Abstract
We evaluated expression of TLR2, TLR4 and proinflammatory genes [NF-κB, TNF-α, cyclooxygenase-2 (COX-2)] in liver samples of patients in different stages of liver disease. Fifteen patients with unexplained transaminases elevation (reference group), 22 with viral chronic hepatitis (hepatitis group), 14 with virus-induced severe fibrosis/cirrhosis (cirrhosis group) and 10 with hepatocarcinoma (hepatocarcinoma group) were consecutively included in the study. Quantification of TLR2, TLR4, NF-κB, TNF-α and COX-2 mRNA was done by real-time RT-PCR and TLR2 and TLR4 protein expression was evaluated by immunohistochemistry. Compared with reference, TLR2 and TLR4 mRNA was increased in hepatitis (TLR2: 2.66 ± 0.69; TLR4: 3.11 ± 0.79; P < 0.05) and cirrhosis (TLR2: 2.14 ± 0.5; TLR4: 1.74 ± 0.27; P < 0.05) and decreased in hepatocarcinoma (TLR2: 0.48 ± 0.15; TLR4: 0.54 ± 0.10; P < 0.05). This associated with increased TNF-α and COX-2 mRNA in hepatitis (TNF-α: 3.24 ± 0.79; COX-2: 2.47 ± 0.36; P < 0.05) and cirrhosis (TNF-α: 1.73 ± 0.28; COX-2: 1.8 ± 0.35, P < 0.05), whereas NF-κB mRNA was increased in hepatitis (2.42 ± 0.31; P < 0.05) and unchanged in cirrhosis (1.34 ± 0.17; P = 0.3). Hepatocarcinoma presented increased COX-2 mRNA (1.63 ± 0.15; P < 0.05) and maintained (at decreased levels) mRNA of NF-κB (0.52 ± 0.12) and TNF-α (0.52 ± 0.12; P < 0.05, all genes). Immunohistochemistry confirmed increased expression of TLR2 and TLR4 in hepatitis and cirrhosis and maintained expression in hepatocarcinoma. Upregulation of TLR2, TLR4 and their proinflammatory mediators is associated with virus-induced hepatic IFC sequence.
Introduction
Chronic liver inflammation, irrespective of the underlying cause (metabolic, immune-driven or virus-induced), leads to fibrosis and/or cirrhosis, which are precancerous states in which the development of hepatocarcinoma is more likely. Some authors call this sequence the hepatic inflammation-fibrosis-carcinoma (IFC) sequence. 1 Nevertheless, the cellular and molecular effectors mediating the interplay between the components of hepatic IFC sequence continue to be largely unknown.
Recently, several studies implicated TLRs as potential key orchestrators of the hepatic IFC sequence.2–5 TLRs are one of the most representative classes of pathogen-associated molecular patterns (PAMPs) receptors that play a critical role in innate immunity activation.6,7 The human TLR family consists of 10 members that enable the innate immunity system to recognize different groups of pathogens while initiating appropriate and distinct immunological responses according to the recognized PAMP.6,7 Besides immune cells, most liver cells (hepatocytes, Kupffer cells and stellate cells) also express TLRs and respond to their ligands.8,9 TLR2 and TLR4 have been the most studied TLRs in liver diseases as they sense bacterial components and, thus, may mediate liver injury associated with increased bacterial translocation that is present in many liver diseases.10–12 TLR2 is essential for the innate immune response to Gram-positive bacteria, being activated by bacterial lipoproteins and peptidoglycan.6,7 TLR4 acts as a receptor for LPS, a cell wall component of Gram-negative bacteria.6,7 Besides exogenous ligands, TLR2 and TLR4 may also sense endogenous ligands initiating danger signals, such as high mobility group box 1, hyaluronan and heat shock protein 60, inducing an inflammatory response in the absence of microbial challenge. 13 Stimulation of these two receptors initiates a signaling cascade that promotes activation of NF-κB and MAPK and, consequently, production of different pro-inflammatory mediators, such as TNF-α and cyclooxygenase-2 (COX-2). 14–18
Recent animal studies and in vitro hepatocyte culture models suggest that TLR2 and TLR4 may play a key role in the hepatic IFC sequence. Modulation of TLR2 and/or TLR4 function was shown to influence liver inflammation in chronic liver diseases, such as alcoholic liver disease (ALD), nonalcoholic fatty liver disease (NAFLD), chronic hepatitis C and chronic hepatitis B.2,3
There is also accumulating evidence that TLR4-induced activation and sensitization of hepatic stellate cells (HSCs) may constitute an important molecular link between hepatic inflammation and fibrogenesis.19–22 Moreover, a recent study has revealed TLRs, in particular TLR4, as major factors linking hepatic chronic inflammation and hepatocarcinoma. 23
However, to date, the suggested implication of TLR2 and TLR4 in the pathogenesis of hepatic IFC sequence is principally based on evidence obtained from animal studies or in vitro hepatocyte culture models. Studies using diseased human liver tissue to confirm or refute the in vitro and animal findings are scarce and have evaluated TLR2 and TLR4 in each stage of IFC sequence separately.
Therefore, in the present study, we evaluated the expression of TLR2 and TLR4 in liver samples from patients in each stage of virus-induced hepatic IFC sequence. The expression of NF-κB, TNF-α and COX-2 was also evaluated in order to characterize their association with TLR2 and TLR4 expression.
Materials and methods
Patients and biological samples
This study included patients from two hospitals of the North of Portugal (Braga Hospital and Portuguese Oncology Institute of Porto). The study protocol respected the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Ethics Committee of Braga Hospital and Portuguese Oncology Institute of Porto. Informed consent was obtained from each patient.
Patients were recruited consecutively during 2009. We defined four groups: reference, hepatitis, cirrhosis and hepatocarcinoma. Reference group included patients followed in the Hepatology Outpatient Clinic of Braga Hospital who underwent liver biopsy because of chronic unexplained transaminase elevation. We excluded from this group patients with alcohol abuse (>30 g/d in males; >20 g/d in females); analytical or histologic findings favoring hemocromatosis; autoimmune hepatitis; primary biliary cirrhosis; primary sclerosing cholangitis; and HIV infection or clinical, analytical, imagiological or histologic evidence of severe fibrosis/cirrhosis (METAVIR F3–4). The hepatitis group included chronic hepatitis B or C patients followed in the Hepatology Outpatient Clinic of Braga Hospital who underwent staging liver biopsy. The cirrhosis group was selected from the same group of patients but with histologic evidence of severe fibrosis/cirrhosis (METAVIR F3–4). The hepatocarcinoma group included chronic hepatitis B or C patients with diagnosis of hepatocarcinoma (according to the EASL 2000 Barcelona Guidelines 24 ) followed in the outpatient clinic of Portuguese Oncology Institute of Porto who underwent surgical resection of hepatocarcinoma. In these groups, patients must have had >18 years serological evidence of chronic hepatitis B (HBsAg+) or C (HCVAb+) and clinical stability. Histologic evidence of cirrhosis and hepatocarcinoma was required in the hepatocarcinoma group. Patients with HIV infection or analytical or histologic findings suggestive of liver disease other than viral chronic hepatitis were excluded.
Before liver biopsy or surgical intervention, blood samples were drawn from fasting patients for routine analysis (complete blood count, glucose, electrolytes, renal and liver function tests, and coagulation study) and viral load quantification. Liver tissue was obtained by percutaneous biopsy using a 16-gauge Menghini needle or by transjugular biopsy. In the hepatocarcinoma group, we obtained hepatocarcinoma tissue [for mRNA quantification (in 6 patients) and immunohistochemical evaluation (in 10 patients)] and adjacent liver tissue (for cirrhosis confirmation) from the surgical specimen. The collected tissue was divided into two fragments: one was immediately placed in RNAlater (Ambion, Madrid, Spain) and stored at −80°C for mRNA isolation and quantification; the other was fixed in 10% buffered formalin and embedded in paraffin for histologic and immunhistochemical analyses.
mRNA isolation and quantification of TLR2, TLR4, NF-κB, TNF-α and COX-2
Specific PCR primers pairs for the studied genes.
F = Forward primer, R = Reverse primer.
Immunohistochemical evaluation of TLR2 and TLR4
Tissue specimens were fixed in 10% neutral buffered formalin for 24 h and paraffin embedded. De-paraffinized tissue slides were submitted to antigen retrieval using a high temperature antigen unmasking technique in a water bath, 95°C in citrate buffer pH 6.0, for 20 min. Endogenous peroxidase activity was blocked by incubating the slides with freshly prepared 0.5% H2O2 in distilled water for 20 min. After washing the slides in distilled water and PBS/0.05% Tween 20 solution, immunostaining was performed using an immunoperoxidase method according to the manufacturer’s instructions. The slides were incubated with normal horse serum (Vector Laboratories, Burlingame, CA, USA) 1/50 in PBS-BSA 1% at room temperature (21–23°C) for 20 min in humid chamber. Sections were then incubated with primary Ab at 4°C for 16–18 h. The following primary Abs were used: rabbit polyclonal Ab anti-TLR2 (H-175, 1 : 50 dilution, Santa Cruz Biotechnology, CA, USA) and rabbit polyclonal anti-TLR4 (H-80, 1 : 100 dilution, Santa Cruz Biotechnology). The slides were then rinsed in PBS/0.05% Tween 20 solution and bound Ab was detected by applying biotinylated secondary Ab (Vectastain Universal Elite ABC Kit, Vector Labs, Peterborough, UK) for 30 min. After washing the slides with PBS/0.05% Tween 20 solution the slides were incubated with ABC reagent (Vectastain Universal Elite ABC Kit) for 30 min. The slides were washed in PBS and incubated for 7 min in 3,3-diaminobenzidine (DAB; Sigma-Aldrich, St Louis, MO, USA) 0.05 g/PBS, 0.03% H2O2. Following counterstaining with hematoxylin for 20 s, the slides were washed for 4 min in water, dehydrated and mounted with Entellan (Merck KGaA, Darmstadt, Germany). Normal gastric mucosa and lymph node tissue were used as negative and positive controls, respectively. An Ab diluent (non-immune IgG, TA-125-UD; Thermo Scientific, Erembodegem, Belgium) was used in some samples as additional negative control, confirming the specificity of our protocol. We evaluated immunostaining of hepatocytes for TLR2 and TLR4 in all the samples. In order to quantify TLR expression in tissue samples, three parameters were considered: (i) sample positivity—a sample was considered positive if hepatocytes were clearly marked by the Ab; (ii) grade of expression—a score of 0–3 was considered according to the number of epithelial cells marked (0 = no cells; 1 = less than 10% of epithelial cells; 2 = 10–75% cells; 3 = more than 75% cells); and (iii) intensity of expression—a score of 0–3 was considered according to a subjective evaluation of the intensity of marked cells (0 = no immunostaining; 1 = weak positive staining; 2 = moderate positive staining; 3 = strong positive staining). Immunohistochemical evaluation was performed independently by two experienced pathologists.
Statistical analysis
Data analysis was performed using the computer software SPSS for Windows (version 17.0; Chicago, IL, USA). Data are presented as mean ± standard error of mean (SEM) or as median and range, according to the type of distribution. Student’s t-test was used for comparison between groups. When necessary, the test was preceded by a natural logarithm transform to obtain a normal distribution. Correlation between TLR2 and TLR4 mRNA expression and viral load, necroinflammatory activity or transaminases levels was evaluated by univariate analysis. Statistical significance was set at P < 0.05.
Results
Baseline characteristics of patients
Baseline characteristics of patients.
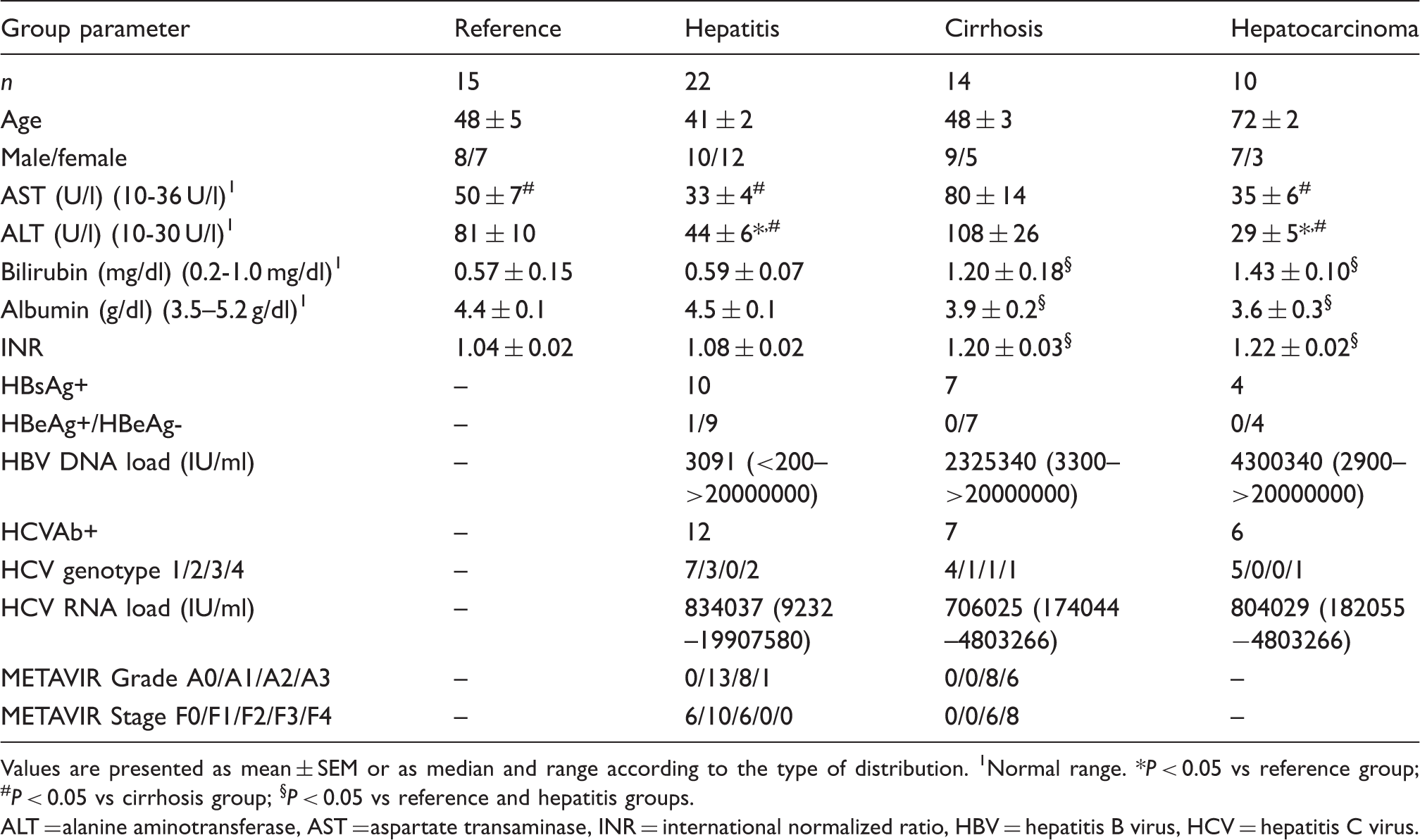
Values are presented as mean ± SEM or as median and range according to the type of distribution. 1Normal range. *P < 0.05 vs reference group; #P < 0.05 vs cirrhosis group; §P < 0.05 vs reference and hepatitis groups.
ALT = alanine aminotransferase, AST = aspartate transaminase, INR = international normalized ratio, HBV = hepatitis B virus, HCV = hepatitis C virus.
mRNA expression of TLR2, TLR4, NF-κB, TNF-α and COX-2
Quantifications of TLR2, TLR4, NF-κB, TNF-α and COX-2 mRNA are shown in Figure 1. In the hepatitis group,
expression of TLR2 (2.66 ± 0.69, P = 0.04) and TLR4 (3.11 ± 0.79,
P = 0.03) were greatly increased. This was associated with increased
expression of NF-κB (2.42 ± 0.31, P = 0.0003), TNF-α (3.24 ± 0.79,
P = 0.02) and COX-2 (2.47 ± 0.36, P = 0.003). Compared
to the reference group, this increased inflammatory profile (with exception of NF-κB)
persisted in the cirrhosis group (TLR2: 2.14 ± 0.5, P = 0.04; TLR4:
1.74 ± 0.27, P = 0.008; NF-κB: 1.34 ± 0.17, P = 0.3;
TNF-α: 1.73 ± 0.28, P = 0.009; COX-2: 1.8 ± 0.35,
P = 0.04), despite a global, but not significant (except for NF-κB),
decrease in expression of all the genes when compared with the hepatitis group. In the
hepatocarcinoma group, all samples were positive for all studied genes. Compared with the
reference group, the hepatocarcinoma group presented a higher mRNA expression of COX-2
(1.63 ± 0.15; P = 0.02), similar to the hepatitis and cirrhosis groups,
and lower mRNA expression of TLR2 (0.48 ± 0.15), TLR4 (0.54 ± 0.10), NF-κB (0.52 ± 0.12)
and TNF-α (0.52 ± 0.12; P = 0.01, all genes). There were no differences
between chronic hepatitis B or C patients (P > 0.05, all genes). We
did not find any difference or tendency when comparing the genetic profile between F0, F1
or F2 patients (P > 0.05, all genes). No correlation was seen between
mRNA expression of any gene and viral load, necroinflammatory activity or transaminases
levels. mRNA quantification of TLR2, TLR4, NF-κB, TNF-α and COX-2, in
the reference (mainly composed of patients with NAFLD), hepatitis (composed of
patients with chronic hepatitis B or C), cirrhosis (composed of patients with
post-chronic hepatitis B or C cirrhosis) and hepatocarcinoma (composed of patients
with post chronic hepatitis B or C cirrhosis-related hepatocarcinoma) groups. Levels
of mRNA are expressed as arbitrary unit (AU) set as the average value of control
group after normalization for GAPDH. Results are presented as mean ± standard error
of mean (SEM). *P < 0.05 vs reference group;
#P < 0.05 vs cirrhosis group;
§P < 0.05 vs hepatitis group.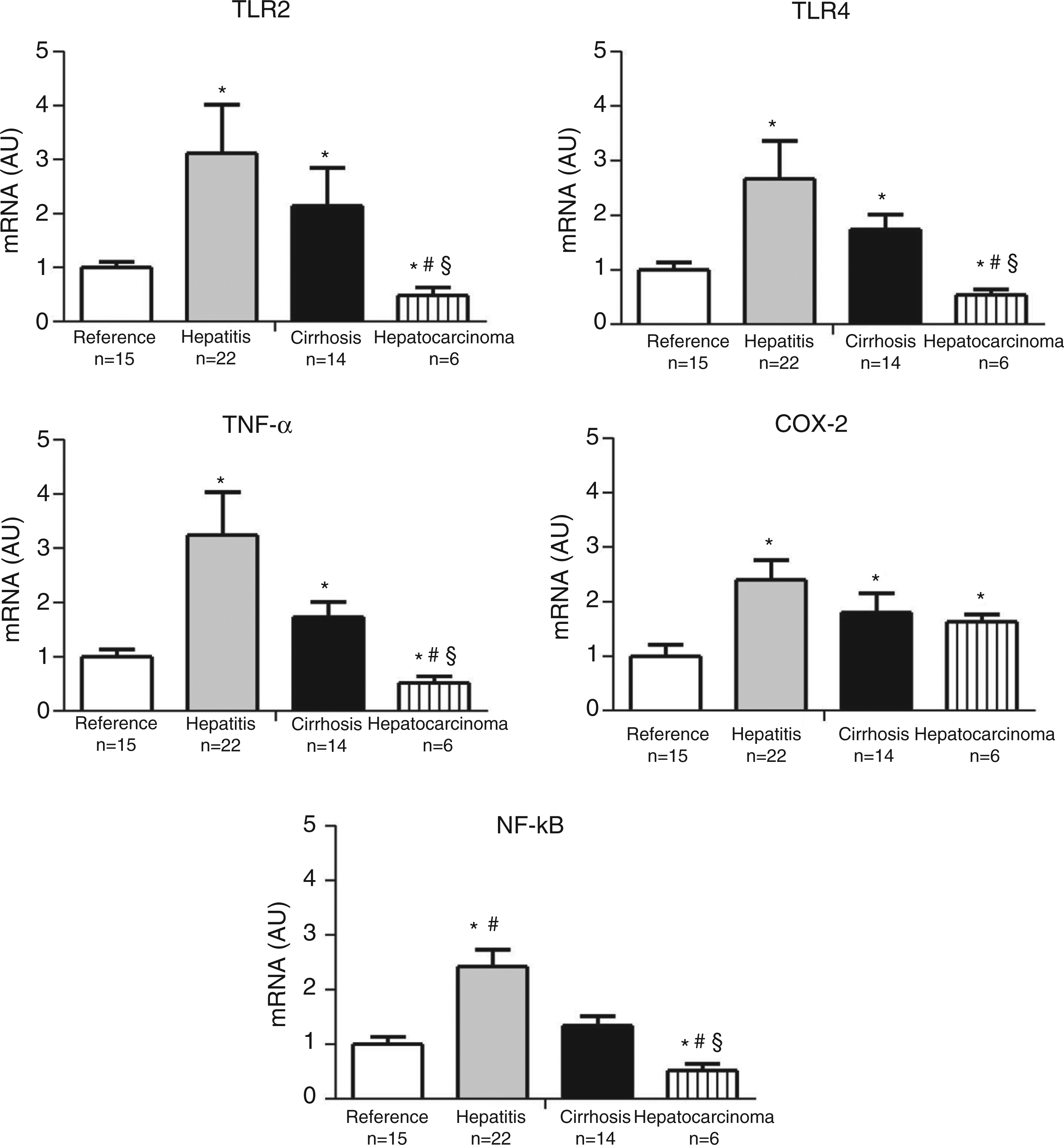
Immunohistochemical evaluation of TLR2 and TLR4
Immunohistochemichal evaluation of TLR2 and TLR4: grade and intensity of expression.
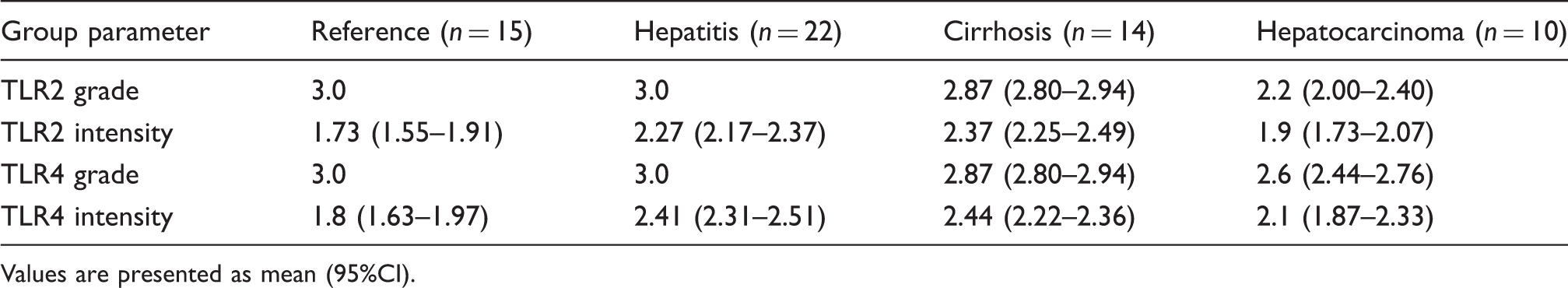
Values are presented as mean (95%CI).
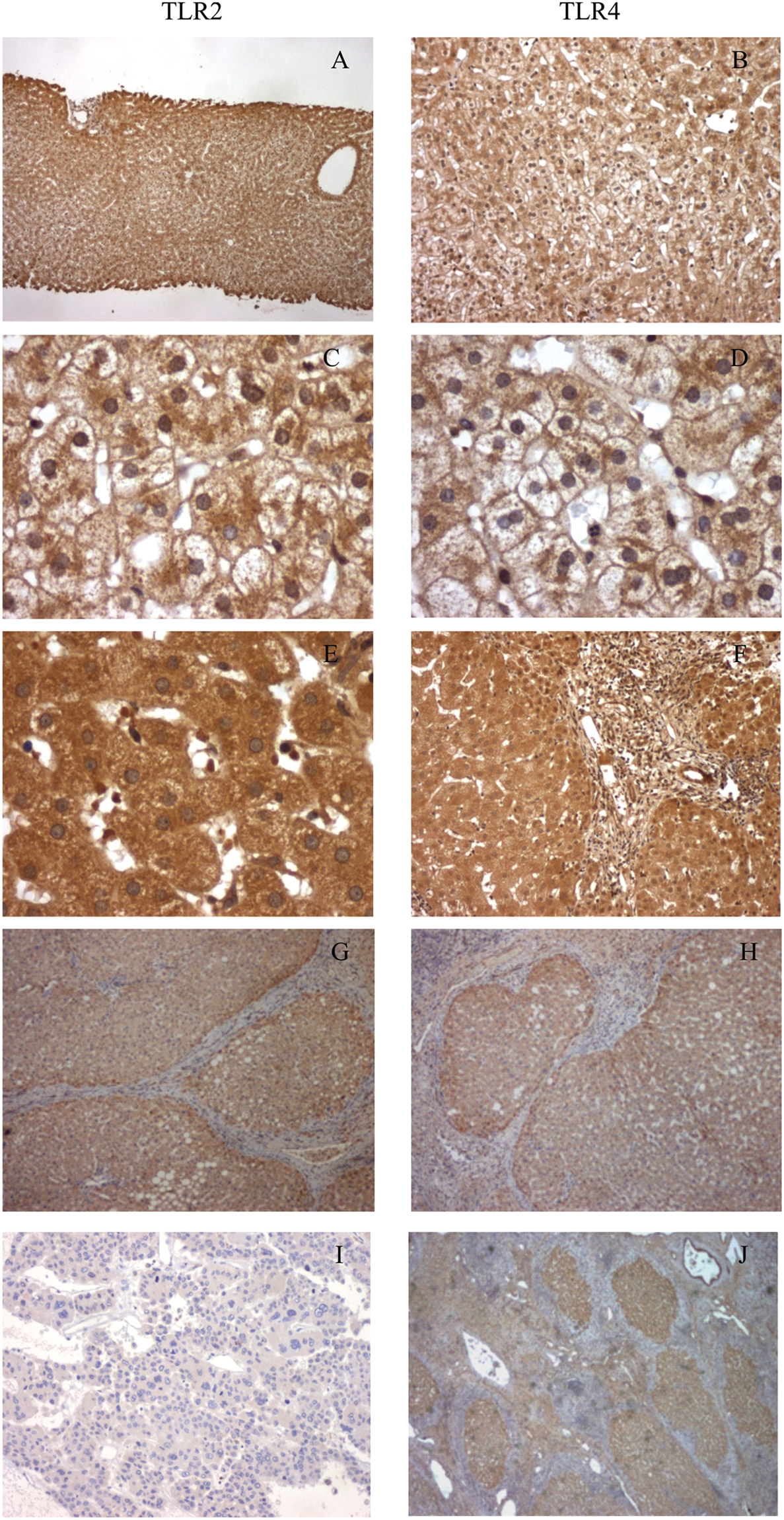
Immunohistochemical evaluation of TLR2 (left) and TLR4 (right). (A, B) Low power field magnification for TLR2 and TLR4 in the reference group (mainly composed of patients with NAFLD): there is a higher intensity staining in acinar zones 1 and 3 than in acinar zone 2. (C, D) High power field magnification for acinar zone 2 in the reference group showing sparse cytoplasmatic staining. (E, F) High and low power field magnification, respectively, in chronic hepatitis B or C (acinar zones 1 and 2) showing diffuse cytoplasmatic staining (in contrast with A–D images). (G, H) Low power field magnification in post-chronic hepatitis B or C cirrhosis: all hepatocytes are stained with a diffuse cytoplasmic staining. (I, J) Low power field magnification in post-chronic hepatitis B or C cirrhosis-related hepatocarcinoma: low intensity staining in poorly-differentiated areas of hepatocarcinoma with multiple bizarre cells (I) in contrast to nodular well-differentiated areas with high intensity staining in hepatocarcinoma cells (J). In most samples of hepatocarcinoma tissue there were well-differentiated areas with high staining (J) alternating with poorly-differentiated areas with low staining (I).
Discussion
In the present study, we evaluated the expression of TLR2 and TLR4 in liver samples from patients in each stage of virus-induced hepatic IFC sequence. We found increased TLR2 and TLR4 mRNA and protein expression in virus-induced chronic hepatitis and cirrhosis, and a maintained TLR2 and TLR4 protein expression in virus-induced hepatocarcinoma.
Although there are several animal and in vitro studies implicating TLRs in the pathogenesis of hepatic IFC sequence, studies using diseased human liver tissue are scarce. Hepatic expression of TLR2 was shown to be maintained in early- and late-stage biliary atresia 25 and HCV cirrhosis,26,27 increased in primary biliary cirrhosis (PBC) and non-alcoholic steatohepatitis (NASH), 28 and decreased 27 or maintained 26 in alcoholic cirrhosis. Hepatic expression of TLR4 was shown to be maintained in early- and late-stage biliary atresia 25 , alcoholic and HCV cirrhosis,26,27 and increased in PBC 28 and NASH.28,29 Besides contradictory results, none of these studies have evaluated simultaneously TLR expression in different stages of liver disease.
To our knowledge the present study is the first to evaluate hepatic TLR2 and TLR4 expression at different stages of the virus-induced hepatic IFC sequence. We found increased TLR2 and TLR4 mRNA and protein expression in virus-induced chronic hepatitis and cirrhosis and a maintained TLR2 and TLR4 protein expression in virus-induced hepatocarcinoma. Thus, upregulation of TLR2 and TLR4 is an early, and persistent, event in the virus-induced hepatic IFC sequence.
Regarding TLR2 and TLR4 protein expression, the differences between the groups were more evident in terms of intensity of expression (which reflects the level of expression per cell) than in terms of grade of expression (which reflects the number of cells expressing the protein). The intensity of TLR2 and TLR4 proteins expression was in line with TLR2 and TLR4 mRNA expression, while the grade of TLR expression changed little between the groups, not accompanying the changes in mRNA expression. This finding suggests that the virus-induced hepatic IFC sequence is associated with changes in the level of TLR2 and TLR4 protein expression per cell and not with changes in the number of cells expressing these proteins.
Interestingly, we found reduced TLR2 and TLR4 mRNA and protein expression in the hepatocarcinoma group when compared with the hepatitis and cirrhosis groups. Although we have no definite explanation for the reduced expression of TLR2 and TLR4 in hepatocarcinoma cells, we believe it is likely a consequence of loss of differentiation of hepatocarcinoma cells. This is suggested by the finding that in most samples of hepatocarcinoma tissue there were well-differentiated areas with high staining alternating with poorly differentiated areas with low staining. This finding likely means that at late stages of the hepatic IFC sequence, the role of these receptors in this sequence becomes smaller.
However, our study has some limitations. Firstly, most patients included in the reference group have evidence of NAFLD and it was demonstrated that NAFLD is associated with increased hepatic TLR2 and TLR4 mRNA expression.28,29 This suggests that the increase in hepatic expression of TLR2 and TLR4 in chronic hepatitis, cirrhosis and hepatocarcinoma may, in fact, be underestimated. Moreover, the hepatitis, cirrhosis and hepatocarcinoma groups included both patients with HBV infection or HCV infection. Nonetheless, statistical analysis revealed no difference between HBV and HCV patients. Moreover, as we included only patients with virus-induced chronic hepatitis in this study, our data cannot be generalized to other chronic hepatic diseases that follow IFC sequence. Another limitation of our study is that the method we used for quantification of protein expression was semi-quantitative. Although this could contribute to the lack of difference in terms of grade of protein expression, we were able to obtain significant differences in terms of intensity of protein expression. The validity of the protein quantification by this method is supported by the overall agreement between protein (grade of expression) and mRNA data. Furthermore, the method was validated in a previous study with consistent results. 30
In the present study we have not explored the mechanisms underlying increased hepatic expression of TLR2 and TLR4. Nonetheless, previous studies have shown that HBV and HCV may upregulate TLR2 and TLR4 through direct and indirect mechanisms. In vitro studies have shown that HCV nonstructural protein NS5A upregulates TLR4 expression and that HBeAg upregulates TLR2 expression.23,31 In chronic hepatitis B and C, besides up-regulated expression of TLR2 and TLR4 by the virus, other factors, such as augmented exposure to their ligands, can also contribute to increased activation of these TLRs, especially in later stages of hepatic fibrosis and cirrhosis. In fact, several studies have demonstrated that bacterial translocation is increased in patients with cirrhosis, resulting in augmented exposure of hepatic TLRs to their ligands.10,11
Herein, we did not search for liver cell-specific expression of TLR2 and TLR4, but instead we have focused on total hepatic mRNA expression and protein expression of hepatocytes. This may be an important issue as it has been demonstrated that HCV and HBV may affect TLRs expression in a cell-specific manner.2–5 Nevertheless, the immunohistochemistry that we performed in this study suggests that, at least in part, this increase in TLRs expression occurs significantly in hepatocytes.
Previous studies on human samples have shown that hepatic expression of TLR2 and TLR4 in HCV cirrhosis was unchanged compared with the reference group, which is in disagreement with our results.26,27 The reasons for this disagreement are unclear, but our data are consistent with a previous in vitro study showing that hepatocyte-specific transgenic expression of the HCV nonstructural protein NS5A upregulates TLR4 expression. 23 Regarding chronic hepatitis B, Visvanathan et al. 31 have shown that expression of TLR2 on hepatocytes and Kupffer cells was significantly reduced in patients with HBeAg-positive chronic hepatitis B in comparison with HBeAg-negative chronic hepatitis B and controls, whereas it was significantly increased in HBeAg-negative chronic hepatitis B compared with controls. The level of TLR4 expression did not differ significantly among the groups. Downregulation of TLR2 was also demonstrated in HepG-2 cells transduced with wild-type HBV (HBeAg-positive) but not in cells transduced with pre-core mutant HBV (HBeAg-negative). Regarding TLR2, our data are consistent with the study by Visvanathan et al. 31 as most of our chronic hepatitis B patients were HBeAg-negative and have increased TLR2. We could not compare HBeAg-positive and HBeAg-negative patients owing to the limited number of HBeAg-positive patients in our study.
In order to clarify TLR2- and TLR4-induced proinflammatory genes expression we also studied NF-κB, TNF-α and COX-2 mRNA expression. We found that hepatic TNF-α and COX-2 mRNA expressions are increased in virus-induced chronic hepatitis and cirrhosis, whereas hepatic NF-κB mRNA is increased in virus-induced chronic hepatitis, but maintained in virus-induced cirrhosis. This is an interesting finding as these proinflammatory genes have been implicated in hepatic inflammation, fibrogenesis and carcinogenesis interplay.1,32,33 Interestingly, hepatocarcinoma samples presented increased COX-2 expression, despite lower expression of other studied genes, suggesting, in line with previous studies, that this enzyme may have an important role in hepatocarcinogenesis. 33 Although we have not investigated the functionality of TLR2 and TLR4, increased expression of TLR2 and TLR4 proteins and of NF-κB, TNF-α and COX-2 (key mediators of TLR2 and TLR4 signaling pathway) mRNA expression suggest augmented signaling of TLR2 and TLR4. When comparing hepatitis with cirrhosis we found a tendency towards lower expression of inflammatory genes mRNA; however, we cannot dismiss the possibility that this tendency is not related to a higher inflammatory cell infiltrate observed in the hepatitis group. In fact, we did not find any clear tendency in the hepatocyte immunohistochemistry results, suggesting that the difference, if any, between the two groups is not significant.
In summary, in patients with HCV or HBV chronic infection, hepatic expression of TLR2 and TLR4 is increased in chronic hepatitis and cirrhosis and is maintained in hepatocarcinoma. This is associated with increased TLR2- and TLR4-induced proinflammatory gene expression. Overall, this study suggests that TLR2 and TLR4 may be key players in the human hepatic IFC sequence associated with viral chronic hepatitis.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgments
We are sincerely grateful to Antónia Teles for her technical support in this study.
Conflict of interest statement
The authors declare that there is no conflict of interest.
