Abstract
The leukocyte Ig-like receptor B4 (LILRB4) is an inhibitory cell surface receptor, primarily expressed on mono-myeloid cells. It contains 2 C-type Ig-like extracellular domains and a long cytoplasmic domain that contains three intracellular immunoreceptor tyrosine-based inhibitory motifs (ITIMs). Data suggest that LILRB4 suppresses Fc receptor-dependent monocyte functions via its ITIMs, but relative contributions of the three ITIMs are not characterised. To address this, tyrosine (Tyr) residues at positions 337, 389 and 419 were single, double or triple mutated to phenylalanine and stably transfected into a human monocytic cell line, THP-1. Intact Tyr389 was sufficient to maximally inhibit FcγRI-mediated TNF-α production in THP-1 cells, but, paradoxically, Tyr337 significantly enhanced TNF-α production. In contrast, bactericidal activity was significantly enhanced in mutants containing Tyr419, while Tyr337 markedly inhibited bacteria killing. Taken together, these results indicate that LILRB4 might have dual inhibitory and activating functions, depending on the position of the functional tyrosine residues in its ITIMs and/or the nature of the stimuli.
Keywords
Introduction
Pairing of immunoreceptor tyrosine-based inhibitory motifs (ITIM) and immunoreceptor tyrosine-based activating motifs (ITAM)-containing receptors is postulated as a central paradigm that regulates the threshold and amplitude of leukocyte activation.1–3 Upon co-ligation with an ITAM-containing activating receptor, tyrosine residues in one or more ITIMs of an inhibitory counterpart are phosphorylated by Src family protein tyrosine kinases recruited to the ITAMs.4–7 Phosphorylated ITIMs act as substrates for Src homology 2 domain (SH2)-containing phosphatases (SHPs, SHIP) that terminate signals by dephosphorylating various tyrosine kinases involved in activation.4–8 However, this paradigm is challenged by recent findings demonstrating that under certain circumstances, ITIMs can mediate activation (termed ‘ITIMA’) and ITAMs can mediate inhibition (termed ‘ITAMI’).3,9–11 Moreover, ITIM-like motifs (YVKM), 12 immunoreceptor tyrosine-based switch motifs (ITSMs, TxYxxI/V),12,13 SH3-binding domains14,15 and proline-rich regions in the intracellular domains of ITIM-containing proteins16,17 contribute to the overall functions of such receptors. These indicate that receptors bearing these consensus motifs may have the capacity to transduce complex regulatory signals.
Leukocyte Ig-like receptor B4 (LILRB4) is an inhibitory cell-surface receptor primarily expressed on monocytes, macrophages and dendritic cells.6–8,18 LILRB4 ligation potently inhibits Fc receptor-mediated cytokine production 6 and clathrin-dependent endocytosis 7 by monocytes, and induces tolerogenic dendritic cells in vitro. 19 Clinically, increased expression of LILRB4 in the kidney is associated with better clinical outcomes in renal transplant recipients. 18 In contrast, increased LILRB4 expression in some cancers, particularly pancreatic cancer, is associated with impaired immune effector responses. 19 Uniquely, LILRB4 contains three ITIMs with sequences 335VTYAKY340, 387VTYAQL392 and 417SVYATL422, two SH3-binding motifs (PxxP) and proline-rich regions. 20 However, contributions of these intracellular consensus sequences to functions of LILRB4 are unclear. To address this, chimeric DNA constructs containing the extracellular and transmembrane domains of IL-2 receptor α-chain and the intracellular domains of LILRB4 ITIMs comprising site-directed mutations of each functional tyrosine (Y) residue to phenylalanine (F) were cloned into a pMH-Neo expression vector (CD25-LILRB4 ITIMs). Studies aimed at defining LILRB4 functions use Ab ligation experiments as its natural ligand(s) are unknown. However, LILRB4-expressing cells, particularly monocytes and monocytic cell lines such as THP-1 also express numerous inhibitory LILRs that have high structural homology to LILRB4,5,19 which may confound results because of potential cross-reaction of anti-LILRB4 Abs with other LILRs. Hence, the reason for the generation of CD25-LILRB4 ITIM chimeric proteins with surface-expressed CD25; THP-1 cells do not express native CD25. Nine constructs—three single mutants, three double mutants, one triple mutant, one wild type and one vector only (mock) construct—were stably transfected into THP-1 cells. Transfected cells were thoroughly quality controlled and then used for experiments aimed at defining the role of each LILRB4 ITIM in regulating Fc receptor-mediated activation. Collectively, the results presented here suggest that LILRB4 is a complex receptor, which, upon co-ligation with Fc receptors, can transduce both activating and inhibitory signalling, depending on the position of the different tyrosine residues in its ITIMs.
Materials and methods
Cells and Abs
THP-1 cells (ATCC clone TIB-202) were cultured in RPMI 1640 medium supplemented with 2 mM l-glutamine, 10% heat-inactivated FBS, 100 U/ml penicillin, 100 µg/ml streptomycin, 1 mM sodium pyruvate, 10 mM HEPES, 0.1% β-mercaptoethanol (all from Life Technologies, Waltham, MA, USA) and 20 mM sodium bicarbonate (Sigma-Aldrich, St. Louis, MO, USA) at 37℃ with 5% CO2. Stably transfected cells were generated as described below and passaged every 2–3 d at a density of 2–3 × 105 cells/ml in complete RPMI medium containing 0.5 mg/ml selection antibiotic, G418 (Life Technologies). Cells between passages 5 and 12 were used for experiments.
Unconjugated mouse anti-human CD25 (Affinity Bioreagents, CO, USA), LILRB4 (kindly donated by Dr Luis Borges, Amgen Inc., Thousand Oaks, CA, USA), mouse IgG1-negative control mAbs (Sigma-Aldrich), PE or FITC-conjugated goat anti-mouse secondary Abs, FITC-conjugated FcγRI and FITC-conjugated mouse IgG1-negative control (all from BD Biosciences, San Jose, CA, USA) were used to analyse surface expression of these receptors by flow cytometry. Sodium azide-free unconjugated mouse anti-FcγRI (R&D Systems, Minneapolis, MN, USA) and mouse anti-CD25 mAb (Affinity Bioreagents) and F(ab)2 fragment goat anti-mouse IgG (Fc-specific; Jackson ImmunoResearch, West Grove, PA, USA) were used for cross-linking and/or co-ligation experiments. Goat anti-SHP-1 (R&D Systems), rabbit anti-SHP-2 (Millipore, Billerica, MA, USA), rabbit anti-SHIP (Millipore), rabbit anti-Syk (Cell Signaling, Danvers, MA, USA), rabbit anti-pSyk (Tyr 525/526) (Cell Signaling), mouse anti-β-actin (Sigma-Aldrich) and HRP conjugated rabbit anti-goat, goat anti-rabbit, goat anti-mouse Abs (all from Bio-Rad, Foster City, CA, USA) were used for Western blotting, 99% pure phorbol myristate acetate (PMA) was purchased from Sigma Aldrich (Castle Hill, Australia).
CD25-LILRB4 chimeric plasmid and site-directed mutant constructs
The extracellular and transmembrane domains of human CD25, including the signal peptide (-21Met to 238Leu), were fused in frame and upstream to the intracellular domain of LILRB4 (258Gln to 425His) by three-step overlap extension PCR. The pMLSV N1/N4-S plasmid (ATCC), containing human CD25 cDNA, was used as template for the first-round PCR amplification using the forward primer (A) 5’-GACCTG
Y337F: 5’-GCAGTGACGT
Y389F: 5’-GGATGTGACCT
Y419F: 5’-GCCTCTCCAGCTGAGCCCAGTGTCT
A unique XmnI site was present in the pMH-Neo vector but absent in the insert containing CD25-LILRB4 chimeric constructs and was used to select mutants from the wild type pMH-Neo-CD25-LILRB4 chimeric plasmid used as a template. The selection primer 5’-AAAGTGCTCATCATTGGA Schematic diagram of CD25-LILRB4 ITIMs plasmid constructs. Plasmid constructs of the extracellular and transmembrane domains of human CD25 α-chain fused to the intracellular domain of human LILRB4 containing various tyrosine-to-phenylalanine site-directed mutants were cloned into pMH-Neo vector mammalian expression vector.
Generation of stably transfected THP-1 cells
Wild type pMH-Neo-CD25-LILRB4 or constructs with one or more Y to F mutation(s), and the pMH-Neo empty vector were stably transfected into THP-1 cells. In brief, cells (2.5 × 105) in completed media excluding antibiotics were seeded onto a 24-well plate at 37℃ with 5% CO2 in air for 1 h and transfected with 1 µg of each DNA construct using Lipofectamine® 2000 (Life Technologies). 22 Selection antibiotic (0.8 mg/ml G418) was added 48 h post-transfection and single cells with high transgene expression selected by limiting dilution at wk 3. Multiple single clones from each stably transfected cell was selected, expanded, quality controlled and multiple aliquots cryopreserved for subsequent functional studies. Quality controls included assessment of the effects of stable transfection with the various mutants on cell viability using trypan blue exclusion, LIVE/DEAD® viability assay kit (Molecular Probes, Eugene, OR, USA) and determine surface expression of the relevant receptors, including the chimeric protein (CD25), FcγRI and native LILRB4, as well as expression of relevant phosphatases including SHP-1, SHP-2 and SHIP-1 by flow cytometry and Western blotting, respectively.
Flow cytometry and Western blotting in transfected THP-1 cells
Surface expression of CD25-LILRB4 ITIM chimeric proteins and relevant surface receptors, including FcγRI and native LILRB4 on THP-1 cells, were assessed by flow cytometry as described previously. 6 In brief, THP-1 cells were washed twice in cold PBS and aliquots (3 × 105 cells in 50 µl PBS) incubated with anti-CD25, anti-FcγRI-FITC, anti-LILRB4 or isotype-matched negative control mouse mAbs (5 µg/ml of each) for 45 min at room temperature (RT; 22–24℃). After one wash in PBS containing 0.5% BSA (Sigma-Aldrich), cells were either incubated with 3 µg/ml fluorochrome-conjugated goat anti-mouse secondary Abs for 1 h on ice, or directly washed and fixed. Cells were washed twice in cold PBS containing 0.5% BSA, then fixed in 1% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA, USA) in PBS. Total of 3 × 104 events were acquired using FACScan™ flow cytometer (BD Biosciences) and data analysed using FlowJo software (Tree star Inc., Ashland, OR, USA).
Baseline SHP-1, SHP-2 and SHIP protein in transfected THP-1 cells were assessed by Western blotting. In brief, cell lysates were prepared in 1% NP-40 containing 150 mM NaCl, 50 mM Tris-HCl, 5 mM EDTA, 10 µM pervanadate (Sigma-Aldrich) and protease inhibitors (Roche Applied Science, Penzberg, Germany). Lysates (20 µg) were loaded onto 10% SDS PAGE gels, separated under reducing conditions, transferred onto polyvinyldifluoride membranes (Millipore), blocked with 5% BSA in TBST buffer (TBS buffer including 0.1% Tween 20) then incubated with unconjugated primary mAb (1 µg/ml) at 4℃ overnight (18 h). After three washes with TBST buffer, membranes were incubated with appropriate HRP-conjugated secondary IgG (H + L) (0.08 µg/ml; Bio-Rad) for 90 min at RT, washed and developed using a chemiluminescent reagent (PerkinElmer, Waltham, MA, USA). Images were acquired using ImageQuant™ LAS 4000 (GE Healthcare Life Sciences, Waltham, MA, USA). Membranes were re-probed with anti-β-actin mAb (1 µg/ml) to confirm equal protein loading.
Co-ligation of FcγRI with CD25-LILRB4 ITIMs on THP-1 cells and TNF-α production
To determine the relative contribution of the three LILRB4 ITIMs in regulating FcγRI-mediated TNF-α production, THP-1 cells with the various mutations were ligated with anti-FcγRI and/or anti-CD25 mAb as described.6,7 In brief, flat-bottomed 96-well plates (Costar®, NY, USA) were coated in duplicates with 50 µg/ml of F (ab’)2 fragment goat anti-mouse IgG (Fc-specific) at 4℃ overnight, washed with 0.9% NaCl once and then coated with 500 ng/ml anti-FcγRI + anti-CD25 IgG1 mAbs or irrelevant control IgG1 mAb in PBS containing 2.5% BSA at 37℃ for 2 h. Unbound Abs were aspirated, wells washed with 0.9% NaCl once and 3.5 × 105 cells in 100 µl of cross-linking buffer (CLB; RPMI supplemented with 10 mM HEPES, 1 mM MgCl2, 0.1 mM CaCl2 and 0.1% BSA) added. Plates were incubated for 30 min at 37℃ with 5% CO2, topped up with 100 µl of CLB containing 20% FBS. After 15 h incubation, 100 µl of cell-free supernatants were collected and used for TNF-α ELISA (DuoSet ELISA kit; R&D Systems). The effects of FcγRI and CD25 after brief (2 min) surface co-ligation of receptors on phosphorylation of Syk was assessed by Western blotting of cell lysates using anti-pSyk (1 µg/ml) and anti-total Syk (0.5 µg/ml) Abs as described by us. 7
Measurement of bactericidal activity in PMA-differentiated, CD25-LILRB4 ITIM transfected THP-1 cells
To assess bactericidal activity, 5 × 104 PMA-differentiated cells were resuspended in 1 ml CLB in sterile FACS tubes and infected with freshly grown, human serum-opsonised live Salmonella Typhimurium at an optimised multiplicity of infection of 50 (bacteria-to-cell ratio of 50:1). After 90 min incubation with the bacteria at 37℃ in 5% CO2 in air, bacteria in the media or those attached to the cell surface were killed by treating cells with 200 µg/ml gentamicin (Life Technologies) for 30 min at 37℃, followed by stringent washes (3×) with PBS. Phagocytosed (intracellular) bacteria were then extracted by lysing the cells using 2% saponin (Sigma-Aldrich) in PBS with constant shaking at 100 rpm and intermittent vortexing for 15 min at RT. Serially diluted lysates were plated onto 10-cm LB agar plates and incubated at 37℃ overnight and bacterial colonies for each sample counted by a blinded observer in five independent experiments and data presented as mean colony numbers ± SEM in which higher counts represented poor bactericidal activity.
Statistical analysis
Relative contributions of the intracellular domains of LILRB4 to its regulatory functions were assessed by comparing data from each clone expressing CD25-LILRB4 ITIM mutants to vector only (mock) transfected cells. One-way ANOVA with Dunnett’s multiple comparison was used to compare CD25 and FcγRI surface expression levels, effects on TNF-α production, bactericidal activity upon co-ligation of FcγRI and CD25-LILRB4 ITIMs. P-Values < 0.05 were considered statistically significant.
Results
Stable CD25-LILRB4 ITIM overexpressing THP-1 cells expressed comparable levels of surface CD25, FcγRI, native LILRB4 and intracellular phosphatases
All mutants expressed high levels of surface CD25 with comparable mean fluorescence intensities (Figure 2a). As expected, vector-only transfected (mock) or non-transfected THP-1 cells did not express CD25 (Figure 2a). Importantly, stable expression of CD25-LILRB4 ITIM chimeric proteins did not significantly alter surface expression of native FcγRI (Figure 2b) or LILRB4 (Figure 2c) when compared with mock transfected or non-transfected cells. Similarly, Western blot analysis of SHP-1, SHP-2 and SHIP indicated that overexpression of (a) particular construct(s) did not significantly alter total amounts of these phosphatases (Figure 2d). LIVE/DEAD viability assays and trypan blue exclusion tests of THP-1 cells stably transfected with the various CD25-LILRB4 ITIM chimeric DNAs consistently showed > 94% viability (ranging from 94.5 ± 1.6 to 95.5 ± 1.2) with little or no differences among different ITIM mutants (Table 1). Expression of similar levels of these relevant proteins and equivalent proportions of viable cells allowed among the various transfected cells allowed unbiased comparisons of functions between the mutants. After performing the comprehensive quality controls described above, 3–5 independent repeat experiments were performed for each functional study using fresh aliquots of cells. All experiments were performed in duplicate.
Expression of surface FcγRI, native LILRB4, CD25-LILRB4-ITIMs chimeric protein and cytosolic SHP-1, SHP-2 and SHIP in stably transfected THP-1 cells. Cell surface staining and flow cytometric analysis of (a) FcγRI native, (b) LILRB4 and (c) CD25 α-chain showing similar levels of surface expression of all receptors in the various CD25-LILRB4 ITIM mutant-expressing THP-1 cells; as expected, no CD25 was expressed on vector only (mock) transfected cells. Data in the insets represent the mean fluorescence intensity of surface expression from five independent experiments ± SD. (d) Representative Western blots of SHP-1, SHP-2, SHIP control in cell lysates from stable mock and CD25-LILRB4 ITIM-transfected THP-1cells demonstrating comparable immuno-reactive bands, indicating overexpression of any of the ITIM mutants did not alter expression level of any of these proteins and allowed unbiased comparison of functions among the various transfected cells. Proportions of viable cells in stably transfected THP-1 cells as determined by LIVE/DEAD assay (n = 6).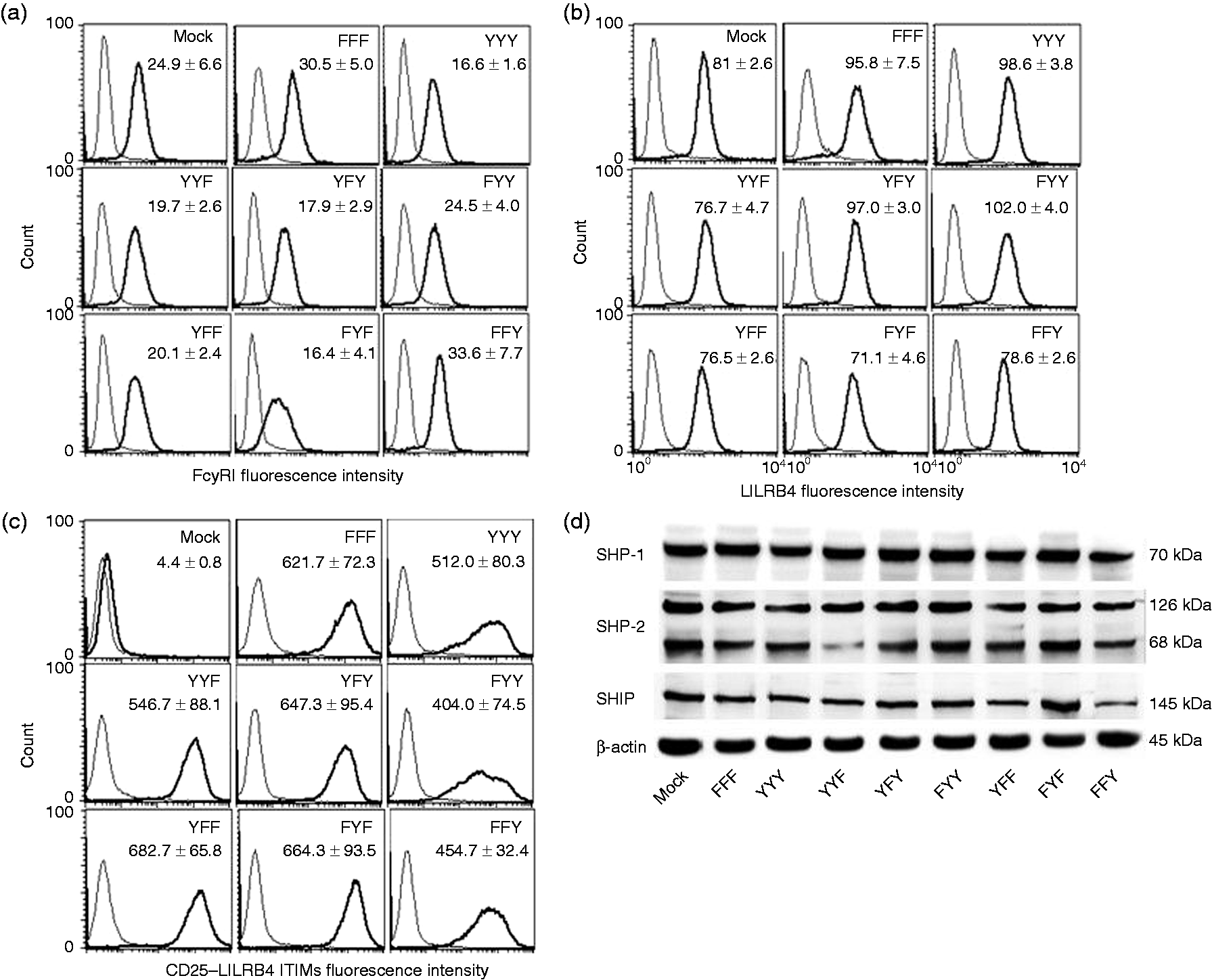
Opposing effects of transfected LILRB4 ITIMs on FcγRI-mediated TNF-α production by THP-1
Co-ligation of FcγRI and CD25 on the surface of vector-only (mock) transfected cells using plate immobilised anti-CD64 and anti-CD25 mAb for 15 h secreted 98.2 ± 8.7 pg/ml of TNF-α into culture supernatant (Figure 3a). As expected, co-ligation of FcγRI and CD25 on cells expressing CD25-LILRB4 ITIM containing all three functional tyrosine residues (YYY; wild type) significantly reduced TNF-α production to 8.2 ± 5.6 pg/ml (Figure 3a). This was a profound 85.3 ± 7.8% suppression of TNF-α release when compared with the mock-transfected cells (Figure 3b). Co-ligation studies using cells expressing CD25-LILRB4 ITIMs with single functional tyrosine residue showed that the middle Tyr389 (FYF) alone was sufficient to cause maximum suppression of TNF-α production to 3.8 ± 3.0 pg/ml (Figure 3a) which was 88.9 ± 8.1% inhibition compared with mock-transfected cells and (Figure 3b). Similarly, the single intact distal Tyr419 (FFY) also suppressed TNF-α production by 74.1 ± 7.6% (19.9 ± 7.0 pg/ml), albeit somewhat less than that achieved with FYF or YYY (Figure 3a, b). In contrast, co-ligation of FcγRI and CD25 on cells expressing the proximal Tyr337 (YFF) significantly enhanced TNF-α production to 150.1 ± 27.6 pg/ml, which was 28.7 ± 2.2% higher than that produced by mock-transfected cells (Figure 3a, b). Consistent with the opposing effects of the proximal Tyr337 (YFF) and the middle Tyr389 (FYF), a construct containing both functional tyrosine residues (YYF) showed markedly attenuated inhibitory effects of 19.8 ± 4.6% (82.7 ± 29.3 pg/ml) (Figure 3a, b), indicating blunting of the inhibitory effects of the middle Tyr389 by the activating function of the proximal Tyr337. Interestingly, unlike its opposing effect to the middle Tyr (FYF), the proximal Tyr was not able to attenuate the suppressive effect of the distal Tyr in cells expressing YFY (Figure 3a, b). FYY also potently suppressed TNF-α production by 70.3 ± 4.2%, which was smaller effect than that achieved by FYF and FFY alone (Figure 3a, b). Surprisingly, co-ligation of FcγRI and CD25 on cells expressing the CD25-LILRB4 ITIM without functional tyrosine residues (FFF) resulted in significantly lower TNF-α production compared with mock-transfected cells (67.6 ± 8.4 vs. 98.2 ± 8.7 pg/ml) (Figure 3a), which was ∼30% lower than mock-transfected cells (Figure 3b).
ELISA of TNF-α production in CD25-LILRB4 ITIM-transfected THP-1 cells upon Ab co-ligation of FcγRI and CD25. (a) Induction of TNF-α production upon surface co-ligation of FcγRI and CD25 for 15 h using plate-immobilised mAbs, which was significantly higher than FcγRI and CD25 co-ligated transfectants expressing YYY, YFY, FYY and FFY, but was significantly lower than YFF-expressing cells. Cells transfected with FFF and YYF also showed decreased levels TNF-α in culture supernatant when compared with mock-transfected cells, but these were not statistically significant. All transfected cells produced ≤ 2.3 pg/ml of TNF-α when incubated with irrelevant IgG1 control and anti-CD25 mAbs that were subtracted from the corresponding data as background (mean ± SEM of four independent experiments in duplicates; ns: not significant; **P < 0.01; ***P < 0.001 compared with mock anti-FcγRI+ anti-CD25). (b) A summary of percentage inhibition or enhancement of TNF-α production by CD25-LILRB4 ITIM-expressing cells relative to mock-transfected cells in which cells expressing YYY, YFY, FYY, FYF and FFY showed inhibition, while YFF showed marked increase in TNF-α production (mean ± SEM of four independent experiments in duplicates). (c) Western blotted using anti-pSyk showing markedly inhibition of FcγRI-mediated Syk phosphorylation upon co-ligation with anti-CD25 mAb in cells expressing CD25-LILRB4 ITIMs containing all three functional Tyr residues (YYY) when compared with mock-transfected and FFF-expressing cells. LILRB4 ITIM mutant with the single intact middle Tyr residue (FYF) also profoundly suppressed Syk phosphorylation in contrast to cells expressing the single proximal Tyr (YFF), which caused increased Syk phosphorylation. Somewhat unexpectedly, Syk phosphorylation was not regulated by FFY, despite its significant suppression of TNF-α production. As expected, all three ITIM mutants expressing two intact Tyr residues markedly decreased Syk phosphorylation. None of the cells showed changes in the expression levels of total Syk upon Ab co-ligation of FcγRI with CD25 (middle panel in each lane). Western blotting against β-actin was used as a loading control (lower panel in each lane; n = 2).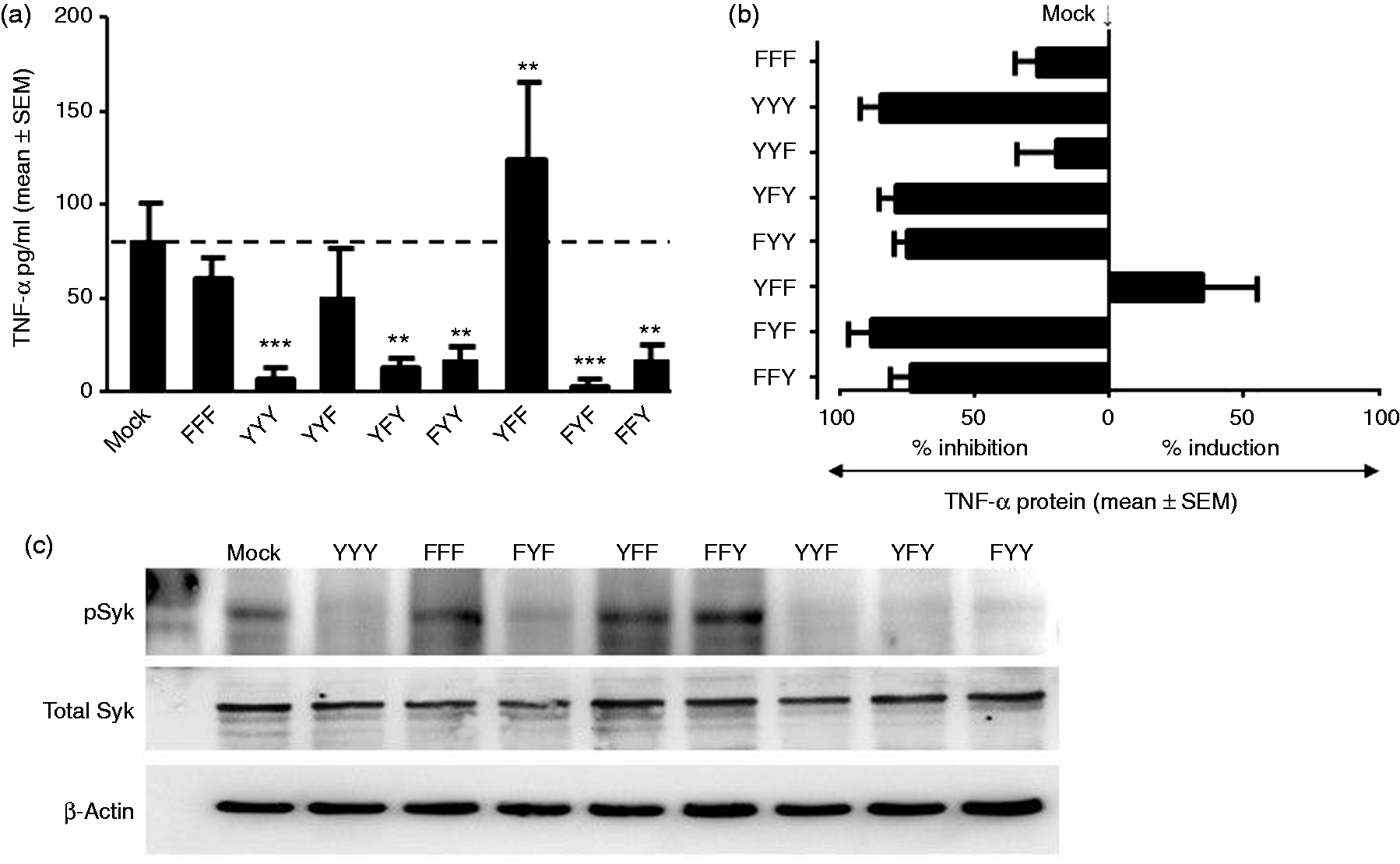
To determine whether the differential regulation of TNF-α production by the various LILRB4 ITIMs in response to FcγRI cross-linking was through variable dephosphorylation of the key upstream signalling molecule Syk, lysates from anti-FcγRI and anti-CD25 co-ligated cells were Western blotted using anti-pSyk and anti-total Syk Abs. Consistent to the profound inhibitory effects on TNF-α production, cells expressing CD25-LILRB4 ITIMs containing all three functional tyrosine residues (YYY) markedly inhibited FcγRI-mediated Syk phosphorylation upon co-ligation with anti-CD25 (Figure 3c). Similarly, LILRB4 ITIM mutant with the single intact middle Tyr residue (FYF) profoundly suppressed Syk phosphorylation (Figure 3c) in agreement with its significant inhibitory effects. In contrast, cells expressing the single proximal Tyr (YFF) increased Syk phosphorylation when compared with FYF (Figure 3c), consistent with its enhancement of TNF-α production. Somewhat unexpectedly, Syk phosphorylation was not regulated by FFY (Figure 3c), despite its significant suppression of TNF-α production. As expected, all three ITIM mutants expressing two intact Tyr residues markedly decreased Syk phosphorylation (Figure 3c).
Expression of CD25-LILRB4 ITIM containing a single intact distal Try significantly enhanced bactericidal activity
Cell lysates from mock-transfected, PMA-differentiated THP-1 cells that were infected with serum-opsonised S. Typhimurium formed an average of 117.5 ± 10 colonies (Figure 4a). Cells transfected with CD25-LILRB4 ITIM containing a single distal Try419 (FFY) formed 33.4 ± 0.9 colonies (Figure 4a), which represented a significant increase in bactericidal activity of 71.6% compared with mock-transfected cells (Figure 4b). In contrast, cells expressing a single proximal Tyr337 (YFF) formed 143.6 ± 23.1 colonies (Figure 4a), indicating significant suppression of bactericidal activity by up to 30% when compared with mock-transfected cells (Figure 4b). Cells expressing FYF formed 80.3 ± 12.2 colonies, indicating a moderate increase of bactericidal activity of 32% (Figure 4a, b). As expected, the significant 70% increase in bactericidal activity promoted by the distal Tyr was markedly blunted by the suppressive effects of the proximal Try in cells expressing both residues (YFY) that formed 96.5 ± 24.1 colonies (Figure 4a), an enhancement of bactericidal activity by 18% and cells expressing the proximal and middle Tyr (YYF) showed moderate enhancement of bactericidal activity by 22% (Figure 4). Surprisingly the presence of both middle and distal Tyr (FYY) enhanced bactericidal activity by only 33.4% (78.4 ± 3.2 colonies), which is equivalent to cells expressing the single middle Tyr (FYF; ∼35% increase) and markedly lower than cells expressing FFY (71% increase) (Figure 4). Cells expressing YYY formed 87.8 ± 6.1 colonies (Figure 4a), representing a 25.7% increase in bactericidal activity (Figure 4b). Somewhat unexpectedly, the ITIM backbone without the functional tyrosine residues (FFF) substantially enhanced bactericidal activity by 28.4 ± 2.7%, although this was not statistically significant (Figure 4).
Bactericidal activity by PMA-differentiated-stably transfected THP-1 cells. (a) Quantification of viable intracellular S. Typhimurium colonies formed in LB agar plates that were inoculated with cell lysates extracted from the various CD25-LILRB4 ITIM-expressing cells in which formation of lower number of colonies compared to mock-transfected cells represented increased bactericidal activity (mean ± SEM; n = 5; *P < 0.05; **P < 0.01). (b) A summary of percentage inhibition or enhancement of bacteria killing by various CD25-LILRB4 ITIM-expressing cells relative to mock-transfected cells; all CD25-LILRB4 ITIM-expressing cells except YFF exhibited variable levels of increased bactericidal activity with the most prominent effect by cells expressing the single distal tyrosine residue (FFY) with > 70% increase in bactericidal activity. In contrast, cells expressing the single proximal tyrosine residue (YFF) showed up to 30% reduction in bactericidal activity (mean ± SEM; n = 5).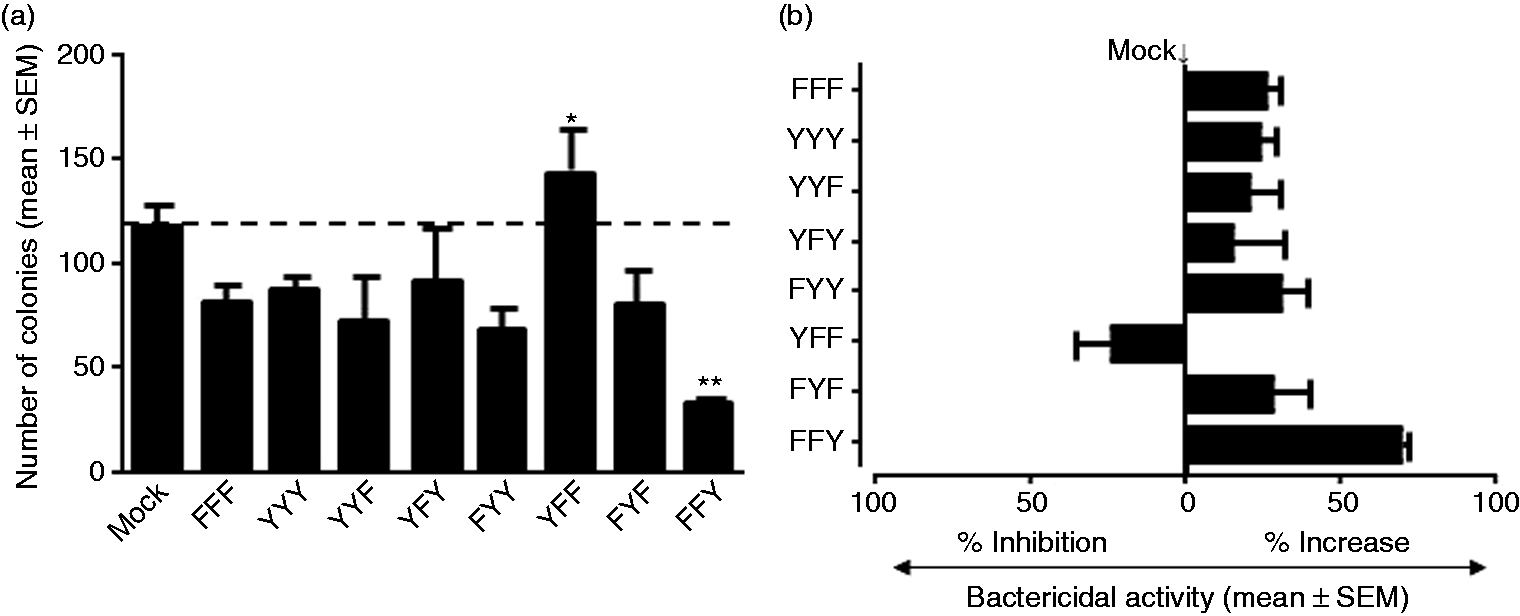
Discussion
Natural ligand(s) for LILRB4 are unknown and most functional studies use LILRB4 Ab ligation. However, cells expressing LILRB4, particularly monocytes and monocytic cell lines, also express numerous inhibitory LILRs that have high structural homology to LILRB4.5,19,23 This may confound results because of potential cross-reaction of anti-LILRB4 Abs with other inhibitory LILRs. We overcame this by transgenic expression of a chimeric protein containing surface CD25 and intracellular LILRB4 ITIMs on THP-1 cells that normally do not express CD25 allowing specific Ab-based ligation experiments. Generation of these CD25-LILRB4 ITIM chimeras also ensured stringent selection of stably transfected cells that expressed similar amounts of each ITIM mutant using comparable CD25 expression level as selection criteria (Figure 2), removing another major variable that could confound comparisons. Moreover, all THP-1 cells stably transfected with the different chimeric constructs have comparable high cell viability (>95%) and expressed similar levels of key relevant molecules, including FcγRI, SHP-1, SHP-2, SHIP and native LILRB4 proteins (Figure 2). Consideration of these parameters permitted unbiased and more valid comparisons of functions among the various mutants.
Effect of CD25-LILRB4 ITIMs in monocyte functions relative to mock-transfected cells (n = 4).
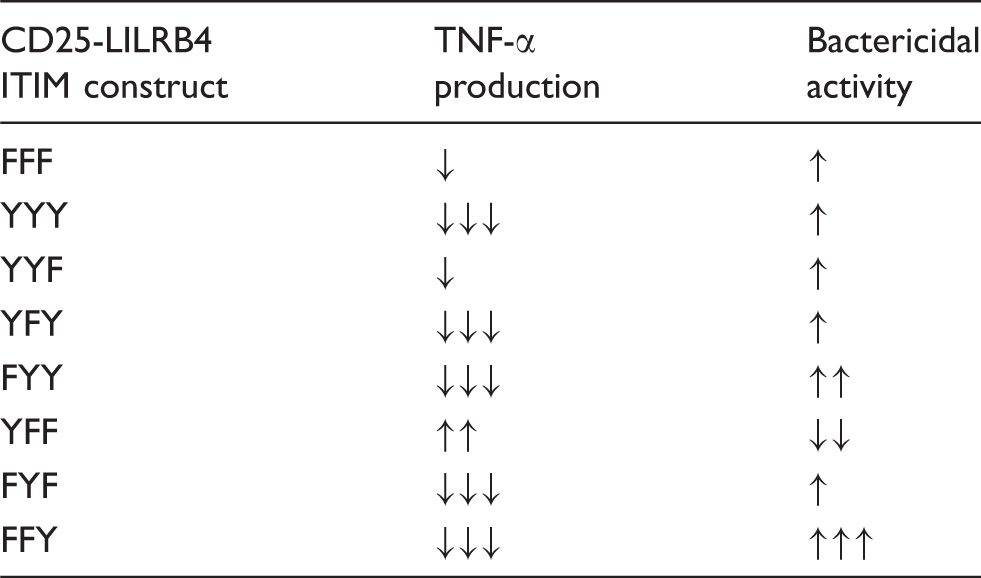
↑: < 33.3%; ↑↑: 33.3 to 66.7%; ↑↑↑: > 66.7%.
↓: < –33.3%; ↓↓: –33.3 to –66.7%; ↓↓↓: > –66.7%.
We previously showed native LILRB4 profoundly suppressed FcγRI-mediated TNF-α production by monocytes by dephosphorylating the proximal key signalling molecule, Syk, in THP-1 cells. 6 Here we found that co-ligation of FcγRI with CD25 in THP-1 cells expressing LILRB4 ITIMs containing the three intact Tyr residues (YYY) completely abrogated phosphorylation of Syk when compared with cells expressing triple-mutant ITIMs (FFF) or mock-transfected cells. Furthermore, cells expressing FYF, YYF, YFY and FYY also showed suppression of Syk phosphorylation in contrast to cells expressing YFF in which phosphorylation of Syk in response FcγRI cross-linking was not affected upon co-ligation with CD25 (Figure 3). These results were consistent with the inhibitory or stimulatory effects of each mutant on TNF-α production, supporting our functional data. Somewhat surprisingly, we found that co-ligation FcγRI with CD25 on cells expressing FFY enhanced Syk phosphorylation, despite causing significant suppression of TNF-α production, which may indicate that LILRB4 ITIMs may regulate both Syk-dependent and Syk-independent signalling pathways depending on the position of Tyr residues.
The opposing effects of certain LILRB4 ITIMs on the secretory functions of innate immune cells was further strengthened by independently validating results in a rat basophilic leukaemia cell line (RBL) that are known to signal through the common γ chain of the FcɛRI. 33 Briefly, RBL cells were first stably transfected with selected CD25-LILRB4 ITIMs, including Mock, FFF, YYY, YFF and FYF, and quality controlled (Supplementary Figure 1a, 1b). Surface FcɛRI was then co-ligated with CD25, and cell activation assessed by measuring percentage release of the pre-formed enzyme β-hexosaminidase (see Supplementary method). We found that co-ligation of FcɛRI with CD25 on cells transfected with CD25-LILRB4 ITIMs containing three non-functional residues (FFF) caused degranulation of RBL cells by an average of 25.4 ± 1.3%, similar to levels observed in mock-transfected cells (22.4 ± 1.8%) (Supplementary Figure 1c, upper panel) and in patterns comparable with TNF-α production by FFF-transfected THP-1 cells (Supplementary Figure 1c, lower panel). Similar to the results in transfected THP-1 cells, cells expressing CD25-LILRB4 ITIMs containing all three intact Tyr residues (YYY) released 6.8 ± 1.6% of total β-hexosaminidase, which was a significant suppression of degranulation by 69% when compared with mock-transfected cells (Supplementary Figure 1c). Furthermore, expression of the single intact proximal Tyr residue had significant stimulatory effects in both RBL and THP-1 cells upon its co-ligation with the Fc receptors (Supplementary Figure 1c). Interestingly, FYF that profoundly inhibited TNF-α production in transfected THP-1 cells significantly increased degranulation of RBL cells (Supplementary Figure 1c), suggesting that the middle Tyr has differing functions on the release of newly synthesised mediators as contrasted to the release of preformed enzymes; however, this warrants further investigation.
Unlike their main inhibitory effects on TNF-α production, most of the LILRB4 ITIMs enhanced bactericidal activity when compared with mock-transfected cells (Table 2). This may indicate that LILRB4 primarily inhibits the secretory functions of monocytes but promotes bacterial clearance. Remarkably, the single proximal Tyr (YFF)-expressing cells that significantly enhanced TNF-α production markedly suppressed bactericidal activity. In contrast, cells expressing the single distal Tyr419 (FFY) showed profound inhibition of TNF-α production but significantly increased bacterial killing. These opposing effects on TNF-α production and bactericidal activity by YFF and FFY suggest that preferential activation of the single proximal Tyr residue may lead to enhanced pro-inflammatory mediator production at the expense of limited bacterial killing, whereas selective activation of the distal Tyr may polarise monocytes towards efficient bacterial clearance with minimal inflammation. Similar polarisation of effector cells occurs following activation of related receptor, LILRA2.34,35 Interestingly, the effect of the single middle Tyr389 (FYF) on bactericidal functions were minimal, despite showing the most dominant inhibitory functions with regards to cytokine production (Table 2), indicating specific regulatory functions.
Unexpectedly, cells expressing the LILRB4 ITIM backbone without functional tyrosine residues (FFF) had measurably reduced FcγRI-mediated TNF-α production and enhanced bactericidal activity (Table 2). This may indicate that additional structural elements within the LILRB4 intracellular domain may also display regulatory functions. Consistent with this, similar inhibitory effects were observed when the tyrosine residue within the single ITIM of the killer inhibitory receptor CD158d was mutated. 36 The structural domains mediating these phosphotyrosine-independent effects are unknown; reports indicate that proline-rich regions16,17,37 and SH3-binding motifs within ITIM backbones may functionally interact with several protein kinases14–17,37 or adaptor proteins, 15 and regulate cellular activation. LILRB4 contains two SH3 binding motifs and a proline-rich region. Whether these structures contribute to its immune regulatory functions remains to be elucidated. It is also noteworthy that the use of the CD25-LILRB4 chimeric receptor system may not fully recapitulate all functions of LILRB4 owing to the potential contribution of the extracellular components of LILRB4 to its overall functions. Future studies are required to validate these and define contribution of the extracellular domains of LILRB4 by site-targeted mutagenesis of the ITIMs of a native LILRB4, which is currently not feasible owing to lack of knowledge of its natural ligands that are required for functional studies. Taken together, results indicate that LILRB4 might have complex inhibitory and activating functions in monocytes depending on the nature of the stimuli and the position of the tyrosine residue in its ITIMs.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to Dr Luis Borges, Amgen Inc. USA for kindly providing us with the anti-LILRB4 monoclonal Ab.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the National Health and Medical Research Council of Australia, Project grant number 568771. The funding body had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
