Abstract
Endotoxin is considered a key signaling molecule in the pathogenesis of sepsis and septic shock. Anti-endotoxin therapies may result in the improvement of a patient’s clinical condition and lower mortality. The pressing clinical challenge is to identify patients for whom endotoxin elimination would be the most beneficial. An endotoxin activity assay (EAA) has been available for detection of endotoxins, allowing selection of patients at high risk of endotoxemia in intensive care units (ICUs). We studied a cohort of 172 consecutive patients who had septic shock on admission to the ICU. Endotoxin activity (EA) was measured with a rapid chemiluminescent EAA, regarded as point-of-care testing. Endotoxemia with a mean EA of 0.59 ± 0.14 EAU was present in 104 patients (60%) and absent in 68 patients (EA = 0.25 ± 0.11 EAU). The risk of endotoxemia increased with the presence of a Gram-negative infection [odds ratio (OR) 3.1, 95% confidence interval (CI) 1.6–5.9; P = 0.001] and bacteremia (OR 3.8, 95% CI 1.6–8.9; P = 0.02) but did not change with a diagnosis of peritonitis (OR 1.03, 95% CI 0.54–1.97; P = 0.90). These findings indicate that anti-endotoxin interventions should be tailored to individual patients based on both clinical conditions and measured endotoxin levels.
Introduction
Septic shock remains a predominant cause of death in intensive care units (ICUs). Endotoxin is a major component of the outer cell envelope of most Gram-negative bacteria and is considered a key signaling molecule in the pathogenesis of systemic infection, sepsis and septic shock. Among infectious triggers, endotoxin is identified as one of the most toxic PAMPs. Intravenous doses as low as 2 ng/kg can cause a systemic inflammatory response in humans with a change in expression of almost 4000 different genes. 1 Higher doses of endotoxin induce sepsis and septic shock, which has a fatal outcome in > 50% of cases.2,3 The higher the endotoxin level, the more severe clinical symptoms are observed. Dysfunction of vital organs and coagulation abnormalities often lead to the development of multiple organ failure and death. 4 Therefore, a therapy aimed at reducing endotoxin level may result in improvement in the clinical condition of patients and a reduced mortality rate. In patients with septic shock in the intensive care unit, anti-endotoxin drug therapies have failed to show significant clinical benefit,5,6 and results of the studies in which circulating endotoxins were eliminated using extracorporeal methods, such as adsorbent-based hemoperfusion or continuous renal replacement therapy, were inconsistent.7,8 However, there may be septic patient groups who would benefit from anti-endotoxin therapies. The pressing clinical challenge is to identify patients for whom endotoxin elimination treatment would be the most beneficial. In many studies the selection of patients for endotoxin elimination therapy was based on the clinical diagnosis or microbiological results, but not on the result of a specific diagnostic test which detected endotoxemia.9–11 Indeed, until recently there has not been a diagnostic assay to measure endotoxin levels in clinical samples, and this is one of the reasons that most previous attempts at anti-endotoxin therapy failed.10–12
The Limulus amebocyte lysate assay (LAL) has been extensively used in research, but the method has never obtained clearance for application with clinical samples owing to the interaction of the assay with different blood components. The utility of the assay to measure endotoxins in blood samples has been limited, owing to the presence of interfering factors in patients’ blood, both inactivators of endotoxin and inhibitors of LAL. 13 In addition, anticoagulants such as heparin, EDTA, and citrate disturb LAL measurement. 14 To overcome technical problems with the assay, extensive time-consuming sample preparation is required, including sample dilution, heat inactivation and centrifugation, and the time to obtain the final result is rather long. These limitations make it difficult to perform LAL-based endotoxin assays as a single measurement, suitable for rapid bedside analysis and prospective patient selection.
A new diagnostic assay, the endotoxin activity assay (EAA), which is available for clinical use, makes it possible to measure the ability of endotoxin to activate the immune system, and thus identify patients that are at high risk of endotoxemia and septic shock in ICUs. 4 This bioassay is based on neutrophil activation by endotoxin in blood samples; it is designed as a point-of-care test and results are available within 30 min.
The aim of this study was to assess the relationship between endotoxin levels measured with a new EAA on admission to the ICU, the clinical diagnosis and the results of microbiological tests in a group of septic shock patients. The unresolved issue is whether the selection of patients for anti-endotoxin therapy can be based solely on the clinical diagnosis.
Materials and methods
The study design was a retrospective, observational analysis of adult patients admitted to the medical–surgical Intensive Care Unit of University Hospital, Wroclaw, Poland, a 966-bed tertiary care center, from 2012 to 2015. We studied a cohort of 172 consecutive patients who had septic shock on admission to the ICU and for whom (1) endotoxin activity was measured and (2) appropriate culture samples were taken on admission to the ICU, according to the Surviving Sepsis Campaign guidelines. 15 Blood samples for endotoxin measurements were collected immediately after ICU admission, no later than 24 h from the diagnosis of septic shock. Septic shock was identified based on the criteria of the American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference as sepsis-induced hypotension (systolic blood pressure < 90 mmHg) that persists despite adequate fluid resuscitation and requires vasopressor support. 16 The control group consisted of 21 consecutive patients who had had a subarachnoid hemorrhage from a ruptured aneurysm (aSAH) and were admitted to the ICU. These critically ill patients suffered brain damage due to aSAH; none of the patients in the control group presented clinical or microbiological signs of infection upon entry to the study. The study protocol was reviewed and approved by the ethics committee of Wroclaw Medical University (KB-529/2015, KB-134/2014) and the need for informed consent was waived. EA measurements are performed routinely at our ICU for patients with a diagnosis of septic shock and for patients with a diagnosis of aSAH using point-of-care testing.
Data collection
Clinical and laboratory data for each patient were collected on the day of ICU admission. Patient clinical status was evaluated with the Acute Physiology and Chronic Health Evaluation II score (APACHE II) and the Sequential Organ Failure Assessment score (SOFA). Systemic inflammatory response parameters such as white blood cell (WBC) count and C-reactive protein (CRP) level were also recorded. As a routine ICU practice, a sample of whole blood and other specimens from sites of suspected infection were collected and sent simultaneously for microbiological testing on the day of admission. Survival status was assessed at discharge from the ICU.
EAA
Endotoxemia was identified with a commercially available, CE, IVD-marked diagnostic EAA (Spectral Diagnostics Inc., Toronto, Canada); the assay received FDA clearance in 2003 and European Regulatory Agency approval in 2004. The EAA is a rapid, in vitro diagnostic test that measures endotoxin activity in whole blood. The clinical samples for the endotoxin assay were collected using an arterial line to a tube (EDTA tube, Vacutainer systems; BD Bioscience, San Jose, CA, USA) within 24 h of ICU admission. Each sample was assayed immediately after collection. The procedure for measuring endotoxin is presented in Figure 1. The test is based on the reaction of endotoxin present in a patient’s blood sample with a monoclonal IgM anti-endotoxin Ab (raised against the lipid A of Escherichia coli J5) present in the reaction tube. A sample of the patient’s blood was incubated with the Ab and then stimulated with a zymosan. Oxygen radicals generated by primed neutrophils produced luminal chemiluminescence, and the signal was recorded with a luminometer (single tube luminometer Smart Line TL; Berthold Detection Systems GmbH, Pforzheim, Germany). Each assay was performed in three tubes: (1) a negative control tube containing blood and luminol–zymosan, (2) a reaction tube containing blood, anti-endotoxin Ab and luminol–zymosan and (3) a positive control containing blood stimulated with a maximum concentration of endotoxin (E. coli 055:B5), anti-endotoxin Ab and luminol–zymosan. The level of endotoxin activity (EA) was calculated by normalizing the chemiluminescence in the reaction sample against the maximum chemiluminescence (positive control); both measurements were corrected for the basal activity chemiluminescence (negative control) using the following formula:
The Endotoxin Activity Assay procedures performed as a point-of-care test.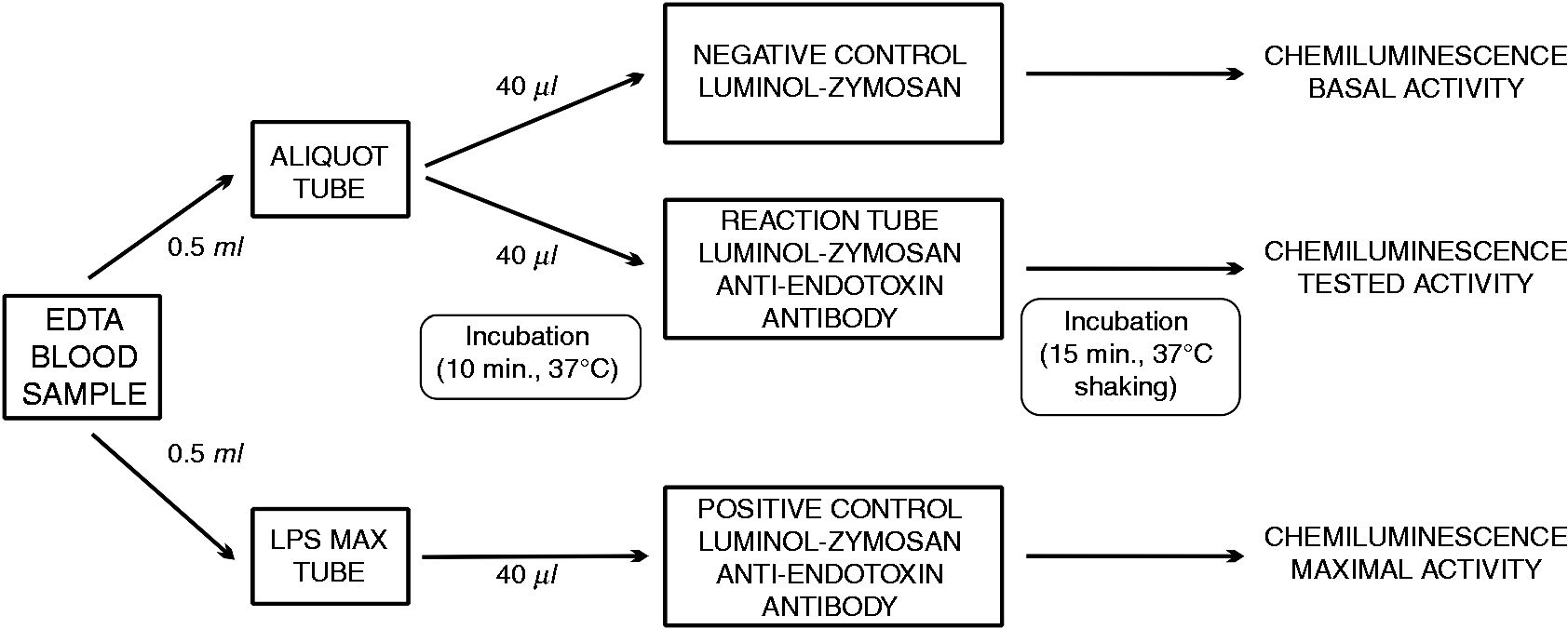
The results are quantitative, expressed in endotoxin activity units (EAU) on a scale from 0 to 1, and they represent the mean value of duplicate analysis from each blood sample. The intra-assay coefficient of variation was 11% and the inter-assay coefficient of variation was 12%. Based on the manufacturer’s information, the endotoxin activity level is considered to be low when it is < 0.4 EAU and a result ≥ 0.4 indicates endotoxemia.
Statistics
The data were analyzed with Statistica 10 (StatSoft, Inc., Tulsa, OK, USA). Continuous variables are presented as means with SD and minimum–maximum values. The Mann-Whitney U-test was used to compare continuous variables. Categorical variables were analyzed using a χ2 test. Kruskal–Wallis ANOVA by Ranks or two-way cross-tabulation table with the Pearson χ2 was used to test if a difference existed between multiple groups. The association between the presence of peritonitis, a Gram-negative infection or bacteremia and endotoxemia was assessed using logistic regression analysis with the likelihood ratio test and was reported as estimated odds ratio (OR) and a 95% confidence interval (CI). The association between the presence endotoxemia and a risk of death was assessed using logistic regression analysis with the likelihood ratio test and was reported as estimated OR and a 95% CI. Statistical significance was determined as P < 0.05.
Results
Characteristics of patients on admission to the intensive care unit.
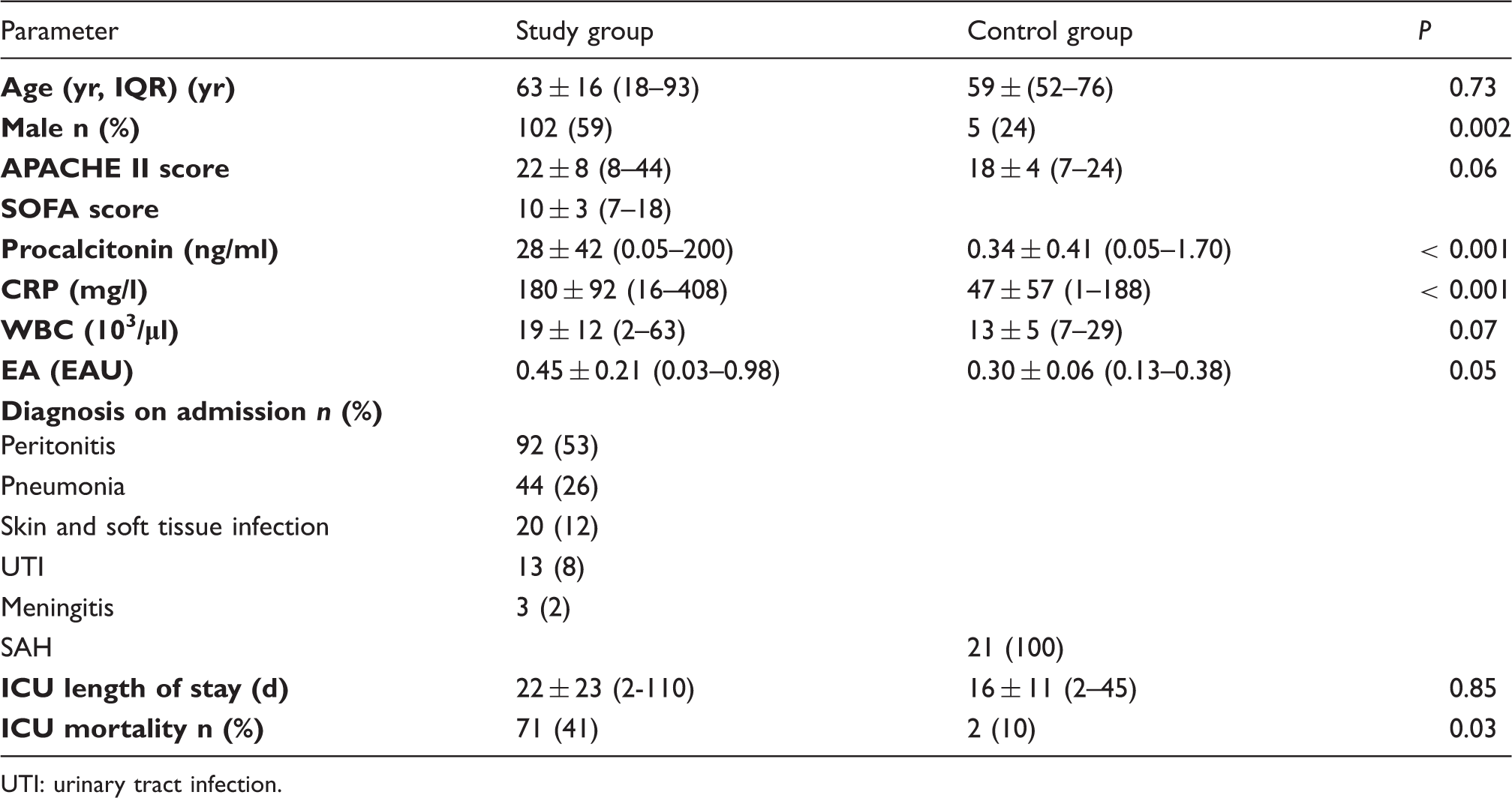
UTI: urinary tract infection.
Endotoxemia and the clinical diagnosis on admission to the ICU
EA in patients with and without peritonitis, with and without a Gram-negative infection, and with and without Gram-negative bacteremia.

EAU: endotoxin activity unit.
Endotoxemia and patient diagnosis on admission.
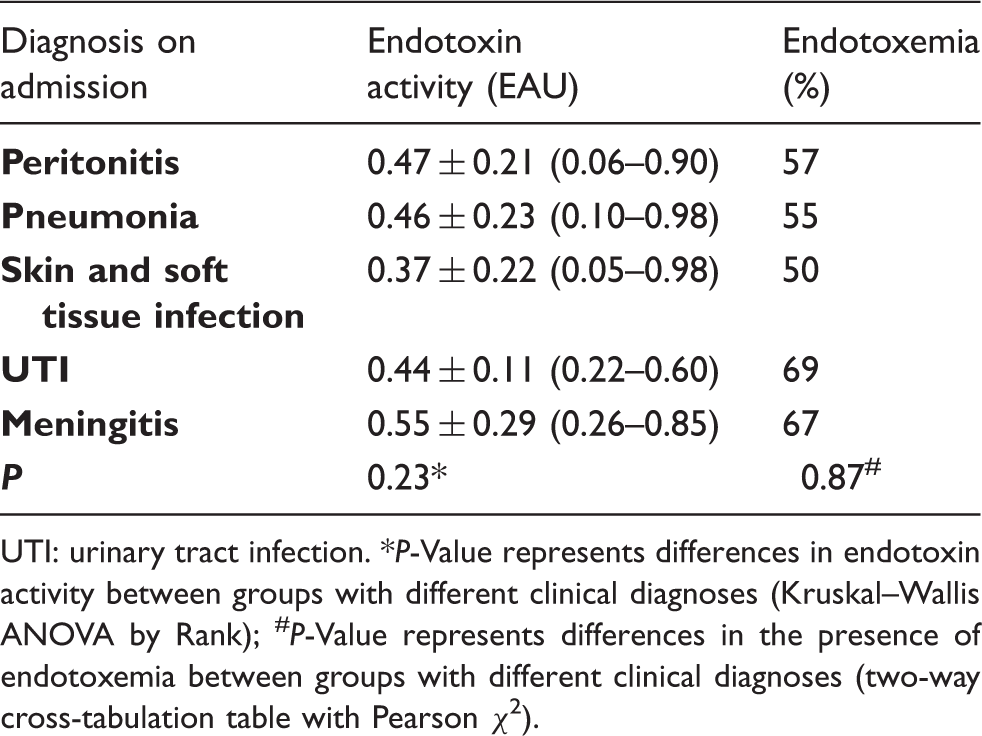
UTI: urinary tract infection. *P-Value represents differences in endotoxin activity between groups with different clinical diagnoses (Kruskal–Wallis ANOVA by Rank); #P-Value represents differences in the presence of endotoxemia between groups with different clinical diagnoses (two-way cross-tabulation table with Pearson χ2).
Endotoxemia and the type of infection
Endotoxemia and pathogens identified as the source of infection in septic shock.
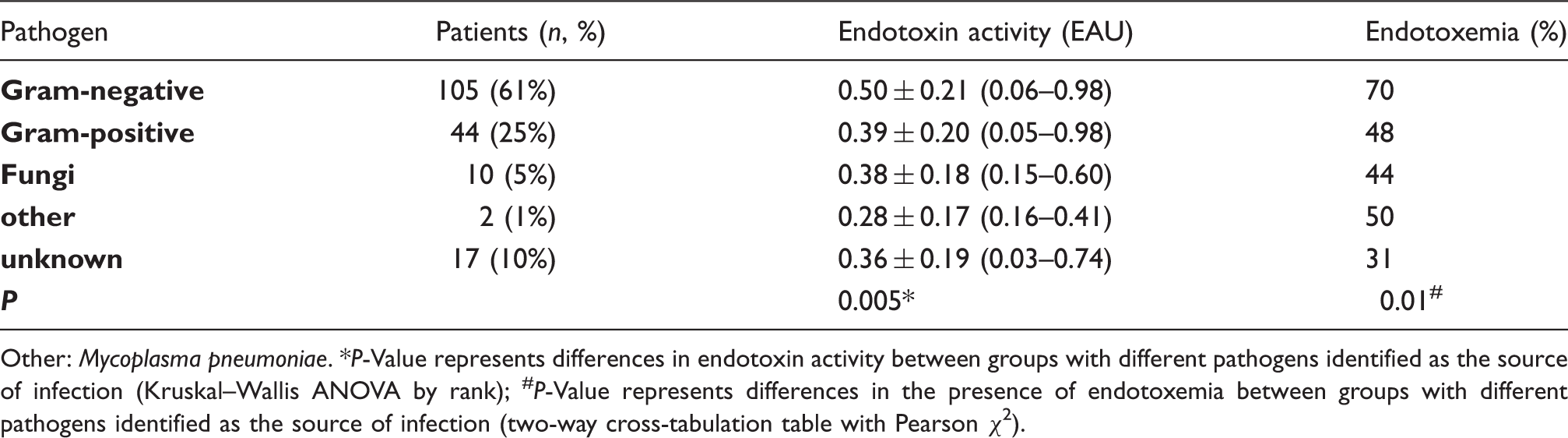
Other: Mycoplasma pneumoniae. *P-Value represents differences in endotoxin activity between groups with different pathogens identified as the source of infection (Kruskal–Wallis ANOVA by rank); #P-Value represents differences in the presence of endotoxemia between groups with different pathogens identified as the source of infection (two-way cross-tabulation table with Pearson χ2).
A Gram-positive source of septic shock was associated with a patient having endotoxemia in 48% of cases; if fungi were found to be the source of infection in septic shock, endotoxemia was detected in 44% of cases. In 17 of patients (10%) pathogen was not identified in the sample collected on admission to the ICU. In these cases, infection was documented clinically: seven patients that had been operated on for peritonitis had an intraoperatively, visually confirmed infection; eight patients had X-ray-confirmed pneumonia; one had clinically documented meningitis; and one had an infected wound.
Discussion
The study shows that endotoxemia is common in patients with septic shock on the day of admission to the ICU and is associated with an increased risk of death. These patients are potential candidates for anti-endotoxin treatment and the current clinical challenge is to identify those for whom endotoxin elimination treatment would be the most beneficial. The rapid EAA performed at the bedside is a useful tool to match the endotoxin elimination therapy to the disease. The results of the assay identified patients with endotoxemia in a very short time, which is crucial in the management of critically ill patients. In making decisions about including patients for endotoxin elimination, conducting an assay should not be precluded by a diagnosis of peritonitis, because endotoxemia has also been observed in many patients with a clinical diagnosis other than peritonitis. Similarly, the assay cannot be replaced with a microbiological test solely for a Gram-negative infection, because endotoxemia has been found in patients with infections other than Gram-negative ones.
Endotoxin elimination treatment is most often applied as an adjuvant therapy in the treatment of post-operative septic shock in patients with an infection originating from the abdominal cavity (peritonitis).9,10 Patients with septic shock from an abdominal origin are the primary candidates for endotoxin elimination therapy, because an abdominal source of septic shock is most likely to be endotoxin driven. Gram-negative bacteria are the predominant species in the intestinal flora. Perforation of the gastrointestinal tract or translocation across a damaged intestinal wall can result in bacteria and bacterial products crossing the intestinal barrier and causing endotoxemia and septic shock. 17 Our study showed that a diagnosis of peritonitis in patients with septic shock was not the sole indicator of all cases of endotoxemia; an elevated endotoxin activity was present in only 57% of these patients. The other 43%, despite clinical signs of both septic shock and peritonitis, had a low level of endotoxins. Thus, administering endotoxin elimination therapy to this group would not be justified, if based only on the diagnosis of peritonitis without the results of an EAA. The results of previously published studies that analyzed the effectiveness of endotoxin elimination have been contradictory. In septic patients with peritonitis, the beneficial effect of endotoxin elimination therapy was observed 24 and 48 h after therapy, as indicated by a significant improvement in the cardiac index, left ventricular stroke work index and oxygen delivery index; however, there was no improvement in mortality rate. 18 In the EUPHAS trial, improvement in the cardiovascular and pulmonary systems was observed 72 h after endotoxin elimination, as indicated by an increase in the mean arterial pressure, a decrease in vasopressor requirements, and an improvement in the PaO2/FiO2 ratio. This improvement in organ function was associated with a significant increase in survival rates. 9
The results from the preliminary studies mentioned above were not confirmed in a recently published, multi-center, randomized clinical trial. 10 In this study endotoxin activity was not measured and only a diagnosis of peritonitis was used as the criterion for endotoxin elimination treatment. No improvement in organ failure or survival rate was observed. A similar conclusion was reached in the largest retrospective analysis of a nationwide Japanese database. 11 There was no beneficial effect of endotoxin elimination treatment in a subpopulation of patients with abdominal septic shock from the perforation of the lower gastrointestinal tract. Based on the results of these studies and the data in our study, starting endotoxin elimination without measuring the actual endotoxin level in the blood might, in some cases, be an unnecessary intervention.
Another approach to inactivate endotoxins in septic patients would be to administer antimicrobial peptides and polymyxins, which are well-known substances that influence the activity of endotoxins by blocking the NF-κB pathway.19,20 Nephro- and neurotoxicity of polymyxins restrict clinical application as a last-resort agent against multi-drug resistant, Gram-negative pathogens.21,22 However, a growing number of multi-drug resistant, Gram-negative infections have influenced the number of studies with polymyxins that neutralize the lethal effects of endotoxins.19,23 Furthermore, multiple clinical trials that assess the efficacy of polymyxins against multiple drug-resistant, Gram-negative pathogens are currently recruiting patients. Nephrotoxic and neurotoxic effects of polymyxin B led to the development of an adsorptive cartridge for endotoxin elimination therapy in which polymyxin B is covalently bound to polystyrene fibers. 24 Immobilization on the surface of an insoluble carrier material prevents release of polymyxin into the bloodstream. This design capitalizes on the endotoxin-binding effects of adsorptive cartridge while minimizing systemic toxicity of antibiotic.
Our study showed that the presence of a Gram-negative pathogen in patients with septic shock was associated with a markedly higher incidence of endotoxemia. The prevalence of endotoxemia was even greater if Gram-negative bacteria were detected in the blood. Endotoxin is a membrane component of Gram-negative bacteria; it is released during multiplication or bacterial death and antibiotics that degrade the structure of the cell wall can also accelerate transient endotoxin release. Regardless of the source of infection, abdominal or not abdominal, the presence of a Gram-negative pathogen is associated with a higher rate of endotoxemia.4,25 Studies on the prevalence of endotoxemia in ICU patients have shown that endotoxin is detected much more often than a culture-proven Gram-negative infection. 26 In the present study, many patients with Gram-positive or fungal infections also demonstrated high levels of endotoxins. Endotoxemia in the absence of viable Gram-negative microorganisms detected in culture can reflect bacterial translocation and may appear in the bloodstream following hypotensive incidents that result in reduced splanchnic blood flow. The bedside measurement of EA levels with an EAA can be helpful in making a decision about starting endotoxin elimination therapy in patients with septic shock and an infection other than a Gram-negative one.
Endotoxin is rapidly cleared from the circulation in healthy people. 27 Detoxification mechanisms are complex and include the uptake of endotoxin principally into the liver, 28 the binding of endotoxin to prevent it from activating a TLR4 29 and the enzymatic degradation of the molecule.30,31 Detoxification mechanisms depend on many factors and can be related to the type of pathogen and the degree of infection, as well as to the patient’s clinical condition and treatment methods. Endotoxin-detoxifying mechanisms may be unable to prevent systemic inflammatory reactions in patients with a severe infection and large individual variations in patients have been observed.32–34 Therefore, intervention such as extracorporeal endotoxin elimination should be tailored to individual patients based on both the clinical conditions and the measured endotoxin levels. The inability to select appropriate populations for intervention on the basis of clinical criteria alone may have an impact on the lack of improvement in the clinical status of patients. The limitation in our study is that the EAA was performed in patients with a diagnosis of septic shock; patients with sepsis on admission to the ICU were not included. Appropriate trials are required to introduce and better define the importance of monitoring endotoxin activity with an EAA in the management of sepsis and septic shock.
Conclusions
The prevalence of endotoxemia is high among patients with septic shock and is associated with an increased risk of death. Circulating endotoxins can be detected using the rapid, whole blood EAA. Anti-endotoxin intervention such as extracorporeal endotoxin elimination should be tailored to individual patients based on both clinical conditions and measured endotoxin levels.
Footnotes
Acknowledgements
The authors would like to thank Mgr. Lukasz Strozecki for the involvement in performing statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in part by the research grant of National Science Center (OPUS 5, Number 2013/09/B/NZ4/01343).

