Abstract
Apidaecin peptides are produced by the honeybee Apis mellifera as a major part of its non-specific defense system against infections. Having verified that the peptides apidaecin 1b and Api88—a designer peptide based on the native apidaecin 1b sequence—are highly active against Gram-negative bacteria, we studied their ability to modulate biological activities of human monocytes and mast cells (MC), two important cell types of the human innate immune system. We could show that both peptides are nontoxic and fairly resistant to degradation in cell culture medium containing 10% FBS. Among the peptides tested we found Api88 to inhibit LPS-induced TNF-α production in a concentration-dependent manner. Resting monocytes did not respond to Api88. Whilst Api88 neither induced migration nor affected the phagocytic activity of monocytes it partially inhibited the generation of reactive oxygen intermediates produced in response to LPS. In human MC, however, Api88 triggered degranulation and the mobilization of intracellular Ca2+-ions. Taken together these data clearly indicate that Api88 is a multifunctional molecule that can modulate biological responses of human monocytes and MC in addition to its antimicrobial activity.
Introduction
Antimicrobial peptides (AMPs) are produced by virtually all species, ranging from plants and insects to humans, as a defense mechanism against invading pathogens, such as bacteria, viruses, fungi and parasites. Although AMPs are diverse in their sequences and structures, certain features are common.1,2 They are generally amphipathic, 12–50 amino acids in length, have at least two positive charges (as arginine or lysine residues) and many contain two or more disulfide bonds. Cationic AMPs are attracted to the negative charge of the microbial membrane and, once attracted, they might disrupt the membrane or, by traversing the membrane, inhibit intracellular targets.3,4 AMPs do not only contribute to the innate host defense by eradicating foreign pathogens, but also modulate immune functions of the host. As regulators of such functions, defensins and cathelicidins, the two major antimicrobial peptide families found in humans and other mammals, have been studied extensively. 5 AMPs are released by a variety of cell types, such as monocytes, mast cells (MC), neutrophils, keratinocytes and epithelial cells6–10 upon stimulation by microbial pathogens. The secreted AMPs can induce chemotaxis in immune effector cells either indirectly by promoting the production of chemokines or directly by serving as chemotactic factors themselves, thereby recruiting immune cells to the site of infection.11,12
Furthermore, AMPs have been shown to play a crucial role in TLR-dependent inflammatory responses. They selectively suppress TNF-α release from human monocytes and macrophages stimulated by LPS or other TLR-agonists, such as lipoteichoic acid (LTA).13–15
It has been suggested that AMPs exert their anti-endotoxic effects, in part, by directly affecting the activation of the transcription factor NF-κB13,14 or, as an alternative, by binding LPS, thereby neutralizing it before it can trigger inflammation.
Other immunomodulatory functions triggered by AMPs include stimulation of wound healing,16–18 promotion of histamine release from MC, 6 extension of the lifespan of neutrophils, 19 stimulation of angiogenesis, 20 and modulation of DC and T cell responses. 21 With the increasing development of antibiotic resistance among bacterial pathogens, AMPs have gained much interest as an alternative for the treatment of infections.
In light of such potential therapeutic applications we have focused our interest on apidaecins—AMPs found in the hemolymph of the honey bee at elevated levels following infections. Apidaecins are 18–20 residue-long, proline-rich peptides with highly conserved PRPPHPR/L C-termini. 22 The less conserved N-terminal part appears to be involved in the modulation of the activity against different Gram-negative bacteria.23,24 Whilst the antimicrobial activities of apidaecins has been well documented,24–26 little is known about their function as immune regulators in mammals and, especially, in humans.
Here, we set out to investigate the effect of apidaecin peptides on the biological response of human monocytes and MC, both cell types involved in the innate immune system. Besides wild type apidaecin 1b (Api1b), we tested the Api1b derivative Api88, which, compared to Api1b, showed increased antibacterial activity. In the presence of LPS, Api88 was found to display anti-inflammatory properties in monocytes whilst it acts as a strong activator of MC degranulation.
Materials and methods
Reagents
Unless stated otherwise, materials were obtained as follows: RPMI 1640 (with
Peptide synthesis
Peptides were synthesized as C-terminal amides or free acids with standard 9-fluorenylmethoxycarbonyl/tert-butyl (Fmoc/tBu)-chemistry at a 25 -µmol scale on a Syro2000 (MultiSynTech GmbH) using DIC activation in the presence of HOBt. 27 Briefly, Api88 and the degradation product Gu-ONNRPVYIPRPRPPHP-OH were synthesized by the same protocol, but, after completion of the synthesis and deprotection of the N-terminal Fmoc-group, it was incubated with 10 equivalents of HBTU in the presence of DIPEA or N-methylmorpholine in DMF to obtain a terminal N,N,N′,N′-tetramethylguanidino-group. 28 All peptides were cleaved with TFA containing 12.5% (v/v) of a scavenger mixture (ethandithiole, m-cresole, thioanisole and water, 1:2:2:2 by volume). After 2 h the peptides were precipitated with cold diethyl ether and finally purified on a C18-phase using a linear aqueous acetonitrile gradient in the presence of 0.1% (v/v) TFA as an ion pair reagent. The purified peptides were characterized by reversed phase (RP)-HPLC and MALDI-TOF/TOF-MS (4700 Proteomic Analyzer; AB Sciex, Darmstadt, Germany).
Antibacterial activity
The minimal inhibitory concentration (MIC) values were determined in triplicate by a liquid broth microdilution assay in sterile 96-well plates (Greiner Bio-One GmbH, Frickenhausen, Germany) using a twofold peptide dilution series starting at a concentration of 128 µg/ml in 1% (w/v) TSB medium (33%) and a total volume of 100 µl per well. The inoculum (50 µl, 1.5 × 107 cells/ml) was prepared by diluting an overnight (18 h) culture grown in nutrient broth (Carl Roth GmbH, Karlsruhe, Germany) with 33% TSB medium. Fifty microliters of these solutions were then added to each well, giving a starting cell concentration of 7.5 × 105 cells/well. The plates were incubated at 37℃ for 18 h. The MIC was defined as the lowest peptide concentration preventing visible bacterial growth.
Cell separation and cell culture
Monocytes were isolated from buffy coat of human blood over a Ficoll-Isopaque (Amersham Biosciences, Freiburg, Germany) density gradient and subsequent counterflow elutriation using the JE-6B elutriation system (Beckman Instruments, Palo Alto, CA, USA), as described previously.
29
The purity of the cell preparation was >90%, as assessed by morphological screening and immunofluorescence staining with a MAb against CD14 (BL-M/G14, DiaMak, Leipzig, Germany). Unless otherwise indicated the monocytes (2 × 106/ml) were suspended in RPMI 1640 medium supplemented with 10% (v/v) FBS, 100 U/ml penicillin and 100 µg/ml streptomycin. For isolation of human lung MC, tumor-free lung tissue obtained from patients with bronchial carcinoma undergoing lobectomy was provided by the Section of Pathology (RC Borstel, Germany). Approval for these studies was obtained from the institutional review board at the University of Lübeck (Germany) and informed consent was provided according to the Declaration of Helsinki. Preparation and purification of MC was performed as described elsewhere.30,31 Briefly, lung specimens were chopped and proteolytically digested. MC were enriched by Percoll gradient centrifugation followed by immunoaffinity magnetic enrichment using anti-phycoerythrin-conjugated microbeads (Miltenyi, Bergisch-Gladbach, Germany) in combination with phycoerythrin-conjugated mAb 97A6 (IOTest, Marseille, France) specific for CD203c. The cells were used not before 2 and not later than 6 wk in culture. During this period, viability (trypan blue exclusion) and functionality (FcεR-mediated degranulation) of the cells was monitored weekly. The purity of MC used for experiments ranged from 88–100%, as assessed by toluidine blue staining. Viability was always >85%, as assessed by trypan blue exclusion. MC (5 × 105/ml) were cultured in Stempro media containing 2 mM
Peptide stability
The stability of Api1b and its derivatives (100 µg/ml), and their degradation during the cell experiments, were studied in cell culture medium in the absence or presence (2 × 106/ml) of monocytes. This assay relied on a previously published protocol to study peptide degradation in sera and bacterial lysates. 27 Briefly, the peptides were incubated in RPMI 1640 medium supplemented with 10% (v/v) FBS, 100 U/ml penicillin and 100 µg/ml streptomycin in the presence or absence of LPS (100 ng/ml) at 37℃. After distinct incubation periods (0 h, 4 h and 16 h) the samples were centrifuged (2000 g, 4℃, 3 min), the supernatants (50 µl) were transferred to polypropylene vials and trichloroacetic acid (12.5 µl, 15%, w/v) was added. The samples were mixed and stored on ice for 15 min. After centrifugation (13000 g, 4℃, 5 min) the supernatants were neutralized with sodium hydroxide solution (1 M) and stored at −20℃ until analyzed by RP-HPLC. The neutralized supernatants (111 µl) were mixed with aqueous acetonitrile (3%, v/v, 89 µl) containing 0.1% (v/v) TFA. After centrifugation (12400 g, 5 min) the supernatant (180 µl) was loaded on the analytical Jupiter C18-column and eluted by a linear aqueous acetonitrile gradient (3% acetonitrile per min) in the presence of 0.1% (v/v) TFA as the ion pair reagent. The absorbance was recorded at a wave length of 220 nm.
MTT assay
The viability of LPS (100 ng/ml) and apidaecin-treated monocytes was determined by the MTT assay, which is a quantitative method based on the metabolic reduction of tetrazolium salt (MTT) into purple formazan-crystals. Monocytes (2 × 106/ml) were incubated in the presence and absence of LPS (100 ng/ml) and apidaecin for 16 h before 0.3 mg/ml MTT was added. After 2 h cells were lysed and incubated for another 6 h before absorbance was read at 570 nm. Each sample was measured in quadruplicate.
Detection of TNF-α and IL-10 in cell culture supernatants
MC (5 × 104/ml) were incubated in the presence of different concentrations of Api88, and monocytes (2 × 106/ml) in the presence and absence of LPS (100 ng/ml) and apidaecins. After 4 h and 16 h the culture supernatants were tested for TNF-α content by ELISA, as described previously 32 and for IL-10 content after 16 h by using the Cytometric Cell Bead Array (CBA; Beckton Dickinson, Erembodegem, Belgium).
Chemotaxis assay
The migration of monocytes in response to chemoattractants was determined using a modified Boyden chamber consisting of a 48-well microchemotaxis chamber (NeuroProbe, Cabin John, MD, USA) as described previously.
33
Briefly, peptides diluted in chemotaxis medium (RPMI 1640 containing 1% BSA, 20 mM HEPES, 2 mM
Phagocytosis assay
Monocytes (2 × 106/ml) were incubated with peptides for 4 h at 37℃ and 5% CO2 before 2 × 106/ml FITC-labeled E. coli particles (Invitrogen™ GmbH, Karlsruhe, Germany) were added for 3 h. The cells were kept on ice for 15 min washed twice with PBS and re-suspended in 200 µl PBS. Immediately after adding 160 µl trypan blue solution the internalization of the particles was measured by flow cytometry (green fluorescence, Fl-1).
Determination of the extracellular reactive oxygen species production
Monocytes (4 × 106/ml) suspended in phenol red-free RPMI 1640 supplemented with 2.5% FBS and 5 mM HEPES were seeded in a volume of 125 µl in a white 96-well plate. Before the addition of LPS (100 ng/ml) and Api88 (100 µg/ml) cells were incubated in the presence of luminol (140 µM) for 40 min. The chemoluminescence induced by the reactive oxygen species (ROS)-dependent oxidation of luminol was recorded continuously for 30 min, with data points being collected every minute (Luminoskan Ascent, Thermo Electron Corporation, Langenselbold, Germany). After the second stimulus recordings were continued for a further 120 min. Duplicates were performed for each set of measurements.
MC degranulation
Degranulation of MC was determined by their release of β-hexosaminidase activity, as described previously.
31
Briefly, pre-warmed MC (2.5 × 104/ml) were activated with Api88, apidaecin revers (Api rev) or anti-IgE at concentrations indicated in the text for 30 min at 37℃ in PBS-Dulbecco/0.1% BSA. Activity of β-hexosaminidase was measured by conversion of 4-nitrophenyl-2-acetamido-2-deoxy-β-
Determination of intracellular free calcium concentrations
Changes in intracellular free calcium concentrations in MC were measured by flow cytometry in fluo-4-loaded cells, as described previously. 34 Briefly, cells (1 × 105/ml) were incubated with 2 µM fluo-4-AM (Molecular Probes) in PBS-D supplemented with 0.9 mM CaCl2 and 0.5 mM MgCl2 for 30 min at 37℃. Free dye was removed by centrifugation and cells were re-suspended in 1 ml aliquots of the same buffer. MC were analyzed after stimulation with Api88, Api rev or anti-IgE-Ab by flow cytometry (BD FACSCalibur, BD Biosciences) at an excitation wavelength of 488 nm and an emission wavelength of 525 nm.
Statistical analyses
Statistical significance was calculated with the tests indicated and classified as follows: *P ≤ 0.05, **P ≤ 0.01 and ***P ≤ 0.001.
Results
Antibacterial activity
Antibacterial activity. Sequences of native Api 1b, Api88 and guanidated Api rev, as well as their minimal inhibitory concentrations (µg/ml) obtained against two E. coli and one K. pneumoniae strains. Antibacterial activity was determined against bacteria using a serial dilution of each peptide in 33% TSB-medium in at least two independent experiments (in triplicate).
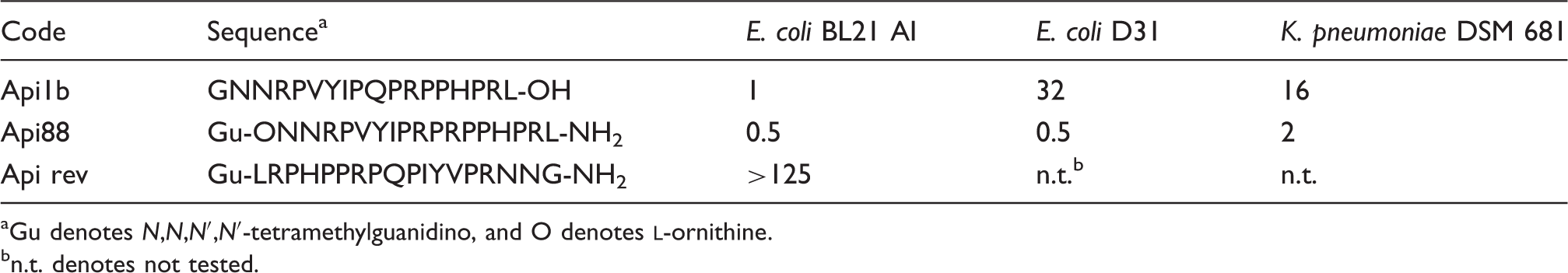
Gu denotes N,N,N′,N′-tetramethylguanidino, and O denotes
n.t. denotes not tested.
Effects of apidaecin peptides on cell viability
Before studying the effect of apidaecin peptides on LPS-induced biological responses in human monocytes we tested whether the peptides affected the viability of cells. Monocytes were incubated with the three peptides (100 mg/ml) Api1b, Api88 and Api rev in the presence and absence of LPS. After 4 h and 16 h viability was measured by using the MTT-assay. None of the peptides displayed any cytotoxic effects on monocytes compared with the untreated control cells (data not shown).
Stability of apidaecin peptides in cell culture medium
As proteases possibly present in serum and medium, or secreted by activated monocytes, may lead to the degradation and inactivation of the peptides, corresponding experiments analyzing peptide stability were performed. Figure 1A shows that of the three apidaecins tested Api88 was the least stable. The degradation rate amounted to 30–40% under all assay conditions, i.e. independent of the presence or absence of cells and LPS. Whilst Api rev was stable in cell-free medium or in the presence of resting monocytes, a significant degradation was observed when the peptide was incubated in the presence of activated monocytes, indicating that the peptide has been cleaved by the action of monocyte-derived proteases. The most stable was Api1b under all assay conditions tested. Analyses of single chromatograms derived from samples of LPS-activated monocytes being incubated with Api88 for 4 h and 16 h (Figure 1B) revealed that Api88 with a retention time of about 20.06 min was degraded slowly, whilst at a retention time of 19.61 min a degradation product appeared. The MALDI-MS of this fraction displayed a strong signal at 2022.22 Da, which matches the N-terminal sequence Gu-ONNRPVYIPRPRPPHP-OH, indicating that the two C-terminal residues of Api88 were cleaved. Taken together, these data demonstrate that the apidaecin peptides are fairly stable during 16 h incubation in serum containing medium and thus can be studied in cellular assays during this period of time.
Serum stability of apidaecin derivatives. (A) Apidaecin derivatives (100 µg/ml) were incubated for 16 h in the presence and absence of monocytes (2 × 106/ml) with or without LPS (100 ng/ml). Peptide concentrations in the supernatants were determined by analytical RP-HPLC. Peak areas of samples measured before incubation were set at 100%. Data shown are the mean ± SEM of three independent experiments. (B) RP-HPLC chromatogram of Api88. Api88 (100 µg/ml) was incubated with human monocytes (2 × 106/ml) in the presence of LPS for 0 h, 4 h and 16 h. Degradation products were analyzed by RP-HPLC. The retention time for the intact Api88 was 20.06 min whilst a degradation product appeared at a retention time of 19.61 min after 4 h and 16 h (arrow). Pictured is one representative experiment after 0 h, 4 h and 16 h (n = 3).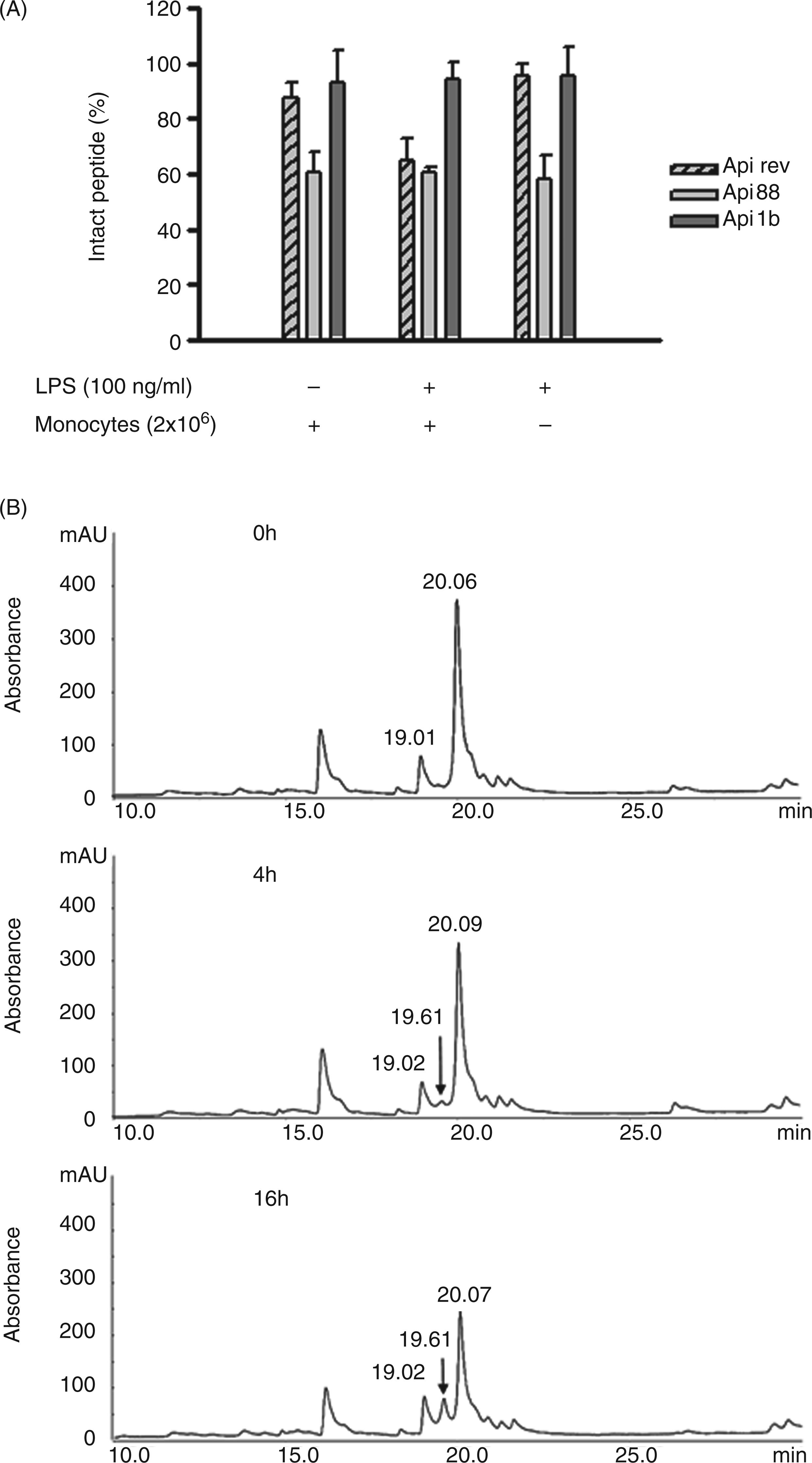
Api88 suppresses LPS- but not Pam3CysSK4-induced TNF-α and increases IL-10 release
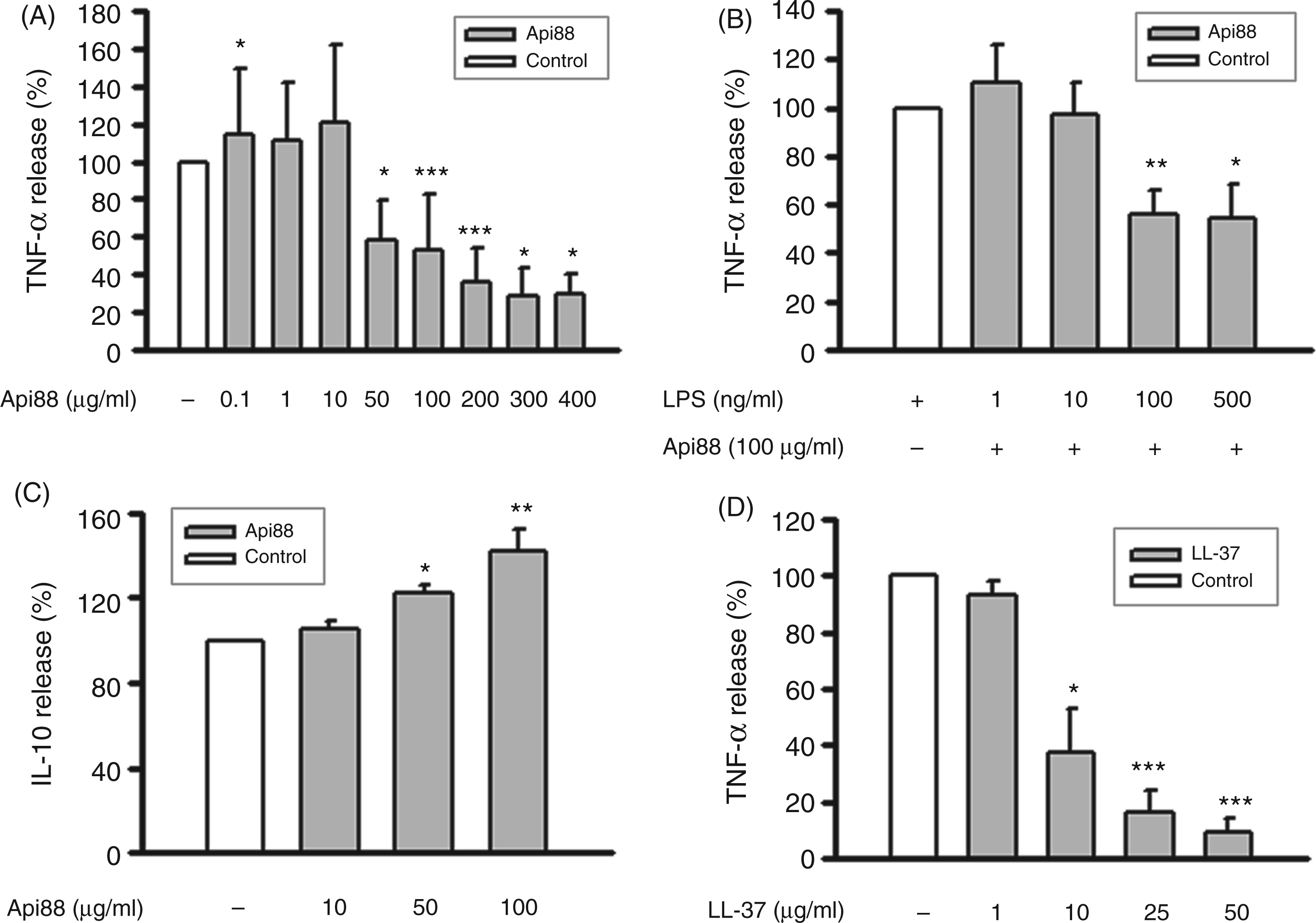
To investigate the effect of apidaecin peptides on the LPS-induced TNF-α production, monocytes were treated with LPS in the presence and absence of the peptides, and the TNF-α concentrations were determined in the cell culture supernatants. The actual TNF-α concentrations produced in response to LPS varied considerably between monocyte preparations of the different blood donors. For example 16 h after stimulation with LPS (100 ng/ml) values of 19.9 ± 22.1 ng/ml (mean ± SD; n = 17) were obtained, with the range being 1.6–76.8 ng/ml. These data reflect the well-known great individual variations. The release of TNF-α was reduced significantly in the presence of Api88 after incubation times of 4 h and 16 h (Figure 2). No TNF-α was detectable when the cells were incubated with the three peptides in the absence of LPS (data not shown). As the suppression of TNF-α production was restricted to Api88, further experiments were carried out mainly with this peptide. To determine the dose dependency of the anti-endotoxin effect, the supernatant was monitored for TNF-α after 16 h of simultaneous stimulation with LPS (100 ng/ml) and Api88 in the range of 0.1–400 µg/ml. Concentrations up to 10 µg/ml (4.3 µM) had no effect on the TNF-α production (Figure 3A). Only at a dose of 50 µg/ml (21.5 µM), and even more at higher concentrations, the TNF-α production was substantially decreased by 40–70%.
Api88 suppresses LPS-induced TNF-α release. Human monocytes (2 × 106/ml) were incubated with LPS (100 ng/ml), apidaecin derivatives (100 µg/ml) (A, B) or the apidaecine degradation product (Gu-ONNRPVYIPRPRPPHP-OH) (100 µg/ml) (C, D) for 4 h and 16 h. TNF-α concentrations in the supernatants were analyzed by ELISA. The TNF-α concentration of monocytes incubated with LPS in the absence of apidaecin (white bar) was set as 100%. Data shown are the mean ± SEM of four (C, D) of at least five (A, B) independent experiments; Mann–Whitney rank sum test calculated to the control. Api88 suppresses LPS-induced TNF-α, but increases IL-10 release. Monocytes (2 × 106/ml) were incubated with LPS (100 ng/ml) and different concentrations of Api88 (A, C), 100 µg/ml Api88 and different concentrations of LPS (B), LPS (100 ng/ml) and different concentrations of LL-37 (D). After 16 h TNF-α concentrations were determined in the supernatants by ELISA (A, B, D) and IL-10 concentrations by cytometric bead array (C). The TNF-α and IL-10 concentrations of monocytes incubated with LPS in the absence of Api88 (white bar) were set as 100%. Data shown are the mean ± SEM of at least three independent experiments; Mann–Whitney rank sum test (A, C) or Student’s t-test (B, D) calculated to the controls.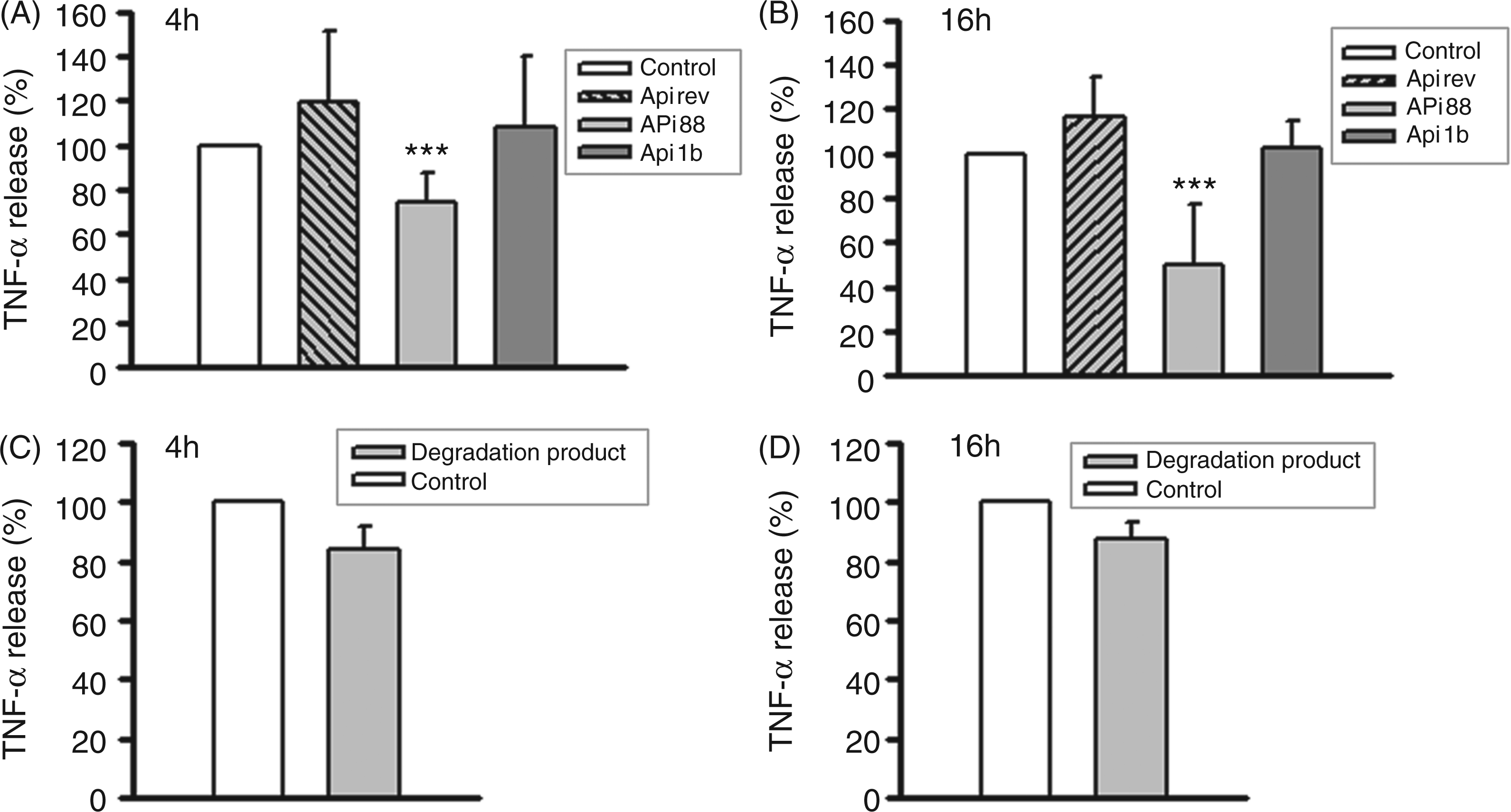
Having shown that a minor part of Api88 was degraded to a product which we identified as Gu-ONNRPVYIPRPRPPHP-OH, we reasoned that the peptide might have a role in suppressing LPS-induced TNF-α production. However, when incubating monocytes in the presence of the peptide and LPS for 4 and 16 h no reduction of TNF-α production was observed (Figure 2C, D) indicating that the inhibitory effect of Api88 was not due to its degradation product.
To test whether the Api88-induced decrease in TNF-α production was due to binding and neutralization of LPS, monocytes were treated with 100 µg/ml Api88 and varying concentrations (1, 10, 100, and 500 ng/ml) of LPS before measuring TNF-α concentrations. We reasoned that should Api88 bind to LPS then a more pronounced inhibitory effect might be expected at lower LPS-concentrations. However, measurements of TNF-α responses were inconsistent with this notion (Figure 3B).
In addition to TNF-α we also measured the IL-10 release of cells stimulated with LPS and Api88 (100 µg/ml) for 16 h. IL-10 is a potent anti-inflammatory cytokine with numerous immunomodulatory effects, including the inhibition of TNF-α production. As seen in Figure 3C, treatment with Api88 led to a significant increase in IL-10 production, indicating that Api88 did not inhibit cytokine production in general.
When replacing LPS by Pam3CysSK4, another activator of monocytes, no inhibitory effect of Api88 on TNF-α production was observed. Compared with TNF-α concentrations measured 4 h after incubation with Pam3CysSK4 (100 ng/ml) alone (control = 100%) the values even slightly rose to 129 ± 20% (mean ± SD; n = 3) when the cells were incubated in the presence of Pam3CysSK4 (100 ng/ml) and Api88 (100 µg/ml). Thus, it seems unlikely that Api88 interferes with biochemical events common to different monocyte activators.
To compare the anti-endotoxin activity of Api88 with that of cathelicidin LL-37, another antimicrobial peptide that has been described to inhibit TNF-α release from human monocytic cells,13,14 monocytes were incubated with LPS and increasing concentrations of LL-37 before screening for TNF-α production (Figure 3D). Human LL-37 suppressed the LPS-induced TNF-α production in a dose dependent manner. At 10 µg/ml (0.56 µM) the release was decreased by more than 60% and at 25 µg/ml (1.39 µM) to 50 µg/ml (2.78 µM) by ≥80%. Considering that about 50 µg/ml (21.8 µM) Api88 was needed to induce TNF-α inhibition it becomes obvious that Api88 is less efficient than LL-37 in suppressing the TNF-α release.
Effect of Api88 on phagocytosis
An important function of monocytes consists in taking up and destroying microorganisms. To determine the role of Api88 in this process we pre-incubated the cells with Api88 or LL-37 for 4 h before measuring the uptake of fluorescence-labelled E. coli particles by FACS analysis. As seen in Figure 4A, when calculating the amount of phagocytosed particles expressed as median fluorescence, Api88 had no influence on the uptake of the particles. Unlike Api88, LL-37 proved to be a potent activator of phagocytosis.
Effect of Api88 on phagocytosis, chemotaxis and ROS-production. (A) Monocytes (2 × 106/ml) were incubated in the presence and absence of Api88 (100 µg/ml) or LL-37 (50 µg/ml) for 4 h before FITC-labeled E. coli particles (2 × 106/ml) were added for 3 h. The uptake of particles (median) was measured by flow cytometry. Data shown are the mean ± SEM of three independent experiments; Student’s t-test calculated to the medium control (=100%; white bar). (B) 5 × 104 cells/well were applied to the upper chamber of a 48-well microchemotaxis chamber (NeuroProbe). Following a 1.5 h incubation migration was assessed by counting the number of migrated cells through the polycarbonate membrane. Migration is expressed as migration index defined as n-fold increase in cell migration in the presence of medium alone. Each bar shows the mean ± SD of three experiments. (C) Monocytes (2 × 106/ml) were treated either simultaneously with LPS (100 ng/ml) and apidaecin derivatives (100 µg/ml), 30 min with apidaecin derivatives before addition of LPS or in reversed order. Results are presented as area under the curve. Data shown are the mean ± SEM of at least three independent experiments; Student’s t-test calculated to the control (no Api88) (=100%; white bar).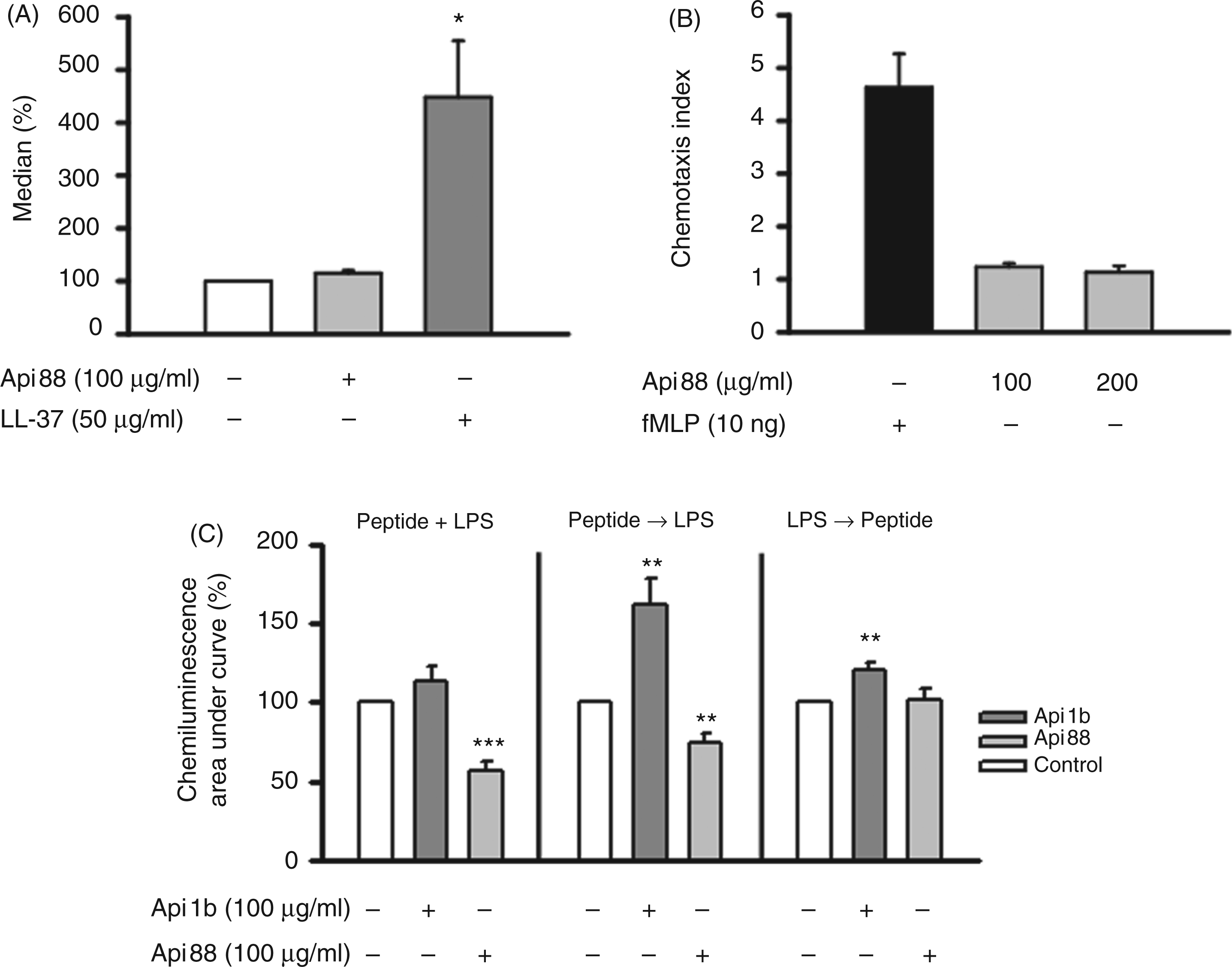
Effect of Api88 on chemotactic activity
An additional function of some AMPs lies in their ability to recruit effector cells, such as neutrophils, monocytes, macrophages, immature dendritic cells (DC) and T cells, to inflammatory sites (for review see Mookherjee and Hancock 12 ).
To test whether Api88 displays chemotactic activity the Boyden chamber assay was used. As shown in Figure 4B there was no increase in migration of monocytes towards Api88 whereas fMLP used as positive control did exhibit chemotactic activity.
Effect of Api88 on LPS-induced ROS production
Generation of ROS is a major microbicidal mechanism employed by monocytes and macrophages. The enzyme involved in the production of ROS is NAD(P)H oxidase, a membrane-located multi-subunit oxidoreductase complex. As seen in Figure 4C, when adding LPS and Api88 simultaneously to the cells, ROS production is inhibited. Pre-incubating the cells with Api88 before the addition of LPS also resulted in reduced ROS production. However, when incubating the cells for 30 min in the presence of LPS prior to the addition of Api88 the effect was abolished. In contrast to Api88, Api1b did not inhibit the LPS-induced release of ROS, but rather increased its production. Api88 by itself had no effect on ROS production (data not shown).
Api88 activates human primary lung MC degranulation
Human MC are able to respond to several AMPs by the release of granule constituents, which is accompanied by changes of the intracellular free calcium concentration. To test whether Api88 displays similar properties, isolated lung MC were incubated with increasing concentrations of Api88 and Api rev, and cell degranulation was determined by measuring the enzymatic activity of β-hexosaminidase released into the supernatant. Compared with anti-IgE-Ab-treated cells, Api88 mediated a moderate, but dose-dependent, liberation of β-hexosaminidase (Figure 5A), which started to reach a significant level at a peptide concentration of 16.7 µg/ml. By contrast, no degranulation surmounting background levels was observed in Api rev-treated cells. MC degranulation induced by Api88 is accompanied by the mobilization of cytosolic free calcium. Whilst challenging cells with Api88 at concentrations of 20 µg/ml or 100 µg/ml resulted in a clear increase of intracellular calcium, only a minor effect was observed at 2 µg/ml Api88 or 100 µg/ml Api rev (Figure 5B). Taken together, these results provide clear evidence that apidaecin acts as a direct activator of human primary MC.
Activation of human lung mast cells (MC) by Api88. IgE-Ab-loaded MC (2.5 × 104/ml) were left untreated (white bar) or stimulated with anti-IgE-Ab (1 µg/ml; black bar), and increasing concentrations of Api88 (grey bars) or Api rev (hatched grey bar). After 30 min, cell-free supernatants were recovered and tested for β-hexosaminidase enzyme activity (A). Data shown are the mean ± SD of four independent experiments; Mann–Whitney rank sum test calculated to the control (untreated sample). Alternatively, mast cells (1 × 105/ml) were loaded with 2 µM fluo-4-AM for 30 min and subsequently challenged with Api88, Api88 rev or anti-IgE-Ab, and [Ca2+]i was analyzed by flow cytometry at an excitation wavelength of 488 nm and an emission wavelength of 525 nm (B). Data from one representative experiment out of three are given.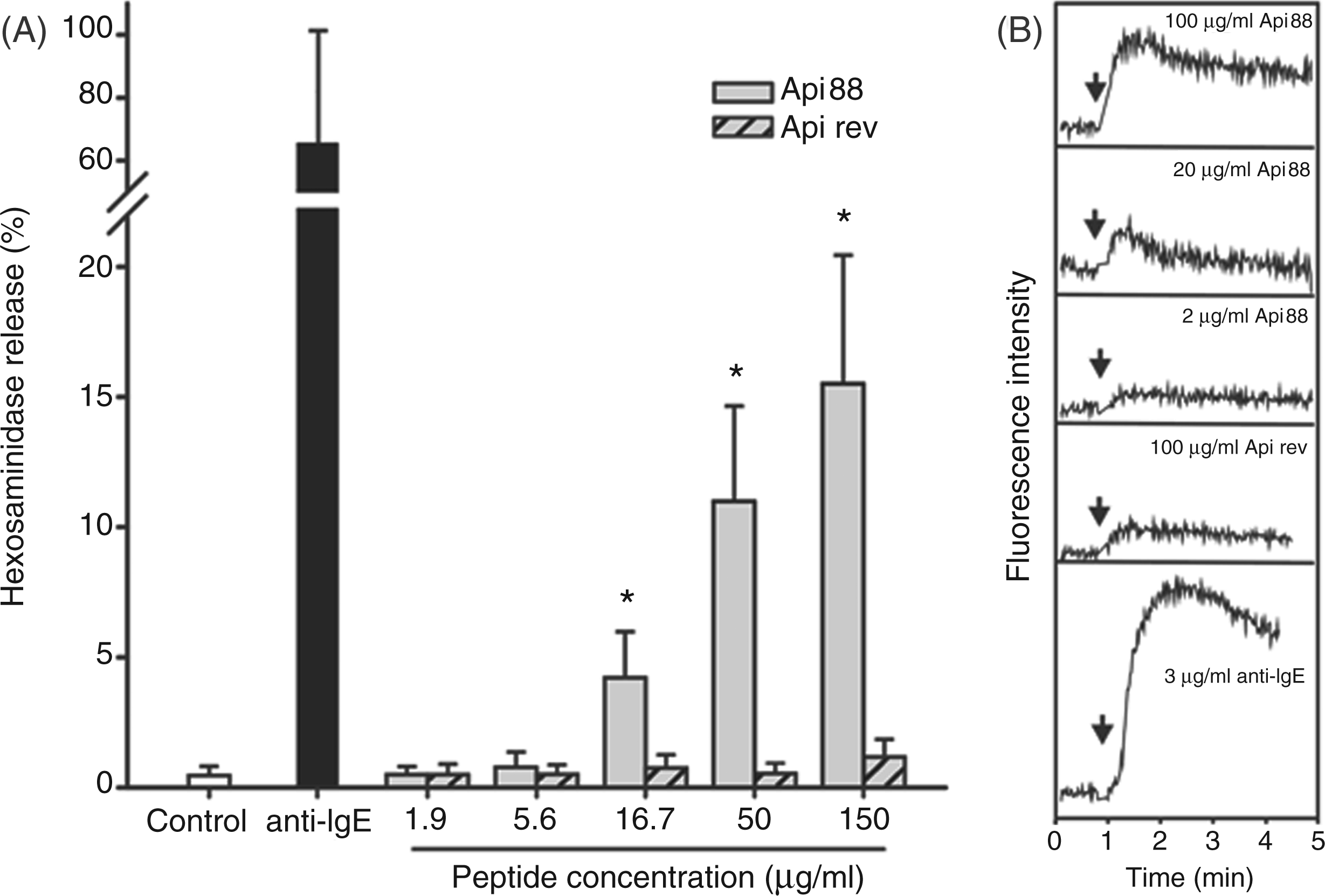
However, when incubating MC (5 × 104/ml) with increasing concentrations of Api88 (16.5, 50, 150 µg/ml) for 4 and 16 h no TNF-α was detectable in supernatants (data not shown), indicating that Api88 does not generally activate human MC.
Discussion
The increasing number of resistance mechanisms in pathogens and the continuous rise of bacterial strains resistant to several antibiotic drug classes pose significant challenges for both nosocomial and community-acquired infections. The annual death rates for nosocomial infections in the USA and the European Union, for example, are estimated to exceed 50,000. 35
One promising approach in treating bacterial infections by novel mechanisms relies on AMPs that can break current resistances by inhibiting novel drugable targets. The designer peptide Api88 is such a promising lead compound besides A3-APO, Bac7 and oncocin Onc72.26,27,36 All these proline-rich AMPs enter the bacteria without any lytic activity on the bacterial membranes and block the intracellular DnaK most likely at the substrate-binding domain. 27 Although these AMPs are less active than small molecule antibiotics in vitro, especially at high salt concentrations, they are highly efficient in murine infection models. This apparent contradiction was explained by additional immune modulatory effects in the animals, although some of the above-mentioned AMPs or their ancestors were isolated from evolutionary distant species, such as insects.
When investigating the potential immunomodulatory properties of native apidaecin 1b and Api88 in human monocytes, we found that Api88 was able to reduce the production of TNF-α—a cytokine that can cause life-threatening shock when released in massive amounts. Recently, and parallel to this study, Tavano et al. found that apidaecin 1b decreased TNF-α production in human monocytes when used at a concentration of 25 µg/ml. 37 In our hands, however, apidaecin 1b did not induce a reproducible inhibition at any concentration tested up to 100 µg/ml. Similar to Api88, the human AMP cathelicidin LL-3713,14 has been reported to selectively suppress the LPS-induced TNF-α release from human monocytes and macrophages and in a mouse/rat endotoxemia model treatment with the insect-derived AMP CEMA 38 or with cathelicidin LL-3715,39 it resulted in a decreased TNF-α production.
Thus, Api88, in line with other AMPs, has the potential to suppress potent and harmful pro-inflammatory responses. The anti-endotoxin properties of cationic host defense peptides have, in part, been attributed to their ability to bind and neutralize LPS.40–43 However, conflicting results are reported concerning the LPS-binding capacity of apidaecin. Whilst Otvos et al. showed that apidaecin could bind to LPS, 44 other groups found that LPS binding was weak or absent.37,45
The data presented herein showing that the inhibitory effect of 100 µg/ml Api88 was abolished at lower LPS concentrations strengthens the assumption that the immune mechanisms of Api88 cannot be explained by mere binding to LPS. Apart from scavenging LPS, other AMPs, such as the cathelicidin LL-37, have been demonstrated to inhibit the LPS-mediated pro-inflammatory response by suppressing the LPS-induced translocation of NFκB subunits to the nucleus.13,14
As Api88 did not inhibit TNF-α production induced by Pam3CysSK4, another monocyte activator that, unlike LPS, is recognized by TLR2 and not by TLR4, one could speculate that Api88 specifically interferes with membrane and/or signalling events following the interaction of LPS with TLR4. Alternatively, Api88 might exert its effect on TLR4-mediated TNF-α production via the inhibition of negative regulators of LPS signalling.
The finding that Api88 reduced TNF-α but enhanced IL-10 production also points to a selective effect of Api88 on LPS-induced cytokine production. IL-10 is a pleiotropic molecule that inhibits the production of pro-inflammatory cytokines, including TNF-α and IL-1β, by activated monocytes/macrophages thereby protecting the host from overwhelming inflammatory responses. 46
In further experiments we asked in how far Api88 can modulate three important functions of monocytes, namely phagocytosis, chemotaxis and microbicidal activity. Whilst Api88 neither interfered with the uptake of E. coli nor served as a chemoattractant for monocytes, it modulated the generation of ROS.
A major antimicrobial mechanism of activated monocytes is the production and intracellular release of ROS. This process, known as respiratory burst, is mediated by the activity of NAD(P)H oxidase, which catalyzes the reduction of molecular oxygen to superoxide anion—a reactive oxygen intermediate that generates other powerful oxidizing agents, such as hydrogen peroxide (H2O2) and hydroxyl radical (OH-). ROS are essential components of early host defense, although excess release of ROS can also cause significant tissue injury during inflammation. Here, we show that Api88 inhibits ROS production when added to monocytes prior to, or together with, LPS. ROS production was not influenced after initiating the oxidative burst, indicating that a direct effect of Api88 on NAD(P)H oxidase once it is activated is unlikely. Api88 could act in a similar way to the porcine cathelicidin peptide PR-39, which has been reported to inhibit NAD(P)H oxidase by blocking assembly via interaction with SH3 domains, which are present in cytosolic subunit p47phox and are responsible for its interaction with the membrane component p22phox. 47
Interestingly, the wild type peptide Api1b, which only differed from Api88 by two amino acids, failed to inhibit ROS production and, when applied either before or after exposure to LPS, even led to an increase in the generation of reactive oxygen species.
As Api1b does not stimulate ROS production by itself (data not shown) one might speculate that in combination with LPS it facilitates NAD(P)H oxidase assembly either by membrane perturbations or by biochemical modifications of the enzyme complex and thus enhances ROS release.
In addition to its capacity to modulate distinct biological responses in human monocytes, Api88 can also regulate human MC functions. According to our data, it induces degranulation and the mobilization of intracellular free Ca2 + in these cells. Interestingly, different to an acute MC activation, Api88 was incapable of inducing a relevant release of TNF-α. Human MC also fail to produce TNF-α in response to LPS under standard culture conditions. In contrast to murine MC, human MC express no, or very low levels of TLR4 and no CD14 on their surface. A response to LPS can only be induced under specific culture conditions and requires an appropriate prestimulation (e.g. with IL-4 or IFN-γ) and the presence of soluble CD14 (own observations).48–50
The activation of MC by host-derived AMPs has been the focus of many recent studies. MC respond to α-defensins (human neutrophil peptides) secreted by neutrophils as well as β-defensins (hBDs) produced by epithelial cells and other cell types by degranulation, cytokine production, prostaglandin D2 synthesis and an increase in vascular permeability.51,52 Moreover, we, and others, could show that cathelicidin LL-37 acts as a potent activator of rat and human MC.31,53 Specific binding molecules involved in AMP-induced MC activation are under intensive investigations. The fast and reversible mobilization of intracellular calcium, as well as the susceptibility of many AMP-mediated functions to pertussis toxin, argue for a participation of G protein-coupled receptors in these processes. Utilization of such receptors has already been described for different cell types. hBDs activate DC and T cells via binding to CCR6, whereas activation on monocytes, as well as on macrophages, is mediated by CCR2.54,55 Furthermore, the cathelicidin LL-37 induces chemotaxis in neutrophils, monocytes and T cells through formyl peptide receptor-like 1 (FPRL1), 56 while MC respond to this AMP by their interaction with the Mas-related gene X2 (MrgX2), a novel G protein-coupled receptor. 57 Recently, and in line with our findings for apidaecin, xenobiotic AMPs derived from different species like frogs (e.g. esculitins, magainins)58,59 or wasps (e.g. eumenitin) 60 have also been shown to activate rat MC in vitro. Owing to their low immune stimulatory properties in humans, xenobiotic AMPs are highly interesting basic compounds for the development of novel types of antibiotics. To prevent potential unwanted side effects like anaphylactic reactions, a fundamental challenge will be the optimization of these peptides to generate components of low MC-stimulating activity.
Taken together, our data, derived from in vitro studies, clearly demonstrate that apidaecin peptides, in addition to their antimicrobial activities, have various effects on cells of the innate immunity. However, elucidation of their biological importance as immune regulators and their potential use as therapeutic agents awaits further in vivo studies.
Footnotes
Funding
This work was funded by the European Union (EFRE) and the Free State of Saxony.
