Abstract
Automobile traffic, industrial processes and natural phenomena cause notable air pollution, including gaseous and particulate contaminants, in urban centers. Exposure to particulate matter (PM) air pollution affects human health, and has been linked to respiratory, cardiovascular and neurological diseases. The mechanisms underlying inflammation in these diverse diseases, and to what extent health effects are different for PM obtained from different sources or locations, are still unclear. This study investigated the in vitro toxicity of ambient course (PM10) and fine (PM2.5) particulate matter collected at seven sites in the urban and periurban zones of Quito, Ecuador. Material from all sites was capable of activating TLR2 and TLR4 signaling pathways, with differences in the activation related to particle size. Additionally, airborne particulate matter from Quito is an effective activator of the NLRP3 inflammasome.
Introduction
Air pollution is a dynamic mixture consisting of gases such as ozone, carbon monoxide and sulfur dioxide, as well as organic compounds and solid pollutants in the form of metals and particulate matter (PM). 1 PM is a suspension of solid and liquid particles in the air. Its composition consists of organic and inorganic particles, such as dust, smoke, soot, sulfates and nitrates. It also has biological components, such as endotoxins, bacteria, pollen, fungal spores and viruses.2–7 Owing to the hazardous characteristics of PM, exposition to these contaminants has been linked to respiratory,8–10 cardiovascular11–14 and neurological diseases.15–18 These health-relevant particles are divided into three main categories on the basis of their diameter: coarse particles, or PM10, are those with an aerodynamic diameter between 10 and 2.5 µm; fine particles, or PM2.5, have diameters < 2.5 µm; and ultrafine particles, PM0.1, have diameters < 0.1 µm. 19 Because smaller particles can travel deeper into the lungs and are often made up of more toxic substances, the World Health Organization considers PM2.5 and smaller to be a particularly important health threat. 20 While PM10 is thought to be relatively less toxic, it can also be deposited into the upper respiratory tract, and several studies have correlated exposition to this material with health problems.20,21
Given the clear influence of air pollution on human health, it is necessary to investigate and characterize the health-affecting mechanisms of airborne contaminants.7,22 At present, the role of specific pathways participating in the propagation of inflammatory effects is still unclear. The first line of defense against the inhalation of particulate matter is the innate immune system, which is characterized by the activation of specific signaling pathways in leukocytes and inflammatory cell recruitment to sites of tissue damage. 23 Ambient air pollutants contribute to the initiation of the inflammatory response and cytotoxic effects within the lung as a result of direct pollutant contact with airway and inflammatory cells, as well as activation of indirect stimuli.12,24 Various macromolecules from exogenous sources, known as pathogen-associated molecular patterns (PAMP) or microbial-associated molecular patterns (MAMPs) or host-derived ligands associated with cell damage and distress, known as DAMPs, can interact as ligands with cell-surface or intracellular receptors (known as PRRs). These interactions lead to the propagation of the inflammatory response, which involves numerous signaling pathways and culminates in the expression of cytokines and chemokines. 25
A well-known inflammatory response pathway is that of the Toll-like receptors (TLR). 26 These receptors are a class of proteins that play a key role in the innate immune system. TLR activation can occur via two pathways: (1) the MyD88-dependent pathway; and (2) the MyD88-independent pathway. These two pathways correspond to early and late-phase NF-κB signaling, and lead to the pathway-specific induction of either pro-inflammatory cytokines and chemokines, or type I IFN. 27 A distinct inflammatory response occurs via the activation of NOD-like receptors (NLR).25,28 This signaling network is divided into several families according to receptor protein characteristics. Certain NLRs, including that of the pyrin-domain-containing protein NLRP3, are able to assemble and oligomerize into a structure known as the inflammasome.29,30 The oligomerization of NLRP3 is understood to require two signals: (1) a priming signal that results in the transcription of inflammasome components, as well as pro-caspase-1, pro-IL-1β and pro-IL-18; and (2) a second signal that promotes indirect activation of the inflammasome, such as ion or membrane perturbations, reactive oxygen species or extracellular ATP. 31 Inflammasome activation leads to the cleavage and activation of caspase-1, which acts on precursor forms to liberate the mature forms of the cytokines of the IL-1 family. 32 The NLRP3 inflammasome has been shown to be activated by particulate and crystalline matter as signal 2.33,34
In recent years, high urban development experienced around the world has resulted in the degradation of air quality caused by air pollutants emitted mainly as a product of burning fossil fuels for transportation, in the generation of electricity and in industrial processes. 20 The pollution process also varies as a result of altitude. Cities located at altitudes >1500 m have lower atmospheric pressures, which means that the partial pressure of oxygen in the air of these cities is lower than in cities that are located at sea level. As a consequence, internal combustion engines have inefficient combustion and increased emissions. A similar phenomenon affects humans; because more air must be inhaled for an equivalent amount of oxygen saturation at high altitude, a higher dose of pollutants is also inhaled 35 and people living at high altitudes may be at higher risk for developing pollution-related ailments. The city of Quito, Ecuador, suffers from air pollution derived from the emissions of gasoline and diesel vehicles, as well as from industrial processes that have grown constantly over the last several decades. 36 Quito is located at an average altitude of 2850 m above sea level and is settled in a long, thin valley surrounded by mountain ranges. This topography impedes the flow of winds that could disperse pollutants,35,36 and favors temperature inversions, which trap the air and its pollutants in the city and allow them to build up. 37 In order to monitor air quality, the city of Quito has a network of air sampling equipment. The monitoring conducted in the city measures several parameters, including PM10 and PM2.5, sedimentable particles and sulfur dioxide. 38 Given its altitude and topography, the diversity of pollutant sources, and its sophisticated monitoring system, Quito is an ideal site to conduct studies about how PM10 and PM2.5 affect human health.
Here we show that course and fine particulate matter from sampling zones around Quito, Ecuador, are associated with innate immune stimulating molecules, including TLR2 and TLR4 ligands, which activate signaling pathways in cultured cells. Additionally, we show that airborne particulate matter from Quito is an effective activator of the NLRP3 inflammasome. Taken together, these data suggest the early signaling mechanisms by which PM can activate pathology-relevant inflammation in individuals exposed to air pollution.
Materials and methods
Air sampling and filter preparation
Air sampling was conducted by the Secretariat of Environment of the Municipality of the Quito Metropolitan District. To gather PM10 particles, air was filtered through manual (semiautomatic) high-volume samplers for particulate matter (Thermo Scientific, Waltham, MA, USA) onto 10-cm microquartz filters (Whatman; GE Healthcare, Pittsburgh, PA, USA). These filters were exposed for 24 h and samples were collected every 6 d. These manual filtering stations were located in the regions denominated Belisario and Jipijapa (both consolidated urban sectors), Los Chillos (conurban sector influenced by industry) and Tababela (rural area, recently influenced by an international airport zone). Additional PM10 and PM2.5 samples were collected with automatic high-volume samplers for particulate matter (Thermo Scientific) onto polyurethane filters (Whatman; GE Healthcare). These filters were exposed for 24 h and samples were collected every 2 wk. Automatic filtering stations for PM10 were placed in the regions denominated Centro y Cotocollao (consolidated urban sectors), Guamaní y Carapungo (developing urban sectors) and Tumbaco (conurban sector); automatic stations for PM2.5 were located in Belisario, Centro, Cotocollao, Carapungo and Los Chillos. Filters were stored in clean plastic bags at room temperature for several wk prior to analysis. Immediately prior to cell-based assays, filter disk samples were prepared in a laminar flow cabinet using sterile protocol. Filters were cut into circular pieces using a 5-mm diameter cork-borer previously cleaned with 70% ethanol and sterilized in an oven at 240 ℃ for 4 h (to eliminate endotoxin). 39 In all assays, blank filters that were not exposed to particles in sampling machines but were otherwise handled similarly were used as controls.
TLR2 stimulation assay
Human embryonic kidney cells (HEK293), stably expressing human TLRs 2 and 9, and a firefly luciferase gene under the control of the NF-κB inducible ELAM promoter were maintained at 37 ℃ in 5% CO2, 40 in DMEM (Gibco, Paisley, UK) supplemented with 10% FBS and penicillin–streptomycin solution (100 U/ml). (Note: in preliminary experiments, these cells showed no response to TLR9 ligands in our hands, suggesting loss of expression of the TLR9 transgene.) Cells were plated at a density of 200,000 cells/well in flat-bottom 96-well plates with a final volume of 100 μl/well. These were stimulated at 37 ℃ in 5% CO2 for 19–24 h with a 5-mm filter disk placed in each well. After cell stimulation, filters were removed using sterile pipette tips and the luciferase activity in the reporter cells was assessed using Tropix Luc-Screen (Applied Biosystems, Foster City, CA, USA) and a Synergy HT plate reader (Biotek, Winooski, VT, USA). Pam2CSK4 (1 µg/ml) (InvivoGen, San Diego, CA, USA) was used as a positive control stimulus for activation of TLR2. 41
TLR4 stimulation assay
HEK293 cells stably expressing both TLR4 and a secreted alkaline phosphatase (SEAP) reporter under the transcriptional control of NF-κB (HEK293-hTLR4-NF-κB) (InvivoGen) were maintained at 37 ℃ in 5% CO2, in DMEM (Gibco) supplemented with 10% FBS and penicillin–streptomycin solution (100 U/ml). Cells were plated at a density of 400,000 cells/well in flat-bottom 96-well plates with a final volume of 100 μl/well. A 5-mm filter disk was placed in each well and incubated at 37 ℃ in 5% CO2 for 19–24 h. A standard curve for endotoxin (Escherichia coli 055:B5 LPS; InvivoGen) was prepared using a serial dilution of LPS in the concentration range of 600 to 2.3 ng/ml and a volume of 100 µl was used to stimulate cells. After stimulation, filters were removed, supernatants were collected and SEAP concentration, representing NF-κB activation, was determined using QUANTI-Blue medium prepared according manufacturer recommendations (InvivoGen). One hundred and ninety μl QUANTI-Blue medium was added to each well of a 96-well flat-bottom plate, together with 30 μl supernatant from stimulated cells. After 15–20 min of incubation at 37 ℃, the SEAP levels were determined by spectrophotometric absorbance at 620 nm in a Synergy HT plate reader (BioTek).
NLRP3 inflammasome activation assay
Immortalized murine bone marrow-derived macrophages from wild type C57Bl6 mice (kind gift from Dr Eicke Latz, University of Bonn) 34 were maintained in DMEM (Gibco) supplemented with 10% FBS and penicillin–streptomycin solution (5%). Cells were plated at a density of 400,000 cells/well in flat-bottom 96-well plates with a final volume of 100 μl/well. Macrophages that were either unprimed or had been primed with 600 ng/ml LPS (E. coli 055:B5 LPS) (InvivoGen) for 3 h were stimulated with either 1 µg/ml silica (Min-U-Sil-5; U.S. Silica Company, Frederick, MD, USA), 4 μM nigericin (Sigma Aldrich, St. Louis, MO, USA), or with a 5-mm filter disk placed in each well, and incubated at 37℃ in 5% CO2 for 19– 24 h. Mouse IL-1β and TNF-α in culture supernatants were measured by ELISA (DuoSet, R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s instructions.
Statistical analyses
For all assays, the graphed data points represent the mean ± SEM of three replicates, repeated in at least three separate experiments. All statistical parameters were calculated using GraphPad Prism 5.0 software (GraphPad Software, San Diego, CA, USA). Statistical significance between groups was determined by two-tailed unpaired t test (P-values < 0.05 were considered significant).
Results
Airborne course particulate matter activates TLR2
PM has previously been shown to be associated with bacterial products.3,4,42 The cell-surface receptor TLR2, dimerized with TLR1 or TLR6, recognizes lipoproteins and lipopeptides of the cell wall of Gram-positive bacteria,
43
as well as a diversity of molecules derived from Gram-negative bacteria,
44
fungi,3,45 and protozoa.46,47 To assess the ability of PM from Quito to activate the TLR2 signaling pathway, we stimulated TLR2-expressing luciferase reporter cells with PM10 particles. Samples from all monitoring stations activated the TLR2 signaling pathway (Figure 1), with the most notable activation coming from samples from the periurban stations of Los Chillos and Tababela. However, blank filters that were handled similarly to samples but not used for air filtering did not activate TLR2 signaling. These results suggest that TLR2-activating microbial products are generally associated with airborne PM10 in the air of the Quito metropolitan area. Alternatively, particle interaction with reporter cells may be able to generate DAMPs sufficient for NF-κB activation in this system.
Course PM (PM10) from Quito monitoring stations is associated with TLR2-activating ligands. Cells expressing TLR2 and a luciferase reporter gene were stimulated with filter disks containing (a) course PM collected by manual sampling stations, or (b) course PM collected by automatic sampling stations. Induction of the luciferase reporter gene was assessed, as compared to unstimulated cells. Blank indicates a filter disk not exposed to PM. Pam2CSK4 is a canonical TLR2 ligand.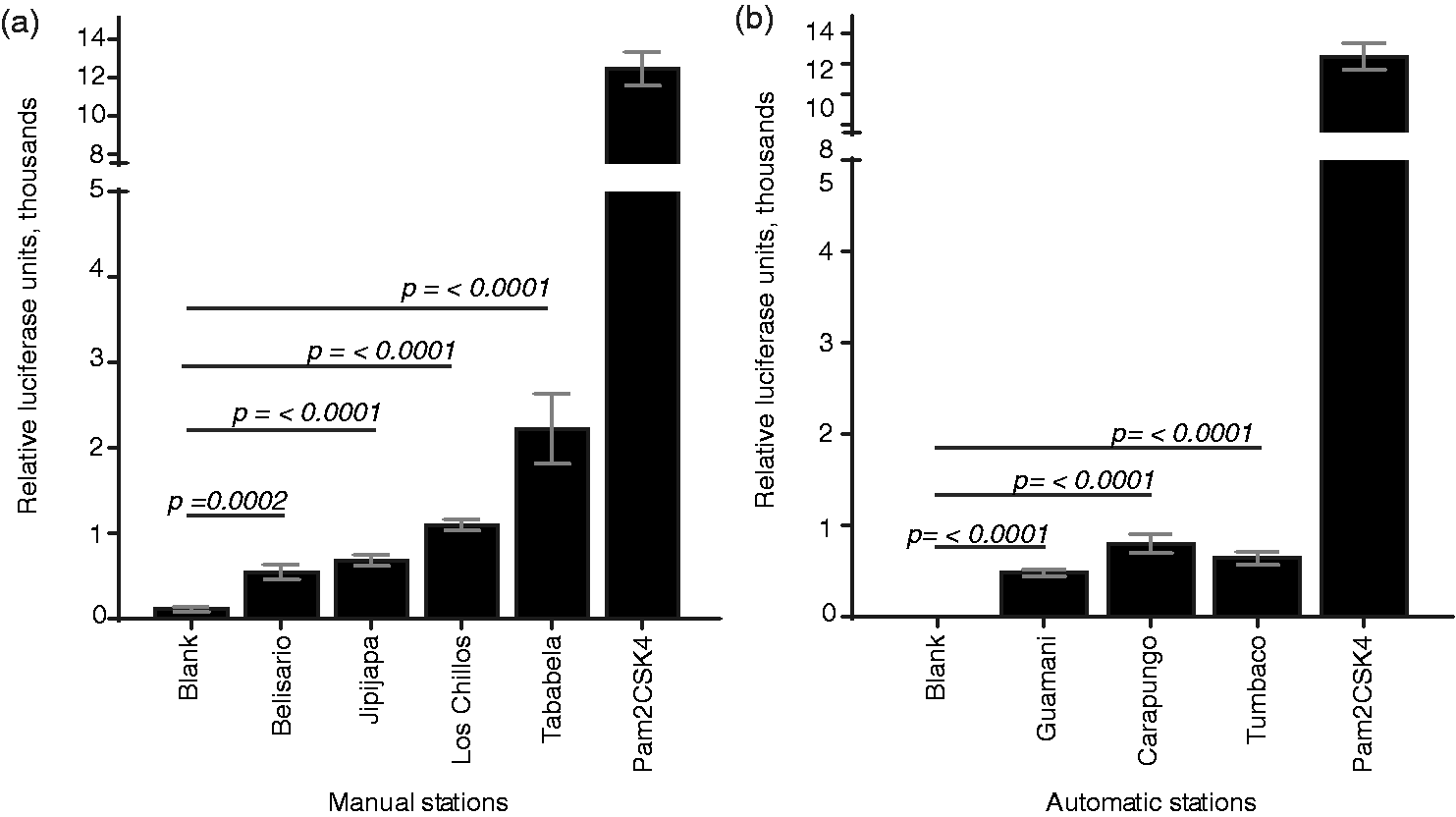
Course and fine PM activates TLR4
It has been suggested that the endotoxin associated with PM can activate an immune response associated with an increase of cytokines.
6
The cell-surface receptor TLR4 can recognize LPS, which is a major component of the outer membrane of Gram-negative bacteria,5,48 as well as several other exogenous and endogenous ligands. To determine the ability of PM from Quito to activate the TLR4 signaling pathway, we stimulated HEK293-hTLR4-NF-κB cells with PM10 collected from manual stations, and with PM10 and PM2.5 particles collected from automatic stations. We compared sample-induced activation of TLR4 with that of a standard curve of known concentrations of bacterial endotoxin, to estimate possible LPS concentrations in sample material. Samples from all monitoring stations demonstrated detectable levels of TLR4 activation, with notable differences between sample stations (Figure 2). For all the stations sampled, the estimated possible endotoxin concentration ranged from 300 ng/ml to 5 µg/ml.
Course and fine PM from Quito monitoring stations activates TLR4. TLR4-expressing reporter cells were stimulated with filter disks containing (a) course PM collected by manual sampling stations, (b) course PM collected by automatic sampling stations, or (c) fine PM collected by automatic sampling stations. Particle-associated TLR4 activating stimuli were compared with a standard curve of soluble LPS (equivalent LPS stimulus).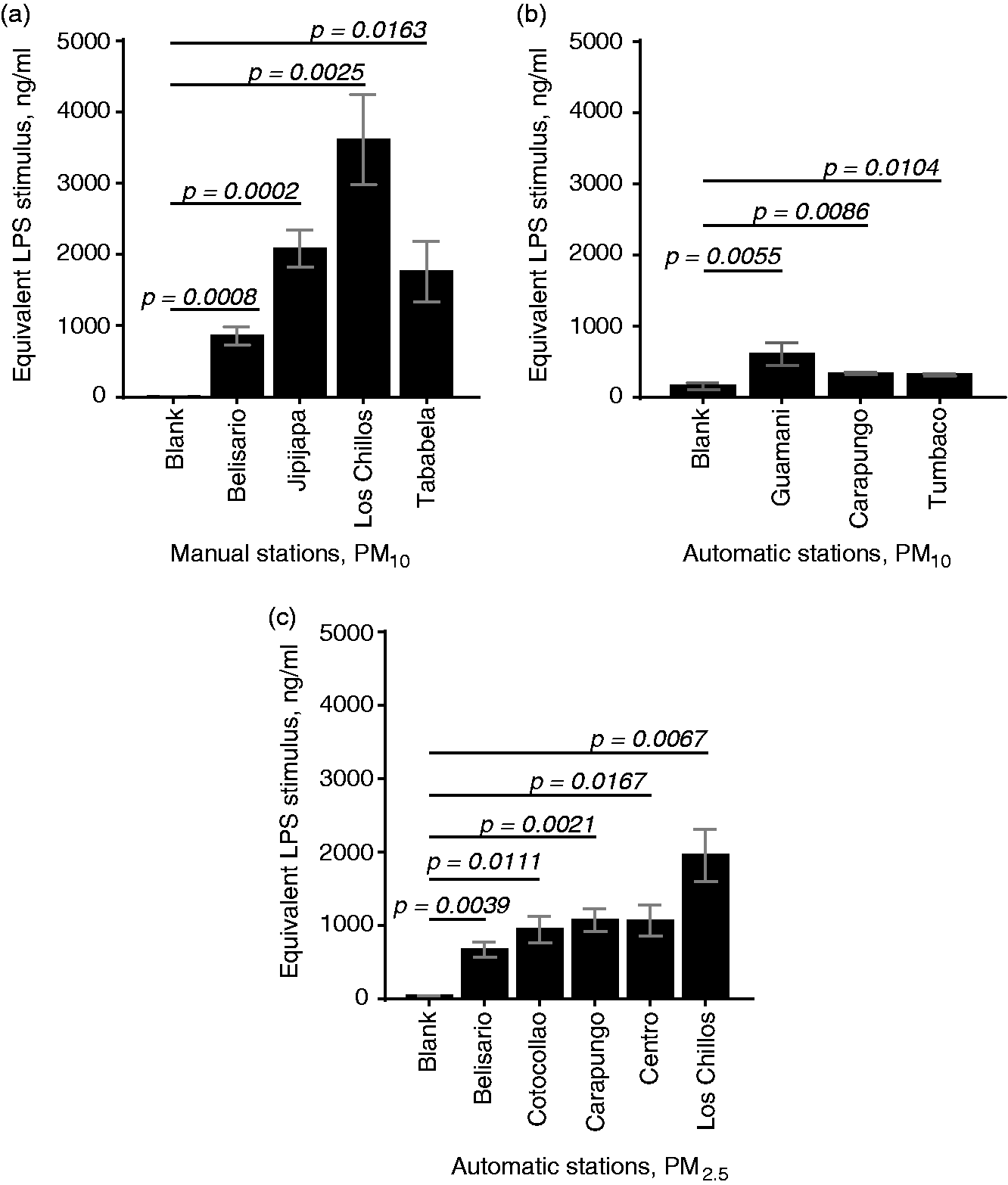
To confirm and identify the microbial origins of the detected TLR2 and TLR4 ligands in samples, we attempted to isolate microbial DNA from the particle collection filters, for genomic analysis via amplification of the 16S rRNA gene. However, even upon isolation from a 98 cm2 area of filter, insufficient DNA was obtained to permit molecular analysis (data not shown).
Airborne PM activates the NLRP3 inflammasome
NLRP3 inflammasome activation requires two stimuli; an initial, “priming” stimulus that activates transcription of inflammasome components and pro-cytokines of the IL-1 family, and a second, “activating” stimulus. Common priming stimuli for the NLRP3 inflammasome include bacterial lipopeptides and LPS, while crystalline and particulate materials are strong activating stimuli.33,34 Owing not only to its particulate nature, but also to its association with TLR2- and TLR4-activating ligands (Figures 1 and 2), we hypothesized that airborne PM could activate the NLRP3 inflammasome, possibly without prior priming. Murine macrophages are a canonical system for testing inflammasome activation and secrete IL-1β in a NLRP3-dependent manner.33,34 To assess NLRP3 activation by PM from Quito air samples, we stimulated murine macrophages with particles, with or without prior priming with LPS. While both unprimed and primed cells secreted negligible amounts of IL-1β in response to blank filters, primed cells secreted significant amounts of IL-1β upon exposure to filters containing PM10. (Figure 3a, b). Notably, unprimed cells stimulated with particles did not secrete IL-1β, suggesting that the TLR-activating ligands associated with PM were insufficient to serve as a priming stimulus for NLRP3 inflammasome activation under these conditions.
Course PM (PM10) from Quito monitoring stations induces cytokine production in murine macrophages. Immortalized murine bone marrow-derived macrophages were left unprimed or primed with LPS and subsequently exposed to filter disks containing course PM from (a, c) manual monitoring stations or (b, d) automatic monitoring stations. Crystalline silica and the pore-forming toxin nigericin were used as positive controls for inflammasome activation. Secretion of (a, b) IL-1β and (c, d) TNF-α into cell culture supernatant was quantified by ELISA.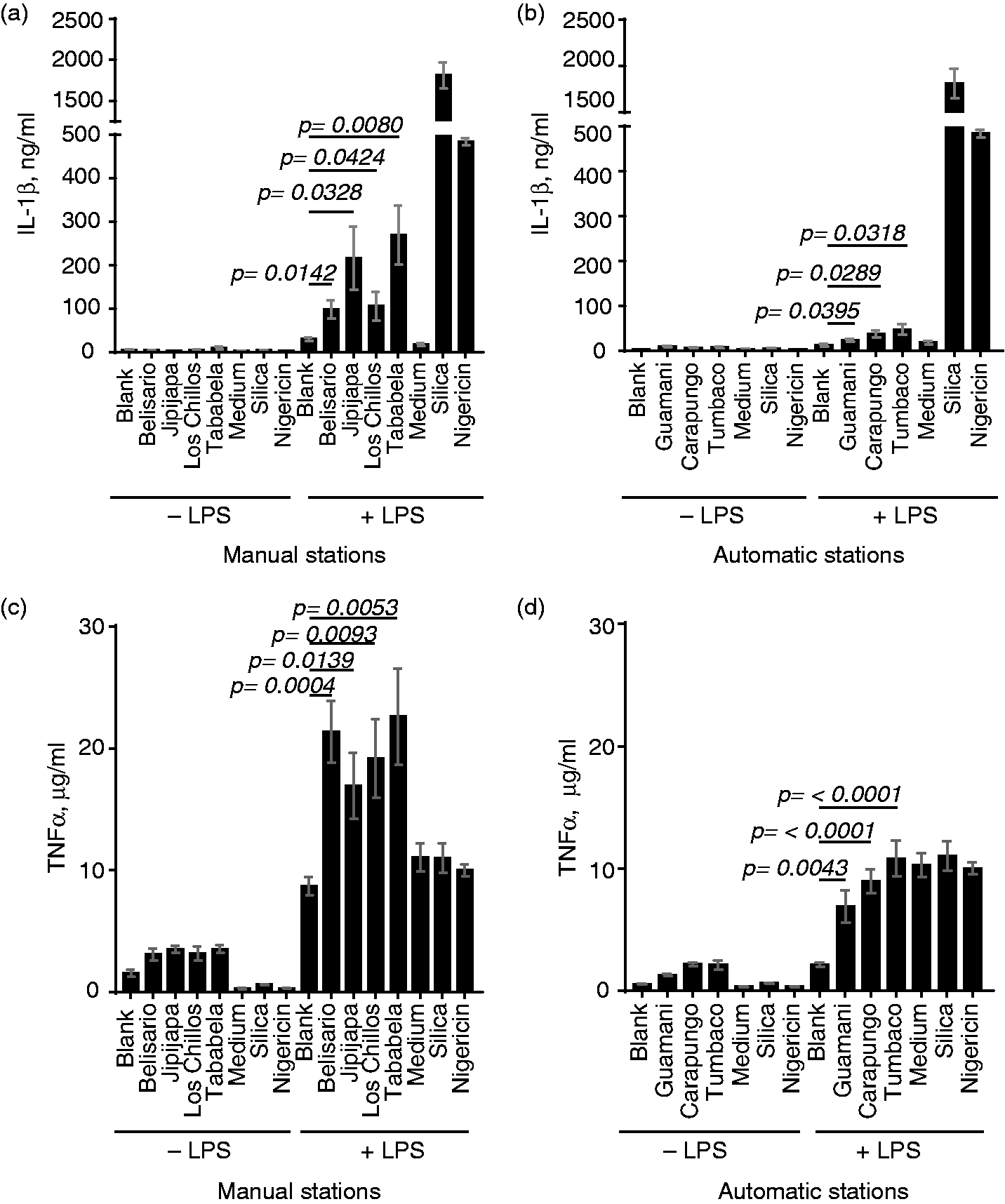
A second cytokine efficiently produced by activated macrophages, independently of inflammasome activation, is TNF-α. In the same assay, both unprimed and primed macrophages produced TNF-α upon exposure to particles (Figure 3c, d), as would be expected for activation of TLRs and other non-inflammasome signaling pathways. Cells treated first with LPS showed additional TNF production upon stimulus with PM, but not with blank filters or the control inflammasome stimuli silica and nigericin, suggesting that particles can synergize with separate microbial stimuli to promote increased cytokine production in macrophages.
Discussion
PM air pollution is regularly monitored in air quality programs and is widely understood to be hazardous for human health. 20 While a great deal of attention has been paid to the formation and role of PM in the context of environmental and climate science, the impact of PM on humans and other mammals is relatively less explored. The majority of existing studies characterize correlation between PM exposure and diverse indicators of pathology and disease, and this has provided a convincing picture of the danger of chronic exposure to all types of PM. However, the molecular mechanisms connecting exposure to pathology are still emerging.
Activation and signaling downstream of the TLRs are among the best-understood processes in leukocyte biology, and these pathways also operate in a variety of stromal cells. 49 The manner in which TLRs 2 and 4 sense microbial cell wall and membrane components is particularly well characterized,50–53 with straightforward cell-based reporter assays available. Thus, we began our exploration of the immune-stimulatory nature of particles in the Quito environment by evaluating their association with microbial components that can activate these receptor signaling pathways. While protocols have been published for isolation of particles from filter material,39,54,55 we found it impossible to separate a sufficient volume of particles from filters without subjecting the particles to treatments that could affect the quality of associated biological material. In preparation for TLR4-activation assays, we endeavored to extract any existing LPS from filter samples using water or 75% ethanol, according to published protocols.23,39,45,56 However, even after significant repetition and standardization of the extraction and stimulation protocols, extracts yielded inconsistent results that were difficult to interpret clearly (data not shown). Owing to these challenges with isolating particles and their biological components from the sampling filters, we chose to conduct assays with small disks of intact filter and the material deposited on it. This technique yielded consistent, robust data for analysis. While the disks themselves were of uniform size, our methods do not consider the relative solubility of the sample material on the disks when placed in aqueous solution such as cell culture medium; concentration of TLR4-activating ligands in the cell stimulation medium was extrapolated by comparing wells containing disks with a sample curve generated by stimulation with commercially prepared LPS in solution and may underestimate total LPS in filter-bound material. Alternatively, immobilization of LPS on a solid substrate may potentiate the activation of TLR4, as clustering of receptors by ligand has been described as a strong activating stimulus of several PRRs of importance in leukocyte activation.40,57–60 This latter case could possibly lead to overestimation of the LPS concentration in this cell-based quantification assay.
It is important to note that TLRs 2 and 4 respond to a variety of ligand molecules, and that the TLR-signaling reporter assays used here cannot distinguish microbially derived stimulatory ligands (PAMPs or MAMPs) from ligands produced indirectly by the cells themselves upon exposure to PM (DAMPs). The association of microbial molecules with airborne particulates is quite well established, despite the technical challenges of isolating whole microbes and their components,48,61 which leads us to assume that the TLR2- and TLR4-activating stimuli are likely to be of microbial origin. Additionally, some common DAMPs that could be released from the cultured HEK293 cells used here (e.g., HMGB1) are thought to interact with PAMPs/MAMPs for receptor stimulation. 62 Interestingly, nickel ions have also been shown to be direct ligands for human TLR4, 63 and mercury has been shown to influence TLR4 expression in an animal system. 64 Composition analysis of PM from Quito has not yet specifically assessed the presence of these inorganic materials, 36 but nickel and heavy metals are understood to be common components of PM derived from the burning of fossil fuels.65,66 In light of this complexity, additional work is needed to clarify definitively the precise molecular ligands involved in activation of immune signaling pathways by PM from Quito. However, the data presented here establish the TLR2 and TLR4 signaling pathways as relevant for cell-based responses to this material.
Given the presence of stimulatory ligands sufficient to activate reporter cells, we were surprised to learn that our PM samples were incapable of providing signal 1 for NLRP3 inflammasome activation. This might have to do with the relative accessibility of receptor-activating microbial components on particles, or the concentration of these components available to the cells. TLR2 and TLR4 activation assays were conducted in cell lines that overexpressed the TLRs on their surface, while inflammasome activation assays were conducted in macrophages expressing endogenous levels of receptors. The TLR ligands on particles, while easily detectable in the reporter assay systems, may not have achieved sufficient concentration in cell culture medium to give a priming stimulus to macrophages. Alternatively, DAMP or inorganic TLR ligands capable of activating overexpressed receptors on reporter cells may not interact in the same manner with TLRs expressed at endogenous levels on murine macrophages. This is particularly relevant when we consider nickel as a TLR4 ligand; nickel ions have been shown to interact with human TLR4 but not with the slightly different amino-acid conformation of murine TLR4. 63
While this study specifically considered PM from the urban and periurban areas of Quito, Ecuador, these findings are generalizable for PM from many urban areas, and the signaling pathways induced by PM are likely to be common mechanisms of pathology underlying the association between PM exposure and subsequent disease. Indeed, several studies have indicated the association of TLR2 and TLR4 ligands with PM from other sources,16,48,67,68 although published data disagree about whether TLR activation upon PM exposure is pro-inflammatory or anti-inflammatory in outcome. PM has also been shown to activate inflammasomes and IL-1 release in other systems,28,69,70 though its role as a provider of signal 1 and signal 2 is still not entirely clear. Regarding the particular high-altitude, equatorial geography of Quito, the extent to which altitude, UV exposure and temperature can alter the composition of PM itself is also somewhat vague; while environmental factors clearly influence particle chemistry, 71 environmental modeling indicates dispersion of particles over vast distances of differing altitudes, 72 complicating the interpretation of the effects of geography on the particle composition itself. Further work focusing on combinations, concentrations, and temporality of exposure to distinct PM components are likely to hold the key to understanding the complex manner in which PM air pollution interacts with human cells to instigate the inflammatory processes that lead to chronic pathology in individuals exposed to polluted air.
The overarching interest in understanding which cell signaling pathways are activated by particles is to propose mechanisms for mitigating particle-induced pathology and improving human health. While limiting the generation of harmful particulates released into the air and reducing human exposure to existing pollution are both ideal solutions for avoiding pathology, there is no sign that air pollution will be completely controlled in the near future. A means for identifying populations at significant exposure risk for PM, as well as studies of the specific composition of the particulates, may provide an intermediate option for reducing the pathology associated with chronic PM exposure. General anti-inflammatory drugs, as well as specific inhibitors of TLR and inflammasome signaling pathways, 73 may provide an option for pathology-reducing treatments in individuals with regular exposure to immunostimulatory PM in air.
Footnotes
Acknowledgements
We thank Dr. Eicke Latz of the Institute of Innate Immunity, University of Bonn, for cell lines, and Drs. Patricio Rojas-Silva and Paúl A. Cárdenas of the Center for Translational Research, Universidad de Las Américas, for critical reading of the manuscript. We are grateful to the personnel of the department of Research, Analysis, and Monitoring (IAMQ) of the Secretariat of the Environment of the Quito Metropolitan District for their assistance in placing and collecting air sampling filters.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded with intramural resources of the Universidad de Las Américas, Quito, Ecuador, and the normal operating budget of the Secretariat of the Environment of the Municipality of the Quito Metropolitan District. CMS receives salary support from the Universidad Tecnológica Equinoccial.
