Abstract
This was a prospective case–control study that measured the prevalence of Chlamydia trachomatis (CT), Neisseria gonorrhoeae (NG) and Mycoplasma genitalium (MG) by an IVD CE multiplex PCR kit in fresh Fallopian tubes (FT) obtained from 96 ectopic pregnancies (EP) and 61 controls in the midluteal phase of the cycle. We later measured the expression profile of IL-6, leukaemia inhibitory factor (LIF) and their signalling molecules, in respect to the type and number of infections, by immunohistochemistry, ELISA and quantitative RT-PCR. The frequencies of CT, and MG mono- and co-infections were significantly higher in EP. IL-6, LIF, their receptors and intracellular mediators were significantly up-regulated at the gene and protein levels in positive compared with negative FTs within each group (P < 0.05). EP tubal samples with co-infections showed the highest significant expression of the candidate cytokines by all techniques (P < 0.05). CT and MG are frequent in EP and up-regulate the tubal expression of IL-6, LIF and their signalling molecules. Both cytokines could be involved in the tubal immune response against bacterial infections, as well as the pathogenesis of EP. Further studies are needed to explore the roles of IL-6 family in infection-induced tubal inflammation and EP.
Keywords
Introduction
Ectopic pregnancy (EP) is a leading cause of maternal morbidity and mortality during the first trimester, representing 1–2% of all pregnancies and the majority (98%) occur at the ampullary region of the Fallopian tube (FT).1,2 The exact pathophysiological mechanisms underlying tubal gestation are still largely unknown. 3 However, upper genital tract infection and the subsequent development of salpingitis are well-established risks for EP. 4
Chlamydia trachomatis (CT) is the most common sexually transmitted bacteria and is associated with major reproductive sequelae in women, including pelvic inflammatory disease (PID), infertility and EP.4,5 Other bacterial infections, including Neisseria gonorrhoeae (NG) and Mycoplasma genitalium (MG), have also been suggested as potential risk factors for EP as several studies detected the microorganisms from women with PID and the prevalence was significantly higher than controls.6,7
Infection-induced tubal damage is believed to create a pathological tubal microenvironment characterised by an abnormal upregulation of several cytokines that may induce tubal receptivity, defective embryo transportation and, consequently, EP.3,8 IL-6 and leukaemia inhibitory factor (LIF) are plethoric cytokines that belong to the IL-6 family. These cytokines bind to their individual receptor and later propagate their signals by forming a complex with the glycoprotein (gp)-130 to trigger downstream signalling cascades, including the Janus tyrosine kinase/signal transducer and activator of transcription (STAT) and extracellular signal-regulated kinase pathways. 9
IL-6 and LIF are involved in the regulation of host innate and humoral immune responses and have been shown to be up-regulated by a variety of sexually transmitted organisms.10–15 Furthermore, the IL-6 family members and their signalling molecules are secreted by the endometrial and tubal cells of several species and have been described as major regulators of endometrial receptivity and blastocyst implantation.16,17 Nevertheless, the available studies on the role of the IL-6 cytokine family members in the pathophysiology of EP are few, their results are conflicting and none of them measured the effect of tubal infection on the production of these cytokines by tubal tissues,8,18,19 except for LIF and its receptor. 12
We have previously reported significantly higher rates of CT and MG by an IVD CE TaqMan PCR kit (Fast-track diagnostics, Junglinster, Luxembourg) in tubes bearing an EP compared with controls. 20 We therefore hypothesise that tubal infection with CT and/or MG may predispose to EP by modulating the expression of IL-6 and LIF within the tube. The present study therefore measured the expression of IL-6, LIF, their corresponding receptors and intracellular mediators (gp130 and STAT3) by infected tubal tissues collected from EP during salpingectomy and at the midluteal phase of the menstrual cycle during total abdominal hysterectomy (TAH) and the results were compared with tubal specimens collected from the same groups and were negative for the pathogens. Understanding the interactions between upper genital tract infection and IL-6 family in the pathogenesis of EP could provide a better tool for the prevention of the adverse reproductive outcomes associated with these microorganisms and/or the early diagnosis/treatment of EP.
Materials and methods
Ethical approval
Ethical approval was obtained from the Ethics Committee of the Faculty of Applied Medical Sciences (AMSEC 10-15-9-2011), Umm Al-Qura University. All tubal samples were collected following obtaining informed written consent from all the study participants.
Study design
This was a prospective case–control study and fresh FT specimens were collected from 96 patients diagnosed with tubal EP and another 61 women at the midluteal phase of the cycle during TAH. The participants were recruited from two Maternity and Children Hospitals (Jeddah and Makkah), and Al-Thager General Hospital (Jeddah) between January 2012 and February 2016 according to previously described inclusion criteria. 20
The diagnosis of EP was made either by transvaginal ultrasound or laparoscopy and salpingectomy was performed on clinical management grounds. The mean age for women with EP was 33.8 ± 8.1 yr (range 26–38 yr) and the average estimated gestational age calculated from the date of admission minus the date of the last menstrual period was 7.5 ± 1.7 wk. The average serum β-human chorionic gonadotropin level was 2686 ± 2349 IU/l.
All women in the TAH group (mean age 39.3 ± 6.2 yr; range 34–42 yr) had regular menstrual cycles, were of proven fertility with no evidence of tubal disease and were not taking exogenous hormones for at least 3 mo prior to surgery. These women had serum progesterone > 25 nmol/l and endometrial biopsies were also immediately collected following TAH to confirm histologically the stage of the cycle, as previously described. 21 Briefly, the luteal phase was characterised by irregular glands in shape and size and full with secretion, lined by columnar epithelium and the stroma was oedematous with cellular decidualisation.
Sampling and processing
All specimens were collected and processed under sterilised conditions. FTs collected from EP were excised at least 1 cm away from the implantation site and the collected tubes from all groups were immediately cut using RNase/DNase-free into small pieces of 1 cm each with one piece from the ampulla being immediately fixed in 10% buffered formalin for immunohistochemistry and the remaining parts were stored at –80℃ in 10 ml RNALater solution (Ambion, Warrington, UK) until processed for DNA, RNA and protein extraction.
Nucleic acids extraction from FT tissues
DNA and total RNA were extracted from a random piece from the ampullary segment of each tubal specimen using AllPrep DNA/RNA Mini Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions and following homogenisation using tissue raptor and sterile plastic tubes with beads (Omni International, Kennesaw, GA, USA). The quality and quantity of extracted DNA and RNA were assessed on a BioSpec-nano machine (Shimadzu, Tokyo, Japan) and typically had an A260:A280 ratio of 1.7:1.9. Extracted DNA from all tubes were diluted to a final concentration of 50 ng/µl and aliquots of the diluted samples were stored at –20℃ until used. Total RNA samples were aliquoted in 20 ng/µl and stored at –80℃ until used for gene expression studies.
Multiplex-PCR for three sexually transmitted pathogens in tubal tissues
The performance of multiplex PCR amplification was done using the FTD Urethritis basic kit (Fast-track Diagnostics, Junglinster, Luxembourg), according to the manufacturer’s protocol, on an ABI 7500 platform from Applied Biosystems (Thermo Fisher Scientific, Warrington, UK). The used kit is IVD CE-certified and included sets of specific primers and TaqMan probes against the three microbes of interest (CT, NG and MG).
The detection limit for all pathogens is 103 copies/ml as reported in the manufacturer’s validation document. Murine cytomegalovirus was provided within the kit as internal control and was added during the extraction step to each FT sample and co-amplified with the target DNA from all specimens. Each PCR reaction consisted of 12.5 μl mastermix, 1 μl enzyme, 1.5 μl of primers and 10 μl (500 ng) DNA. The amplification protocol was 42℃ for 15 min hold, 94℃ for 3 min hold and 40 cycles (94℃ for 8 s and 60℃ for 34 s). For those samples that were positive for NG, the results were validated by the FTD Gonorrhoeae confirmation kit (Fast-track Diagnostics) to confirm the results as per the manufacturer’s instructions and as previously described. 20
The results were validated according to the manufacturer’s instructions using the enclosed positive and negative controls. We also performed an extra validation step for all negative samples to confirm the results by spiking them with the provided positive DNA controls at 4:1 ratio for a sample (8 µl) and positive control (2 µl) as previously described. 20
Immunohistochemistry
The primary Abs (Santa-Cruz Biotechnology, Burlingame, CA, USA) for the detection of IL-6, IL6Rα, LIFR, gp130 and STAT3 proteins were polyclonal rabbit IgG Abs, while goat polyclonal IgG Abs were used to detect LIF. An avidin–biotin HRP technique was applied to localise the molecules of interest using ImmunoCruz Goat or Rabbit LSAB Staining System (Santa-Cruz Biotechnology) and by following the manufacturer’s protocol. The concentration was 1:200 for the IL-6 and gp130 Abs, while for the remaining Abs a concentration of 1:150 was used. The negative control slides consisted of a section of the tissue block being studied, which was treated identically to all other slides, with the exception that the primary Abs were omitted and replaced with corresponding primary normal goat (sc-2028) or rabbit (sc-2027) IgG Abs (Santa-Cruz Biotechnology) to control for non-specific binding of the detection system.
The sections were observed on an EVOS XL Core microscope (Thermo Fisher Scientific, Warrington, UK) at magnifications of × 100, × 200 and × 400 to evaluate and score the immunostain. Each section was examined by two observers who were blinded to the source of tissue, and the intensity of staining was assessed in five random fields of each section at × 200 magnification and by using ‘H score’, which was calculated as follow:3,5 H score = ∑Pί (ί + 1), where ί represents the intensity of staining [(0) = negative; (1) = equivocal; (2–4) = weak; (5–7) = moderate; and (8–10) = strong] and Pί is the percentage of cells (0–100%) stained at each intensity. In the case of a wide disagreement between both observers, the slides were reanalysed by a third independent reviewer. The final results were calculated by averaging the individual observer readings.
ELISA
A random ampullary specimen of 100 mg per tube was homogenised in 3 ml RIPA lysis buffer containing protease inhibitors (Santa-Cruz Biotechnology) to extract total protein. All samples were centrifuged at 15,400 g for 30 min at 4℃ and small aliquots (500 μl) of the resultant supernatant were placed in sterile Eppendorf tubes and the concentrations of total proteins in tissue homogenates were measured using the BioSpec-nano at 280 OD. All protein samples were diluted to a final concentration of 250 µg/ml total protein using normal sterile saline.
The concentrations of IL-6 and LIF proteins in the tissue homogenates were measured by specific ELISA kits (R&D systems, Minneapolis, MN, USA) and all samples were processed according the manufacturers’ instructions on a fully automated ELISA system (Human Diagnostics, Wiesbaden, Germany). As reported by the manufacturer, the IL-6 kit had a detection range between 3.12 and 300 pg/ml, sensitivity 0.7 pg/ml, intra-assay precision 4.4% and inter-assay precision 3.7%. The LIF kit had a detection range between 31.2 and 2000 pg/ml, sensitivity of 8 pg/ml, intra-assay and inter-assay precisions of 5.4% and 3.9%, respectively.
Quantitative RT-PCR
Synthesis of cDNA was done by transcribing 200 ng total RNA using a high-capacity RNA-to-cDNA Reverse Transcription Kit (Thermo Fisher Scientific). PCR reactions were carried out in triplicate wells on ABI 7500 system using power SYBR Green master mix (Thermo Fisher Scientific). The PCR reaction for each well included 10 µl SYBR Green, 7 µl DNase/RNase free water, 1 µl from each primer (5 pmol) and 1 µl cDNA (25 ng). The amplification was performed under the following conditions: 40 cycles (95℃/15 s and 60℃/1 min). Two negative controls were included, one with minus-reverse transcription control from the previous reverse transcription step and a minus-template PCR, which contained all the PCR components but water was used as a template.
The sequences of PCR primers used for the detection of 18S, IL-6, LIF, their corresponding receptors (IL6RA and LIFR), gp130 and STAT3 genes in tubal samples, including the corresponding genes accession numbers.

Statistical analysis
SPSS version 16 (IBM, Armonk, NY, USA) was used for the statistical analysis of the results. χ2 or Fisher’s exact tests were used for frequency analysis and calculation of crude odds ratio (COR) with 95% confidence interval (CI) following cross-tabulation. Binary regression was also used to estimate the odds ratio (OR) and 95% CI of each microorganism, as well as single/multiple infection on the development of EP. One-way ANOVA followed by Tukey post hoc test or Kruskal–Wallis followed by Dunn’s post hoc test was used to compare differences between the different groups in the expression of IL-6, LIF, their corresponding receptors and intracellular mediators proteins and mRNAs depending on the data homogeneity. All tests performed were two-sided and P < 0.05 was considered significant.
Results
Rates of sexually transmitted pathogens
The participants were 96 (61.1%) females diagnosed with tubal EP and 61 control women (38.9%) in the midluteal phase of the cycle and who were recruited at the time of TAH. Histological dating of all endometrial samples obtained from the menstrual cycle group correlated with the date of last menstrual period reported by each participant (data not shown).
The three microbes were detected in tubal specimens from the study groups either as single or multi-infections. All wells that included the positive controls provided with the used kit showed appropriate signals and Ct value as per the manufacturer’s validation criteria. Additionally, there was no signal detected in the negative control wells. Negative tubal samples that were spiked with the provided DNA of the pathogens of interest also showed a positive signal reassuring the observed results of this study.
The total number of women positive for any of the three microorganisms of interest either as single or co-infections was 32 (20.4%) cases. The rates were 8.3% (n = 13) for single infection and 12.1% (n = 19) for co-infections. The co-infection group consisted of three positive cases for all bacteria and 16 patients positive for two pathogens. The rates of infection according to the type of bacteria were 17.8% (n = 28) for CT, 3.8% (n = 6) for NG and 12.7% (n = 20) for MG.
Distribution of positive cases of CT, NG and MG in EP and control (total abdominal hysterectomy) groups according to single and co-infections with OR and 95% CIs.
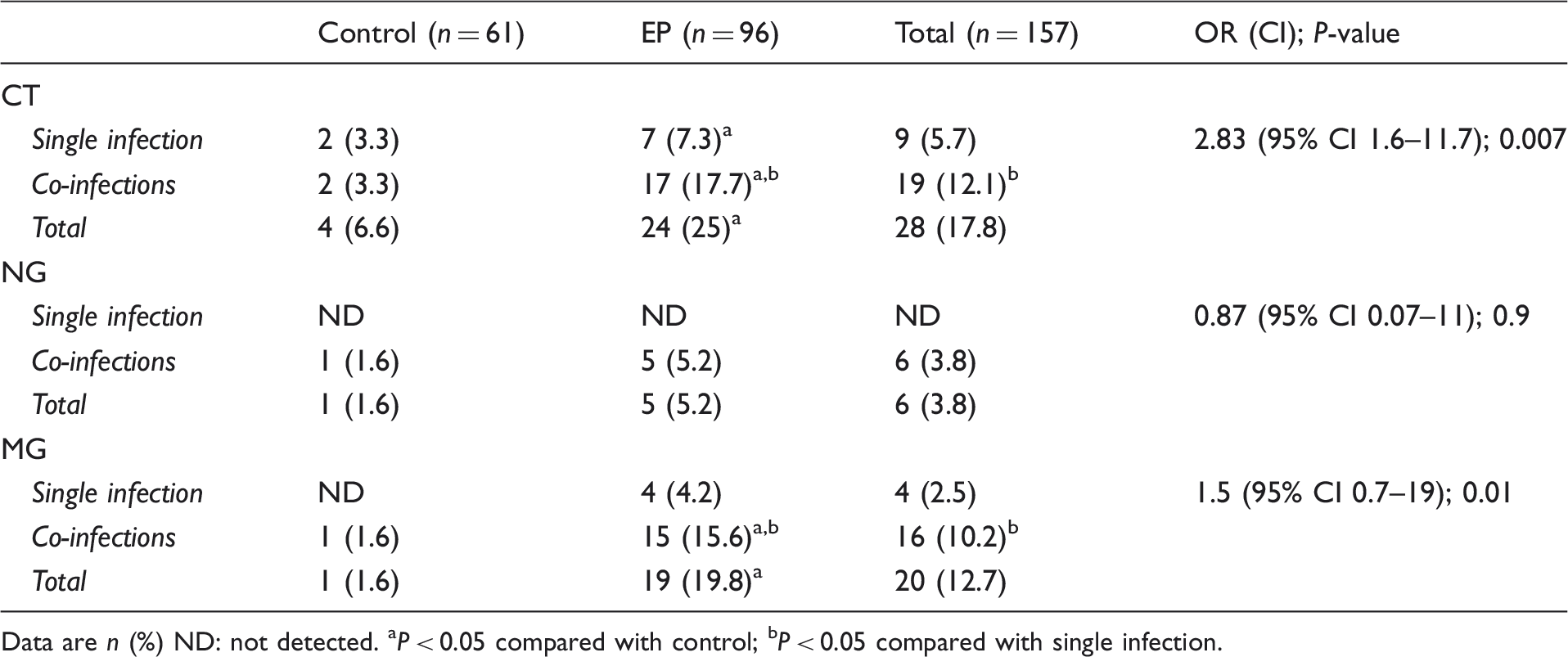
Data are n (%) ND: not detected. aP < 0.05 compared with control; bP < 0.05 compared with single infection.
The frequencies of monoinfection (11.5% vs. 3.3%; COR 3.5, 95% CI 0.8–15.2; P = 0.04) and co-infections (17.7% vs. 3.3%; COR 5.4, 95% CI 1.3–22.5; P = 0.006) were also significantly higher in the EP group compared with the TAH group. However, NG was not detected as a single infection in both the EP and TAH groups, while MG was only detected as monoinfection in the EP group (Table 2). Binary logistic regression showed that monoinfection (OR 4.6, 95% CI 0.98–21.6; P = 0.04) and multiple infections ( ≥ 2 pathogens; OR 7.1, 95% CI 1.6–32.1; P = 0.01) were associated with a significantly higher risk of EP (Table 2).
The TAH and EP groups were then categorised according to the results of PCR for further analysis of tubal expression of the candidate genes and proteins into negative-control TAH (NC; n = 57), positive control TAH (PC; n = 4), negative EP (N-EP; n = 68), CT-monoinfection EP (CT-EP; n = 7), MG-monoinfection EP (MG-EP; n = 4) and multi-infection EP (≥ 2 pathogens; M-EP; n = 17) groups.
Immunohistochemistry
Age, gestational age, serum β-human chorionic gonadotropin (hCG), immunohistochemistry scores for IL-6, LIF, their receptors and intracellular mediators in FT collected from infection-free (NC) TAH, TAH positive for ≥ 1 pathogen (PC), EP negative for the microbes (N-EP), EP with CT-EP or MG-EP mono-infection and EP positive for ≥ 2 microbes (M-EP) groups.
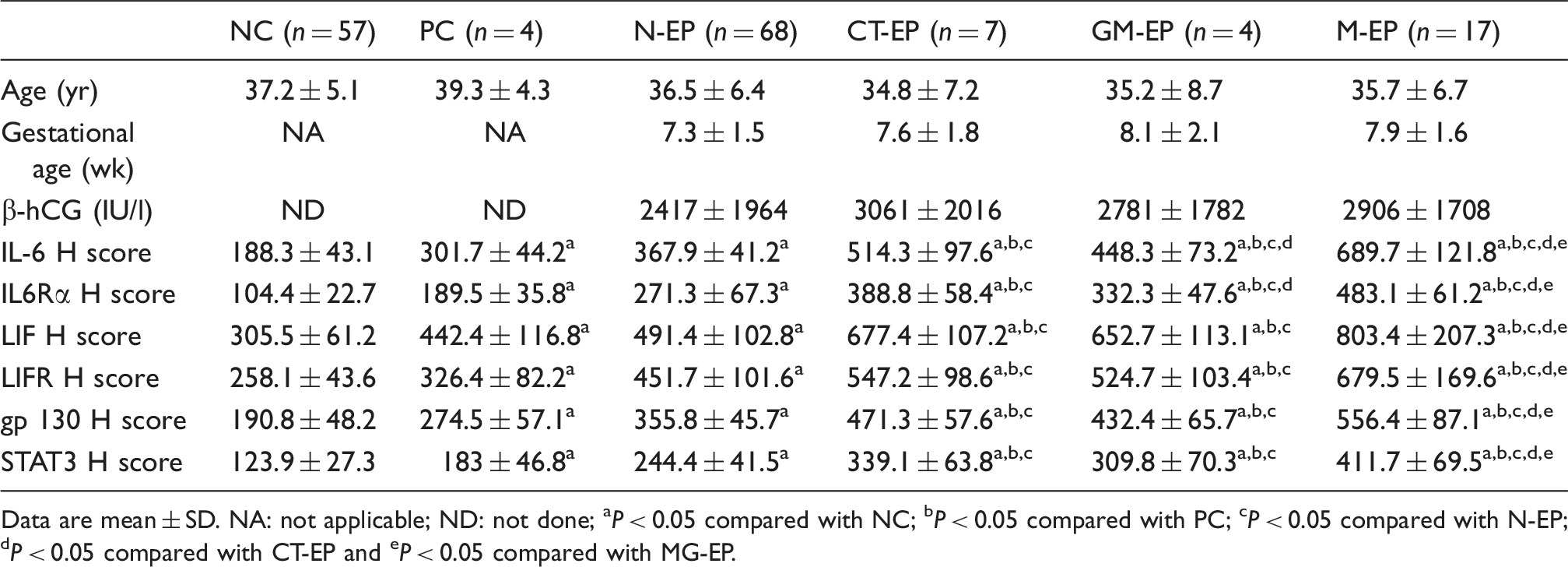
Data are mean ± SD. NA: not applicable; ND: not done; aP < 0.05 compared with NC; bP < 0.05 compared with PC; cP < 0.05 compared with N-EP; dP < 0.05 compared with CT-EP and eP < 0.05 compared with MG-EP.
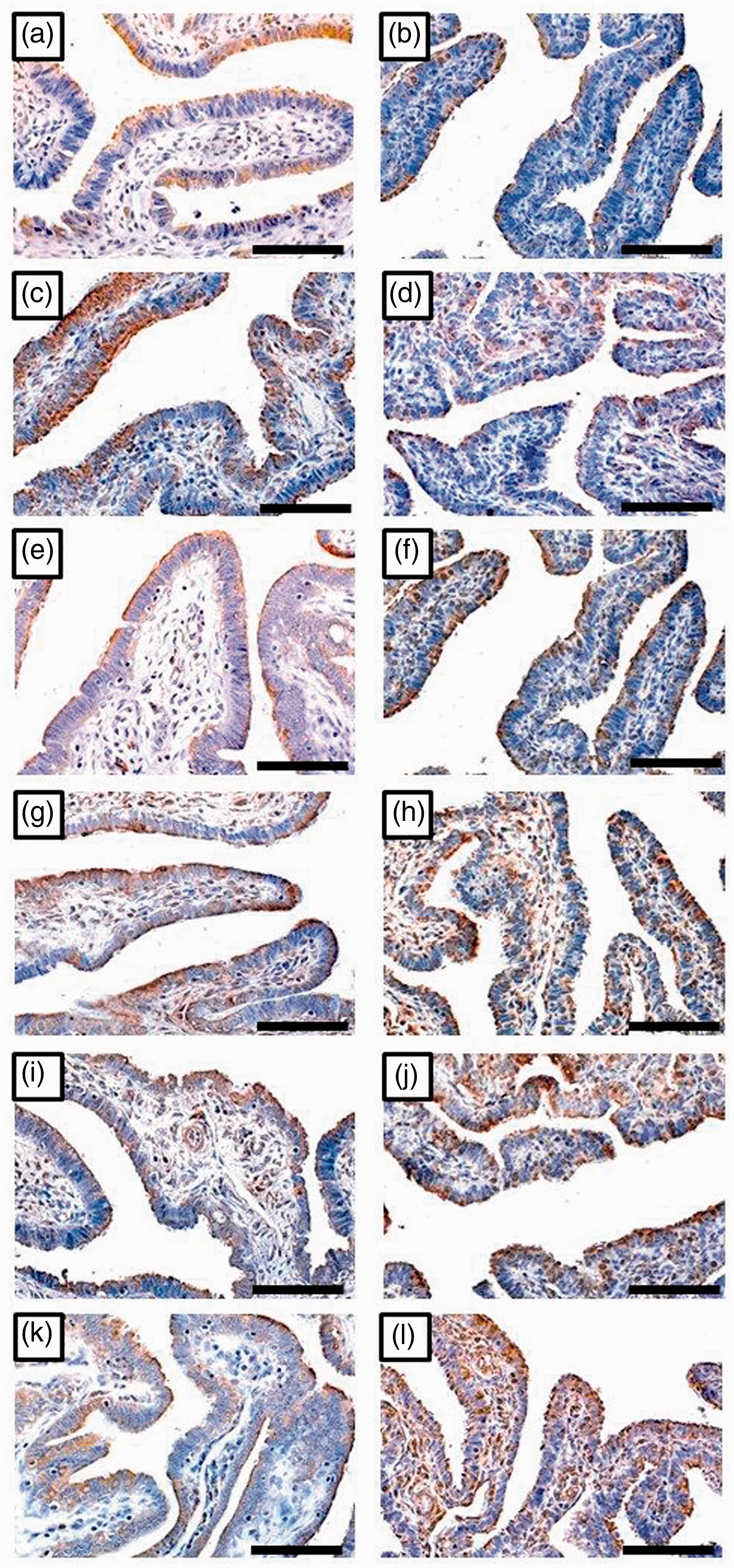
In the EP groups, there was also a significant increase in the immunostain of IL-6 (P = 0.005) and its receptor (P = 0.004) between CT-EP and MG-EP monoinfection groups (Figure 1g–j). However, there was no significant difference in the immunostain scores for LIF (P = 0.1), LIFR (P = 0.3) (Figure 2g–j), gp130 (P = 0.2) and STAT3 (P = 0.1) (Figure 3g–j) between the two monoinfection groups of EP (Table 3). The highest H scores for the target molecules were observed in the M-EP group and they were significantly higher compared with the other study groups (Table 3).
Immunohistochemistry localisation of IL-6 (left column) and IL6Rα (right column) in tubal samples collected from TAH and negative for infection (a, b; n = 57), TAH with ≥ 1 infection (c, d; n = 4), EP negative for infection (e, f; n = 68), EP with CT monoinfection (g, h; n = 7), EP with MG monoinfection (i, j; n = 4) and EP with ≥ 2 infections (k, l; n = 17) groups (×400 magnification, scale bar = 5 µm). Immunohistochemistry localisation of LIF (left column) and LIFR (right column) in tubal samples collected from TAH and negative for infection (a, b), TAH with ≥ 1 infection (c, b), EP negative for infection (e, f), EP with CT monoinfection (g, h), EP with MG monoinfection (i, j) and EP with ≥ 2 infections (k, l) groups ( × 400 magnification, scale bar = 5 µm). Immunohistochemistry localisation of gp130 (left column) and STAT3 (right column) in tubal samples collected from TAH and negative for infection (a, b), TAH with ≥1 infection (c, d), EP negative for infection (e, f), EP with CT monoinfection (g, h), EP with MG monoinfection (i, j) and EP with ≥ 2 infections (k, l) groups ( × 400 magnification, scale bar = 5 µm).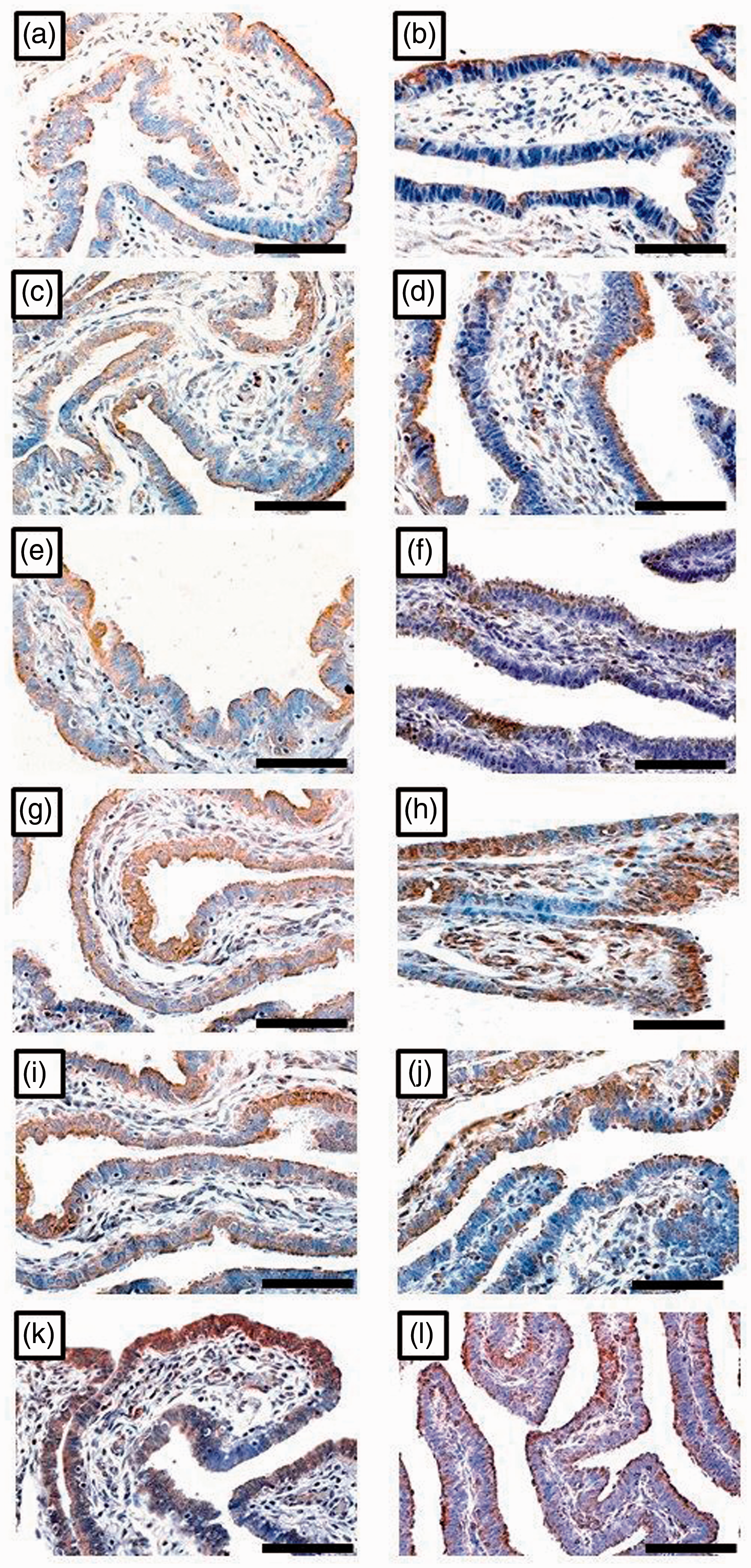
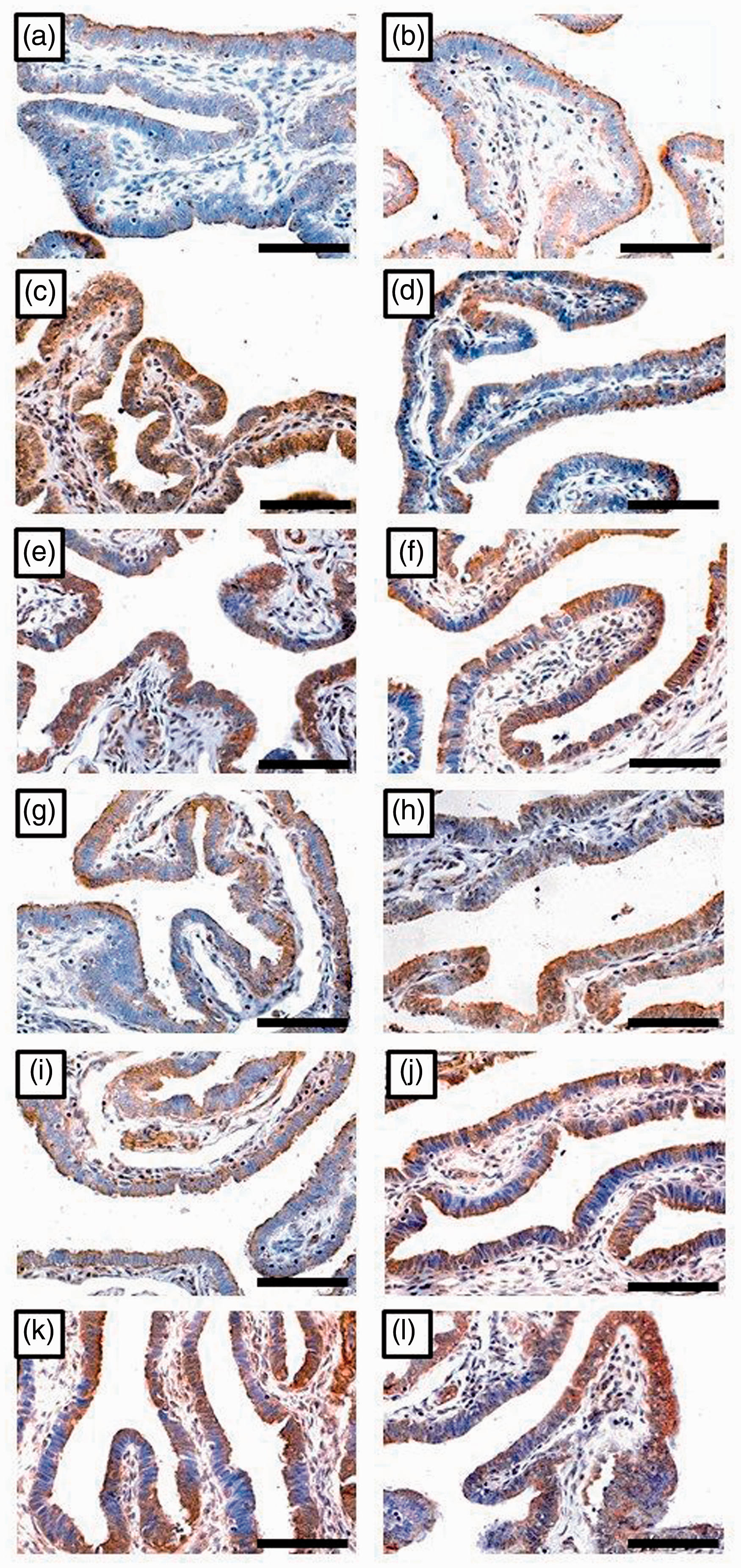
ELISA
Tubal specimens collected from TAH and were positive for an infection had significantly higher concentrations of IL-6 (P = 0.004) and LIF (P = 0.02) proteins compared with the NC group (see Figure 5). N-EP group also showed significantly higher concentrations for IL-6 (P = 0.009) and LIF (P = 0.03) compared with the NC group. Nevertheless, the tissue concentrations of both cytokines were comparable between the N-EP and PC groups (Figure 4). Similar to the results of immunohistochemistry, The MG-EP group showed a significantly lower concentrations of IL-6 (P = 0.006), but not LIF (P = 0.4), compared with the CT-EP group. Furthermore, the highest concentrations of all cytokines in tubal tissue homogenates were detected in the M-EP group compared with all other groups of the study (Figure 4).
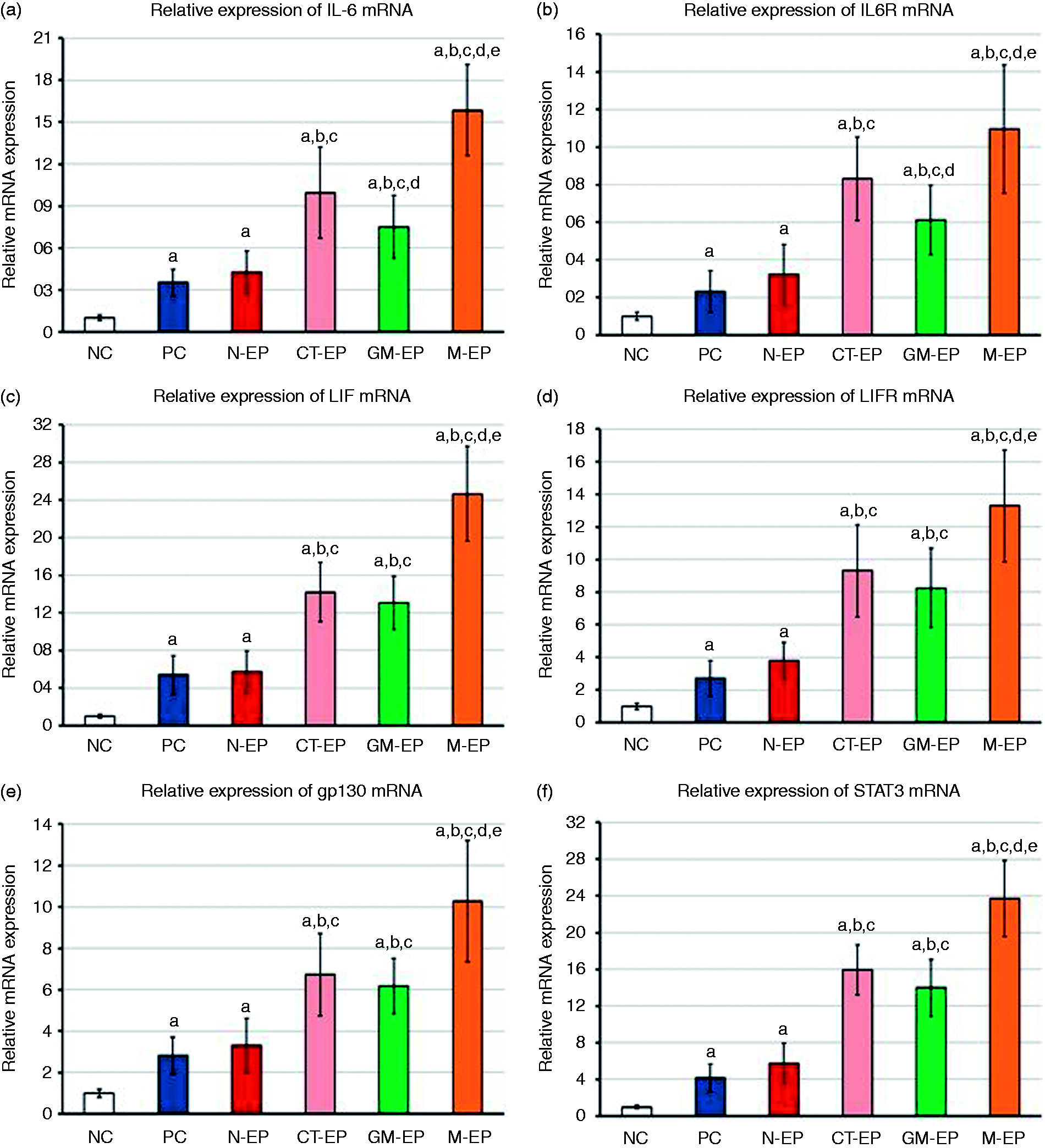
Mean ± SD of tissue homogenates concentrations of (a) IL-6 and (b) LIF in FT samples collected from TAH and negative for infection (NC), tube samples collected from TAH and positive for ≥ 1 infection (PC), EP negative for infection (N-EP), EP with CT monoinfection (CT-EP), EP with MG monoinfection (GM-EP) and EP concurrently positive for ≥ 2 infections (M-EP) groups (aP < 0.05 compared with NC group, bP < 0.05 compared with PC group, cP < 0.05 compared with N-EP group, dP < 0.05 compared with CT-EP group and eP < 0.05 compared with MG-EP group). Mean ± SD of mRNA relative expression of (a) IL-6, (b) IL6RA, (c) LIF, (d) LIFR, (e) gp130 and (f) STAT3 in FT samples collected from TAH and negative for infection (NC; n = 57), tube samples collected from TAH and positive for ≥ 1 infection (PC; n = 4), EP negative for infection (N-EP; n = 68), EP with CT monoinfection (CT-EP; n = 7), EP with MG monoinfection (GM-EP; n = 4) and EP concurrently positive for ≥ 2 infections (M-EP; n = 17) groups (aP < 0.05 compared with NC group, bP < 0.05 compared with PC group, cP < 0.05 compared with N-EP group, dP < 0.05 compared with N-EP group and eP < 0.05 compared with MG-EP group).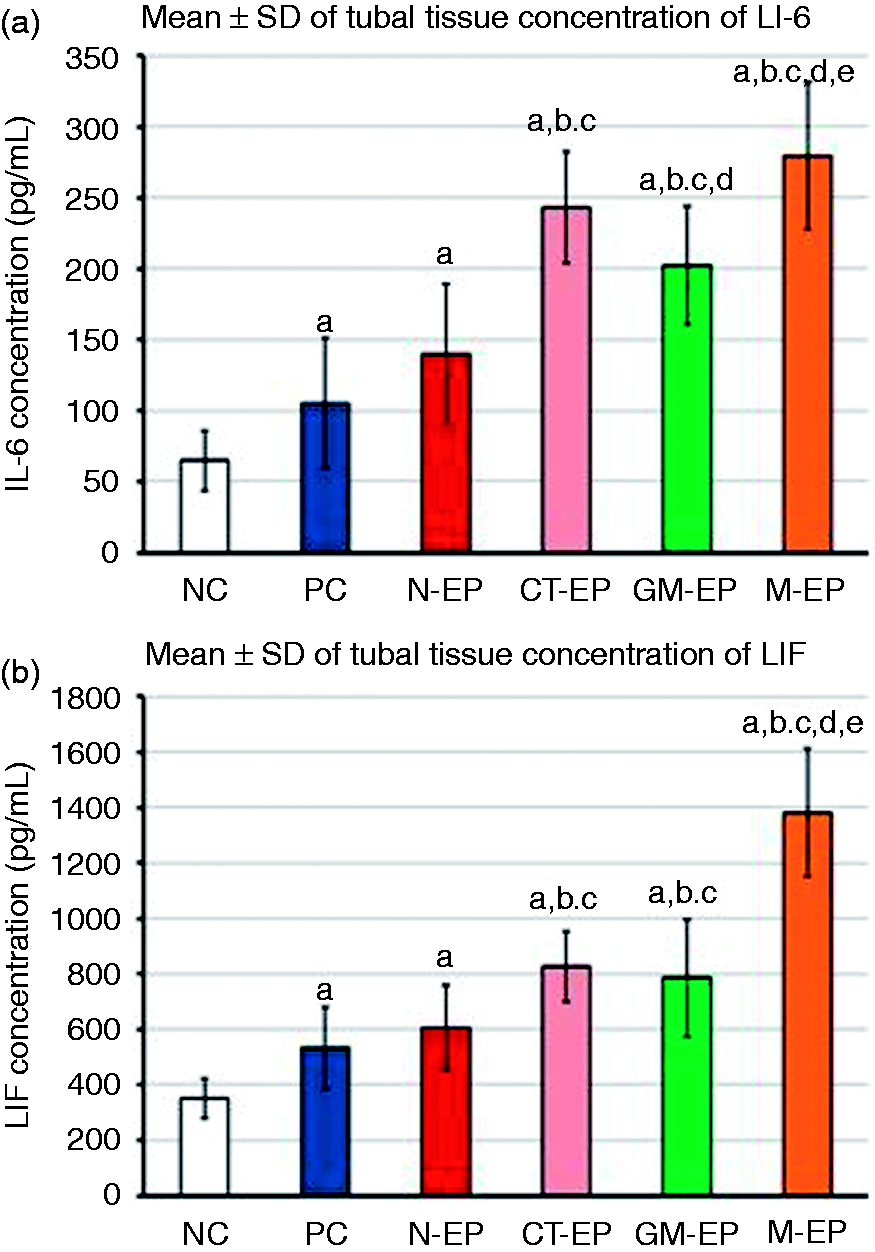
Quantitative RT-PCR
The relative expression of the mRNAs of IL-6 (P = 0.004), IL6RA (P = 0.006), LIF (P = 0.01), LIFR (P = 0.04), gp130 (P = 0.007) and STAT3 (P = 0.02) was significantly higher in the PC samples compared with NC group (Figure 5). Furthermore, a significant increase in the expression of IL-6 (P = 0.003), its receptor (P = 0.0001), LIF (P = 0.0002) and LIFR (P = 0.003), gp130 (P = 0.002) and STAT3 (P = 0.0006) mRNAs was also observed in the N-EP group compared with NC. No difference was observed in the mRNA expression of all molecules between the N-EP and PC groups (Figure 5). The CT-EP group had significantly higher expression of IL-6 (P = 0.003) and IL6RA (P = 0.01) compared with the MG-EP group. Both monoinfection groups also showed a significant increase in all molecules compared with the N-EP group (Figure 5). Similar to the protein results, the highest significant fold-increase for all molecules was detected in the M-EP group compared with all other study groups (Figure 5).
Discussion
Herein, our aim was to measure the effect of tubal infection with CT, NG and MG on the expression of IL-6, LIF and their intracellular signalling molecules by human endosalpinx. The rates of the pathogens of interest were measured by multiplex TaqMan PCR in FT specimens obtained from EP and the results were compared with control tubes collected at midluteal phase of the cycle during TAH. Our results showed that CT and MG, but not NG, infections were significantly more frequent in the EP group compared with control. Additionally, the majority of cases were multi-infections, which was associated with a significantly greater risk of developing EP.
The participants were then classified according to the type and number of detected microbes and the expression of IL-6, LIF and their intracellular signalling mediators was measured at the protein and gene levels and the results were compared between the TAH and EP groups. To the best of our knowledge, this is the first study to report an increase in the expression of several members of IL-6 family of cytokines and their signalling molecules within human FTs bearing an EP and that were concurrently positive for CT, NG and/or MG by PCR. All the studied molecules were also significantly increased in tubes obtained from negative EP samples compared with those non-infected tubes collected at the mid-luteal phase of the cycle. These observations were detected at the mRNA and protein levels, as confirmed by quantitative RT-PCR, immunohistochemistry and ELISA.
Our results are in agreement with the notion that the microbiological aetiology of PID and/or EP is a poly-microbial process. This study also suggests that tubal IL-6, LIF and their signalling molecules could be involved in the immune response and/or tubal damage induced by CT and MG bacterial infections of upper genital tract. Furthermore, the aberrant increase in the production of the cytokines of interest by the tubal epithelial cells could contribute to the pathogenesis of tubal pregnancy.
A history of PID increases the risk of EP by 7.5-fold,22–24 and 30% of women could develop PID and salpingitis subsequent to infection with CT. 25 Other studies have also reported an association between PID and EP with NG.26,27 Additionally, several other non-chlamydial/non-gonococcal pathogens have been detected in a variety of clinical specimens from female upper genital tract and have been suggested as potential contributors in the pathogenesis of EP. The link between MG infection and the development of PID, endometritis, tubal factor infertility and EP has been proposed by the serological and PCR findings of several studies that have shown significantly higher frequencies of MG infection in the case groups compared with controls.6,7,28,29
Studies from Saudi Arabia have also shown that almost 50% of women diagnosed with EP reported a history of PID and/or infertility.30–33 Unfortunately, discussing sexually transmitted diseases in Islamic nations, especially in Saudi Arabia, is a very sensitive topic and therefore little is known about the role of sexually transmitted microbes in the pathogenesis of EP. 34 However, the few available governmental reports from the kingdom have shown that the annual incidence of chlamydial and gonococcal infections between 1995 and 1999 were 14.8 and 5.2 per 100,000 population, respectively. 35 Furthermore, the majority of patients were Saudi females (92%) of reproductive age. 36
The present findings are in agreement and correlate with the abovementioned reports as they showed significantly higher rates of CT and MG in specimens obtained from EP. However, there was no significant difference in the frequencies of NG between EP and the control group. The present study suggests that CT and MG, but not NG, infections could be major contributors in the pathogenesis of EP in Saudi Arabia. Our findings also reinforce the recent demands to introduce a national screening programme for the detection of sexually transmitted infections in the kingdom.34,37–39 Additionally, the observed high rates of multi-infection in the current study is also aligned with the assumption that the microbiological aetiology of EP is multi-microbial that could benefit from applying syndromic approaches for the diagnosis and/or treatment.26,36,40
Unfortunately, the present study does not provide data on possible confounding causes that could have contributed in the acquisition of upper genital tract infection among our study population (e.g. number of sexual partners, socio-economic factors, etc.), mainly owing to the aforementioned social and culture barriers within the Saudi community regarding sexual behaviour and health.34,37,39 Therefore, more studies are compulsory to measure potential socio-demographic risk factors that could be associated with the acquisition of these microorganisms.
The members of the IL-6 family of cytokines are involved in the regulation of host innate and humoral immune responses against a variety of pathogens. In this regard, concentrations of IL-6,11,41,42 LIF12,18 and gp-130 43 increased significantly, both in vitro and in vivo, following infection with CT. The expression of IL-6 has also been shown to increase following infection of vaginal and cervical epithelial cells with MG in vitro.13,14,44 Similar observations have also been reported for the cytokine with NG infection.15,45,46 However, there are no currently published data on the role of LIF in the immune response against MG and NG.
Our results correlate with the previous reports as a significant increase was observed in the expression IL-6, LIF and their signalling molecules at the gene and protein levels in tubal samples collected from EP and TAH, and were positive for CT and/or MG compared with those tissues that were negative for the pathogens. However, there was no significant difference in the expression of LIF and its receptor between CT and MG monoinfection groups. These findings suggest that the cytokines of interest and their signal mediators are involved in the tubal immune response against CT and MG infections. It also appears that tubal infection with CT could have a more potent effect than MG on the endosalpingeal expression of IL-6 as there was a significant difference between the two monoinfection groups of EP. However, we were not able to confirm the EP results by comparing between both bacteria in the TAH group, owing to the small number of positive cases, and also there were no cases of MG monoinfection in non-pregnant women. Similarly, NG was only detected with co-infections. Therefore, additional studies with larger numbers of patients are needed to measure the effects of the three pathogens on the expression of IL-6 during upper genital tract infection.
Additionally, the highest expression for IL-6 and LIF was detected in the multi-infection group from EP. Hence, we postulate that tubal infection with CT and/or MG may induce upregulation of IL-6, LIF and their related molecules as part of the cellular immune response that can lead to tubal malfunction and subsequently EP. Proposed pathogenic mechanisms for EP include abnormal tubal transportation of gametes and/or increase in tubal receptivity for embryo implantation due to pathological alterations in tubal secretions following inflammation.3,4 In this context, the IL-6 family members are crucial regulators of endometrial receptivity and embryo implantation, and implantation failure has been associated with abnormal decrease in the expression of these cytokines by the endometrium.16,47,48
Nonetheless, the available data on their role(s) in the pathophysiology of EP are very limited and the results are controversial. The expression of IL-6, LIF and their signalling molecules have been localised in human FTs,49,50 and LIF is the most studied member of the IL-6 family in EP. Although a significant increase in the expression of both LIF18,51 and its receptor12,52 in tubes bearing an EP has been demonstrated, other studies also reported a non-significant increase in LIF protein in tubal gestation samples.12,53 Notably, all the studies applied only IHC for the detection of LIF protein and none of them measured the mRNA expression of the cytokine in the collected samples.
IL-6, which inhibits tubal ciliary beats in vitro, 54 was only investigated by a single study and has been shown to increase in tubal samples collected from EP and the highest expression was detected in tissues adjacent to the implantation site. 19 However, the authors failed to detect significant alterations in the protein expression of IL6Rα receptor. In contrast, a later report that only measured the mRNA, but not protein, expression of IL-6 receptor and gp130 in tissues collected from tubal gestation documented a significant increase in the receptor, as well as a significant decrease in gp130 mRNA. 8
The present study simultaneously measured the expression of several members of IL-6 family and their signalling components at both the protein and gene levels and showed a significant increase in the expression of all molecules in EP. A possible explanation for the discrepancies in the results of the cytokines of interest between the studies could be associated with the interpretation of immunohistochemistry results despite being a widely used technique in clinical and medical research. 55 An alternative explanation for the observed conflicting findings could be related to the role of IL-6 and LIF in the regulation of cellular immune responses. Remarkably, none of the earlier reports correlated their findings with possible tubal infection except that reported by Li et al., 12 who have shown a significant increase in the expression of LIF and its receptor in tubes with detected infection. Nevertheless, our study is the first to confirm the findings of IHC by alternative methods and the results from each technique were concurring. Additionally, the results of the cytokines were analysed with considerations of a variety of tubal infections.
In conclusion, infections with CT and MG are frequent in FTs bearing an EP and the majority of cases had co-infection. Both pathogens increase the tubal expression of IL-6, LIF and their related molecules which appear to play a role in the regulation of cellular immune response against tubal bacterial infections and/or could be involved in the pathogenesis of EP by promoting tubal receptivity and embryo implantation. Further in vitro and in vivo studies are compulsory to illustrate the roles of IL-6 family in the immune response against upper genital tract infection and the pathogenesis of EP.
Footnotes
Acknowledgment
The authors would like to thank Ms. Athar Khojah and Ms. Noha Al-Qadi from the Laboratory Medicine Department, Faculty of Applied Medical Sciences, Umm Al-Qura University, for performing the ELISA experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the National Science, Technology and Innovation Plan (MARRIFAH)—King Abdul Aziz City for Science and Technology (KACST), Kingdom of Saudi Arabia, Award Number (11-MED2067-10).
