Abstract
The present study was aimed at investigating whether dietary anemonin could alleviate LPS-induced intestinal injury and improve intestinal barrier restoration in a piglet model. Eighteen 35-d-old pigs were randomly assigned to three treatment groups (control, LPS and LPS+anemonin). The control and LPS groups were fed a basal diet, and the LPS + anemonin group received the basal diet + 100 mg anemonin/kg diet. After 21 d of feeding, the LPS- and anemonin-treated piglets received i.p. administration of LPS; the control group received saline. At 4 h post-injection, jejunum samples were collected. The results showed that supplemental anemonin increased villus height and transepithelial electrical resistance, and decreased crypt depth and paracellular flux of dextran (4 kDa) compared with the LPS group. Moreover, anemonin increased tight junction claudin-1, occludin and ZO-1 expression in the jejunal mucosa, compared with LPS group. Anemonin also decreased TNF-α, IL-6, IL-8 and IL-1β mRNA expression. Supplementation with anemonin also increased TGF-β1 mRNA and protein expression, Smad4 and Smad7 mRNA expressions, and epidermal growth factor and epidermal growth factor receptor (EGFR) mRNA expression in the jejunal mucosa. These findings suggest that dietary anemonin attenuates LPS-induced intestinal injury by improving mucosa restoration, alleviating intestinal inflammation and influencing TGF-β1 canonical Smads and EGFR signaling pathways.
Introduction
Pulsatilla chinensis is a traditional Chinese herb that has been used in China for the treatment of enteritis and bacillary dysentery for many years.1,2 The important bioactive compounds are anemonin, protoanemonin and saponin, which have been shown to possess anti-inflammatory, antibacterial, antiviral and antitoxic properties.3,4 Anemonin is thought to be the primary compound responsible for the multiple activities of the plant.4–6 Recently, a growing body of evidence has indicated that anemonin can be effective in protection in a number of injury models.7–9 It has also been reported that anemonin exerts a beneficial role in ameliorating mucosal damage by enhancing the repair in trinitrobenzene sulfonic acid-induced colitis in rats. 10 However, no research has been conducted on the beneficial effects of anemonin on intestine injury in weaned piglets.
After injury, mucosal repair is a complex event that is highly regulated by multiple growth factors in the damaged intestine.11–13 TGF-β1 and epidermal growth factor receptor (EGFR) signaling pathway were suspected to play an important regulatory role in restoration of damaged intestine.14–16 TGF-β is a multifunctional polypeptide growth factor that has a central role in modulating gut mucosal cell growth, differentiation, migration and epithelial restitution.14,17 The TGF-β signaling pathway is mediated by the Smad family of proteins. 18 The role of TGF-β1 in the restoration of barrier integrity has been demonstrated in our previous studies. 16 Expression of epithelial growth factor (EGF) and EGFR were reported to correlate positively with the recovery of the small intestinal mucosa.15,19,20 However, until now, few data are have been available on the effects of anemonin on TGF-β1 and EGF, and their receptors and downstream signaling pathways in weaned pigs intestine after injury.
We hypothesized that anemonin may ameliorate LPS-induced intestinal injury and improve restoration by regulating the TGF-β1 and EGFR signaling pathways. In the present experiment, Escherichia coli LPS was administered to establish the model of gut injury. The purpose of the present study was to test this hypothesis and to elucidate the underlying mechanisms.
Materials and methods
Animals and treatment
Ingredients and chemical composition of the weaned diet on an as-fed basis.
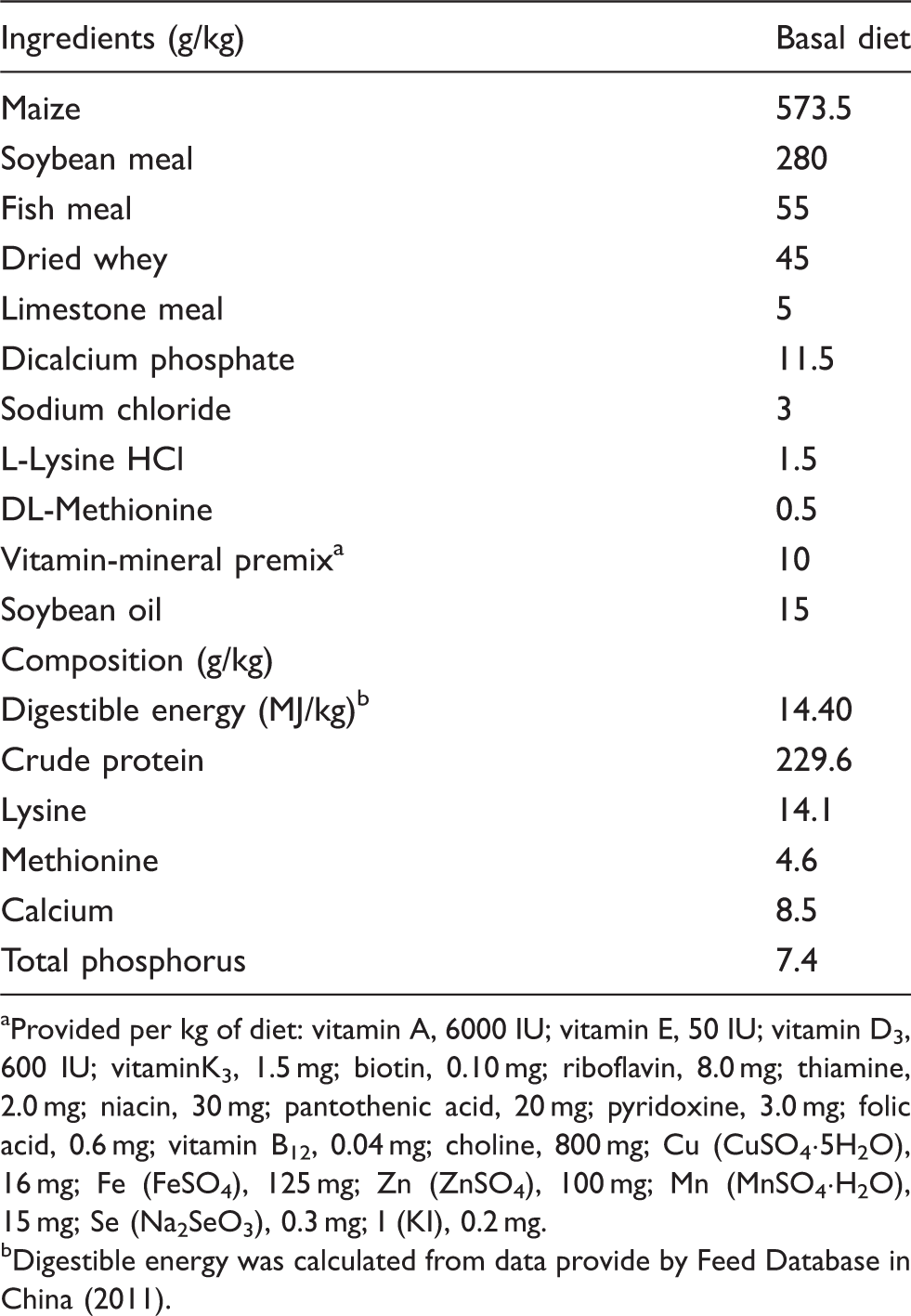
Provided per kg of diet: vitamin A, 6000 IU; vitamin E, 50 IU; vitamin D3, 600 IU; vitaminK3, 1.5 mg; biotin, 0.10 mg; riboflavin, 8.0 mg; thiamine, 2.0 mg; niacin, 30 mg; pantothenic acid, 20 mg; pyridoxine, 3.0 mg; folic acid, 0.6 mg; vitamin B12, 0.04 mg; choline, 800 mg; Cu (CuSO4·5H2O), 16 mg; Fe (FeSO4), 125 mg; Zn (ZnSO4), 100 mg; Mn (MnSO4·H2O), 15 mg; Se (Na2SeO3), 0.3 mg; I (KI), 0.2 mg.
Digestible energy was calculated from data provide by Feed Database in China (2011).
Sample collection
Four h post-injection of LPS or saline (on d 21), piglets were killed under anesthesia with an i.v. injection of sodium pentobarbital (50 mg/kg BM) and the gastrointestinal tract quickly removed. Segments of proximal jejunum were harvested immediately after killed and prepared for Ussing chamber studies. Adjacent specimens were fixed in 10% buffered formalin until morphology measurements. Mucosal scrapings from the remaining jejunum were collected, rapidly frozen in liquid nitrogen and stored at –80℃.
Intestinal morphology and barrier function
After fixation, the intestinal segments were dehydrated, embedded in paraffin, sectioned into 5-µm samples and stained with hematoxylin and eosin. 22 Villus height and crypt depth were measured in three intestinal cross-sections per animal with at least 10 well-oriented crypt villus units for each cross-section using image analysis (Leica Imaging Systems, Wetzlar, Germany) and averaged for each sample. Transepithelial electrical resistance (TER) and mucosal-to-serosal permeability to 4-kDa FITC dextran (FD4; Sigma-Aldrich, St. Louis, MO, USA) were determined in vitro in a Ussing chamber system, according to the procedures outlined by Moeser et al. 23 Briefly, segments of jejunum were stripped from the seromuscular layer in oxygenated (95% O2/5% CO2) Ringer’s solution and then mounted in the Easy Mount Ussing chamber system with a multi-channel voltage-current clamp (model VCC MC6; Physiologic Instruments). The clamps were connected to acquire and analyze software (Physiologic Instruments, San Diego, CA, USA) for automatic data collection. After a 30-min equilibration period on the Ussing chambers, TER (Ω·cm2) was recorded at 15-min intervals over a 2-h period and then averaged to derive the TER values for a given pig. FD4 (Sigma-Aldrich) was added on the mucosal side at a final concentration of 0.375 mg/ml. Mucosal-to-serosal flux of FD4 (µg/cm2/h 1 ) was monitored from the serosal side at 30-min intervals for 120 min. The concentrations of FD4 in the serosal side were measured by fluorescence microplate reader (FLx800; Bio-Tek Instruments Inc., Winooski, VT, USA). The flux over the 2-h period was calculated.
Protein expression analysis by Western blot
The Western blot analysis was performed according to the procedures outlined by Hu et al. 22 Briefly, after electrophoresis the proteins were transferred to polyvinylidene difluoride membranes (Millipore, Bedford, MA, USA). The membranes were incubated with primary Ab at 4℃ for 10 h and then with the secondary Ab for 2 h at room temperature. The primary Abs [occludin, claduin-1, zonula occludens-1 (ZO-1), TGF-β1, β-actin] were purchased from Santa Cruz Technology Inc. (Santa Cruz, CA, USA). The secondary Ab was HRP-conjugated anti-rabbit Ab (Cell Signaling Technology, Danvers, MA, USA). Western blot was detected with an enhanced chemiluminescence detection kit (Amersham, Arlington Heights, IL, USA), photographed by a ChemiScope 3400 (Clinx Science Instruments, Shanghai, China) and analyzed using Quantity One software. β-Actin was used as an internal control, which exhibited no difference among each group. The relative abundance of each target protein was expressed as target protein/β-actin protein ratio. The protein expression of all samples was expressed as fold changes, calculated relative to the control group.
mRNA expression analysis by RT-PCR
Forward and reverse primers used for real-time PCR.

Statistical analysis
Data were analyzed using the SAS statistical package (SAS Institute, Cary, NC,USA), with each animal considered an experimental unit. Data were subjected to ANOVA. Differences between the other means were tested using Duncan’s multiple range tests. Differences were considered significant at P < 0.05.
Results
Intestinal morphology and barrier function
Effects of dietary anemonin on jejunum morphology and barrier function of piglets.
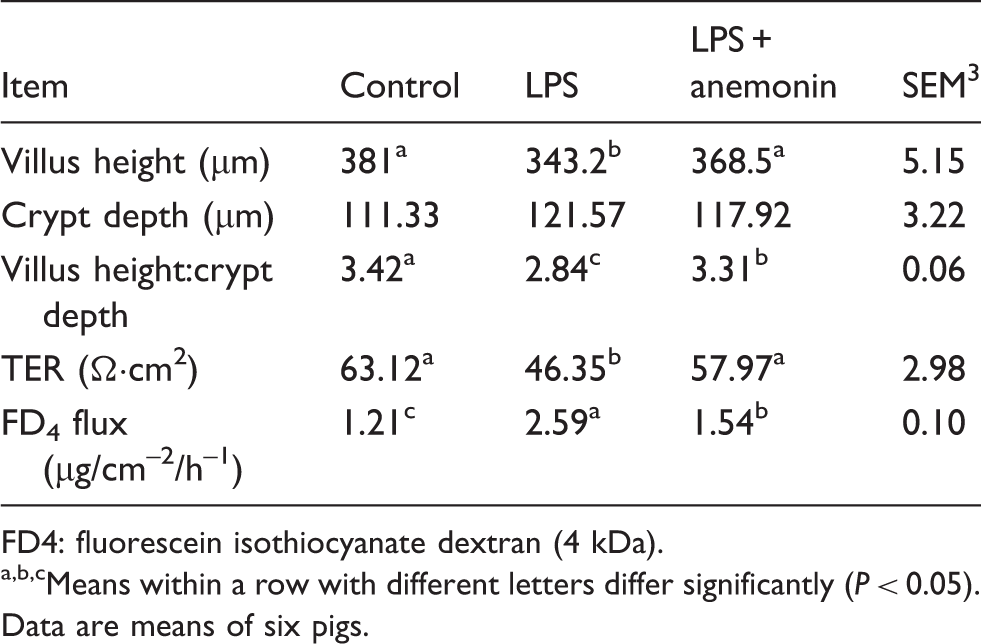
FD4: fluorescein isothiocyanate dextran (4 kDa).
Means within a row with different letters differ significantly (P < 0.05). Data are means of six pigs.
Tight junction protein expression
Figure 1 shows the tight junction protein levels of occludin, claudin-1 and (ZO-1 in the jejunal mucosa. As compared with the control group, LPS challenge decreased (P < 0.05) protein expressions of occludin, claudin-1 and ZO-1. However, dietary supplementation with 100 mg anemonin/kg diet resulted in an increase (P < 0.05) in protein expressions of occludin and claudin-1 compared with the LPS group.
Effects of dietary anemonin on protein expression of occludin, claudin-1 and ZO-1 in the jejunal mucosa of piglets. (a) Representative blots of occludin, claudin, ZO-1 and β-actin in the jejunal mucosa of piglets. (b) Relative tight junction proteins expression in jejunal mucosa of piglets. Values are means and SD, represented by vertical bars. a,bMeans with different letters differ significantly (P < 0.05). The control sample was used as the reference sample. The protein expression of all samples was expressed as fold changes, calculated relative to the control group. LPS; LPS + anemonin. Control (non-challenged control) piglets receiving a control diet and injected with 0.9% sterile saline; LPS (LPS-challenged control), piglets receiving the same control diet and injected with E. coli LPS; LPS + anemonin (LPS challenged + 100 mg/kg anemonin), piglets receiving a 100 mg/kg anemonin diet and injected with LPS.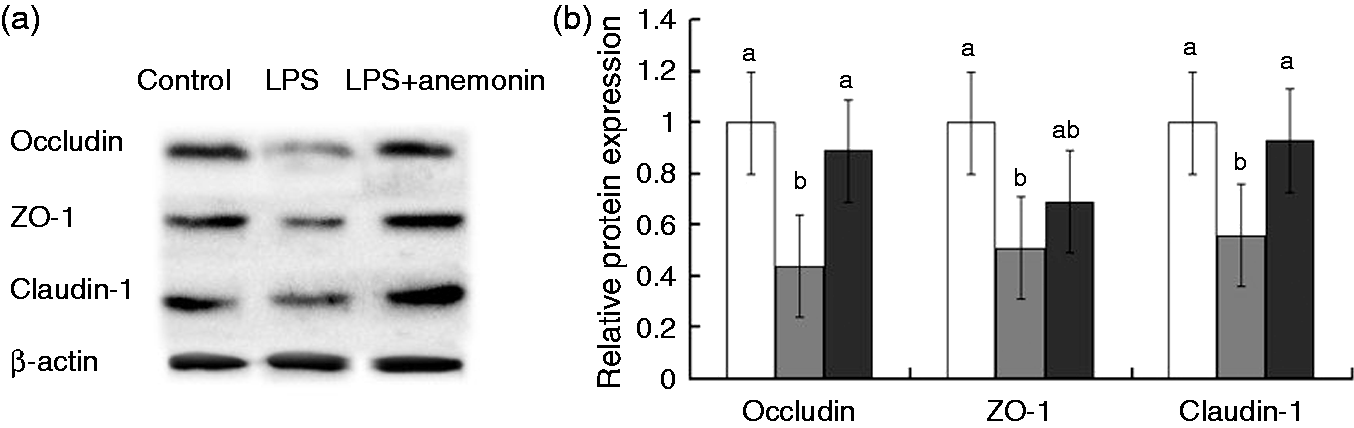
Pro-inflammatory cytokine mRNA
Effects of dietary anemonin on cytokine mRNA levels on the jejunal mucosa of piglets. a
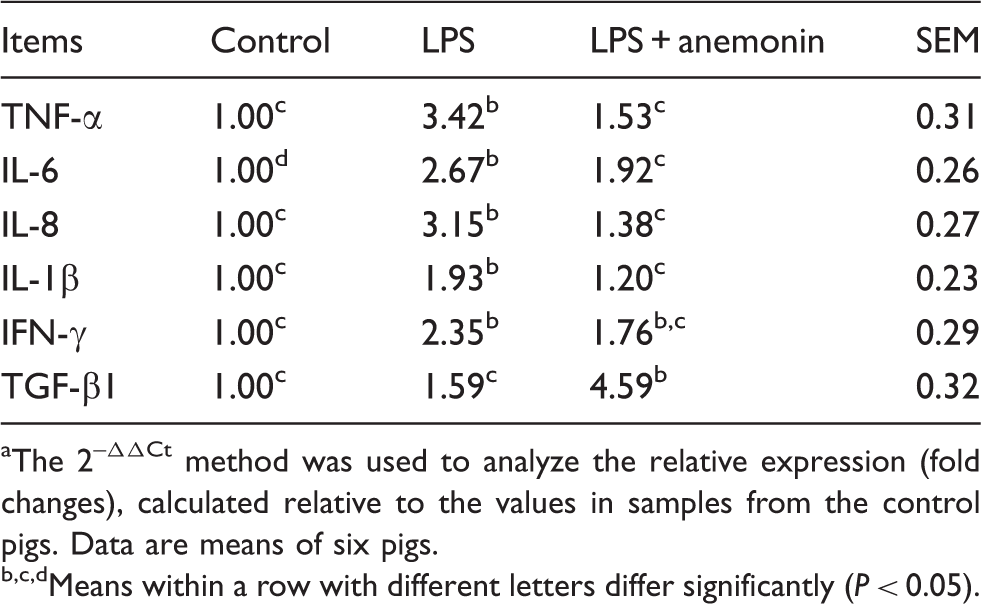
The 2–ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the values in samples from the control pigs. Data are means of six pigs.
Means within a row with different letters differ significantly (P < 0.05).
TGF-β1 expression
Figure 2 presents the protein expression of TGF-β1 in the jejunal mucosa of piglets. Piglets challenged with LPS did not differ (P > 0.05) from the control group with regard to TGF-β1 protein expression. Dietary supplementation with 100 mg anemonin/kg diet significantly increased (P < 0.05) the expression of TGF-β1 compared with the LPS and control groups. Consistent with the increased protein expression of TGF-β1, Table 4 shows that 100 mg anemonin/kg diet supplementation also significantly improved (P < 0.05) the mRNA expression of TGF-β1.
Effects of dietary anemonin on the protein expression of TGF-β1 in the jejunal mucosa of piglets. (a) Representative blots of TGF-β1 expression and β-actin. (b) Relative TGF-β1 protein expression in jejunal mucosa of piglets. a,bMean values with different letters were significantly different (P < 0.05). Values are means and SD, represented by vertical bars. The control sample was used as the reference sample. The protein expression of all samples was expressed as fold changes, calculated relative to the control group. Control (non-challenged control), piglets receiving a control diet and injected with 0.9% sterile saline; LPS (LPS-challenged control), piglets receiving the same control diet and injected with E. coli LPS; LPS + anemonin (LPS challenged + 100 mg/kg anemonin), piglets receiving a 100 mg/kg anemonin diet and injected with LPS.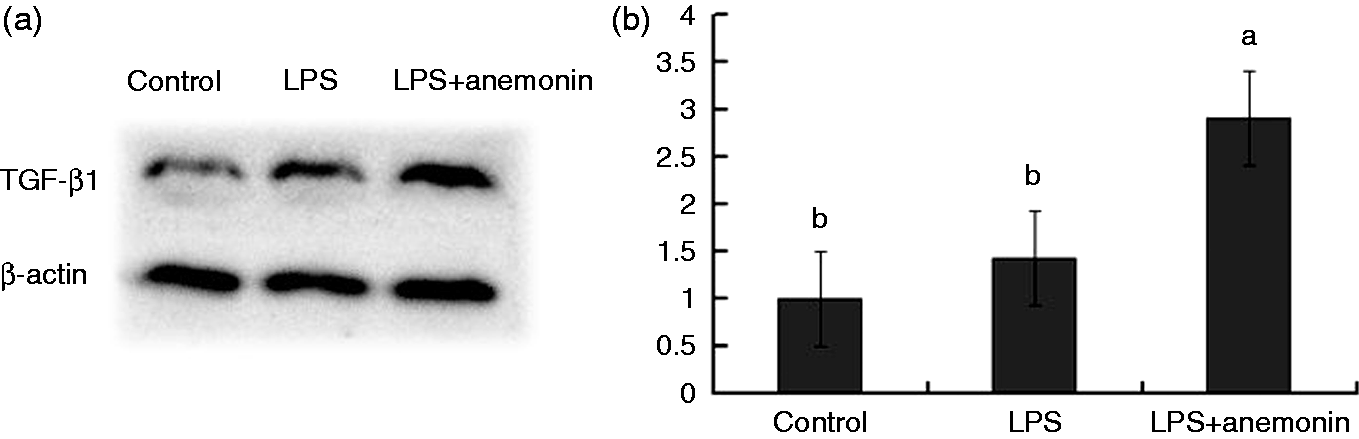
mRNA expression of Smads
Effects of dietary anemonin mRNA expressions of Smad signals on the jejunal mucosa of piglets. a
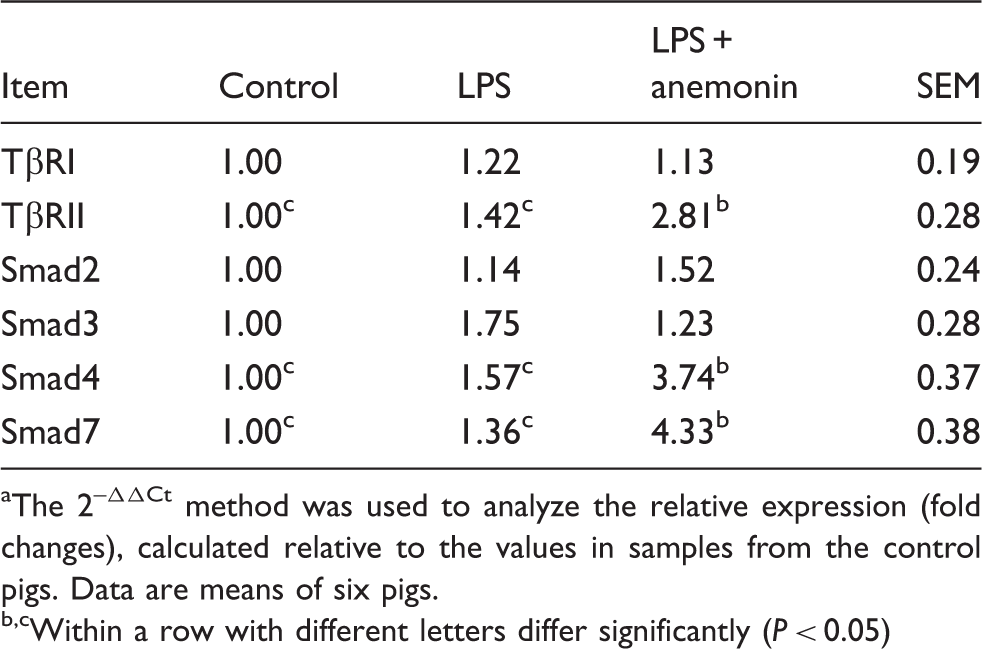
The 2–ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the values in samples from the control pigs. Data are means of six pigs.
Within a row with different letters differ significantly (P < 0.05)
mRNA expression of EGFR signal pathways
Effects of dietary anemonin on EGF and EGFR mRNA expression in the jejunal mucosa of piglets. a
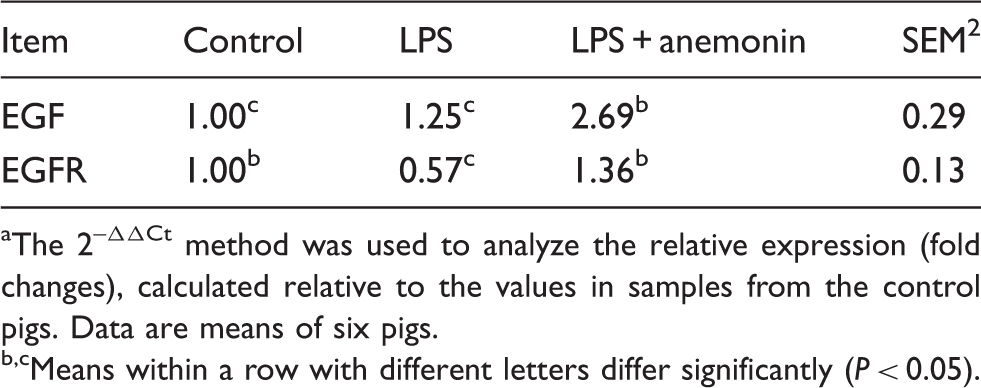
The 2–ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the values in samples from the control pigs. Data are means of six pigs.
Means within a row with different letters differ significantly (P < 0.05).
Discussion
Many herbal plant-derived products have been proven to be natural, less toxic and residue-free in certain doses, and are thought to be ideal feed additives to animal diets.24,25 Pulsatilla chinensis is a traditional Chinese medicine that has been widely used in China for thousands of years to treat gastrointestinal disorders such as gastroenteritis and diarrhea. Pulsatilla chinensis extracts contain abundant natural bioactive compounds such as anemonin, protoanemonin, okinalin and saponin, which have been shown to possess anti-inflammatory, antibacterial, antiviral, antitoxic and immune-enhancing properties.3,4 Recently, increasing evidence suggested that P. chinensis extracts can ameliorate mucosal damage and enhance repair in sodium dextran sulfate-induced colitis in mice. 10 Anemonin is thought to be the primary compound responsible for these multiple activities.4,7,9 Anemonin (the dilactone of cyclobutane-1,2-diol-1,2-diacrylic acid) has been isolated from P. chinensis, Drymaria diandra, Knowltonia capensis, Clematis chinensis and Ranunculaceous plants. 6 It is an innocuous compound that is further hydrolyzed to a non-toxic carboxylic acid. 26 There is evidence that anemonin has significant therapeutic effects on chicken endotoxin damage, and the beneficial effects of anemonin may be related to the enhanced capacity of antioxidant. 27 Moreover, anemonin can promote the expression of PCNA, EGFR, TGF-α and TGF-β1, which are important intestinal mucosa repair factors in diarrheal mice. Also, anemonin has been demonstrated to prevent the expansion of apoptotic neuronal death effectively in MCAO-induced cereal I/R rats. 9 It has also been reported that anemonin prevented intestinal microvascular dysfunction activated by LPS through antagonizing the LPS by inhibiting, in part, the production of NO, ET-1 and ICAM-1 in RIMECs to exert some beneficial therapeutic action in intestinal inflammation. 4 So far, there has been no study to evaluate whether dietary supplementation anemonin could alleviate gut injury and enhance intestine repair in weaned piglets chronically challenged with E. coli LPS. LPS-induced intestine injury a well-established animal model used to study infant nutrition and gastrointestinal physiology.21,28
In the present study, LPS challenge exhibited a decrease in villus height and increase in crypt depth in jejunum of piglets, which indicated that LPS caused intestinal mucosa damage. Our results are consistent with the findings of Liu et al. 21 and Hou et al. 28 Dietary supplementation 100 mg anemonin/kg diet ameliorated LPS-induced intestinal jury by decreased jejunal crypt depth and increased villus height and villus height:crypt depth ratio after LPS challenge, which indicated that supplementation improved intestinal morphology after damage. Our data were supported by the results of Xu et al., 29 who reported anemonin alleviated mouse intestinal ultrastructure after exposure to PRV and E. coli infection. Liu et al. 30 also reported that Merbal medicine (Chinese Pulsatilla root) improved mucosa structure by increasing villus height and decreasing crypt depths in the small intestine of canine. Consistent with improved intestinal morphology, the present study also demonstrated that anemonin improved intestinal barrier function, reflected by increased TER and decreased FD4 flux in jejunum of piglets compared with the LPS group. These findings support the notion that anemonin beneficially ameliorates the LPS-induced damage in intestinal structure and function. According to these results, we speculated that enhancing the restoration of epithelium after injury might be the mechanism by which anemonin protects intestinal integrity and barrier function in an LPS-induced injury model.
The intestinal barrier is mainly formed by a layer of epithelial cells joined together by tight junctions. 31 Of them, occludin, claudins and ZO-1 are thought of as the major integral membrane proteins, which participate in tight junction structural integrity by binding to the actin cytoskeleton. 32 LPS has been reported to disrupt barrier function in a time- and dose-dependent manner, and also induces a redistribution of tight junction proteins (occludin, claudin-1and ZO-1) from the intercellular junctions.28,33 In the current experiment, our results convincingly indicated that LPS reduced the abundance of occludin, claudin-1 and ZO-1 in the small intestinal mucosa of piglets. Consistent with the improved intestinal morphology and barrier function, it is noteworthy that anemonin supplementation increased the protein expression of occludin, claudin-1 and ZO-1 in the small intestine of LPS-challenged piglets. Similarly, Zhang and Jiang reported that Pulsatilla alcohol extract protected intestinal epithelial tight junctions in rats with trinitrobenzene sulfonic acid-induced colitis. 10 Consequently, in the present study, anemonin may have partially improved the intestinal integrity and barrier function via increasing tight junction protein expression.
Cytokines also play an important role in the regulation of the intestinal barrier integrity. 34 Overproduction of pro-inflammatory cytokines has a negative influence on gut integrity and epithelial function.35,36 In the present study, pro-inflammatory cytokine (TNF-α, IL-1β, IL-6, IFN-γ and IL-8) gene expression was elevated in the jejunal mucosa of piglets challenged with LPS. Consistent with improved intestinal integrity, dietary anemonin decreased cytokine expression (TNF-α, IL-6, IL-1β, IL-8). Currently, there is an abundance of research on the modulation of intestinal pro-inflammatory mediators through supplementation of anemonin.5,21 In line with our findings, Sang et al. reported that the protective role of Pulsatilla extract in LPS-induced inflammatory responses is partially due to its inhibitory effects on the overproduction of pro-inflammatory mediators, including pro-inflammatory cytokines (TNF-α, IL-1β, IL-6). 8 Therefore, in the current study, feeding pigs with an anemonin-supplemented diet may improve intestinal integrity, partially by inhibiting the production of pro-inflammatory mediators.
TGF-β is considered to be the key regulator of intestinal injury restoration as most of the cytokines that promote epithelial restitution act through a TGF-β-dependent mechanism. 16 It has been reported to play an important role in the post-weaning adaptation process in the intestine of pigs.14,16 In the present study, we found that anemonin increased the TGF-β1 content in the jejunum mucosa of piglets, both in gene and protein expression, compared with the LPS-challenged pigs. Similarly, Liu et al. 7 found that the anemonin from P. chinensis prevents diarrhea in mice by regulating TGF-β1 expression. Increased TGF-β1 due to the traditional medicine shikonin was also reported to regulate positively wound healing after injury. 37 It is likely that the increased TGF-β1 concentration induced by anemonin was directly responsible for the restoration of the intestine barrier function. Therefore, we hypothesize that the beneficial role of anemonin in intestinal barrier protection may be partially influenced by TGF-β1.
The canonical TGF-β signaling pathway is mediated by the Smad family of proteins. 18 Smad proteins—the only substrates of TβR kinase—are critical mediators of the TGF-β signaling transducer. When TGF-β reaches the membrane of target cells, it binds directly to TGF-β type II receptors (TβRII), which leads to the recruitment of TGF-β type I receptors (TβRI). TβRII then trans-phosphorylates TβRI, enabling the TβRI kinase domain to act on cytoplasmic proteins and thereby propel downstream signaling actions. Smad2 and Smad3 are receptor-regulated Smads (R-Smads). Following stimulation by TGF-β, Smad2 and Smad3 become phosphorylated. Phosphorylated Smad 2/3 can complex with Smad4 (the common Smad mediator), and then translocate to the nucleus and regulate gene expression. 38 To determine whether the TGF-β1 signaling pathway is involved in anemonin exerting beneficial effects on LPS-induced intestinal injury, we evaluated the canonical downstream substrates of the TGF-β signal (TβRI, TβRII, Smad2, Smad3, Smad4 and Smad7) and observed that anemonin supplementation increased the gene expressions of TβRII, Smad4 and Smad7 after LPS challenge, indicating that the canonical Smads signaling pathway was activated. However, mRNA expression of Smad2 and Smad3 in the intestinal mucosa did not vary significantly compared with the LPS and control groups. In line with our study, another study has shown that TGF-β1 protects the immature gut from injury via Smad protein-dependent signaling pathways. 39 Consequently, we speculate that anemonin-improved intestinal barrier restoration in LPS-induced intestine injury may partially be induced through the TGF-β1 Smad pathway, either directly or indirectly.
EGF
In conclusion, dietary supplementation with an 100-mg anemonin/kg diet is beneficial for alleviating intestine injury and improving mucosal repair in piglets. Possible mechanisms for the actions of anemonin may include the following: (1) reduction of pro-inflammatory cytokines (indicated by decreased TNF-α, IL-1β, IL-6 and IL-8); (2) alleviation of intestinal injury (indicated by alleviation of LPS-induced damage in the intestinal structure and barrier function, and increased expression of tight junction protein levels of occludin and claudin-1 in the jejunal mucosa of the LPS-challenged piglets); and (3) improvement of mucosal repair via TGF-β1 canonical Smad pathways and EGFR signaling (indicated by increase in TGF-β1 and EGFR expression in the jejunm mucosa). These findings may have important implications for treating intestinal damage in humans and other animals.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was jointly supported by National Natural Science Foundation of China (31472103), and the Special Fund for Agroscientific Research in the Public Interest (201403047).
