Abstract
Hemoglobin and its structures have been described since the 1990s to enhance a variety of biological activities of endotoxins (LPS) in a dose-dependent manner. To investigate the interaction processes in more detail, the system was extended by studying the interactions of newly designed peptides from the γ-chain of human hemoglobin with the adjuvant monophosphoryl lipid A (MPLA), a partial structure of lipid A lacking its 1-phosphate. It was found that some selected Hbg peptides, in particular two synthetic substructures designated Hbg32 and Hbg35, considerably increased the bioactivity of MPLA, which alone was only a weak activator of immune cells. These findings hold true for human mononuclar cells, monocytes and T lymphocytes. To understand the mechanisms of action in more detail, biophysical techniques were applied. These showed a peptide-induced change of the MPLA aggregate structure from multilamellar into a non-lamellar, probably inverted, cubic structure. Concomitantly, the peptides incorporated into the tightly packed MPLA aggregates into smaller units down to monomers. The fragmentation of the aggregates was an endothermic process, differing from a complex formation but rather typical for a catalytic reaction.
Introduction
LPS are constituents of the cell envelope of most Gram-negative bacteria. Owing to the profound immunostimulatory and detrimental actions at higher doses in mammalian species, mediated by particular structural classes, for example the prototypic enterobacterial chemotypes, LPS is synonymously referred to as endotoxins. Endotoxins activate the innate immune system and contribute to the activation of the mammalian adaptive immune system. During severe Gram-negative infections endotoxins play a key role in the development of septic shock. 1 However, lower concentrations and also partial structures of LPS, such as 4′-monophosphoryl lipid A (MPLA), exhibit beneficial effects for the host. These effects include enhanced resistance to infection and protection against malignancy, owing to their striking immune stimulatory and modulatory activities.2,3 The harmful, as well as the beneficial, responses are not directly induced by LPS and its immunostimulatory partial structures but rather mediated by endogenous immunomodulatory molecules of the host, such as TNF-α, members of the IL family (IL-1, IL-6, IL-8, IL-10, IL-12), IFNs, reduced oxygen species, and bioactive lipid products, such as those originating from the arachidonic metabolism like prostaglandins, formed in the membranes of the host cells. These mediators are released not only by monocytes/macrophages, but also by other cell types like vascular, epithelial, and polymorphonuclear cells.4,5 Members of the TLR family have been shown to represent a pivotal class of signalling receptors in mammalian innate immunity, and TLR4 has been documented to be the transmembrane receptor mediating cellular activation by LPS/lipid A. 6 For signalling, TLR4 needs the co-expression of an adaptor protein called MD-2, which displays high-affinity association to the TLR4 ectodomain and represents the central LPS/lipid A binding constituent of the cellular LPS receptor system. 7 Using radiolabelled LPS, a physical contact between LPS, TLR4 and MD-2 could be demonstrated only when a further molecule, termed CD14, was present. 8 CD14 has been shown to be involved in the binding of LPS to TLR4 and activation of cells by LPS. CD14 is located as a glycosylphosphatidylinositol-anchored membrane protein (mCD14) on the surface of monocytic cells, polymorphonuclear leukocytes, some B-lymphocytes and epithelial cells.9–11
Preparations of MPLA, the partial structure of lipid A, are obtained by mild acid hydrolysis of LPS, for example from Salmonella Minnesota or by chemical synthesis. 12 Compared with lipid A, native or synthetic MPLA exhibits only low endotoxicity and preserved beneficial immunostimulatory properties, including adjuvant activity. Therefore, the vaccine adjuvant MPLA was termed a ‘detoxified form of endotoxin’. An MPLA-type preparation from Salmonella Minnesota R595, designated MPL™, represents the first TLR agonist to be approved for use as vaccine adjuvant in humans. 13 Here, we used a synthetic MPLA that differs from that of Escherichia coli only by the length of the 3-OH acyl chain at the 3’ position of the diglucosamine backbone, that is, possessing 14:0 instead of 12:0.
In biophysical investigations it could be shown that all endotoxically highly active lipid A structures display a particular ‘endotoxic conformation’, that is, a conical shape of the lipid A part corresponding to a higher cross-section of the hydrophobic as the hydrophilic region. 14 Connected with the conical shape is an inclination of the lipid A diglucosamine backbone with an inclination angle of larger than 50° with respect to the membrane normal, that is, the direction of the acyl chains. 15 For the endotoxically inactive or low active forms, such as MPLA, the molecular conformation becomes more cylindrical, connected with a much lower inclination angle around 30–35°. 15
Hemoglobin (Hb) is a tetrameric heme protein complex with a molecular mass of about 64,500 Da. Its main biological function is the transport of oxygen (O2) in the circulation. 16 Adult human Hb (HbA) consists of two α- and two β-chains, with each of these subunits containing one heme group as a prosthetic group. Fetal Hb (HbF) consists of two α-chains and two γ-chains in their heme-complexed forms. In human ontogenetic development HbA is synthesized in the bone marrow in the postnatal lifespan, whereas HbF is primarily produced in the liver and spleen of the fetus. 17 In the following, the term HbF denotes its tetrameric forms, as well as heme-free HbF. α-, β- and γ-chains are the monomeric heme-free globin chains derived from HbA and/or HbF.
HbF of sheep or human origin enhances the LPS- and lipid A-mediated induction of cytokines from murine macrophages and human peripheral blood monocytes in vitro. In addition, LPS-induced modulation of innate and adaptive immune responses by HbF was observed in vivo.18,19 This amplifying activity could be shown to reside mainly in the γ-chain of HbF (Hbg). One explanation for this enhancing interaction between Hbg and lipid A is considered to be contributed by a Hb-induced conformational change of lipid A, thus conferring the enhanced biological activity. In line with this model, biophysical analyses showed that Hb, as well as purified Hb chains, lead to an increasing conversion of the endotoxic conformation of lipid A into a more conical shape upon intercalation of Hb and γ-globin molecules into endotoxin aggregates followed by dispersion of the LPS supramolecular structures into smaller-size units.20,21
In a preceding study, we analyzed the mechanisms that governed the enhancing activities on the endotoxin-induced cytokine production of a human Hbg (huHbg)-derived peptide in comparison with an inhibiting peptide from the Pep19-2.5 series. 22
Sequences of the Hbg peptides.
Materials and methods
Approval for these studies was obtained from the Institutional Ethics Committee at the University of Lübeck (Lübeck, Germany; Az. 12-202A) according to the Declaration of Helsinki. All donors gave written informed consent.
Hbg peptides and MPLA
All synthetic peptides were prepared and obtained from EMC Microcollections GmbH (Tübingen, Germany). The short-length peptides (15–16 amino acids) were purified by precipitation and their structures were verified by MS analysis. The long-length peptides were purified by HPLC and shown to have a purity of >95%. The peptides of the chosen synthetic library were dissolved in DMSO at a concentration of 10 mM. All other peptides were dissolved in 0.9% NaCl at a concentration of 10 mM. The peptides were named as Hbg-##. The ##-numbers refer to the peptide number.
Purified native Hbγ chains were prepared by JP Mach. These Hbγ preparations were dissolved in 0.9% NaCl at a concentration of 100 µM. MPLA was obtained from Avanti Polar Lipids (Alabaster, AL, USA). MPLA was dissolved in aqua ad injectabilia (B. Braun Melsungen AG, Melsungen, Germany) by eight cycles of vortexing (15 min) and incubation in a water batch sonicator (15 min) at a concentration of 250 µg/ml.
Cell cultures
Human mononuclear cells (MNC) were isolated from peripheral blood of healthy donors by density-gradient centrifugation on Ficoll gradient media (PAA Laboratories GmbH, Pasching, Austria). The cells were suspended and cultured in RPMI-1640 medium, containing 1 µg/ml penicillin and 100 U/ml streptomycin. Purified monocytes were isolated from MNC by counterflow centrifugation using a Beckman JE-6B-elutriator (Beckman Instruments, Munich, Germany). The monocyte fraction collected consisted of >95% monocytes, as determined by FACS analysis.
Fifty μl of a Hbg peptide solution were incubated with 50 µl MPLA solution in a U-form 96-well microcell culture plate #3799 (Corning Costar GmbH, Bodenheim, Germany) for 15 min at 37℃. After this incubation period 20 µl heat-inactivated (56℃/30 min) human serum or RPMI-1640 medium were added. Finally, 80 µl MNC cell suspension was added to give a final concentration of 1–2 × 106 cells for culture in the absence of human serum, or 1 × 106 cells for culture in the presence of serum. After a culture time of 20 h the cells were spun down and the culture supernatants were harvested. The cytokine concentrations in the supernatants were determined by matched antibody pairs for ELISA, obtained from Invitrogen GmbH (Darmstadt, Germany). Each value represents the mean ± SD of duplicate cultures.
For stimulation of T lymphocytes, human MNC were cultured in RPMI-1640 medium, containing 1 µg/ml penicillin, 100 U/ml streptomycin and 10% inactivated human serum at a concentration of 0.5–1.0 × 106 cells per ml in flat-bottom 96-well tissue culture plates. To investigate the adjuvant effect of MPLA and Hbg peptides, the recall antigen purified protein derivative of Mycobacterium tuberculosis (PPD; Statens Serum Institute, Copenhagen, Denmark) was used. After 6 d of culture, cells were labeled with 3H-Thymidine (3HTdR; 2 Ci/mmol, 0.2 µCi/culture) and after an additional day of culture the cells were harvested on glass-filter mats for measurement of incorporated radioactivity into the DNA. The results are expressed as mean ± SD of duplicate cultures.
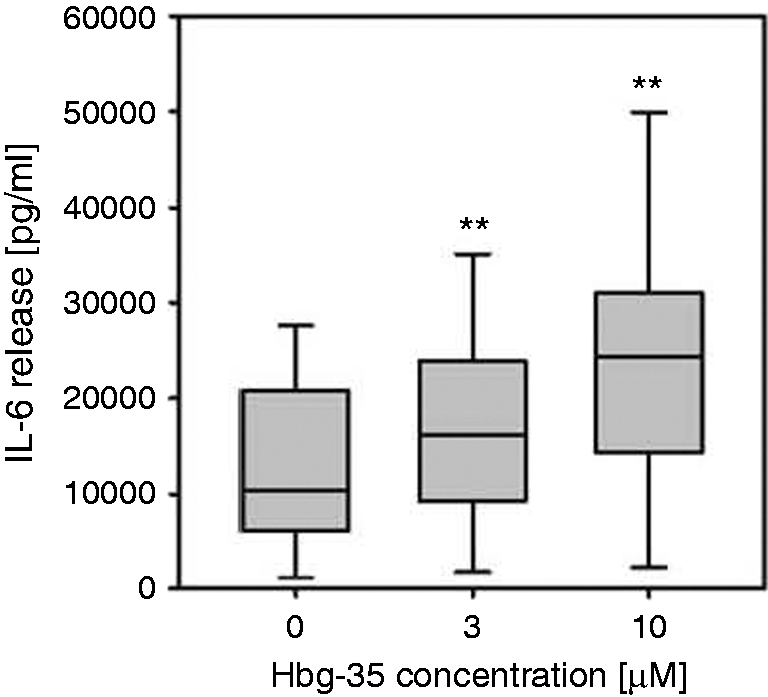
Statistical analyses were performed using paired Student's t-test or Mann–Whitney Rank Sum Test as appropriate with the aid of Sigma Plot software. Differences were considered to be significant at P < 0.05. As a typical example of the statistical analyses, in Figure 1 the dependence of the IL-6 secretion on the concentration of Hbg35 is presented, exhibiting the details of the statistical evaluation.
Freeze-fracture transmission electron microscopy
A small amount of the samples was sandwiched between two copper profiles and frozen by plunging the sandwiches immediately into a liquified ethane/propane mixture cooled in liquid nitrogen. Fracturing and replication were performed at –150℃ in a BAF 400 T freeze-fracture device (BAL-TEC, Balzers, Liechtenstein) equipped with electron guns and a film sheet thickness monitor. For replication Pt(C) was evaporated under an angle of 35° and (C) under 90°. The replicas were placed on copper grids and cleaned with a chloroform/methanol mixture. Further details are published elsewhere. 23
Small angle X-ray scattering
Small angle X-ray scattering (SAXS) measurements were performed at the European Molecular Biology Laboratory outstation at the Hamburg synchrotron radiation facility HASYLAB using camera X33.
24
Scattering patterns in the range of the scattering vector 0.1 < s < 1.0 nm–1 (s = 2 sin θ/λ, 2θ scattering angle and λ the wavelength = 0.15 nm) were recorded at 40℃ with exposure times of 1 min using an image plate detector with online readout (MAR345; MarResearch, Norderstedt, Germany). The lipid concentration was 20 mM in all cases. The s-axis was calibrated with Ag-behenate, which has a periodicity of 58.4 nm. The diffraction patterns were evaluated as described previously,
25
assigning the spacing ratios of the main scattering maxima to defined three-dimensional structures. The lamellar and cubic structures are the most relevant here. The following features characterize the supramolecular structure:
Lamellar: the reflections are grouped in equidistant ratios, that is, 1, 1/2, 1/3, 1/4, and so on, of the lamellar repeat distance dl. Cubic: the different space groups of these non-lamellar three-dimensional structures differ in the ratio of their spacings. The relation between reciprocal spacing shkl = 1/dhkl and lattice constant a is
(hkl = Miller indices of the corresponding set of plane).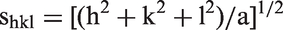
Förster (fluorescence) resonance energy transfer spectroscopy
Intercalation of Hbg-32 into MPLA aggregates was determined by Förster resonance energy transfer (FRET) spectroscopy applied as a probe dilution assay. MPLA was labelled with the donor dye NBD-phosphatidylethanolamine (NBD-PE) and the acceptor dye rhodamine-PE. 26 The donor dye was excited at 470 nm, and its emission intensity measured at 531 nm. In the FRET methodology, the donor emission fluorescence intensity is transferred in a radiation less process to the acceptor, which has as an excitation wavelength corresponding to the donor emission wavelength (531 nm). The Hbg peptide was added to MPLA (0.01 mM) at a final concentration of 0.01 or 1 mM. Intercalation was monitored as the increase of the ratio of the donor emission intensity Id at 531 nm to that of the acceptor emission intensity Ia at 593 nm (FRET signal) in a time-dependent manner.
Atomic force microscopic analyses
Atomic force microscopic (AFM) allows a direct look onto the morphology of lipid aggregates on a molecular scale under almost physiological conditions. This allows depth profiles of MPLA dispersions down to a resolution limit of lower than 1 nm to be obtained. 27 For the analysis, MPLA dispersions were prepared at a concentration of 25 µM in physiological saline in the absence and presence of an equimolar content of Hbg-32.
Isothermal titration calorimetry
Microcalorimetric measurements of peptide binding to MPLA were performed on a MCS isothermal titration calorimeter (Microcal Inc., Northampton, MA, USA) at 37℃ as recently described.25,28,29 MPLA (0.05 mM)—prepared as described above—was dispensed into the microcalorimetric cell (volume 1.3 ml) and the peptide solutions (1 mM) were filled into the syringe compartment (volume 100 µl). After temperature equilibration, the peptides were titrated in 3-µl portions every 5 min into the lipid-containing cell under constant stirring, and the heat of interaction after each injection measured by the isothermal titration calorimetry (ITC) instrument was plotted vs. time. In this assay, endothermic reactions between the two ligands lead to peaks upward and exothermic reactions to one downward.
Fourier-transform infrared spectroscopy
The infrared spectroscopic measurements were performed on an IFS-55 spectrometer (Bruker, Karlsruhe, Germany). Peptide and MPLA plus peptide mixtures were dispersed in 20 mM HEPES buffer, pH 7.0, and spread on a ZnSe attenuated total reflectance unit. After evaporation of free buffer solution, 100 interferograms of the samples were accumulated, apodized, Fourier-transformed and converted to absorbance spectra. The infrared spectra were evaluated in the range of the amide I vibration (predominantly C = O stretching vibration), the peak positions of which are characteristic for the secondary structure of the Hbg-32 peptide. Usually, α-helical structures are found at a peak position of 1655–1662 cm–1, while β-sheet structures show a main absorbance maximum around 1630 cm–1. 30
Results
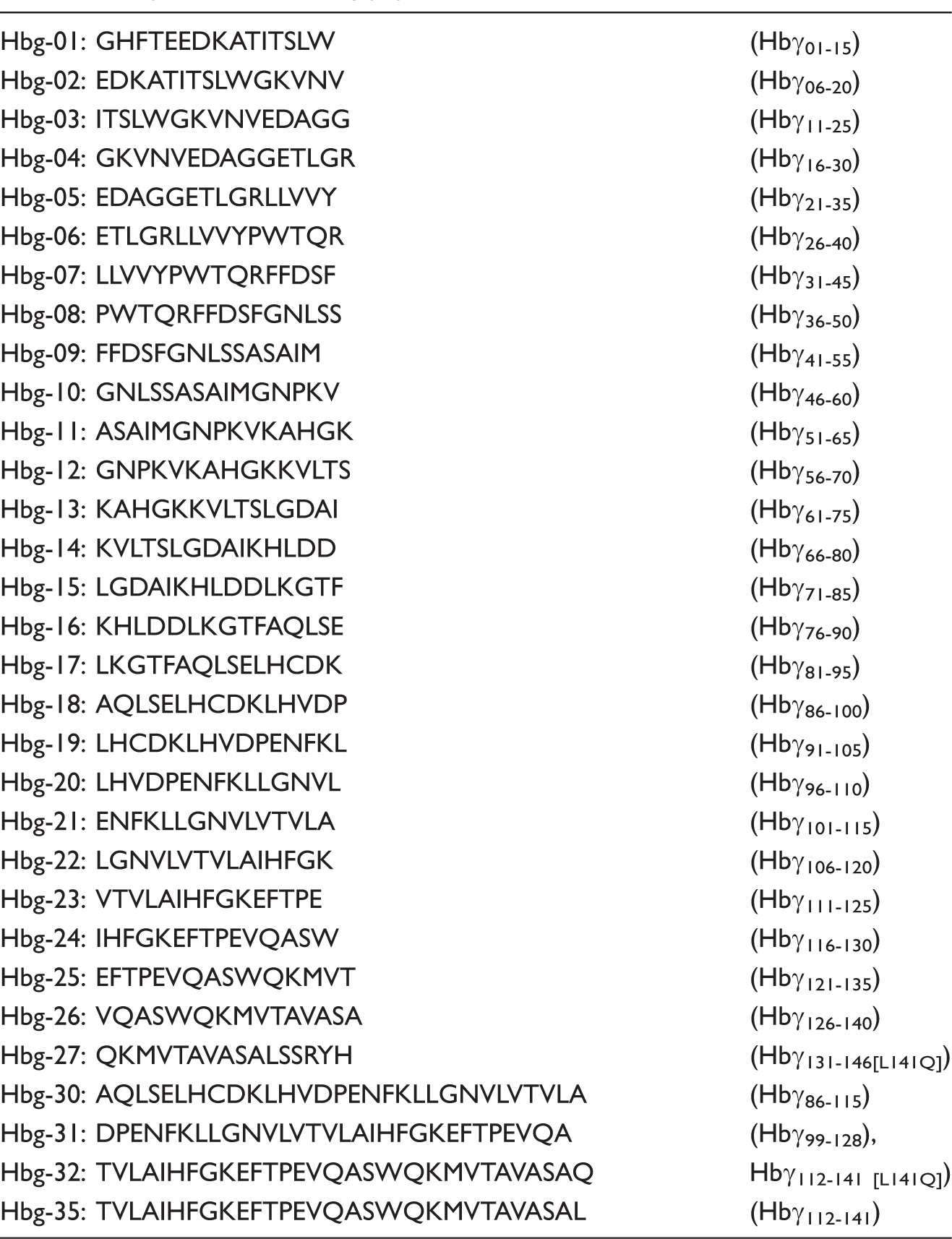
Based on the amino acid sequence of huHbg, a library of 27 overlapping synthetic peptides was generated. Several of these huHbγ-derived peptides were found to express an enhancing activity during stimulation of MNC with MPLA (not shown). In particular, peptides No. 23 (Hb γ11–125) and No. 24 (Hb γ116–130) exhibited profound enhancing effects on MPLA-induced cytokine release in human MNCs (see Table 1 for sequences). None of the native peptides expressed any cytokine-induction capacity in the absence of MPLA (Figure 2A). The partially overlapping peptides Hbg-23 and Hbg-24 showed the most prominent enhancing actions on the immunostimulatory activities of MPLA. We therefore synthesized a peptide covering the sequences of Hbg-23 and Hbg-24, VTVLAIHFGKEFTPEVQASW, which was designated as Hbg-34 (Hbγ1111–130). This peptide expressed a similar degree of amplifying the activity of MPLA as Hbg-23 and Hbg24 (not shown). Because most of the peptides derived from the C-terminal region of huHbg (peptide No. 20–27) displayed enhancing effects on the bioactivities of MPLA, three longer and partially overlapping peptides, designated Hbg-30–105 (Hbγ86–115), Hbg-31–106 (Hbγ99–128) and Hbg-32–107 (Hbγ112–141 [L141Q]) were synthesized in addition (see Table 1).
Enhancing effects of Hbg1-peptides on stimulation of human MNC with MPLA. (A) Human MNC were stimulated with MPLA (3 µg/ml) in presence or absence of the Hbg1-derived peptides Hbg-23, Hbg-24, Hbg-30–105, Hbg-31–106 or Hbg-32 (3 and 10 µM) (black columns). Control cultures were kept without MPLA (open columns). (B) Cytokine release after a culture period of 20 h, the IL-1α, IL-6, IL-8 and TNF-β concentration in the supernatant was determined by ELISA. (C) Comparison of the enhancing effects of Hbg-32 and native Hbg1 chain protein in the absence and presence of 10% human serum on stimulation of human MNC with MPLA. (D) Comparison of the enhancing effects of Hbg32 and Hbg35 on the stimulation of human MNCs by MPLA. After a culture period of 20 h, the IL-6 concentration in the supernatant was determined by ELISA. Each value represents the mean ± SD of duplicate cultures. In all figures, the horizontal broken line indicates the zero level in the absence of peptides.
Compared with Hbγ-23, the prolonged peptide Hbg-32–107 was found to display enhancing effects concerning MPLA-induced immune stimulation (Figure 2A). In previous experiments it was demonstrated that the amplifying activity of native huHbg in the LPS- or MPLA-induced release of cytokines by MNC is only observed in the absence of human serum during culture of the cells. In contrast, the synthetic peptides Hbg-23 and Hbg-32–107 showed enhancing effects in the absence, as well as in the presence, of human serum in the MPLA-induced stimulation of MNC (not shown). Our data indicate that Hbg peptides and, in particular, Hbg-32 display enhancing actions in the MPLA-mediated induction of IL-6 and IL-8 release in MNC. As shown in Figure 2B, this amplifying effect of Hbg-32 (batch 113) was not only limited to IL-6 and IL-8 release, but was also found with respect to IL-1β and TNF-α release.
Finally, we compared the enhancing effects of Hbg-32 in MPLA-induced immune activation with the amplifying actions of two preparations of the complete Hbg chain, namely Hbg-133 and Hbg-140. The experiments confirmed our prior findings that—in contrast to Hbg-32—native Hbg expresses no enhancing activity in vitro in the presence of human serum. Moreover, in comparison to native Hbg, Hbg-32–107 was found to express higher enhancing activity during stimulation of MNC by MPLA in the absence of supplemented human serum (Figure 2C). These results show that the level of enhancing activity of Hbg-32 with MPLA exceeded that of the total Hbg chain.
At this stage of our investigations, it was recognized that owing to an error in data transfer the C-terminal amino acid residue had been incorrectly selected in the synthesis of the Hbg-32-peptide. Instead of leucine (L141), as present in the native Hbg sequence, a glutamine (Q) residue had been introduced in Hbg-32. Consequently, we have synthesized a huHbg-derived peptide with the correct C- terminal amino acid residue as present in the human globin-γ chain designated as Hbg-35 (see Table 1). As we could show, the enhancing effect of this peptide on the MPLA-induced cytokine release in MNCs was very similar to that of Hbg-32 and was also found to be expressed in presence or absence of human serum (Figure 2D). It can be concluded that the C-terminal polar/neutral glutamine, as present in Hbg-32, can be replaced by the nonpolar/neutral leucine without impairing the enhancing activity of the synthetic peptide effect on the MPLA-induced activation of MNCs.
In addition to displaying amplifying effects in the stimulation of MNC by MPLA, Hbg-23 and Hbg-32 were also found to enhance the MPLA-mediated activation of purified human monocytes (Figure 3A).
Enhancing effect of Hbg-32 peptide on stimulation of human monocytes with MPLA and LPS. (A) Purified human monocytes (> 95%) were stimulated with MPLA (1 µg/ml) in presence or absence of the Hbg1-peptide Hbg-32 (1, 3 and 10 µM) (black columns). Control cultures were kept without MPLA (open columns). (B) Human MNC were stimulated with MPLA (3 µg/ml) or LPS (1 ng/ml) in presence or absence of Hbg-32 (1, 3 and 10 µM) (black columns). Control cultures were kept without MPLA or LPS (open columns). After a culture period of 20 h, the IL-6 concentration in the supernatant was determined by ELISA. Each value represents the mean ± SD of duplicate cultures.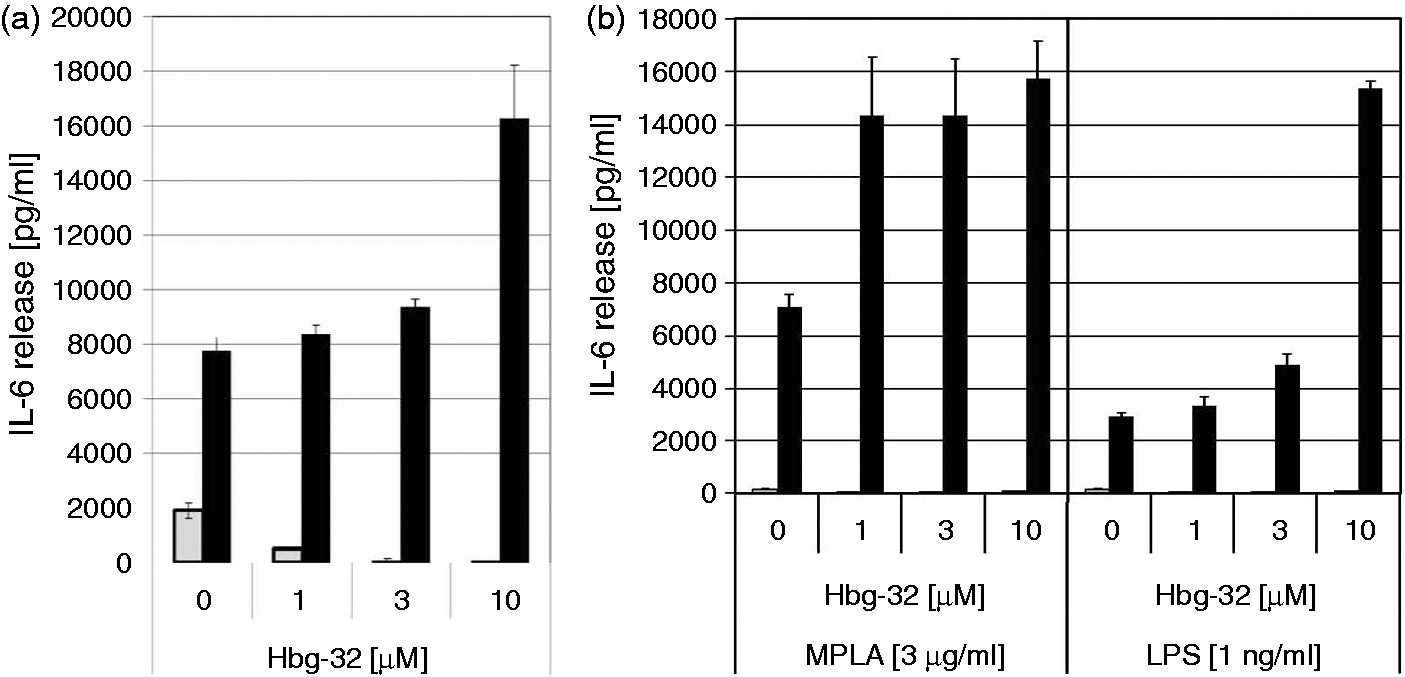
MPLA is a partial structure of LPS. We therefore investigated whether Hbg-32 also displays enhancement of MNC stimulation by complete LPS compared with the MPLA-related effects of this peptide. As shown in Figure 3B, the immunostimulatory activity of the LPS preparation was also found to be enhanced by Hbg-32.
We next investigated the combined effects of Hbg-35 of MPLA and in the activation of T-lymphocytes with recall antigens. There is a close interaction between the innate immune system, as presented by monocytes and dendritic cells, for example, and the adaptive immune system, as presented by T lymphocytes and B lymphocytes. For our investigations we used the model of activation of T-memory lymphocytes in a cell preparation of human MNC with PPD of M. tuberculosis, which is used for the tuberculin skin test. In this cell system only memory T lymphocytes respond in a specific way to these antigens. After stimulation of human MNC with PPD it was found that MPLA has a very effective adjuvant activity on the stimulation of T memory cells with the recall antigens. As little as 10 ng/ml MPLA is sufficient to exert a nearly optimal adjuvant effect (Figure 4A). Also, a marginal proliferation of T lymphocytes with MPLA alone was observed. The adjuvant effect of MPLA on PPD-induced T lymphocyte proliferation was found be enhanced by the addition of Hbg-35 (Figure 4B), indicating that the composition comprising of MPLA plus Hbg-35 also confers a higher stimulatory effect on the adaptive immune system compared with the single component formulation of the MPLA adjuvant
Adjuvant effect of MPLA during stimulation of memory T-cells. (A) Adjuvant effect of MPLA during stimulation of memory T cells with recall antigens. Human MNC were stimulated with PPD of M. tuberculosis (1 µg/ml) in the presence or absence of MPLA (10, 100 or 1000 ng/ml). (B) Enhancing effect of Hbg-35 on the adjuvant activity of MPLA during stimulation of human memory T cells with PPD. Human MNC were stimulated with PPD of M. tuberculosis (1 µg/ml) in the presence or absence of MPLA (10 ng/ml) and Hbg-35 (1 or 3 µM). After 6 d of culture, cells were labeled with 3H-Thymidine (3HTdR; 2 Ci/mmol, 0.2 µCi/culture) and after an additional day of culture the cells were harvested on glass-filter mats for measurement of incorporated radioactivity into the DNA. The results are expressed as mean ± SD of duplicate cultures.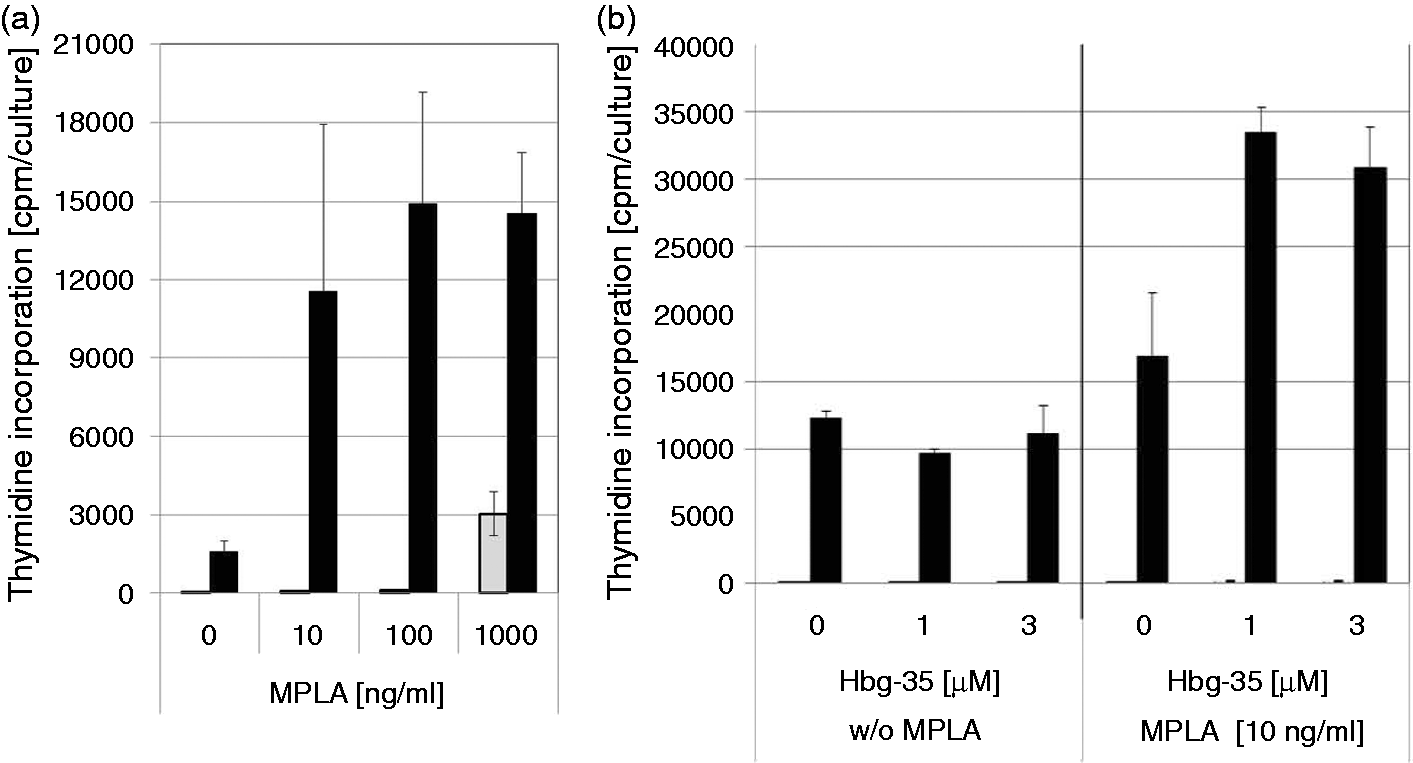
Mechanism of the enhancing effects of synthetic Hbγ-derived peptides on immunoactivation by MPLA
The biophysical assays were essentially performed employing peptides Hbg-31 and Hbg-35, shown to be highly active in enhancing the MPLA-induced cytokine release in MNCs. From these data it should be possible to investigate the molecular mechanism of the enhancing effects of the Hbg peptides on the immunostimulatory activities MPLA in more detail.
The aggregate structure of endotoxins is a determinant of its ability to induce cytokines in immune cells. This aggregate structure was characterized by performing synchrotron radiation SAXS in the absence and presence of peptides. The structure of MPLA alone was found to be largely multilamellar (Figure 5A), corresponding to an aggregation form with no or low bioactivity. This can be deduced from the occurrence of one strong peak at 4.11 nm corresponding to the distance of neighbouring reflecting bilayers of tightly packed MPLA multilamellae.
SAXS pattern of (A) MPLA alone or in the presence of (B) Hbg-32. The logarithm of the scattering intensity is plotted vs. the scattering vector s ( = 1/d, d = spacings). In the absence of the peptide, the sharp reflection at 4.11 nm indicates the existence of the multilamellar periodicity, in the presence of the peptide the occurrence of the various additional reflections is characteristic for the existence of a cubic aggregate structure.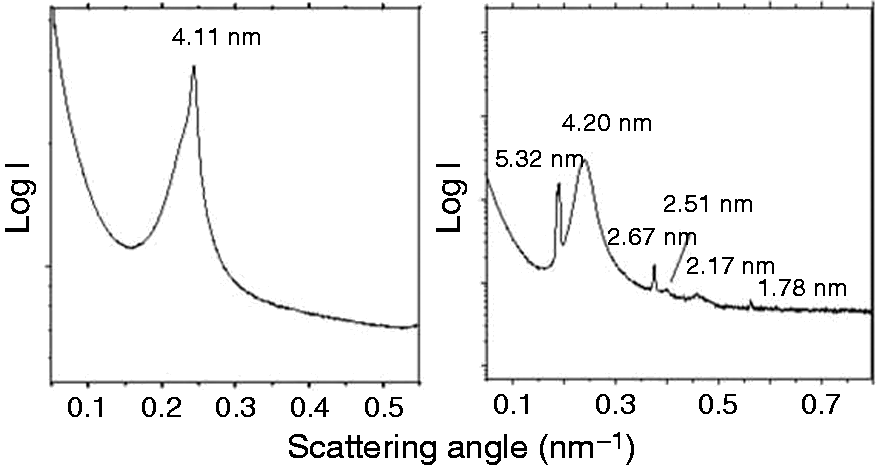
In the presence of the peptide Hbg-32, this aggregate was found to be converted into one with cubic symmetry (Figure 5B), which can be derived from the reflections at 5.26, 2.51, 2.18, 1.75 and 1.64 nm. The further reflections at 5.20 and 2.64 nm should result from the remaining lamellar phase with the periodicity dl = 5.20 nm and 2.64 nm = dl/2. Cubic phases previously have shown to represent the bioactive form of endotoxins. 31
The morphology of MPLA aggregates was studied by an independent method: freeze-fracture electron microscopy. This allows the maintaining of the structures of the lipids by shock-freezing the samples. The results for MPLA alone were indicative of very densely and lamellar packed aggregates with large sizes in the micrometer range, in accordance with the above results (Figure 6A). In the presence of the peptide Hbg-32, a strong reduction of the particles sizes was observed, accompanied by a change in morphology, leading to the dissolution of the packing arrangement into smaller size spherical-like aggregates in the range from 20 to some hundred nm (Figure 6B).
Freeze fracture electron microscopy of (A) MPLA (1 mM) alone and in the presence of (B) Hbg-32 at an equimolar concentration. In the absence of the peptide, the MPLA aggregates are densely packed forming large multi-bilayered arrangements. In the presence of the peptides, theses aggregates are dispersed and become more spherical.
AFM was employed in addition to analyze the morphology of lipid aggregates on a molecular scale under near physiological conditions. This allows depth profiles of MPLA dispersions down to a resolution limit of lower than 1 nm to be obtained. The data show for pure MPLA, spread on a mica plate, the existence of densely packed multilamellar aggregates with some hundred nm extension in the z-direction. The peptide, spread cautiously onto the lipid dispersion, interacts by only passive diffusion with the lipid aggregates. In this technique, mixing processes between the two compounds, which normally occur on a time-scale of minutes, can be slowed down to some hours, thus allowing a detailed analysis of the time kinetics of the interaction process.
As can be seen for the MPLA assembly in the presence of Hbg-32 (Figure 7A,B, recorded after 1 h) the tightly packed layers disaggregate dramatically by breaking down the multilamellae into bilayered structures. The mode of action of the peptide is its transmembraneous incorporation into the MPLA bilayers, thus reducing the sizes of the lamellar units considerably.
AFM of MPLA in absence or presence of Hbg-32. (A) AFM picture of MPLA (25 µM) on mica. Presented is the depth profile of a x − y = 5.0 × 5.0 µm2 section of the lipid aggregate. The different colors stand for the different heights of the MPLA assembly showing compact layers between z = 50 and 300 nm. (B) AFM picture of MPLA (25 µM) in the presence of an equimolar content of Hbg32 on mica. Presented is the depth profile of a 5.0 × 5.0 µm2 section of the lipid aggregate. The different colors stand for the different heights of the MPLA assembly with maximum heights up to 10 nm. The more than 90% of the plane shows bilayered structures of approximately 5-nm thickness.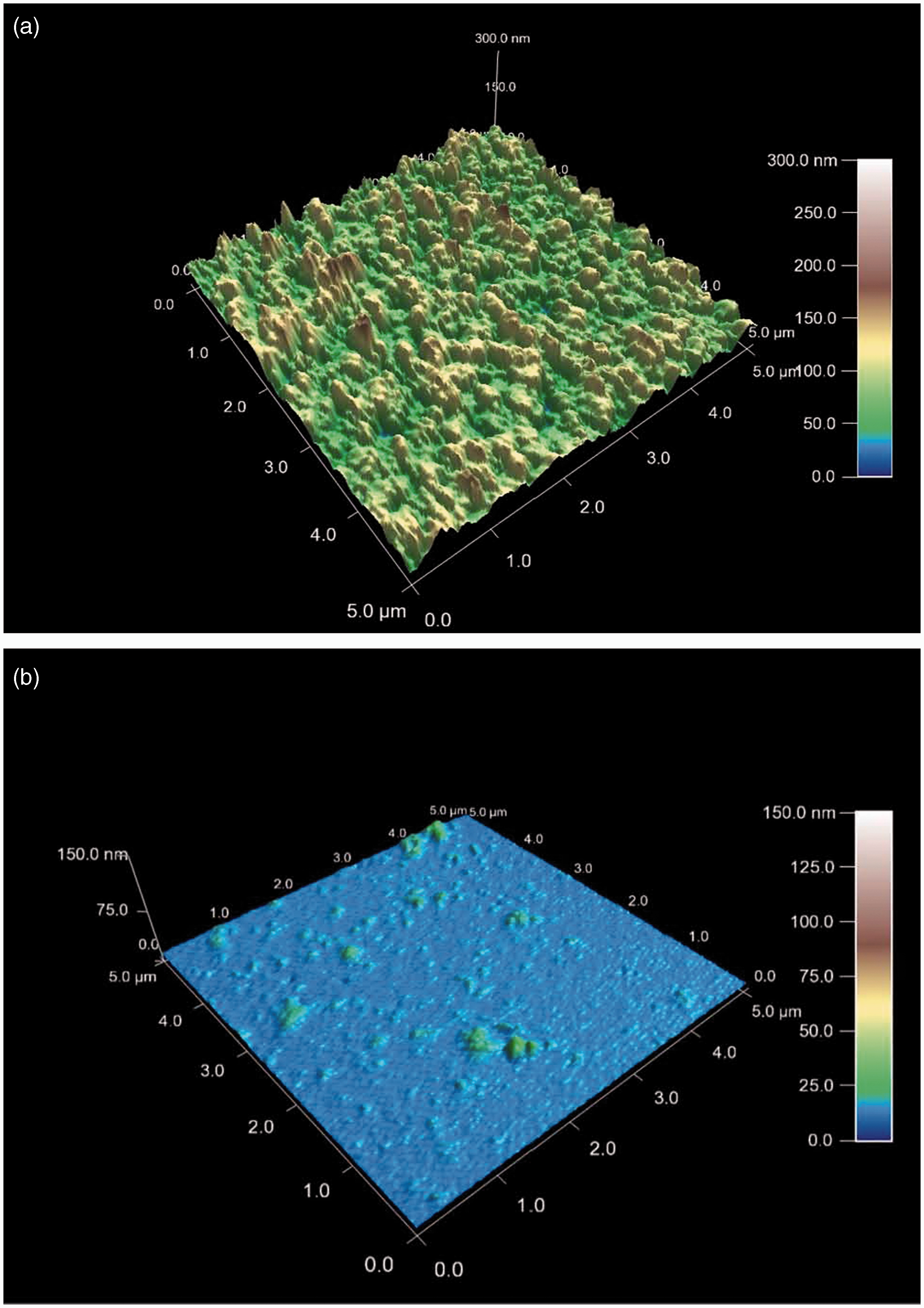
The ability of the huHbg-derived synthetic peptide to incorporate into the MPLA aggregate structures was shown to represent an important property for the enhancing effects of Hbg-32 on the cytokine-inducing activities of MPLA. FRET spectroscopy was applied to monitor the intercalation behaviour of the Hbg-32 peptide into MPLA aggregates. A considerable intercalation of the peptide into MPLA already at a [MPLA]:[Hbg-32] molar ratio of 10:1 was found, being much more pronounced at an equimolar concentration (Figure 8).
FRET spectroscopy of MPLA aggregates, to which Hbg-32 was added. The MPLA aggregates were labeled with the dyes NBD-PE (donor) and RhoPE (acceptor), and the intensity ratio ID /IA is a sensitive measure of the incorporation of an external compound such as the Hbg-32 peptide observed here. Isothermal calorimetric titration of monophosphoryl lipid A MPLA with Hbg-35. Isothermal calorimetric titration of MPLA with Hbg-35 in dependence on time (top) and the resulting enthalpy change ΔH was determined as a function of the molar ratio peptide to MPLA. The peptide was titrated in 3-µl portions every 5 min into the MPLA-containing cell under constant stirring, and the heat of interaction after each injection measured by the ITC instrument was plotted vs. time (bottom).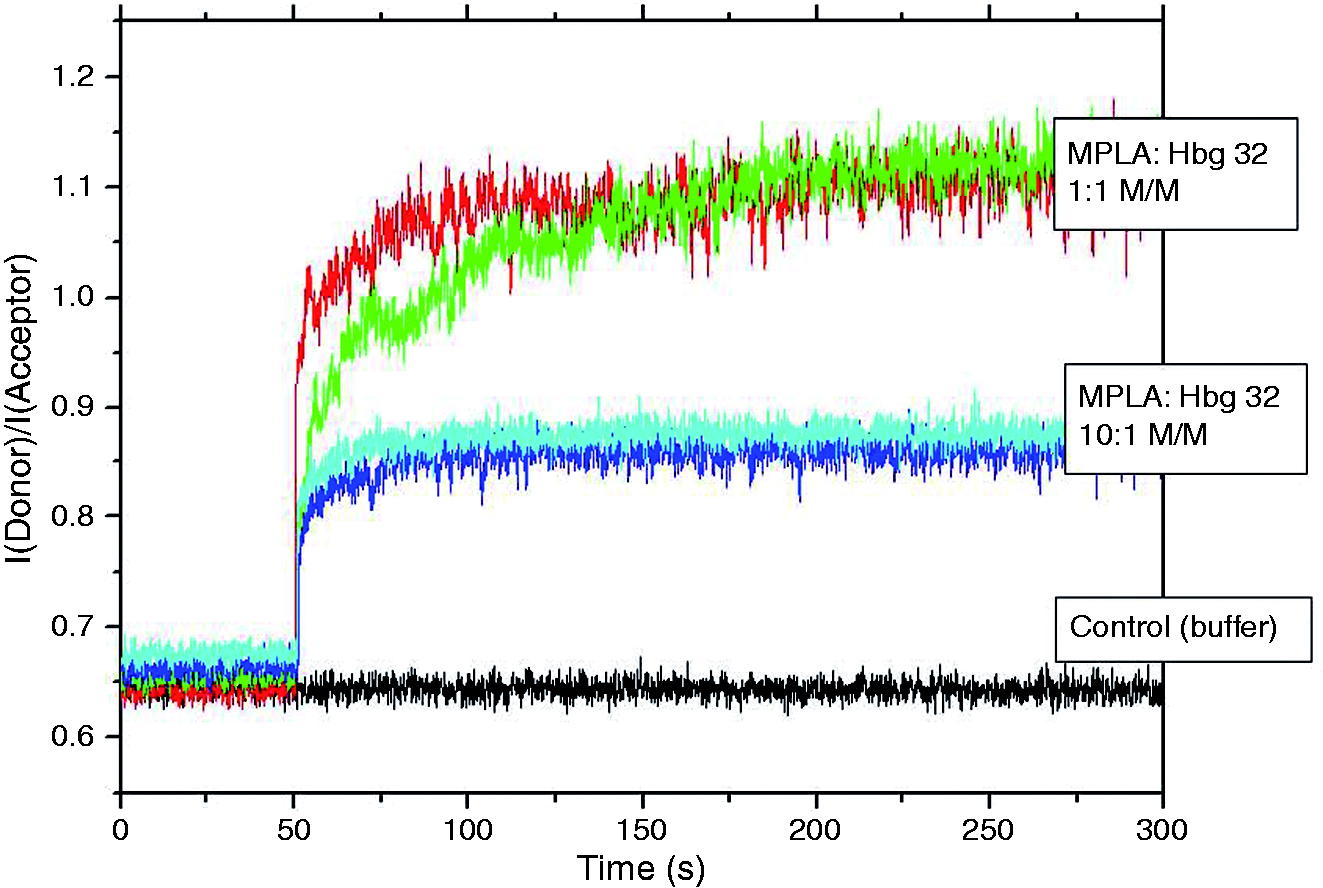
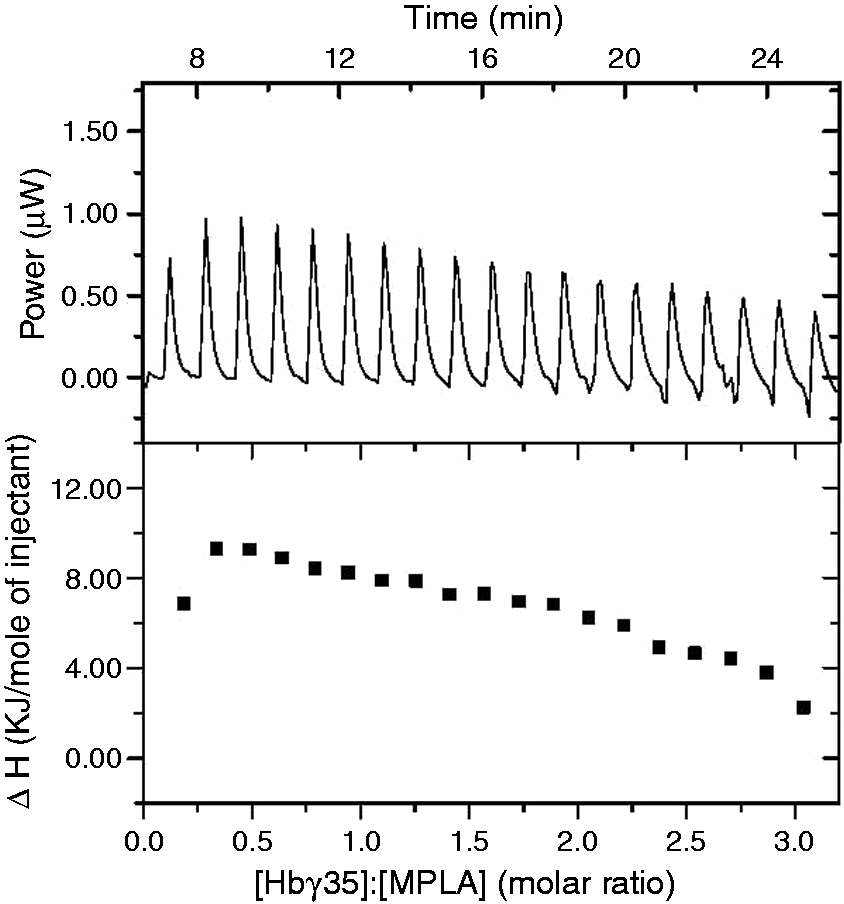
With ITC, the thermodynamics of the interaction of two different molecular species can be deduced. In the ITC analysis the molecular interactions of peptide Hbg-35 with MPLA were investigated. The data on analyzing the power of the interaction and the enthalpy change associated with varying the [Hbg-35]:[MPLA] molar ratio are shown in Figure 9. These results revealed that (i) only endothermic reactions (peaks are directed upwards) occurred; and (ii) that there was only a slight decrease of the enthalpy change vs. time and molar ratio. This observation was also found to hold true for even higher molar ratios, that is, the interaction of the peptide with the MPLA aggregates showed a non-saturable behavior. Finally, for obtaining information on the secondary structures of the peptide, Fourier transform infrared spectroscopy was applied. In Figure 10, the spectra of the Hbg35 (top) and that of a Hbg-35:MPLA mixture (bottom, 1:1 molar) are plotted. As can be seen in both figures, a main absorbance maximum around 1659 cm–1 and a second peak around 1628 cm–1 was observed, which are indicative of a main α-helical structure and a minor β-sheet component, respectively. Importantly, the addition of the lipid does not influence the peptide’s secondary structure. This was in accordance with the data presented above, as a direct binding of the peptide rather than a catalytic reaction would lead to a change of its secondary structure.
Infrared spectrum for Hbg-35 alone and in the presence of MPLA. The infrared spectrum was measured in the range of the amide I vibrational band (predominantly C = O stretching vibration) between 1700 and 1590 cm–1 for Hbg-35 alone (top) and in the presence of an equimolar content of MPLA (bottom). The absorbance maxima around 1659 and 1628 cm–1 are indicative of the existence of α-helical and β-sheet secondary structures, respectively. Statistical analysis of the effect of Hbg-35 on stimulation of human MNC with MPLA. Human MNC were stimulated with MPLA (3 µg/ml) in presence or absence of Hbg-35 (3 µM and 10 µM). After a culture period of 20 h, the IL-6 concentration in the supernatant was determined by ELISA. The data are expressed in box plots. Box plots graph data as a box representing statistical values. The boundary of the box closest to zero indicates the 25th percentile, a line within the box marks the median and the boundary of the box farthest from zero indicates the 75th percentile. Error bars above and below the box indicate the 90th and 10th percentiles. Each box represents statistical values from 12 individual experiments having duplicate cultures. **P < 0.001 as determined by paired t-test using analysis with Sigma Plot.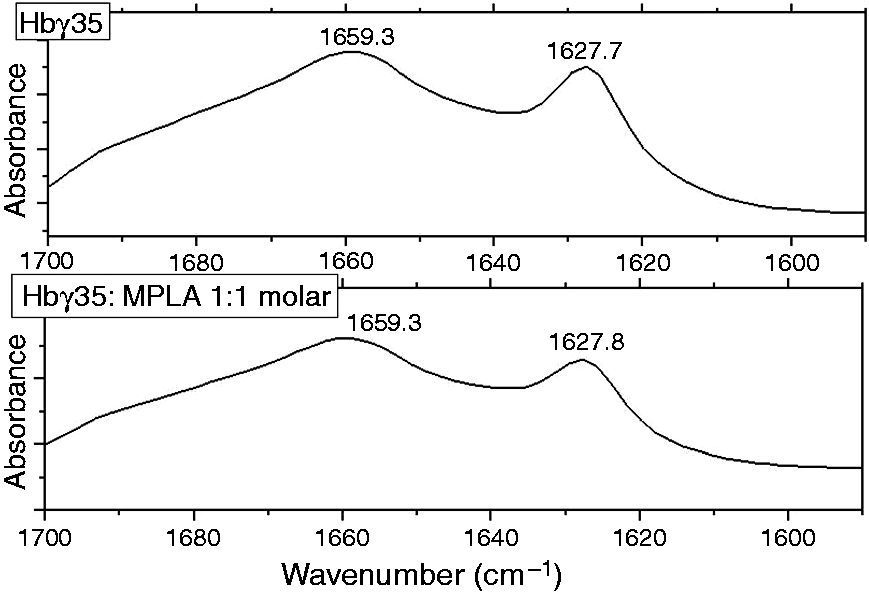
Discussion
In the present paper we have shown that selected synthetic peptides derived from the human γ-chain of Hb have the ability to increase the immunostimulatory activities of MPLA in a dose-dependent manner. These enhancing effects of the selected Hbg peptides were found in virtually all of the in vitro assays employed, that is, for the MPLA-induced cytokine release by mononuclear cells (Figure 2) and isolated monocytes (Figure 3A), as well as for the adjuvant activities of MPLA in the human memory T-cells test (Figure 4).
The increase of diverse biological activities of LPS by HbA and substructures thereof is a phenomenon described by Levin and co-workers in the 1990s. These effects comprised stimulation of immune cells, increase of reactivity in the Limulus test and endothelial cell tissue factor, and enhanced lethal toxicity in animals.32–35 Interestingly, it was even observed that biologically inactive samples such as penta-acyl LPS and tetra-acyl lipid A could convert into a biologically active form in the presence of α,α-cross linked Hb, 21 as evidenced by the increase in TNF-α secretion of human MNC, and this holds true also for other penta-acylated compounds such as those from Chlamydia trachomatis, and is even found for particular non-LPS amphiphiles, a bacterial lipopeptide and a lipid A-like phospholipid. 18 The principles that are responsible for these increases of activity, are characterized by the following major rules: (1) cell activation by LPS, MPLA and the other amphiphiles occurs via highly aggregated samples; (2) LPS and other biologically active amphiphiles have a non-lamellar, essentially cubic, inverted aggregate structure; (3) biologically inactive LPS and other amphiphiles with penta- or tetra-acylated lipid A or MPLA have a multilamellar aggregate structure.
The increase in bioactivity leads to a disaggregation of the amphiphiles. Concomitantly, there is an increase in the expression of non-lamellar aggregate structures. It should be noted that these facts refer to the initial step of cell activation, whereas there is experimental evidence that the final step of signal transduction could be a monomeric endotoxin bound to the TLR4/MD2 complex. 36
As indicated by the panel of biophysical tests employed in this study (Figures 5–10), these processes apparently also play a decisive role in the enhancing effects of HbF and the corresponding set of synthetic peptides on the immunoactvities of MPLA, which, per se, was found to display only weak to moderate in vitro activities compared with LPS (Figures 2–4). Transition of a multilamellar into a non-lamellar phase (Figure 5), dispersion of the highly aggregated states (Figure 6) leading to very small aggregates, probably also into monomers (Figure 7). The AFM data (Figure 7), the electron microscopy data with freeze-fracture (Figure 6) and the SAXS experiments (Figure 5) now give a clear picture of the observation that the peptides cause an increase in biological activity. The considerably smaller MPLA particles formed in the interactions with HbF, the Hb gamma globin chain or the corresponding synthetic peptides may bind in a more efficient manner to serum and membrane proteins such as CD14 and LBP, and may, in particular, provide increased levels of monomerized MPLA to the TLR4/MD-2 signaling system compared with the larger aggregates of MPLA present in the original aqueous formulation of this pivotal immunostimulant and candidate vaccine adjuvant. The interaction process of MPLA with the peptide is connected with a strong intercalation of the peptide molecules into the aggregates (Figure 8). This interaction is non-saturable and endothermic (Figure 9), and corresponds to a more catalytic rather than to a typical binding reaction, which would form a complex. This is in contrast to the action of antimicrobial peptides with endotoxins, for which exothermic reactions take place, leading to saturation of binding. 25 Interestingly, in the case of inhibiting peptides such as Pep19-2.5,25,29 these also incorporate into target membranes such as LPS and/or phospholipid containing bilayers but not as a transmembraneous step as in the case of the huHbg peptides.
The reasons for the activity-enhancing property of the human Hbg peptides in contrast to the activity-decreasing action of the Pep19 series could also be connected to the secondary structures of the peptides, with a mainly α-helical structure of the former and a pure β-sheet structure of the latter (Figure 10, see also Heinbockel et al. 22 ). These also remain invariant in the presence of the binding partners. The disaggregation of the tight MPLA stacks seems only be possible with helical structures spanning the membranes and thus fragmenting them, as seen in Figure 7, while β-sheet structures bind to the LPS surfaces with a penetration of the C-terminal end sticking into the membrane.
Footnotes
Acknowledgements
We gratefully acknowledge the technical assistance of Nina Grohmann, Nina Hahlbrock and Suhad Al-Badri.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. Financial support was kindly provided by Clinique La Prairie (Clarens, Switzerland).
