Abstract
Monocytes and neutrophils are important players in the innate immune response and cooperate during infection and inflammation. In our study we analyzed the effects of neutrophil degranulation products (polymorphonuclear granulocytes degranulation products, PMN-DGP) on the activation, the adhesion and the migration of three bovine monocyte subsets, as well as their effects on monocyte-macrophage differentiation. Cross-linking of surface CD18 molecules on bovine PMN resulted in the release of primary, secondary and tertiary granules as well as of secretory vesicles. PMN-DGP induced a significant Ca2+-influx in classical (classical monocytes, cM) and intermediate monocytes (intermediate monocytes, intM) but not in non-classical monocytes (non-classical monocytes, ncM). A selective and up-regulated expression induced by PMN-DGP was only seen for CD11a and CD31 on intM. PMN-DGP induced a selective migration of intM in vitro. The presence of PMN-DGP during the differentiation of cM or intM into macrophages resulted in increased expression of membrane CD163 and reduced expression of MHC-II molecules. PMN-DGP-derived macrophages produced more IL-12 and IL-10 and showed enhanced phagocytosis and ROS production capacities. In conclusion, PMN-DGP selectively attract bovine intM and skew the functional maturation of cM and intM.
Introduction
Monocytes and neutrophils are important players in the immune response and cooperate during infection and inflammation. Blood monocytes are a heterogeneous population of circulating cells with distinct phenotypic and functional properties.1,2 Based on the cell surface expression of CD14 and CD16, human 1 and bovine 2 monocytes are subdivided into classical (cM, CD14++ CD16-), intermediate (intM, CD14++ CD16+) and non-classical (ncM, CD14+ CD16++) monocyte subsets.
Recent studies have shown that human and mouse ncM in the steady-state crawl along the endothelium of blood vessels and thereby take a patrolling function of healthy tissues.3,4 In response to tissue damage or infection, these patrolling monocytes have been shown to rapidly migrate out of the circulation and invade the damaged site. However, this early efflux of patrolling ncM has been shown to be transient and these cells are rapidly replaced by recruited neutrophils.5,6
Early recruited neutrophils are believed to contribute to the recruitment of a second wave of monocytes by several mechanisms including the release of neutrophil granule proteins.7-9 Recent research in human and mouse provides evidence for the importance of PMN granule proteins in the interaction with distinct monocyte subsets. 8 It has been demonstrated that PMN-DGP specifically induce the adhesion and recruitment of human and murine classical monocytes.7,10
During the migration from blood to tissue, PMN sequentially release their different granule types, via which they communicate with other immune cells, in particular monocytes and macrophages.11-15 These granule types, stored in 4 different compartments, differ in their ability to be mobilized. Secretory vesicles have been shown to be released upon the interaction of neutrophil with endothelial cells, tertiary granules are discharged during neutrophil trans-endothelial migration. Being only released once the neutrophil has been activated in the extravascular tissue, primary and secondary granules are the last granule types released. 16
Following extravasation, the differentiation of monocytes into macrophages can be guided by different local mediators like cytokines, chemokines and microbial products resulting in different functional macrophage subtypes. 17 Recent studies in human have revealed a polarizing effect of degranulation products of human neutrophils on the differentiation of human monocytes into macrophages. 18
As bovine monocyte subsets display distinct functional differences compared to their human and murine counterparts,2,19 we addressed the question whether bovine monocyte subsets differ in their responsiveness to PMN-DGP.
Material and methods
All procedures involving animals were carried out in accordance with German legislation on animal welfare. The commissioner for animal welfare at the University of Veterinary Medicine, Hannover, Germany, approved the mentioned procedures and permission was granted by the Niedersächsisches Landesamt für Verbraucherschutz und Lebensmittelsicherheit (LAVES).
Animals and blood separation
Blood was obtained from non-pregnant, non-lactating healthy German Holstein cows (10 cows aged between 3–14 yr) housed in the Clinic for Cattle, University of Veterinary Medicine, Hannover, Germany. Used animals were not within the transition period. Blood was drawn into heparinized vacutainer tubes from the vena jugularis externa.
Separation of bovine mononuclear cells (MNC) and neutrophils (PMN)
Bovine MNC and PMN were separated according to Sipka et al. 20 Briefly, blood was layered on Ficoll-Isopaque (PAA, Pasching, Austria) and centrifuged at 10℃ for 30 min at 1000 g. The interphase containing MNC was washed three times in PBS (500 g, 200 g and 100 g, 4℃). To separate bovine PMN, the PMN-erythrocyte containing phase was suspended in distilled water for 20 s and adding double concentrated PBS restored tonicity. This was repeated (usually twice) until complete erythrolysis. PMN purity assessed flow cytometrically according to their FCS/SSC properties always exceeded 90%. The mean viability of PMN as determined by exclusion of propidium iodide (2 µg/ml, Calbiochem, Germany) ranged 92% ± 4%. PMN were finally suspended in Hank's balanced salt solution (HBSS) at 5 × 106 cells/ml.
Flow cytometric analysis of adhesion molecules on monocyte subsets
Peripheral blood mononuclear cells (4 × 105) from healthy cows were incubated with mAbs specific for bovine CD62L (clone CC32, 1:100 final), bovine CD11b (clone CC126, 1:100 final), sheep CD31 (clone CO.3E1D4, 1:100 final), human CD49d (clone HP2/1, 1:100 final), bovine CD18 (clone HuH82A, 1:100 final) (all from AbD Serotec; cross-reactive with homologous bovine molecules) or bovine CD11a (clone HUH73A; Kingfisher Biotech, USA, 1:100 final) in PBS containing BSA (5 g/l) and NaN3 (0.1 g/l). After 30 min incubation (4℃), cells were washed once and incubated (30 min in the dark, 4℃) with a secondary RPE-conjugated goat anti-mouse IgG Ab (IgG, Clone pAK, Dianova). Labeled cells were washed once and incubated with human CD14 Ab (clone TÜK4, Alexa Fluor 647, 1:50 final) and human CD16 Ab (clone KD1, FITC, 1:200 final). After 30 min in the dark (4℃), labeled cells were washed once and analyzed flow cytometrically (BD Accuri C6 flow cytometer, BD Biosciences). Dead cells were excluded from the analysis by adding propidium iodide (PI, 2 µg/ml). At least 100 000 MNC or 20 000 monocytes were collected and analyzed with the CFlow Software, Version 1.0.264.15 (BD Biosciences).
Degranulation of bovine neutrophils
For the preparation of PMN-DGP, degranulation was induced through Ab cross-linking of the integrin β2-chain CD18 as described before. 21 Isolated bovine PMN were incubated with murine mAbs against CD18 (2 μg per 106 PMN). We first tested three different clones of primary CD18-specific mAbs for their ability to activate bovine PMN upon cross-linking with a goat anti-mouse IgG F(ab′)2. MAb clones tested were: mouse anti-dog CD18 (clone CA1.4E9; AbD Serotec) and mouse anti-bovine CD18 (clone HuH82A and BAQ30A; Kingfisher, USA). After incubation for 30 min at room temperature (RT, 20℃), cells were washed twice to remove unbound Ab and were suspended in HBSS. Pretreated PMN were then subjected to CD18 cross-linking by addition of goat F(ab′)2 anti-mouse IgG (diluted 1/20; Jackson ImmunoResearch Laboratories). Alternatively, separated PMN were stimulated with ionomycin (1 µg/ml, Sigma chemicals). After 10 min incubation at 37℃ PMN were centrifuged (300 g, 10 min) and cell-free supernatant containing PMN-DGP was collected and stored at –20℃ until use. For shape-change analysis (change of forward scatter), PMN were suspended in HBSS and analyzed flow cytometrically [at least 30.000 viable PMN (PI negative) were acquired, BDAccuri™ C6 flow cytometer, BD Biosciences].
Analysis of PMN degranulation
For the analysis of neutrophil degranulation the marker enzymes myeloperoxidase (MPO), lactoferrin, and alkaline phosphatase were assessed in the supernatant of activated neutrophils as markers of azurophilic, specific and tertiary granules, respectively. The release of PMN secretory vesicles was analyzed flow cytometrically (at least 30.000 viable PMN were acquired) by determining CD16 expression on activated neutrophils after labeling the cells with a murine mAb specific for CD16 (see below). For determining alkaline phosphatase enzymatic activity, 50 µl PMN supernatant per well of a 96 well ELISA plate was incubated with 50 µl of p-nitrophenyl phosphate (pNPP; Interchim, France) solution (1 mg/ml in assay buffer: 100 mM Tris-HCl, pH 9.5, 100 mM NaCl, 5 mM MgCl2). After 15 min incubation at RT, the reaction was stopped by the addition of 50 µl of 2 N NaOH, and the plates were read in an ELISA microplate reader (ELX 800, BioTek, Germany) at 405 nm. The activity of myeloperoxidase (MPO) was determined according to a previously described method. 22 Briefly, 50 μl PMN supernatant per well of a 96 well ELISA plate was incubated with 100 μl/well peroxidase substrate buffer (33.3 mmol/l citric acid, 66.7 mmol/l NaH2PO4, pH 5.0) supplemented with 130 µg/ml 3,3', 5,5'-tetramethylbenzidine (TMB, Sigma, Germany) and 0.01% (v/v) H2O2 (Sigma-Aldrich, Germany). After 20 min at RT in the dark the addition of 75 μl 0.5 M H2SO4 stopped the reaction. Optical densities were read in an ELISA microplate reader (ELX 800, BioTek, Germany) at 450 nm and 630 nm.
For measuring lactoferrin release in the cell-free neutrophil supernatants a commercially available ELISA kit for bovine lactoferrin (Bovine Lactoferrin ELISA Quantitation Set, Bethyl Laboratories, Montgomery, TX) was used. Briefly, 100 μl of diluted coating Ab (4 µg/ml in coating buffer: 50 mM carbonate-bicarbonate, pH 9.6) was added to each well of 96-well ELISA-plate and incubated for 1 h. Plates were washed five times with phosphate buffer [2.5 mmol/l NaH2PO4, 7.5 mmol/l Na2HPO4, 145 mmol/l NaCl, 0.1% (v/v) Tween 20, pH 7.2] and each well was filled with 100 μl of the cell–free supernatant from stimulated PMN. Serial dilutions of recombinant bovine lactoferrin were included in each assay to construct a standard curve. After incubation (1 h, RT) plates were washed five times with phosphate buffer and wells were filled with 100 μl of diluted horseradish peroxidase (HRP) detection Ab (goat anti bovine lactoferrin). After 1 h at RT the plates were washed and subsequently filled with 75 μl/well peroxidase substrate buffer (33.3 mmol/l citric acid, 66.7 mmol/l NaH2PO4, pH 5.0) supplemented with 130 µg/ml TMB and 0.01 % (v/v) H2O2 (all chemicals from Sigma-Aldrich, Germany). After 20 min at RT in the dark addition of 75 μl 0.5 M H2SO4 to each well stopped the reaction. Optical densities were read in an ELISA microplate reader (ELX 800, BioTek, Germany) at 450 nm and 630 nm. Bovine lactoferrin concentrations were calculated by referring to the standard curve.
Separation of bovine monocyte subsets
Bovine monocyte subsets were separated as described recently. 2 Briefly, after density gradient separation of heparinized blood, mononuclear cells were labeled with a FITC-conjugated CD16-specific Ab (clone KD1, AbD Serotec, 1:30 final) for 15 min (4℃) and subsequently with anti-FITC MultiSort MicroBeads (Anti-FITC MultiSort Kit; Miltenyi Biotec, 8 µl/1 × 107 MNC for 20 min, 4℃) to separate MNC into a CD16-positive fraction (containing intM and ncM) and a CD16-negative fraction (containing cM and lymphocytes) using a magnetic column. To release the anti-FITC MultiSort MicroBeads from the selected CD16-positive fraction, cells were incubated with the MultiSort release reagent (included in the anti-FITC MultiSort Kit) for 10 min. CD16-positive cells (positively selected) and CD16-negative cells (in the flow throw) were subsequently incubated with anti-CD14 MicroBeads (Miltenyi Biotec, 10 µl/1 × 107 cells for 20 min, 4℃) and separated according to the manufacturer's instructions into 3 monocyte subsets. CD14-positive cells were positively selected from the CD16-negative fraction (cM) and the CD16-positive fraction (intM). Remaining cells in the flow throw of the CD16-positive fraction presented ncM being CD14-negative and CD16-positive. All steps of monocyte subset isolation were performed at 4℃. After each labeling step, the cells were washed with PBS-EDTA (300 g, 10 min). Negatively and positively selected cells, being already labeled with CD16-FITC Abs during the separation process, were checked flow cytometrically for their viability and purity after labeling the cells with a PE-conjugated mouse anti bovine CD14 mAb (clone TÜK4, AbD Serotec) and adding propidium iodide (2 µg/ml). This separation procedure yielded a purity of 94.9 % ± 5.4% for cM, 89.9% ± 4.5% for intM and 93.3% ± 2.2% for ncM. The mean viability was 92% ± 5% for all subsets.
For the separation of CD14-positive monocytes (including cM and intM) bovine MNC were incubated with CD14-MicroBeads (Miltenyi Biotec) and MACS-separation was performed according to the manufacturer's instructions.
Ca2+ influx in monocyte subsets
Ca2+ influx in bovine monocyte subsets was analyzed according to Hussen et al. 19 Briefly, bovine MNC in PBS were labeled with mouse anti-bovine CD14-Alexa647 (Tük4, 1:125 final) and mouse anti human CD16-PE (KD1, 1:200 final) to define monocyte subsets (all Abs were from AbD Serotec). Cells were washed two times in 5 ml of membrane immunofluorescence (MIF) buffer [PBS containing BSA (5 g/l) and NaN3 (0.1 g/l)] (300 g, 4 min, 4℃) and suspended in HBSS without phenol red (5 × 106/ml). Cell suspension (1 ml) was incubated with 2 µl of the calcium-sensitive fluorochrome Fluo 4 (final 1:10 in PBS, 100 nmol/l, Invitrogen, Karlsruhe, Germany) (30 min, 37℃). After two washings in HBSS (300 g, 6 min, RT) cells were adjusted to 5 × 106/ml and 100 µl was transferred to 1.5 ml Eppendorf tubes containing propidium iodide (100 µl, 2 µg/ml) to exclude dead cells. Green fluorescence was monitored flow cytometrically. Twenty s after start of measurement (baseline), cells were stimulated with 100 µl PMN supernatant and flow cytometric assessment continued for 2 min. The response towards ionomycin (1 µg/ml final, Sigma-Aldrich, Germany) served as a positive control, supernatant from PMN cultures in HBSS served as negative control.
In forward (FSC) versus side scatter (SSC) dot plots, MNC were gated based on their characteristic scatter properties and monocytes subsets were defined by gates in a CD14/CD16 dot plot. Green fluorescence (FL1) was plotted against time. Using a quadrant, a horizontal line was adjusted above the FL1 baseline. Directly after addition of stimuli, the percentage of cells above the horizontal line served as a measure for Ca2+ influx in bovine monocytes.
In vitro migration assay of monocyte subsets
Monocyte migration was evaluated in a transwell system (6.5 mm-diameter polycarbonate membranes with 5.0 µm-diameter pores; 24 well plate, Corning). MACS-separated bovine monocyte subsets (2 × 105 in 200 µl DMEM medium without serum) were placed in the upper chambers. The lower chambers were filled with 500 µl PMN supernatant. After 4 h incubation at 37℃, non-migrating cells were collected from the upper and migrating cells from the lower chamber. After counting the cells flow cytometrically, 23 migration rates 24 [the percentage of migrating cells in the lower chamber of total recovered cells (sum of cells in the lower and the upper chamber)] were calculated and presented for each subset.
Generation of monocyte-derived macrophages (MdM) in vitro
Monocyte subsets after MACS separation were suspended in Iscové medium supplemented with 100 U/ml penicillin/streptomycin and 10% FCS, adjusted to 2 × 105 cells/ml and seeded in 24-well plates (2 × 105 cells/well, Fisher Scientific GmbH, Schwerte, Germany) for 24 h at 37℃ (5% CO2 in air). After this resting period, culture medium was removed and cells were further incubated without or with cell-free supernatant containing PMN-DGP throughout the in vitro culture period. On d 4 of culture, PMN-DGP was removed and fresh medium was added to the cells. Day 4 MdM were further incubated for 4 h at 37℃ (5% CO2 in air) and the supernatant was collected after plate centrifugation at 300 g for 10 min.
Immunophenotyping of monocyte-derived macrophages
Monocyte-derived macrophages (2 × 105) were incubated with the fluorochrome-conjugated mAbs, mouse anti porcine CD163 (clone 2A10/11, 1:50 final) and mouse anti-ovine MHC-II (clone 37.68, 1:40 final) in PBS containing BSA (5 g/l) and NaN3 (0.1 g/l). Both Abs are cross-reactive with homologous bovine molecules (AbD Serotec). After 30 min in the dark (4℃), labeled cells were washed once and analyzed flow cytometrically. Dead cells were excluded from the analysis by adding propidium iodide (PI, 2 µg/ml, Calbiochem, Germany). At least 10 000 macrophages were collected and analyzed with the CFlow Software, Version 1.0.264.15 (BD Biosciences).
Cytokine ELISA
Previously described cytokine ELISA procedures were used to detect bovine IL-1225 and IL-1026 in macrophage culture supernatants. Briefly, 96-well microtiter plates were coated with 100 µl (4 µg/ml) mouse anti-bovine IL-12 (clone CC301, AbD Serotec) or IL-10 (clone CC318, AbD Serotec) overnight (18 h) at 4℃. Plates were washed with PBS Tween 20 (0.05%) and blocked with 150 µl fish skin gelatin (2%) for 1 h at 25℃. Culture supernatants (100 µl) were added to the plates. Bovine IL-12 or IL-10 protein standard (Innovative research, USA) was serially diluted and added (100 µl) to the wells. Plates were incubated for 1 h at 25℃. After washing with PBS Tween 20 (0.05%), plates were incubated with 75 µl (2 µg/ml) of biotin-labeled mouse anti-bovine IL-12 (clone CC326, AbD Serotec) or biotin-labeled mouse anti-bovine IL-10 (clone CC320, AbD Serotec) at 37℃ for 1 h. Plates were washed, and streptavidin HRP (1:1000 in blocking buffer, GE Healthcare Lifesciences, UK, 100 µl) was added. After 1 h at RT the plates were washed and subsequently filled with 75 μl/well peroxidase substrate buffer (33.3 mmol/l citric acid, 66.7 mmol/l NaH2PO4, pH 5.0) supplemented with 130 µg/ml TMB and 0.01% (v/v) H2O2 (all chemicals from Sigma-Aldrich, Germany). After 20 min at RT in the dark the addition of 75 μl of 0.5 M H2SO4 to each well stopped the reaction. Optical densities were read in an ELISA microplate reader (ELX 800, BioTek, Germany) at 450 nm and 630 nm. Bovine IL-12 and IL-10 concentrations were calculated by referring to the standard curves.
Phagocytosis assay
Phagocytosis was analyzed by flow cytometry after incubation of macrophages with FITC-labeled Escherichia coli. Labeling of E. coli was performed as described previously 27 with modifications. Briefly, E. coli (Institute of Microbiology, University of Veterinary Medicine, Hannover, Germany) were inoculated into broth and incubated overnight at 37℃ in a shaker. The bacteria were washed three times in PBS and fixed for 1 h at 4℃ with 4% paraformaldehyde in PBS. Fixed bacteria were incubated for 12 h with FITC (Sigma-Aldrich, St. Louis, Missouri, USA) (25 µg/ml). Bacterial cell count and labeling efficiency ( ≥ 80%) were determined flow cytometrically. Day 4 macrophages, after washing off the PMN-DGP, were plated in 96 well plates (1 × 105/well) and incubated with non-opsonized bacteria (25 bacteria/cell) for 40 min (37℃, 5% CO2). After incubation, cells were washed (300 g for 10 min) with PBS. To exclude dead macrophages and adherent bacteria from the analysis, PI (2 µg/ml final), which is unable to pass through the plasma membrane of viable phagocytic cells, was added to the cells. Phagocytosis activity was defined flow cytometrically as the percentage of green fluorescing cells among viable cells. At least 20.000 viable macrophages were acquired.
Generation of reactive oxygen species (ROS)
ROS generation was induced in 96-well round-bottom microtiter plates (Corning, NY, USA). Day 4 macrophages (1 × 105/well) in RPMI medium were incubated with killed non-opsonized E. coli bacteria. Day 4 macrophages, after washing off the PMN-DGP, were stimulated with bacteria (50 bacteria/cell) for 15 min (37℃, 5% CO2). For the detection of ROS, dihydrorhodamine 123 (DHR 123, Mobitec, Goettingen, Germany) was added to the cells (750 ng/ml final). After incubation, cells were washed with RPMI medium and suspended in medium containing PI (2 µg/ml final) to exclude dead cells from the analysis. The relative amount of generated ROS was determined flow cytometrically by the median green fluorescence intensity of gated cells after acquisition of 20 000 viable macrophages.
Statistical analyses
Statistical analysis was carried out using the software Prism (GraphPad software). Results are expressed as means ± S.E. of the mean (SEM). Differences between means were tested with t-test (comparison between two groups) or the one-factorial analysis of variance (ANOVA) (comparison between more than two groups) and Bonferroni's correction for normally distributed data. Results were considered statistically significant at a P-value of less than 0.05.
Results
Cross-linking of CD18 on bovine neutrophils induces their degranulation
To collect PMN-DGP, PMN-degranulation was induced through Ab cross-linking of surface CD18 molecules using a CD18 primary Ab and a secondary goat anti-mouse IgG F(ab′)2-fragment.
21
Cellular activation of PMN was analyzed by flow cytometric assessment of shape change.
28
Shape change was assessed as the change in median FSC of PMN. Based on side scatter parameters, a gate was drawn to exclude doublets (SSC-A vs. SSC-W). While no significant differences could be seen between the median FSC of PMN in HBSS (1.98 ± 0.01 × 106) and those of PMN incubated with the primary CD18 Ab alone (1.99 ± 0.02 × 106) or with the secondary goat anti-mouse IgG F(ab′)2 Ab alone (1.99 ± 0.01 × 106), cross-linking of CD18 Ab with the secondary goat anti-mouse IgG F(ab′)2 Ab resulted in increased median FSC (2.19 ± 0.02× 106) indicating a shape change of bovine PMN (Figure 1a).
Different activating capacities of CD18-specific mAbs for bovine PMN. Bovine PMN were incubated with three different mouse mAbs specific for CD18. Bound mAbs were cross-linked after addition of goat anti-mouse IgG F(ab′)2 for 10 min. Alternatively, PMN were stimulated with ionomycin for 10 min or left completely untreated. PMN activation was analyzed by flow cytometric assessment of their shape change after drawing a gate to exclude doublets (SSC-A vs. SSC-W). (a) Representative dot plots of one animal showing a shift in the FSC of activated PMN resulting in a higher percentage of cells in the lower right quadrant. (b) The mean forward scatter (FSC A) of PMN was assessed and data of 7 tested animals were presented as mean ± SEM (* = P < 0.05 in comparison to HBSS control).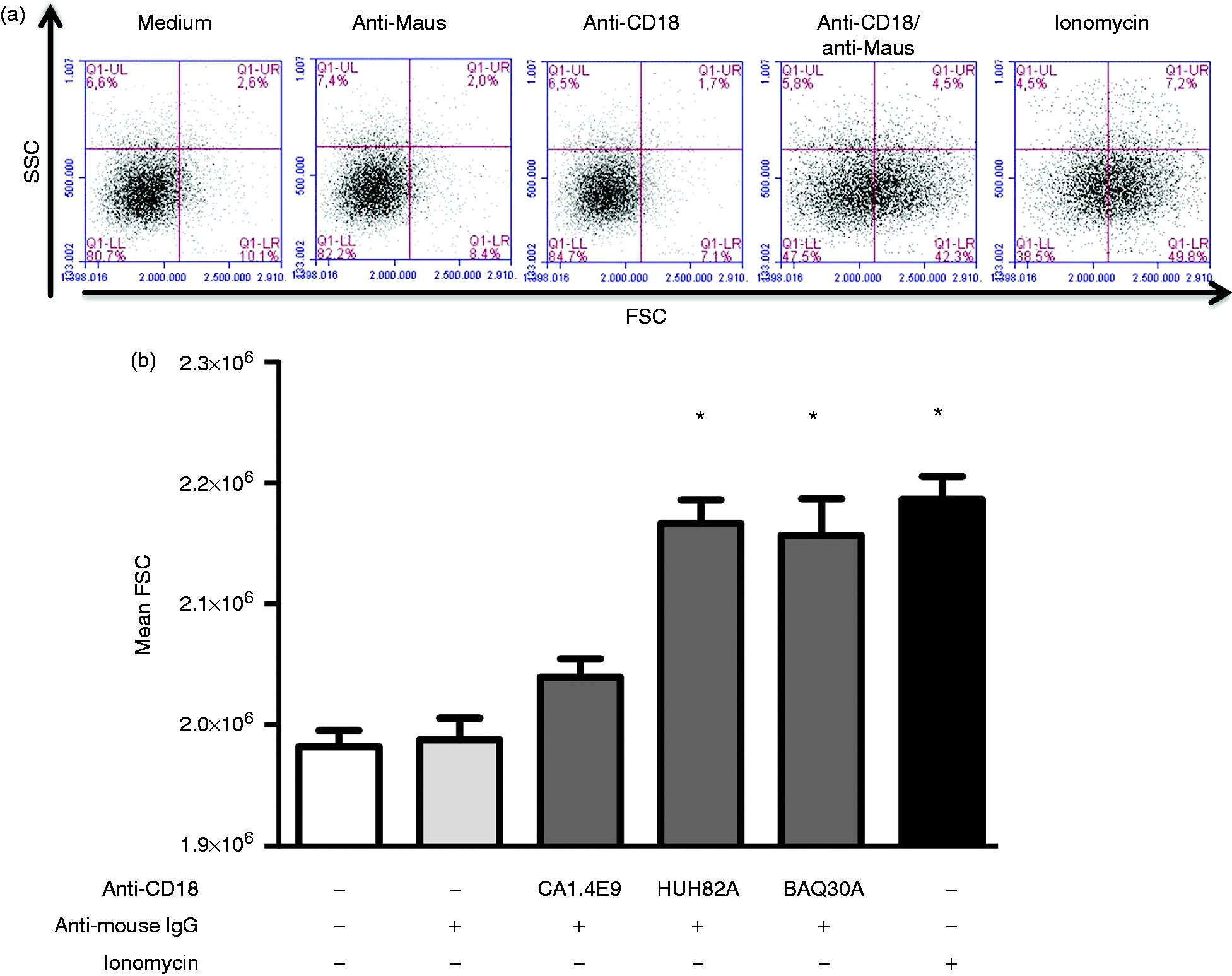
Looking for a CD18 Ab clone for the functional activation of PMN, we first tested three different clones of the primary CD18-specific mAb for their ability to activate bovine PMN upon cross-linking with a goat anti-mouse IgG F(ab′)2. While the cross-linking of the anti-canine CD18 Ab did not induce a shape change of bovine PMN, the cross-linking of two bovine CD18-specific Abs induced a shape change of bovine PMN (P < 0.001), which was comparable to activation with ionomycin (Figure 1b). For further experiments to induce PMN-degranulation, the clone HuH82A was chosen, which also induced more MPO release than the other two clones (data not shown).
In opposite to the incubation of PMN in HBSS, with the primary CD18 Ab alone or with the secondary goat anti-mouse IgG F(ab′)2 Ab alone, cross-linking of CD18 on the surface of PMN resulted in the up-regulation (P < 0.05) of PMN CD16 expression (Figure 2a). Supernatants of cross-linked PMN showed an increase in the enzymatic activity of alkaline phosphatase (Figure 2b) and MPO (Figure 2d) (P < 0.5). Also, the concentration of bovine lactoferrin was higher in supernatants of cross-linked PMN in comparison to supernatant of PMN in HBSS or PMN incubated with the primary CD18 Ab alone or with the secondary goat anti-mouse IgG F(ab′)2 Ab alone (Figure 2c). As a positive control, stimulation of PMN with ionomycin resulted in upregulation of CD16 on PMN, increased alkaline phosphatase and MPO enzymatic activity of the supernatant as well as a higher concentration of lactoferrin in the supernatant of stimulated PMN (P < 0.05) (Figure 2a–d). The detection of alkaline phosphatase, lactoferrin and MPO in the cell-free supernatants of cross-linked PMN indicates the release of their tertiary, secondary and primary granules. We subsequently used these cell-free supernatants as PMN-DGP to study their potential to modulate the activation, the migration and the differentiation of bovine monocyte subsets.
Analysis of PMN degranulation products. Bovine PMN were pretreated with mouse mAb against CD18 and were then subjected to CD18 cross-linking through addition of goat anti-mouse F(ab′)2 for 10 min. Alternatively, PMN were stimulated with ionomycin for 10 min. The release of PMN secretory vesicles was analyzed flow cytometrically by determining CD16 expression (a) on activated neutrophils (n = 5 animals) after labeling the cells with a murine mAb specific for CD16.The enzymatic activity of alkaline phosphatase (b) was determined in the supernatant of degranulated PMN by colorimetric assays (n = 6 animals, optical densities, OD). Bovine lactoferrin release (c) in the supernatants of degranulated PMN (n = 6 animals) was measured by ELISA. Myeloperoxidase enzymatic activity (d) was determined in the supernatant of degranulated PMN by colorimetric assays (n = 6 animals, optical densities, OD). All data are shown as mean ± SEM (* = P < 0.05 in comparison to HBSS control).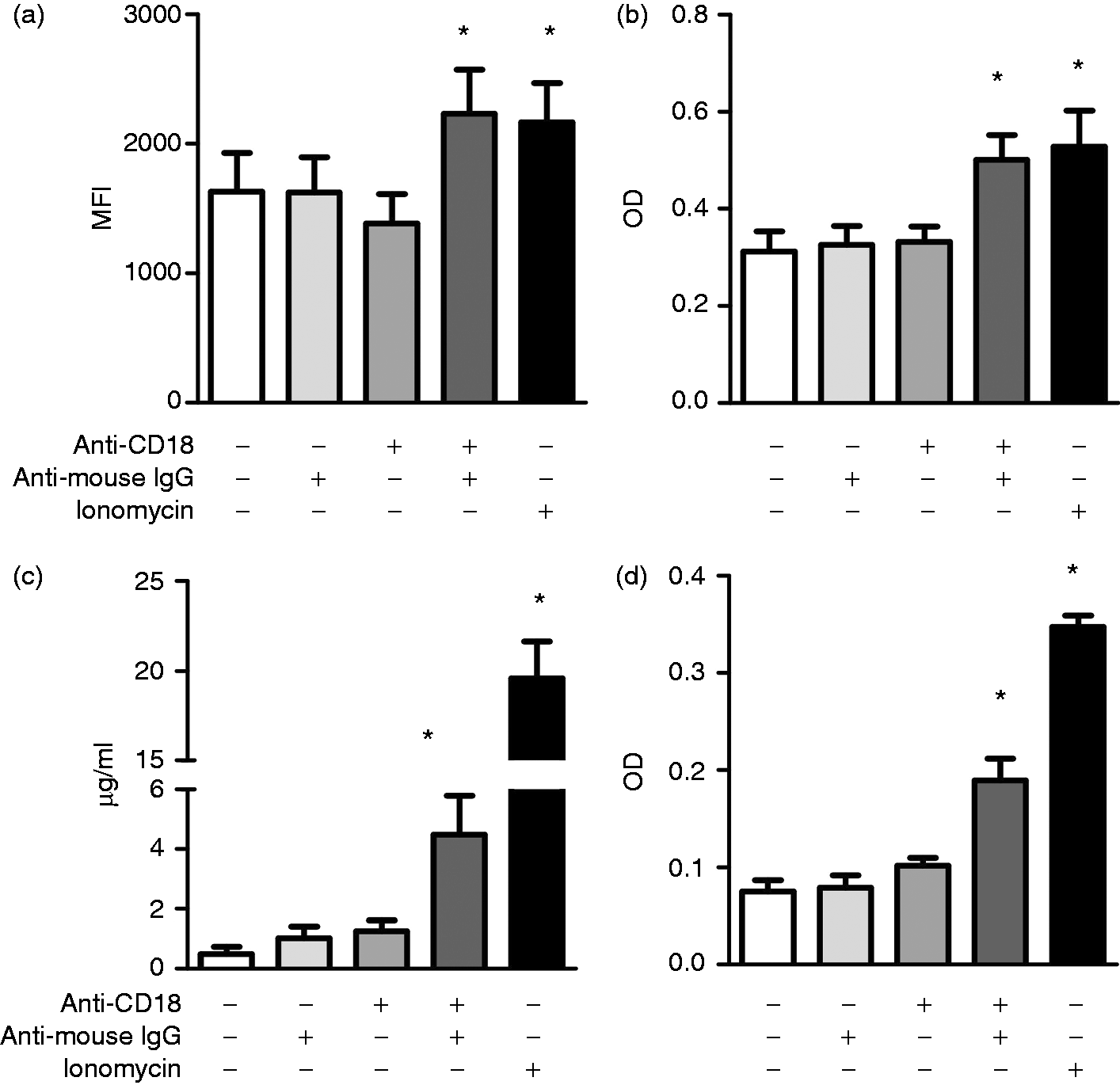
Neutrophil secretions induce Ca2+ mobilization in bovine classical and intermediate monocyte subsets
To see whether PMN-DGP have an activating potential for distinct monocyte subsets we analyzed Ca2+ mobilization in bovine cM, intM and ncM after stimulation with PMN-DGP. Compared to supernatant of PMN in HBSS, supernatants of CD18 cross-linked neutrophils (PMN-DGP) induced a comparable rise of intracellular Ca2+ in bovine classical and intermediate monocytes (Figure 3, P < 0.05). Notably, non-classical monocytes did not respond towards PMN-DGP (Figure 3), although all monocyte subpopulations responded in the same way towards ionomycin stimulation. The contaminating F(ab′)2 fragment of the secondary, cross-linking Ab did not exhibit a significant Ca2+ influx-inducing potential (3.1 ± 0.3, mean ± SEM) in bovine monocytes in comparison to supernatant from non-stimulated PMN cultured in HBSS (2.0 ± 0.4, mean ± SEM).
PMN-DGP-induced Ca2+-influx in bovine monocyte subsets. A) Bovine mononuclear cells (MNC) were stained with mAbs specific for CD14 and CD16 and were preloaded with Fluo-4. (a) Green fluorescence (FL1) was plotted against time. For gated bovine monocyte subsets, Ca2+influx in classical (cM), intermediate (intM) and non-classical monocytes (ncM) was measured as the percentage of cells above the horizontal line after addition of the stimulus (arrows). (b) The percentage of responding monocytes towards HBSS (supernatant from PMN cultures in HBSS, negative control), ionomycin (positive control) or PMN-DGP is presented as mean ± SEM (n = 6 animals; * = P < 0.05 in comparison to HBSS control).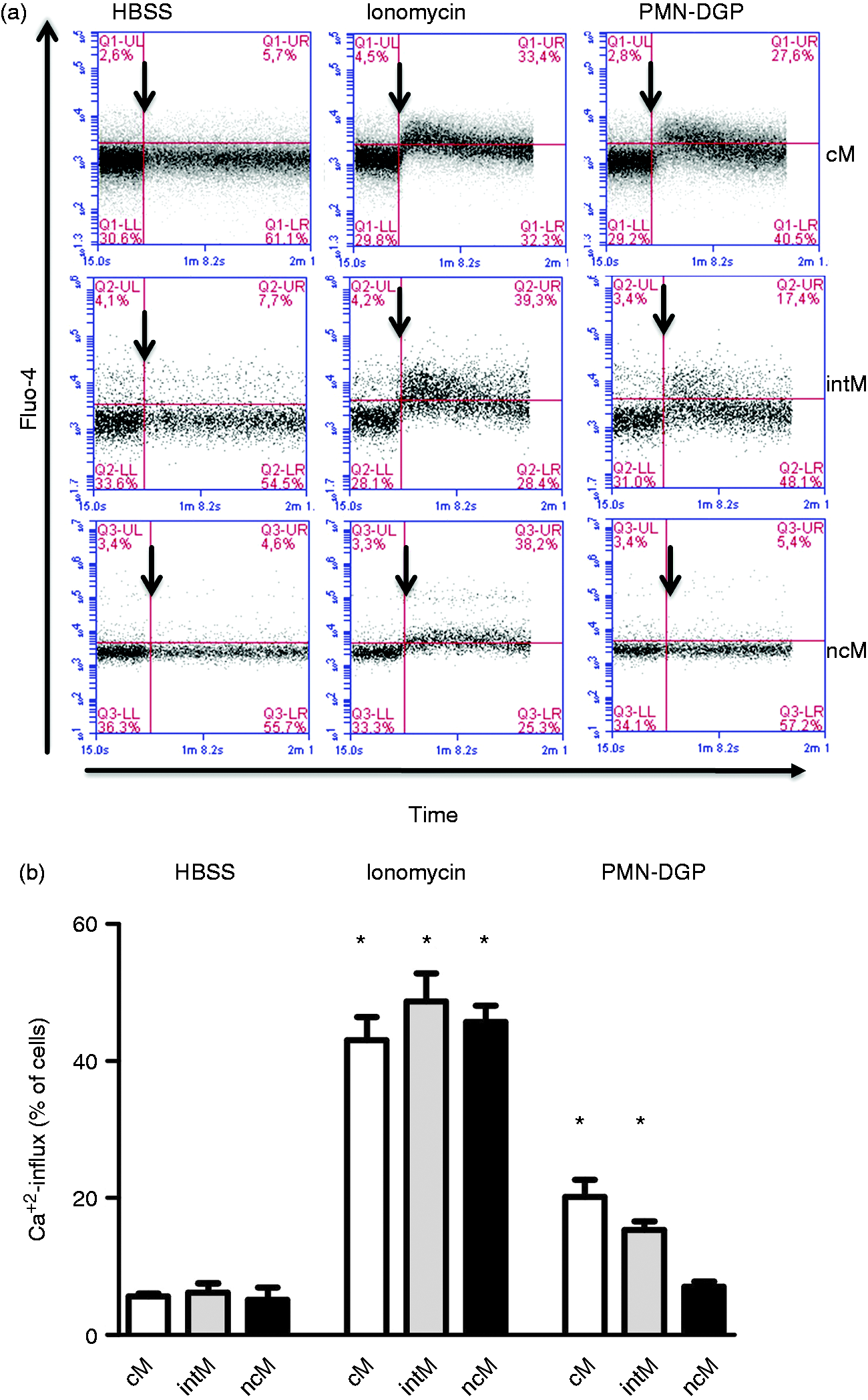
Neutrophil secretions modulate the expression of adhesion molecules on bovine monocyte subsets
Looking for a possible role of PMN-DGP in the adhesion and migration of bovine monocyte subsets, the surface expression of adhesion molecules on bovine monocyte subsets was determined flow cytometrically. Classical monocytes and intM showed a 3-4 times higher expression (mean fluorescence intensities, MFI) of CD62L and CD11b compared to ncM (P < 0, 05, Figure 4). The surface expression density (MFI) of CD11a and CD49d was lowest on cM, higher on intM and highest on ncM (P < 0.05). CD31 and CD18 were higher expressed (MFI) on intM compared to cM or ncM (P < 0.05; Figure 4).
Flow cytometric analysis of adhesion molecules on bovine monocyte subsets. Bovine MNC were labeled with mouse mAbs specific for CD14 and CD16 to define bovine monocyte subsets. MNC were also labeled with mouse mAbs specific for CD62L (a), CD11b (b), CD31 (c), CD18 (d), CD11a (e) or CD49d (f) and labeled cells were analyzed on the flow cytometer. After dead cell exclusion with propidium iodide, gates were made on cM, intM or ncM subsets in a CD14 versus CD16 dot plot. Mean fluorescence intensities of adhesion molecules were determined for gated bovine cM, intM and ncM (mean ± SEM, n = 13 animals; * = P < 0.05).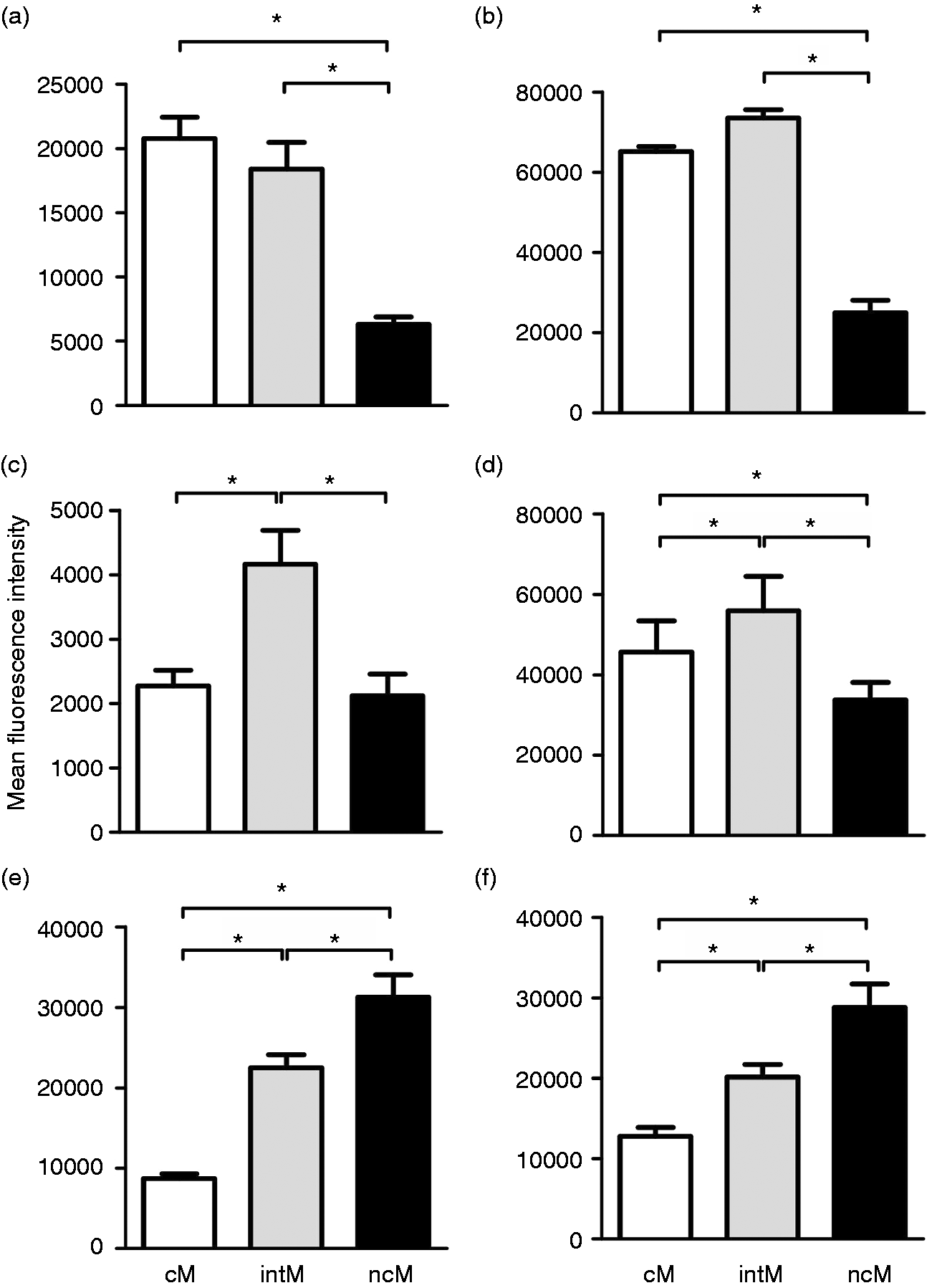
The incubation of bovine monocytes with PMN-DGP resulted in the up-regulation of CD11a and CD31 on intM (P < 0.005). The expression of CD18, CD11b, CD49d or CD62-L was not affected (P > 0.05) by PMN-DGP (Figure 5).
PMN-DGP-induced modulation of adhesion molecule expression on bovine monocyte subsets. Bovine MNC were incubated without or with bovine PMN-DGP for 2 h. Incubated cells were then labeled with mouse mAbs specific for CD14 and CD16 to define bovine monocyte subsets. MNC were also labeled with mouse mAbs specific for CD62L (a), CD11b (b), CD31 (c), CD18 (d), CD11a (e) or CD49d (f) and labeled cells were analyzed on the flow cytometer. MFI were determined for gated bovine cM, intM and ncM. MFI values of cells in control set ups (MNC in RPMI medium) were set to 100 % (mean ± SEM; n = 13 animals; * = P < 0.05).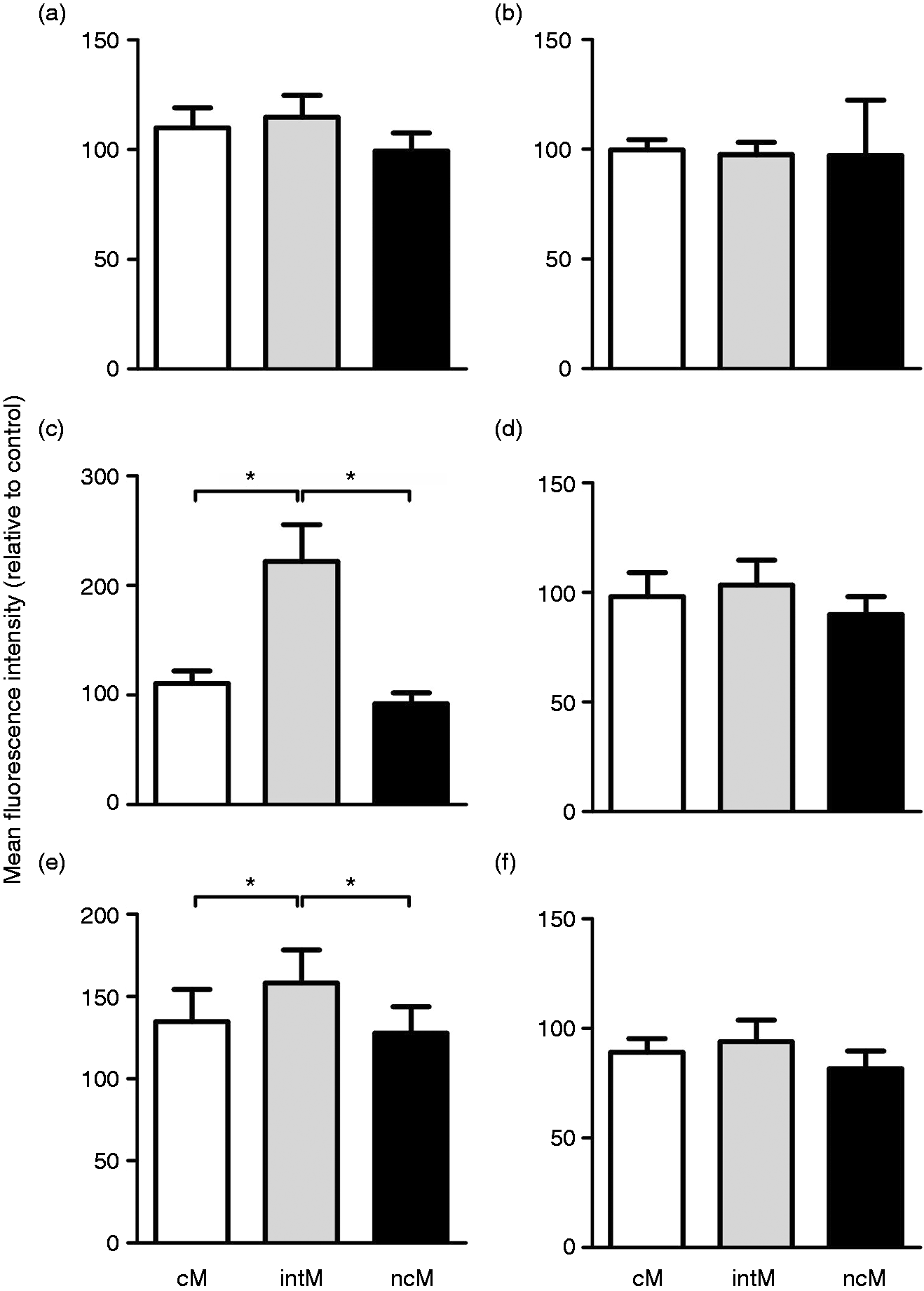
Neutrophil secretions induce a selective migration of bovine intermediate monocytes in vitro
To investigate the role of PMN in the selective recruitment of distinct monocyte subsets, the migration of bovine monocyte subsets toward PMN-DGP was evaluated using a transwell migration system. Although cM showed a higher random migration rate (46% ± 4%; mean ± SEM) toward HBSS, no increase in their migration rate (56% ± 10%; mean ± SEM) could be induced by PMN-DGP (P > 0.05, Figure 6a). For ncM, with lower random migration rates about (22%± 9%; mean ±SEM), PMN-DGP showed no effect (P > 0.05) on the migration rates of ncM (24% ± 9%; mean ± SEM). IntM showed the lowest migration rates toward HBSS (14% ± 6%; mean ± SEM) and displayed a enhanced migration (32% ± 7%; mean ± SEM) towards PMN-DGP (P < 0.05) (Figure 6).
PMN-DGP-induced migration of bovine monocyte subsets. MACS-separated bovine monocyte subsets were analyzed in a transwell system. Lower chambers were filled with 500 µl HBSS or PMN degranulation products (PMN-DGP). After 4 h non-migrating (upper chamber) and migrating cells (lower chamber) were counted flow cytometrically. A) Migration rates of monocyte subsets were calculated as the percentage of migrating cells of total cells (sum of cells in the lower and upper chamber). Data are presented as mean ± SEM (n = 5 animals; * = P < 0.05).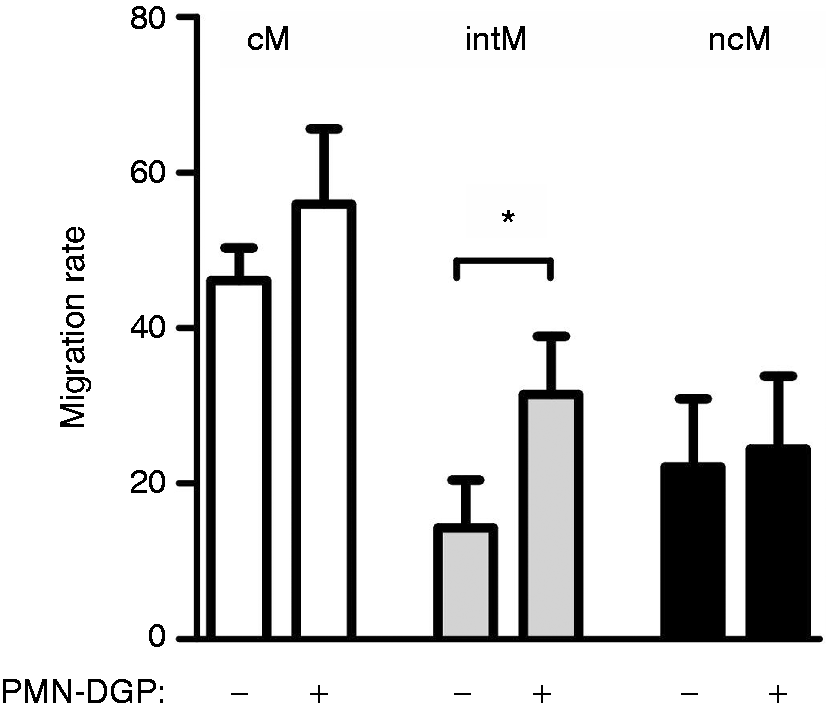
Neutrophil secretions guide the differentiation of bovine monocytes into macrophages with a distinct phenotype and cytokine profile
For bovine monocyte subsets responding towards PMN-DGP (cM, intM), the phenotype of monocyte-derived macrophages differentiated in the presence of PMN-DGP was determined by analyzing the surface expression of CD163 and MHC-II molecules. In the absence of PMN-DGP during the differentiation, cM-derived and intM-derived macrophages displayed a similar expression density of MHC-II molecules. CD163 expression was higher on cM-derived macrophages (P < 0.05, Figure 7). Compared to the differentiation in Iscové medium, the presence of PMN-DGP during the differentiation of monocytes into macrophages enhanced the expression of CD163 and reduced the expression of MHC-II on macrophages differentiated from both monocyte subsets (P < 0.05) (Figure 7).
Effects of PMN-DGP on the phenotype of bovine monocyte-derived macrophages. MACS-separated bovine cM and intM were differentiated in Iscové medium or in Iscové medium containing PMN-DGP for 4 d in vitro. Day 4 macrophages were labeled with mAbs specific for MHC class II (a) and CD163 (b). MFI after flow cytometrical analysis are presented as mean ± SEM (n = 9 animals; * = P < 0.05).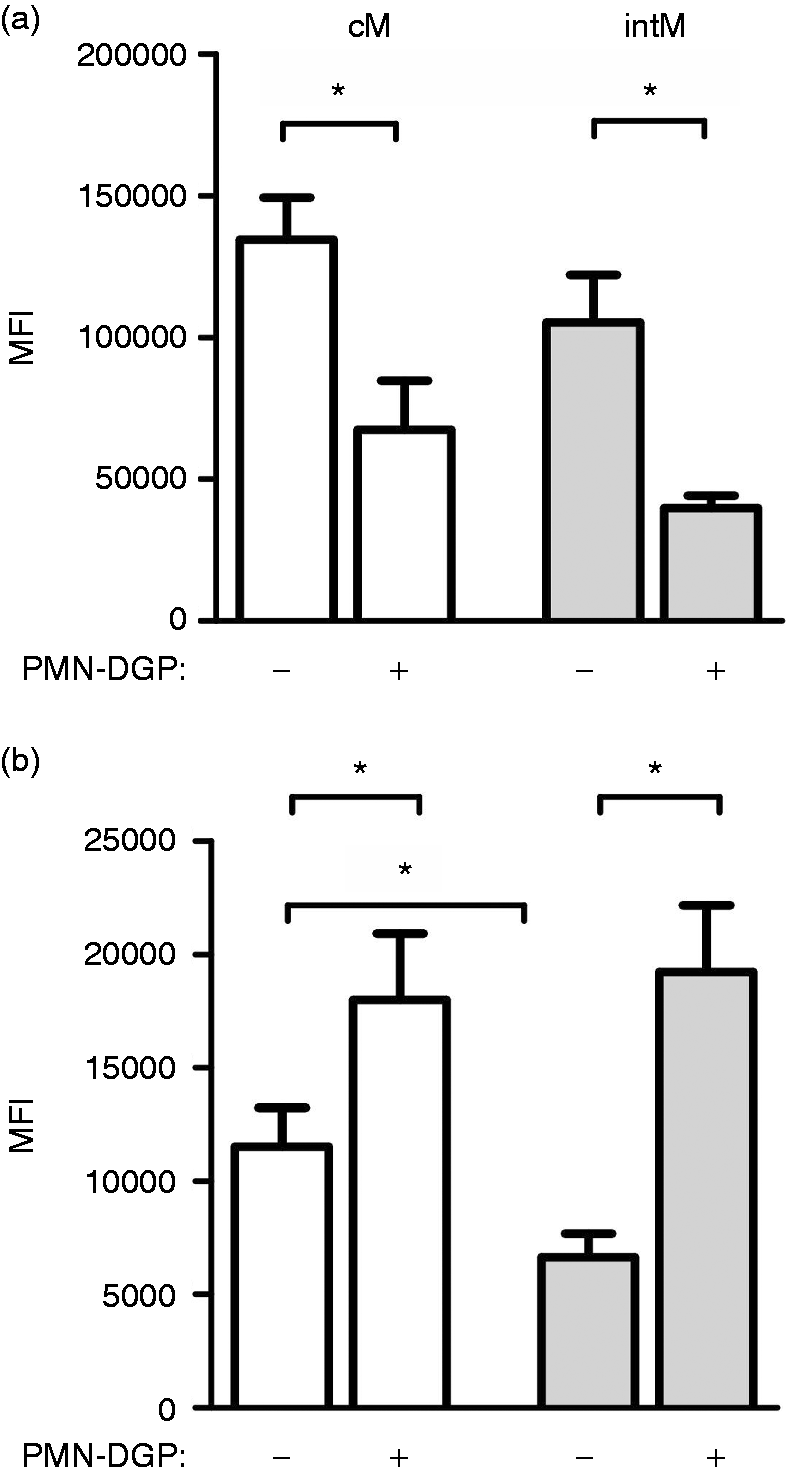
Both cM- and intM-derived macrophages generated in the presence of PMN-DGP secreted more IL-12 and IL-10 than macrophages differentiated in Iscové medium (P < 0.05) (Figure 8). The tendency of PMN-DGP guided intM-derived macrophages to produce less IL-10 and IL-12 than cM-derived macrophages was not significant (Figure 8).
Effect of PMN-DGP on cytokine production of bovine monocyte-derived macrophages. MACS-separated bovine cM and intM were differentiated in culture Iscové medium or in Iscové medium containing PMN-DGP for 4 d in vitro. IL-12 (a) and IL-10 (b) concentration in culture supernatants were determined by ELISA (n = 5; means ± SEM; * = P < 0.05)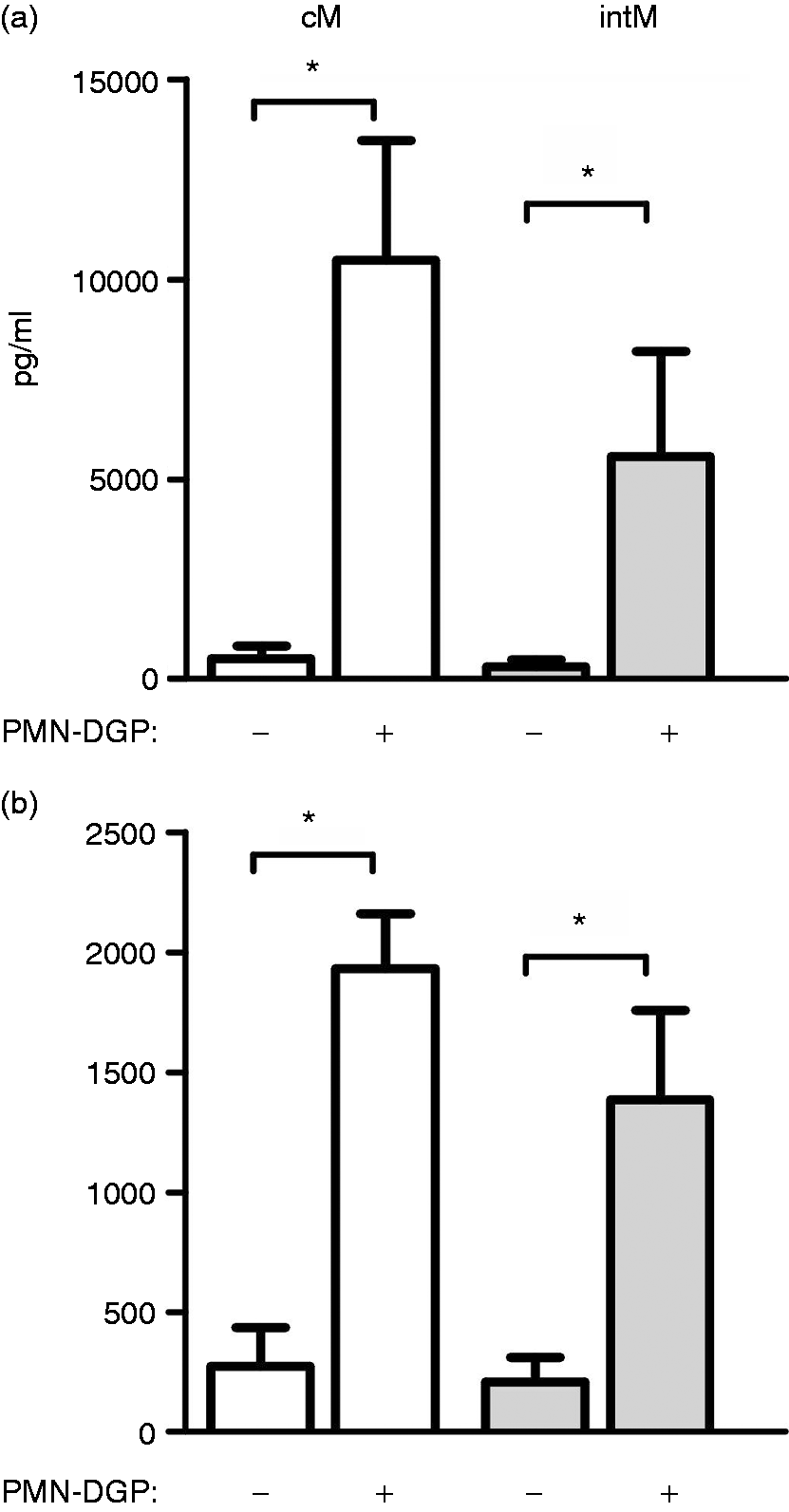
Monocyte-derived macrophages differentiated in the presence of neutrophil secretions show enhanced antimicrobial capacities
The results (Figure 7 and 8) showing no differences between cM- and intM-derived macrophages prompted us to investigate the antimicrobial capacities of CD14-positive monocyte-derived macrophages (including the cM and intM subset). The presence of PMN-DGP during monocyte differentiation into macrophages resulted in increased (P < 0.05) percentage of E. coli-positive macrophages (Figure 9a). Also, the generation of reactive oxygen species in response to E. coli stimulation was higher (P < 0.001) in macrophages generated in the presence of PMN-DGP (Figure 9b).
Effect of PMN-DGP on the antimicrobial function of bovine monocyte-derived macrophages. MACS-separated bovine CD14-positive monocytes were differentiated in culture medium (Iscové) or in medium containing PMN-DGP for 4 d in vitro. (a) Day 4 macrophages were incubated with killed, FITC-labeled E. coli. The percentages of phagocytosis-positive cells among viable (PI-negative) cells were determined flow cytometrically. Data from 3 tested animals were presented as mean ± SEM. (b) Generation of ROS was assessed flow cytometrically after incubation of d 4 macrophages with killed E. coli in the presence of DHR-123 for 30 min. Data are shown as mean ± SEM (n = 3 animals; * = P < 0.05).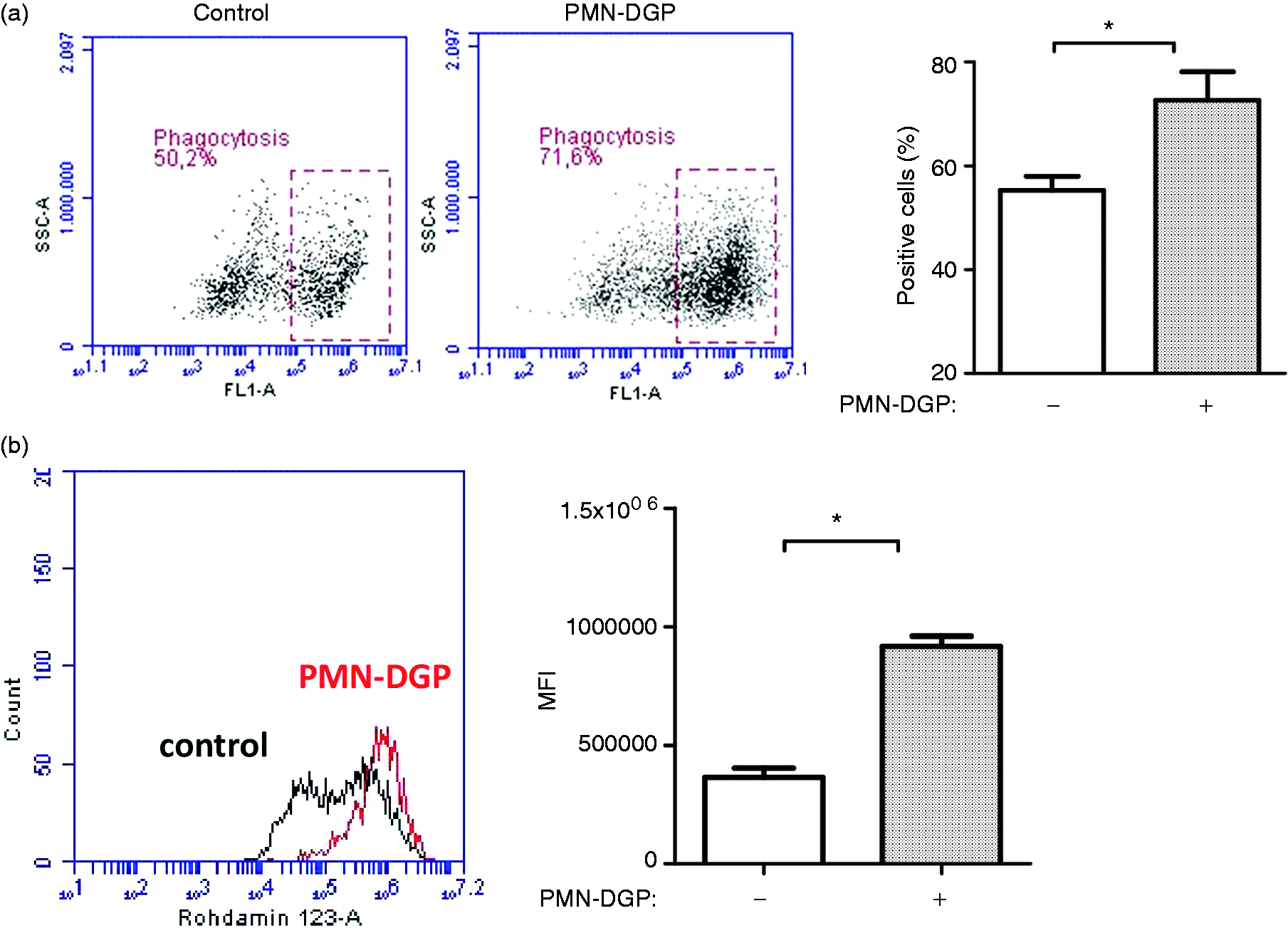
Discussion
Since the monocyte extravasation cascade involves a series of sequential interactions between monocytes, endothelial cells, and chemoattracting agents 29 we first addressed the expression of adhesion molecules on bovine monocyte subsets.
Similar to their human and murine counterparts, 30 bovine cM showed the highest expression of L-selectin (CD62L) when compared with intM and ncM subsets. This is also the case for the CD11b (Mac1) expression level. Thus, the expression of adhesion molecules on cM and intM as well as the lower expression of CD11b and CD62L on ncM is comparable to the human system.4,30
The crawling behavior of murine and human ncM along endothelial cells in blood vessels is mediated by the integrin LFA1 (CD11a).3,4 In addition, the very late antigen-4 (VLA-4 or CD49d), which promotes monocyte adherence to endothelial vascular cell adhesion molecule-1 (VCAM-1) was shown to be highest expressed on human ncM.4,31 The highest expression level of LFA1 and VLA-4 on bovine ncM suggests a similar patrolling function of this monocyte subset in the bovine system.
However, in contrast to human and murine monocytes where the ncM subset was shown to express the highest level of the platelet endothelial cell adhesion molecule (PECAM1, CD31),30,32 we found the highest level of PECAM1 on bovine intM monocytes. PECAM1 triggers both the adhesion of leucocytes to endothelial cells and integrin-mediated migration into surrounding tissues.33,34 Thus, one may speculate that this molecule primarily contributes to the accumulation of the bovine intM rather than the cM or ncM subset.
PMN which dominate the early immune response to acute infection and injury,6,16 contribute to the recruitment of a second wave of monocytes 35 by releasing their preformed PMN granule proteins that initiate monocyte adhesion and recruitment. 10
Although CD18 cross-linking 21 resulted in the release of secretory vesicles, tertiary, secondary, and primary granules of PMN, this approach takes not into account that PMN release their granule contents sequentially, which may also result in other kinds of monocyte modulations observed here. On the other hand, the chosen approach minimizes unwanted effects on monocytes of more natural PMN stimuli, which would be still present in the supernatants of activated PMN. The contaminating F(ab′)2 fragment of the secondary, cross-linking Ab did not exhibit any Ca2+ influx-inducing potential. Therefore, effects of PMN-DGP on bovine monocytes can be regarded as specific for PMN granule contents.
Whereas studies in human 36 and mice 10 indicate the involvement of neutrophil degranulation products in the recruitment of classical monocytes nothing is known about the differential PMN-DGP responsiveness of bovine monocyte subsets. PMN-DGP induced a selective migration of bovine intM in vitro. This is also supported by the PMN-DGP induced selective upregulation of CD31 and CD11a on intM.
Interestingly, this migration and adhesion molecule inducing potential of bovine PMN-DGP was only observed for intermediate monocytes, although PMN-DGP induced Ca2+ influx in both classical and intermediate monocytes. This is roughly in parallel with the murine system, where stimulation with PMN-DGP resulted in Ca2+-mobilization in inflammatory monocytes, 10 which include both the murine classical and intermediate monocytes. The lack of a Ca2+ inducing potential of PMN-DGP for bovine ncM is paralleled by findings in the human system. 1
Following extravasation, monocytes differentiate into different macrophage populations after conditioning by local mediators. 17 Having shown via Ca2+influx induction that both cM and intM are targets of PMN-DGP, we addressed the question whether PMN-DGP, when present during the in vitro differentiation, also guide the differentiation of these monocytes into macrophages with distinct phenotype and function. Although the induced up-regulation of CD163 and the down-regulation of MHC class II on bovine DGP-differentiated macrophages indicates a M2 phenotype, 37 the increased production of the inflammatory cytokine IL-12 as well as the anti-inflammatory cytokine IL-10 argues against a polarizing effect of PMN-DGP on monocyte differentiation into macrophages as supposed by others.38-40 For instance, human monocytes were polarized towards the M1 phenotype by the cathelicidin LL-37, a degranulation product of human neutrophils. 18 Whether such effects are balanced by the actions of other PMN degranulation products or whether the bovine LL-37 homologue BMAP-27 induces the same kind of polarization is not known. It may also be, that the PMN-DGP-induced mixed M1/M2 phenotype of bovine macrophages depends on a species-specific composition of PMN granule molecules. This composition differs between human and bovine neutrophils. 35 In addition, bovine neutrophils contain a unique large granule type, which is not evident in human cells.41,42 As CD163 was differently expressed on cM- and intM-derived macrophages generated in the absence of PMN-DGP, the comparable expression of CD163 between PMN-DGP-guided cM- and intM-derived macrophages indicates no selective effect of PMN-DGP on the differentiation of bovine cM or intM into macrophages.
As no phenotypic or functional differences could be seen between PMN-DGP-guided cM- and intM-derived macrophages, we analyzed the antimicrobial capacity of macrophages differentiated from bovine CD14-positive monocytes, including cM and intM, in presence of PMN-DGP. The enhanced capacity of such macrophages to phagocytize and to generate reactive oxygen species is in agreement with a previous study with human and murine macrophages, where PMN-DGP enhanced the phagocytosis and the production of ROS after stimulation with E. coli. 43 Thus, although many direct and indirect antimicrobial activities have been described for PMN-DGP, 44 our results clearly indicate a direct effect of PMN-DGP on macrophages rather than a mere opsonization effect.
Conclusion
In conclusion, the selective PMN-DGP induced up-regulation of adhesion molecules on intM and the migration inducing potential of PMN-DGP for intM suggests that degranulating neutrophils take part in the selective attraction of the bovine intermediate monocyte subset. PMN-DGP do not polarize monocyte-derived macrophages but result in the differentiation towards a mixed macrophage phenotype with enhanced antimicrobial functions.
Footnotes
Acknowledgements
We highly acknowledge the skilful assistance of Silke Schoeneberg and Udo Rabe.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
