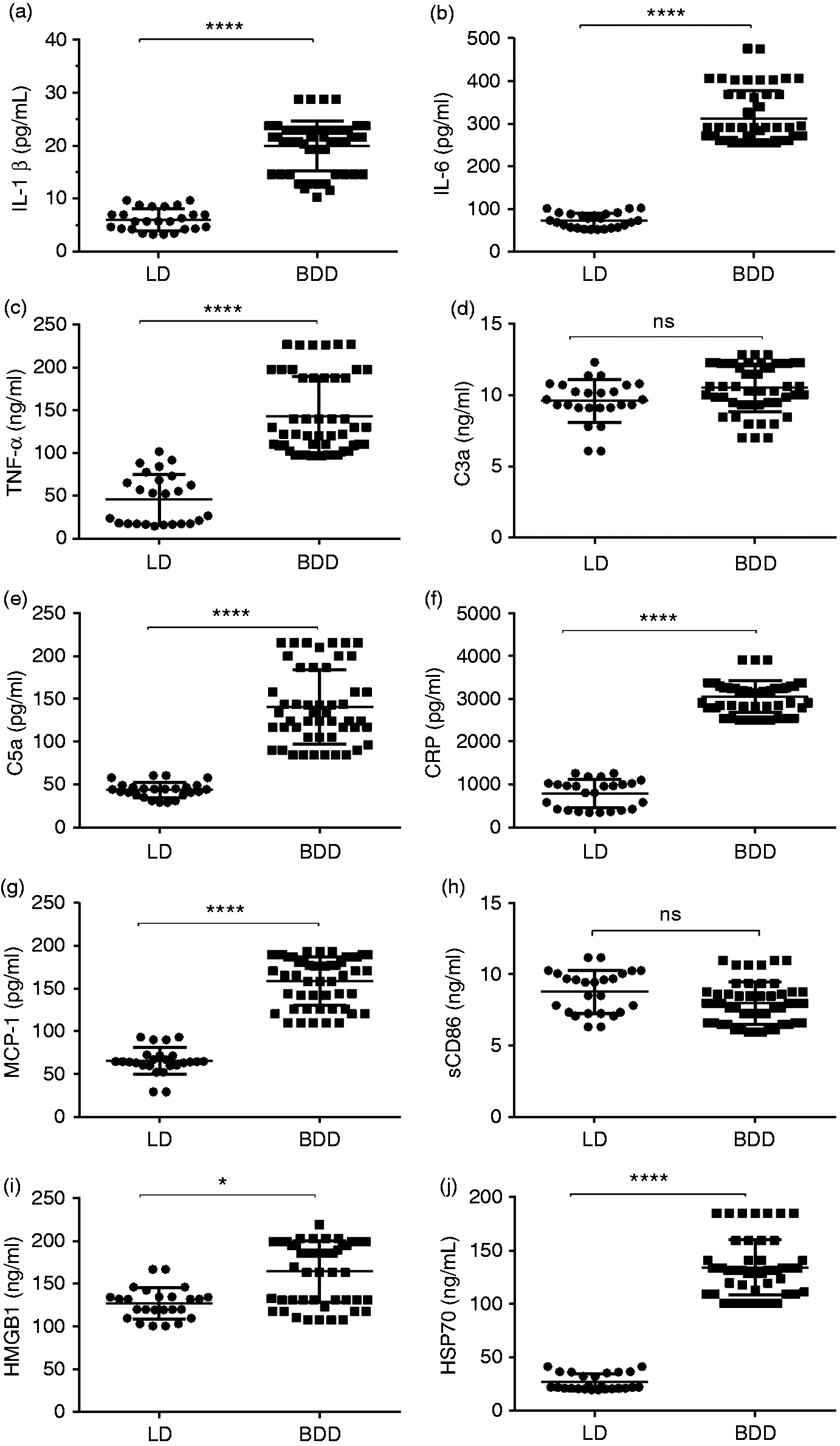
Higher expression of TLR2 and TLR4 on the peripheral blood mononuclear cells of BDDs
Peripheral blood mononuclear cell samples stained with specific anti-human TLRs Abs were analysed by FACS Calibur using CELLQuest. Monocytes and lymphocytes were gated according to their side scatter and forward scatter flow cytometry profiles and their expression on TLR2 and TLR4 (Figure 3a, b). Ten thousand events were acquired for the lymphocyte and monocyte cellular gates. Mean channel fluorescence intensity was used to identify the level of cell surface TLR expression. As shown in Figure 3(C–f), a constitutive and significant expression of TLR2 and TLR4 proteins was evidenced on the surface of lymphocytes and monocytes in BDDs (P < 0.05).
Flow cytometry panel gating strategy for surface TLR2 and TLR4. Peripheral blood mononuclear cell samples were stained with isotype control Abs or Abs to TLR2 (IgG2a) and TLR4 (IgG2a) as described. Lymphocytes and monocytes were separated by FSC/SSC gating in (a) LDs and (b) BDDs. Histograms represented the TLR2 and TLR4 expression on the (c) lymphocyte and (d) monocytes in BDDs and LDs. Data analysis by FlowJo (version 7.6.1) showed significant increases in the levels of (e) TLR2 and (f) TLR4 expression on monocytes and lymphocytes (data not shown) in BDDs compared with LDs (Mann–Whitney U-test). Data are presented as mean ± SEM, and significant values are represented as *P < 0.05 and **P < 0.01.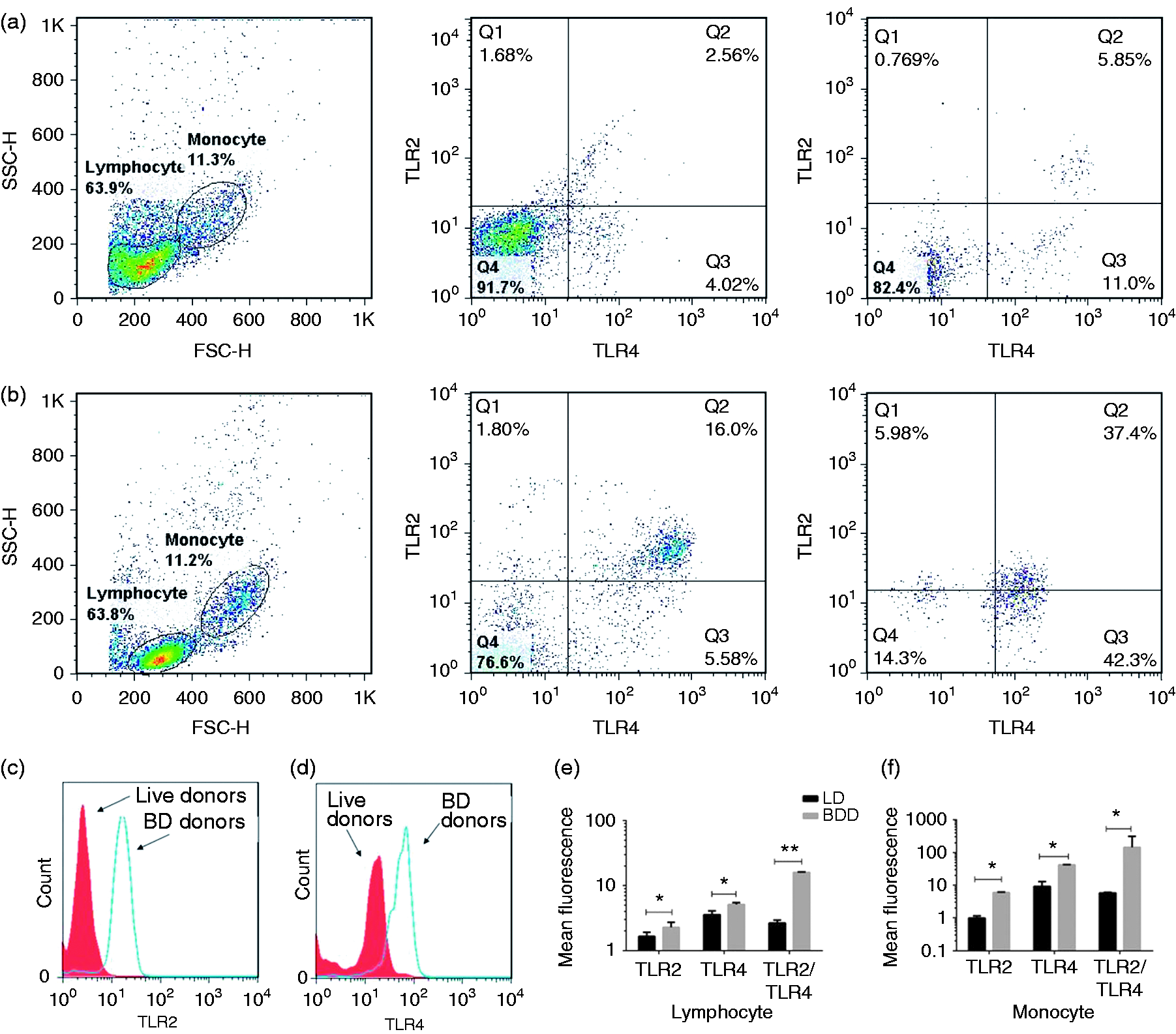
Discussion
To our knowledge, this is the first study to investigate the effect of common MP on innate immune-related genes.
Up-regulation of pro-inflammatory mediators and Ag-independent inflammatory responses following brain death adversely affect the quality of peripheral organs and increase rate and intensity of host allo-responsiveness after engraftment.8,25 Experimental and clinical studies have shown that haemodynamic and hormone-replacement therapy improve graft quality and reduce serum creatinine and graft function.26,27 In addition, some studies have demonstrated that MP therapy can reduce the amount of inflammatory mediators and improve the outcome of transplantation.28,29
Treatment of BDDs with MP 250 mg initially and 100 mg/h until organ harvesting, significantly reduced soluble IL-2, MCP-1, Il-6, TNF-α and gene expression of CD80, ICAM-1, IL-6 and TNF-α. However, this high and subsequent infusion did not reduce expression of TLR2, TLR4, CCL19 and CC21, secreted chemokines involved in immunoregulatory and inflammatory processes. 30 Krüger et al. found that TLR4 was up-regulated in kidney biopsies from BDDs compared with LDs. 14 However, Yang et al. have reported that HMBG1 mediates ischaemia–reperfusion injury by TRIF adaptor independent of TLR4 signalling. 15 In combination with these studies, we showed a significant increase in TLRs 1, 2, 4, 5, 6, 7 and 8, CD14, a co-receptor of surface TLRs, and TICAM1 in BDDs who received MP (15 mg/kg/d). However, there was no statistical significance seen in the TLR3 and TLR9 data between the two groups. Moreover, our flow cytometry analysis identified relevant significant increase in TLR2 and TLR4 on the surface of PBMC and soluble HMGB1 and HSP70 drawn from BDDs. Results from our study revealed that MP therapy was not able to down-regulate MYD88, NF-κB1, NFKB1A and IRAK1. One reason for this finding may be the inadequate effects of MP on the production of DAMPs in cadaver donors. Therefore, ligation of multiple TLRs by HMGB1 and HSP70 result in up-regulation of TLRs and downstream signalling pathways.31,32 Nijboer et al. showed no significant difference in gene expression of MCP-1, IL-1β, IL-6, TNF-α and ICAM-1 between BDDs and LDs. 9 However, our results from PCR array data analysis and immunoassay detected significant increases in mRNA expression in IL-1β, TNF-α and soluble TNF-α, IL-6 and IL-1β, but not MCP-1, in the BDD group. Furthermore, we found a significant mRNA expression of IL-18 and caspase-1, an enzyme involved in inflammasome formation, and in the maturation form of IL-1β and IL-18 in BDDs compared with LDs. In this regard, Can et al. have suggested that MP reduced severity of spinal cord injury by inhibiting activated leukocytes, but not by inhibiting the production of TNF-α. 33
Koo et al. and Schwarz et al., in separate studies,10,11 reported no significant differences in ICAM-1 and VCAM-1 amounts in BDDs compared with LDs. In contrast, we found a significant difference in ICAM-1 levels in BDDs compared with LDs.
The JAK–STAT signalling pathway represents an early adaptation from extracellular signals such as cytokines and their receptors to the nucleus, resulting in DNA transcription and expression of genes involved in inflammation, proliferation, differentiation and apoptosis. 34 Besides increased levels of inflammatory cytokines, we observed significant mRNA expression of IL-1R1, STAT3 and JAK2 in BDDs. Karin has reported that protein–protein interaction between activated glucocorticosteroid receptor and transcription factors such as AP-1, NF-κB and some STAT proteins (STAT3, STAT5 and STAT6) result in modification of the expression of inflammatory genes. 23 However, our findings showed MP therapy did not significantly reduce expression of NF-κB1, NF-κB1A, STAT3 and JAK2.
The complement system is an essential part of innate immunity that participates in defence against infection, homeostatic processes and reperfusion injury.35,36 Our study showed that the soluble levels of C3a and C5a were higher in the BDD group. However, mRNA expression of C3 was not significant in either group. Perhaps the explanation for this inconsistency is that that prednisolone treatment reduces C3 expression in kidney but not in liver. 24 It can be concluded that gene expression and protein production of complement components in the liver were not affected by MP. Accordingly, serum levels of CRP, which is mainly produced by the liver, were significantly increased in cadaver donors, whereas there was no significant difference in mRNA expression of CRP in the BDD group compared with LDs.
APCs like dendritic cells play a key role in translation of innate to adaptive immunity. 37 Surface co-stimulatory molecules (CD80, CD86), MHC class I and CD40L are involved in dendritic cell activation and maturation. 38 Therefore, we evaluated dendritic cell expression of surface activation markers (CD80, CD86, CD40) and CD40L, HLA-A, HLA-E and soluble CD86 (sCD86) in samples drawn from BDDs and LDs. The difference was found to be statistically significant in the case of CD86, CD40, CD40L, HLA-A and HLA-E in BDDs. Despite a higher amount of sCD86 in BDDs, there was no significant difference in the two groups.
Conclusion
Despite treatment with MP, analysis of gene expression profiles showed an inflammatory condition in BDDs at the point of organ harvesting. This may be due to inadequate doses or inefficient effect of MP on oxidative stress and production of damaged-associated molecules. However, the best doses and the best time to give steroid pulses before organ surgery are not clear. Therefore, several methodological challenges in donor management research need to be addressed for future well-designed randomized controlled trials.
Footnotes
Acknowledgments
We thank Samaneh Aghajanpour for her help in optimising some of the PCR array analyses performed in this study. We also thank Adeleh Adelipour and Maryam Pourhosein for their help in sample collection and management of patients.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Iran National Sciences Foundation (INSF) of vice-presidency for Sciences and Technology (grant number: 93033237).
